Back to Journals » Clinical Ophthalmology » Volume 13
Optic disc pit maculopathy: tamponade of maculoschisis
Authors Morris RE , Hashimi H , McFarland AJ , Kuhn F, Sapp M, Oltmanns M
Received 16 April 2019
Accepted for publication 26 July 2019
Published 5 September 2019 Volume 2019:13 Pages 1735—1741
DOI https://doi.org/10.2147/OPTH.S212421
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Video abstract presented by Robert E Morris.
Views: 1763
Robert E Morris,1–3 Hannah Hashimi,4 Andrew J McFarland,1,2,5 Ferenc Kuhn,2,6–7 Mathew Sapp,1–3 Matthew Oltmanns1–3
1Retina Specialists of Alabama, Birmingham, AL, USA; 2Helen Keller Foundation for Research and Education, Birmingham, AL, USA; 3Department of Ophthalmology, University of Alabama at Birmingham (UAB), Birmingham, AL, USA; 4School of Medicine, UAB, Birmingham, AL, USA; 5Retina Associates, Winchester, VA, USA; 6Milos Eye Hospital, Belgrade, Serbia; 7Zagorskeigo Eye Hospital, Krakow, Poland
Correspondence: Robert E Morris
Retina Specialists of Alabama, 2208 University Blvd, Birmingham, AL 35233, USA
Tel +1 205 558 2598
Fax +1 205 558 2596
Email [email protected]
Purpose: To present rapid and safe closure of the pit-macula communication (PMC) by core vitrectomy and adequate duration gas tamponade as our preferred method of resolving optic disc pit (ODP) maculopathy and to define the term “maculoschisis” in ODP maculopathy as an alternative to the term “schisis-like.”
Patient and methods: A twenty-four-year-old female presented with an optical coherence tomography (OCT) confirmed ODP and a “giant” communicating maculoschisis cavity. Central macular thickness (CMT) measured 906 microns, and macular volume was twice normal, at 20.8 mm3. Snellen corrected visual acuity was 20/70. Two months after initial vitrectomy performed elsewhere with short-term gas tamponade (SF6 20%), CMT and visual acuity were not significantly improved. Combined lens extraction/intraocular lens placement and repeat vitrectomy with C3F8 15% gas tamponade were performed, with one supplemental (office) gas injection.
Results: OCT imaging six weeks postoperatively showed definitive closure of the PMC with CMT reduced by 405 microns. Sequestered from its ODP source, foveal schisis fluid then resolved by 12 weeks postoperatively. At final follow-up 3.4 years postoperatively, the macula remained dry with a CMT of 322 microns and a concave foveal contour. Macular volume was reduced to (a normal) 10.2 mm3 and visual acuity had improved to 20/25.
Conclusion: No report heretofore has documented rapid, sustained closure of the PMC by gas tamponade as the preferred method of expeditiously resolving ODP maculopathy. However, tamponade PMC closure sequesters ODP fluid and uniquely provides early assurance of ultimate maculopathy resolution. In all other techniques, PMC closure is a trailing phenomenon and success remains uncertain during months to a year or more of (unsequestered) fluid resolution. We suggest that more invasive techniques (laser barrier application to the peripapillary choroid, vitreoretinal interface maneuvers, and pit-plugging) be withheld unless a recurrence is detected during subsequent examinations.
Keywords: optic disc pit, ODP, schisis, maculoschisis, ODP maculopathy, vitrectomy, tamponade, optical coherence tomography, OCT
Introduction
Optic disc pit (ODP) maculopathy was first described clinically in 1882 by Wiethe.1 For more than a century thereafter, macular fluid associated with ODP was referred to as macular detachment and was thought to directly exit an optic disc pit into the subretinal space.2,3 In 1988, Lincoff et al used stereo fundus photography to identify “schisis-like” intraretinal channels communicating with the pit and central, outer layer lamellar holes providing an indirect pathway for the accumulation of subretinal fluid.4 A decade later, with the advent of clinical optical coherence tomography (OCT) imaging, these observations were confirmed with maculoschisis (from the Ancient Greek “skhisis”, meaning splitting) being the most common form of ODP maculopathy and with (schisis) fluid usually entering the subretinal space through an outer layer hole distant from the pit.5,6
Vitrectomy is the most common form of ODP maculopathy treatment, with a 50–90% success rate of achieving foveal attachment when combined with various adjunctive techniques.7,8 But fluid resolution is usually prolonged, often requiring from 7 months to more than a year, while success remains uncertain.8,9 And in non-tamponade techniques, the pit-macula communication (PMC) remains open, closing only passively as a trailing phenomenon. Although seemingly obvious, no report has heretofore (OCT) documented tamponade closure of the PMC itself as the safest, quickest, and most certain method of resolving ODP maculopathy.
