Back to Journals » Local and Regional Anesthesia » Volume 12
Obesity And Obstetric Anesthesia: Current Insights
Authors Taylor CR , Dominguez JE , Habib AS
Received 11 June 2019
Accepted for publication 18 October 2019
Published 18 November 2019 Volume 2019:12 Pages 111—124
DOI https://doi.org/10.2147/LRA.S186530
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Stefan Wirz
Cameron R Taylor, Jennifer E Dominguez, Ashraf S Habib
Department of Anesthesiology, Division of Women’s Anesthesia, Duke University, Durham, NC 27710, USA
Correspondence: Ashraf S Habib
Department of Anesthesiology, Division of Women’s Anesthesia, Duke University, 2301 Erwin Road, Durham, NC 27710, USA
Tel +1 919 668 2024
Fax +1 919 681 4698
Email [email protected]
Abstract: Obesity is a significant global health problem. It results in a higher incidence of complications for pregnant women and their neonates. Cesarean deliveries are more common in obese parturients as well. The increased burden of comorbidities seen in this population, such as obstructive sleep apnea, necessitates antepartum anesthetic consultation. These patients pose unique challenges for the practicing anesthesiologist and may benefit from optimization prior to delivery. Complications from anesthesia and overall morbidity and mortality are higher in this population. Neuraxial anesthesia can be challenging to place in the obese parturient, but is the preferred anesthetic for cesarean delivery to avoid airway manipulation, minimize aspiration risk, prevent fetal exposure to volatile anesthetic, and decrease risk of post-partum hemorrhage from volatile anesthetic exposure. Monitoring and positioning of these patients for surgery may pose specific challenges. Functional labor epidural catheters can be topped up to provide conditions suitable for surgery. In the absence of a working epidural catheter, a combined spinal epidural anesthetic is often the technique of choice due to relative ease of placement versus a single shot spinal technique as well as the ability to extend the anesthetic through the epidural portion. For cesarean delivery with a vertical supraumbilical skin incision, a two-catheter technique may be beneficial. Concern for thromboembolism necessitates early mobilization and a multimodal analgesic regimen can help accomplish this. In addition, thromboprophylaxis is recommended in this population after delivery—especially cesarean delivery. These patients also need close monitoring in the post-partum period when they are at increased risk for several complications.
Keywords: obesity, neuraxial anesthesia, labor analgesia, cesarean delivery
Introduction
Obesity continues to affect the general population worldwide, and its impact on the maternal-fetal unit represents a significant global health problem. In the United States, it is estimated that more than half of all pregnant women are obese.1 The World Health Organization uses the body mass index (BMI) to stratify individuals into the following classes: normal BMI 18.9–24.9 kg/m2; overweight BMI 25–29.9 kg/m2; and obese BMI > 30 kg/m2.2 Obesity is further sub-classified as class 1 for BMI 30–34.9 kg/m2, class 2 for BMI 35–39.9 kg/m2, and class 3 for BMI > 40 kg/m2.2 The following classification system also exists for individuals with class 3 obesity: morbid obesity applies to BMI 40–49.9 kg/m2; super obesity to BMI 50–50.9 kg/m2; and super-super obesity for BMI ≥ 60 kg/m2.3 There is currently no alternate obesity classification for parturients, although some groups have suggested using cut-off values 5 kg/m2 higher during pregnancy.4
Obesity is a systemic disease associated with multiple comorbidities (Table 1), and a higher risk of obstetric and peripartum complications (Table 2). Obesity has been associated with prolonged labor5 that is associated with complications for both mother and fetus. Obese parturients are also at an increased risk of requiring a cesarean delivery. Tabet et al6 examined 121,092 nulliparous women stratified by pre-pregnancy BMI and reported an odds ratio (95% confidence interval) for cesarean delivery of 1.50 (1.41, 1.59) for overweight women and 2.06 (1.91, 2.21) for obese women. Other large cohorts have reported similar findings.7,8
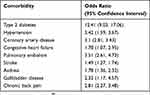 |
Table 1 Common Comorbidities Associated With Obesity Compared To Non-Obese Individuals |
 |
Table 2 Obstetric Complications In The Obese Compared With Non-Obese Parturients |
In 1993, Hood et al9 examined obstetric and anesthetic outcomes in obese parturients compared with their non-obese counterparts. They reported a higher rate of cesarean delivery, higher rates of initial epidural failure, and increased rates of postoperative complications. A study in 2015 by the same group reported that morbidly obese parturients were still more likely to undergo cesarean delivery, have longer first stage of labor, higher fetal weights, and an increased risk of neuraxial failure together with longer neuraxial procedure times.4,10
Several studies have reported an increased risk of maternal death for obese parturients. Schellpfeffer et al reviewed all pregnancy-related deaths in Wisconsin between 2006 and 2010 and showed that 76% of maternal deaths occurred in overweight or obese women.11 A review of maternal deaths in California from 2002 to 2005 also found that women who died were more likely to be obese or morbidly obese.12 Similarly, the United Kingdom’s confidential inquiry into maternal deaths showed that the majority of women who died from 2003–2013 were overweight or obese.13,14
Obesity has also been implicated as a factor in anesthesia-related maternal deaths. The last three reports from the Confidential Enquiries into Maternal Deaths in the United Kingdom concluded that six of the 17 women who died due to anesthesia-related complications were obese.13–15 In the United States, Mhyre and colleagues16 reviewed anesthesia-related mortality between 1985–2003 in Michigan and found that six of the eight women that died were obese.