We here present the rare image of an ODP discharging fluid into a “giant” outer layer maculoschisis cavity, 906 microns high, extending throughout the central macula, doubling the normal macular volume. With this case, we show that safe and relatively rapid closure of the PMC was accomplished by vitrectomy and adequate duration gas tamponade alone, with relatively prompt subsequent resolution of foveal schisis fluid and a small subfoveal detachment.
Patient and methods
A twenty-four-year-old woman with no family history of eye disease presented with visual loss of at least two month’s duration in the right eye. Her left eye was normal to examination.
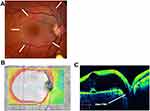
The right eye corrected visual acuity was 20/70 using a Snellen chart, and the anterior segment was normal. There was a clear and solid vitreous with no posterior vitreous detachment and a normal peripheral retina. A large pocket of fluid was seen at the macula, contacting a subtle optic disc pit visible at the inferior disc margin (Figure 1A). The disc was otherwise normal, and there was a small, non-communicating choroidal coloboma 3 mm inferior to the optic disc, commonly seen in ODP eyes.
OCT imaging showed a 906-micron high maculoschisis cavity in the outer retina, extending throughout the central macula with a small, central foveal detachment beneath a microscopic outer layer hole. Macular volume was 20.8 mm3 in the ODP right eye and 11.2 mm3 in the normal left eye. There was a wide-open communication between the schisis fluid and a definite pit (Figure 1B and C).
A 25-gauge vitrectomy was performed by a surgeon in the patient’s hometown, with attempted drainage of macular fluid through a small retinotomy inside the distal superotemporal arcade. Laser treatment was placed at this retinotomy and 20% SF6 tamponade was placed to encourage flattening of the schisis cavity postoperatively. Despite a week of full-time postoperative prone positioning and persistent nighttime prone positioning, the central macular thickness (CMT) remained little changed at 871 microns with the pit communication still wide open two months postoperatively.
After counseling and informed consent, we then performed a clear lens extraction with placement of a posterior chamber intraocular lens, enabling immediate return to college studies with no prone positioning during daylight hours; reduction of six diopters of myopia; and avoidance of secondary cataract from gas contact. In the same operation, a 25-gauge vitrectomy with placement of more prolonged C3F8 15% tamponade was performed. A small epimacular membrane that had developed parafoveal subsequent to previous vitrectomy was also removed. Post-operative instructions were to position facing downward only while sleeping.
Results
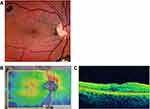
Six weeks later, the CMT had decreased from 906 microns at presentation to 501 microns, macular volume had decreased from 20.8 mm3 at presentation to 15.3 mm, and the PMC was closed (Figure 2A and B). As a further assurance of adequate tamponade, an office-based, sequential liquid/gas exchange was then performed using a 27-gauge needle placed through the inferior pars plana, adding 3 cc of C3F8 30% as the prior tamponade resolved.
Twelve weeks postoperatively, the maculoschisis had resolved at the fovea as it contracted inferonasally, and the PMC remained closed (Figure 3A and B). The patient moved to another city and was lost to frequent follow-up until 3.4 years after the initial presentation. The maculoschisis fluid had completely resolved and the fovea was dry and concave with visual acuity improved to 20/25. The macular volume had diminished to a normal 10.2 mm3 and the CMT was 322 mm (Figure 4A–C).
Discussion
Reports in the OCT era have shown a myriad of ODP maculopathy anatomic variations,10,11 with most cases including “schisis” fluid within the macula. Schisis (Greek skhisis) is technically a word fragment and in medicine is combined with an anatomical prefix, such as palatoschisis, iridoschisis, retinoschisis, or maculoschisis, to denote a clefting or separation of tissues.
We here define maculoschisis as the accumulation of reversible intraretinal fluid in the macula, separating inner and outer retinal layers, with preserved axonal connections. These delicate connections distinguish maculoschisis from (peripheral) retinoschisis, in which all such connections are permanently broken. This explains partially preserved visual acuity in maculoschisis and provides the basis for improvement in central vision upon fluid resolution. Unlike peripheral retinoschisis, maculoschisis is a secondary form of schisis with fluid entering the retina from an adjacent pit and/or resulting from traction maculopathy.12,13
This report uniquely demonstrates a very large maculoschisis cavity in the outer retina with a prominent PMC. In contrast to many other ODP reports that show shallow schisis channels overlying relatively large macular detachments,11,14 here a “giant” schisis cavity developed and apparently remained a pure maculoschisis until finally breaking through a microscopic outer layer hole to form a small detachment at the fovea (Figure 1C), as first described by Lincoff et al.4
Sobol et al showed that the natural course of ODP maculopathy results in 80% of eyes having 20/200 visual acuity or less, at an average follow-up of nine years.15 Because of both increased recognition of this poor prognosis and advances in vitrectomy technology, treatment of ODP maculopathy has been a subject of increasing interest in the vitrectomy/OCT era.
All recently reported treatments have included vitrectomy with some form of adjunctive procedure. But due to its rarity and poorly understood pathogenesis, there have been no clinical trials of proposed ODP maculopathy treatments and no treatment consensus other than vitrectomy has developed.
Many ODP treatment reports mention in passing that a posterior vitreous detachment was created, as though it was simple, innocuous, and required.16 But in young ODP maculoschisis eyes (with typically strong cortical vitreous adherence to ILM overlying tenuous neural connections), producing cortical vitreous detachment can be especially hazardous (Figure 5A and B)17 and should be performed with extreme caution. In contrast, gas tamponade actually safeguards fragile maculoschisis tissue (Figure 6).
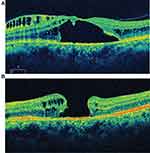 |
Figure 5 (A) Preoperative OCT showing intact foveal tissue, albeit with schisis and subretinal fluid. (B) OCT image of macular hole that developed after vitrectomy for ODP maculopathy.Note: Copyright © 2013. Dove Medical Press. Reproduced from Tzu JH, Flynn HW, Berrocal AM, Smiddy WE, Murray TG, Fisher YL. Clinical manifestations of optic pit maculopathy as demonstrated by spectral domain optical coherence tomography. Clin Ophthalmol. 2013;7:167–172.17Abbreviations: OCT, optical coherence tomography; ODP, optic disc pit. |
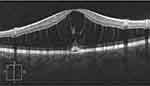 |
Figure 6 Within the maculoschisis cavity, tenuous axonal strands remain, linking the inner and outer retinal layers. |
Core vitrectomy with an effort to remove only cortical vitreous that separates easily relieves possible occult anteroposterior vitreous traction as a triggering mechanism.18 Moreover, there is no significant evidence for tangential traction in ODP maculopathy requiring cortical vitreous separation from the macular surface as a condition of success.19,20 ILM peeling, which we introduced in 1990,21 poses further iatrogenic tractional hazard and is not required.
Conclusion
Thirty years ago, Morris and Witherspoon reported the first successful treatment series for this rare condition, achieving complete resolution of ODP maculopathy in 8 of 8 eyes using core vitrectomy, gas tamponade, and laser retinopexy underlying the papillomacular bundle.20 Four of the 8 eyes required a second tamponade and/or laser treatment, immediately upon clearance of macular gas tamponade, based on persistent fluid seen over the laser treatment on contact lens examination. They thus permanently closed the PMC in all eyes by carefully creating a physical barrier to the egress of pit fluid.
With OCT imaging, we have since learned that once flattened by vitrectomy and adequate duration gas tamponade, the PMC typically remains closed without the need of laser retinopexy,16 and all sequestered ODP maculopathy fluid subsequently resolves. OCT imaging also helps guide tamponade so as to be utilized long enough, and only long enough, to achieve PMC closure. Adequate duration tamponade may require pseudophakia as the price of most safely regaining optimal macular function, depending on patient tolerance of positioning.
In summary, the novel insight from this report is that PMC closure by vitrectomy with tamponade alone, without higher risk peripapillary laser or macular dissection techniques, is effective and is the only reliable, early indicator of ultimate success, obviating months of outcome uncertainty. Subsequent OCT monitoring then serves to confirm progressive resolution and detect recrudescence. This is now our preferred ODP maculopathy treatment, with higher risk laser retinopexy and/or vitreoretinal interface surgery reserved only for a rare recurrence.
Informed consent
The patient described has given written informed consent to publication of this report, including images.
Acknowledgment
The authors gratefully acknowledge Jessica Haynes and Chris Sullivan for their extraordinary research assistance, editorial services, and administrative support. They are also grateful to Paul Kaufman, MD, and Ms. Kat Barber for imaging and examination assistance to achieve final follow-up. Funding was partially provided by the Helen Keller Foundation for Research and Education through a grant from the Pete Hanna Charitable Trust. This retrospective case report is exempt under regulations put forth by the University of Alabama in Birmingham Institutional Review Board.
Disclosure
The authors report no conflicts of interest in this work, financial, or otherwise.
References
1. Wiethe T. Ein- Fall von angeborener deformitat der schnervenpapile. Arch F Augenh. 1882;11:14–19.
2. Kranenburg EW. Crater-like holes in the optic disc and central serous retinopathy. Arch Ophthalmol. 1960;64:912–924. doi:10.1001/archopht.1960.01840010914013
3. Gass JD. Serous detachment of the macula. Secondary to congential pit of the optic nervehead. Am J Ophthalmol. 1969;67(6):821–841. doi:10.1016/0002-9394(69)90075-0
4. Lincoff H, Lopez R, Kreissig I, Yannuzzi L, Cox M, Burton T. Retinoschisis associated with optic nerve pits. Arch Ophthalmol Chic Ill 1960. 1988;106:61–67. doi:10.1001/archopht.1988.01060130067030
5. Krivoy D, Gentile R, Liebmann JM, Stegman Z, Walsh JB, Ritch R. Imaging congenital optic disc pits and associated maculopathy using optical coherence tomography. Arch Ophthalmol Chic Ill 1960. 1996;114(2):165–170. doi:10.1001/archopht.1996.01100130159008
6. Rutledge BK, Puliafito CA, Duker JS, Hee MR, Cox MS. Optical coherence tomography of macular lesions associated with optic nerve head pits. Ophthalmology. 1996;103(7):1047–1053. doi:10.1016/s0161-6420(96)30568-x
7. Steel DHW, Suleman J, Murphy DC, Song A, Dodds S, Rees J. Optic disc pit maculopathy: a two-year nationwide prospective population-based study. Ophthalmology. 2018:125(11):1757–1764. Epub 2018 Jun 7. doi:10.1016/j.ophtha.2018.05.009
8. Avci R, Kapran Z, Ozdek Ş, et al. Multicenter study of pars plana vitrectomy for optic disc pit maculopathy: MACPIT study. Eye Lond Engl. 2017;31(9):1266–1273.
9. Hirakata A, Inoue M, Hiraoka T, McCuen BW. Vitrectomy without laser treatment or gas tamponade for macular detachment associated with an optic disc pit. Ophthalmology. 2012;119(4):810–818. doi:10.1016/j.ophtha.2011.09.026
10. Imamura Y, Zweifel SA, Fujiwara T, Freund KB, Spaide RF. High-resolution optical coherence tomography findings in optic pit maculopathy. Retina. 2010;30(7):1104–1112. doi:10.1097/IAE.0b013e3181d87ecb
11. Sanghi G, Padhi TR, Warkad VU, et al. Optical coherence tomography findings and retinal changes after vitrectomy for optic disc pit maculopathy. Indian J Ophthalmol. 2014;62(3):287–902012. doi:10.4103/0301-4738.111191
12. Morris R, Kuhn F, Witherspoon CD. Hemorrhagic macular cysts. Ophthalmology. 1994;101(1):1. doi:10.1016/s0161-6420(94)31162-6
13. Witherspoon CD, Morris R, Kuhn F, Taylor SW. The traction maculopathies: macular pucker, macular hole, cellophane maculopathy, vitreomacular traction syndrome. In: Boyd BS, Boyd S, editors. Highlights of Ophthalmology: Retinaland Vitreoretinal Surgery: Master the Latest Techniques. Panama: Highlights Press; 2002;333–352.
14. Bottoni F, Cereda M, Secondi R, Bochicchio S, Staurenghi G. Vitrectomy for optic disc pit maculopathy: a long-term follow-up study. Graefes Arch Clin Exp Ophthalmol. 2018;256(4):675–682.
15. Sobol WM, Blodi CF, Folk JC, Weingeist TA. Long-term visual outcome in patients with optic nerve pit and serous retinal detachment of the macula. Ophthalmology. 1990;97(11):1539–1542. doi:10.1016/s0161-6420(90)32380-1
16. Abouammoh MA, Alsulaiman SM, Gupta VS, et al. Pars plana vitrectomy with juxtapapillary laser photocoagulation versus vitrectomy without juxtapapillary laser photocoagulation for the treatment of optic disc pit maculopathy: the results of the KKESH International Collaborative Retina Study Group. Br J Ophthalmol. 2016;100:478–483. doi:10.1136/bjophthalmol-2015-307128
17. Tzu JH, Flynn HW, Berrocal AM, Smiddy WE, Murray TG, Fisher YL. Clinical manifestations of optic pit maculopathy as demonstrated by spectral domain optical coherence tomography. Clin Ophthalmol. 2013;7:167–172. doi:10.2147/OPTH.S37157
18. Gondorfer A, Kampik A. Role of vitreoretinal interface in the pathogenesis and therapy of macular disease associated with optic pits. Ophthalmologe. 2000;97:276–279.
19. Ooto S, Mittra RA, Ridley ME, Spaide RF. Vitrectomy with inner retinal fenestration for optic disc pit maculopathy. Ophthalmology. 2014;121(9):1727–1733. doi:10.1016/j.ophtha.2014.04.006
20. Cox MS, Witherspoon CD, Morris RE, Flynn HW. Evolving techniques in the treatment of macular detachment caused by optic nerve pits. Ophthalmology. 1988;95(7):889–896. doi:10.1016/s0161-6420(88)33078-2
21. Almony A, Nudleman E, Shah GK, et al. Techniques, rationale, and outcomes of internal limiting membrane peeling. Retina. 2012;32(5):877–891. doi:10.1097/IAE.0b013e318227ab39
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.