In this chapter, we will discuss considerations for predelivery planning, anesthetic techniques for vaginal and cesarean delivery with a focus on neuraxial techniques, and postpartum care to optimize the care and safety of the obese parturient.
Anesthetic Consultation And Pre-Delivery Planning
Professional medical societies in both the United States and United Kingdom recommend a multidisciplinary approach to the management of obese parturients.17,18 The published recommendations from the Royal College of Obstetricians and Gynaecologists (RCOG) in conjunction with the Centre for Maternal and Child Enquiries and the American College of Obstetricians and Gynecologists (ACOG) include preconception counseling, screening for obesity using BMI, monitoring BMI throughout pregnancy, and implementing stringent limits for weight gain during pregnancy. Both RCOG and ACOG further recommend a formal anesthesia consultation for obese women, with the RCOG specifying that this applies to those with class 3 obesity in the third trimester.17
The anesthetic consultation provides an opportunity to obtain a thorough medical history and to screen for comorbidities. This time is well-spent conducting a comprehensive physical exam that includes a pulmonary and cardiovascular exam, as well as an airway exam. The obstetric anesthesiologist may use this visit to discuss anesthetic options for labor analgesia, as well as cesarean delivery and to elicit any history of difficult neuraxial placement or difficult airway. The parturient should be counseled that neuraxial analgesia placement may be challenging. For this reason, it is important to emphasize that these patients should plan to request neuraxial analgesia early in labor to allow sufficient time for placement and confirmation of block efficacy and reliability, and to decrease the risk of needing general anesthesia for an emergency cesarean delivery. This meeting should also be conducted early enough in pregnancy to allow for additional screening if warranted, such as screening for obstructive sleep apnea (OSA), and optimization of any maternal comorbidities.
Obesity is an important risk factor for OSA.19 One recent prospective study of late second and third trimester parturients with BMI ≥ 40 kg/m2 reported an OSA prevalence of 24%,20 whereas OSA is much less common in the general pregnant population with an 8% third-trimester prevalence reported in one recent large prospective study.21 Several studies suggest that OSA is associated with adverse pregnancy outcomes, particularly hypertensive disorders of pregnancy and gestational diabetes.21,22 OSA screening tools validated in non-pregnant populations have not been shown to be useful in several cohorts of pregnant women.20,23 However, some studies suggest that advanced maternal age, morbid obesity, chronic hypertension, and loud, frequent snoring are important risk factors for OSA in pregnancy.20,21,23,24
Anesthetic Considerations For Labor Analgesia
Neuraxial techniques are preferred for women undergoing delivery by any mode—this is especially true for the obese parturient. In the obese parturient, the placement of a well-functioning epidural catheter is one of the safest methods of providing labor analgesia. In addition to providing good pain relief, labor epidural analgesia can be converted to surgical anesthesia if a need for cesarean delivery arises. In addition, given the increased risk of fetal macrosomia, a well-functioning epidural catheter can be helpful in the management of a shoulder dystocia. Therefore, regular assessment of the labor epidural catheter is paramount to ensure that the block can be reliably extended to provide adequate surgical anesthesia if needed. When evaluating the epidural block, one that fails to achieve a sensory level of T10 or one that requires frequent top-ups may not ultimately allow for surgical anesthesia.25 As such, any epidural catheter that is not providing adequate labor analgesia should be replaced early to minimize the risk of failure should a cesarean delivery be required. Furthermore, obese parturients with OSA are likely to be more sensitive to the respiratory depressant of intravenous opioids if used for labor analgesia.
In addition to epidural analgesia, other neuraxial options for labor analgesia include combined spinal epidural (CSE), dural puncture epidural or continuous spinal technique.
CSE provides rapid onset of dense labor analgesia. However, one concern with the CSE technique is the “untested catheter.” In this scenario, the epidural catheter is not tested or used until the spinal component of the CSE wears off, leaving the possibility that the epidural catheter may fail to provide adequate anesthesia if a need for a cesarean delivery arises in this high-risk patient population. Multiple studies, however, suggest that the epidural failure rate when placed as part of a CSE technique is lower than that of a straightforward continuous epidural technique, as obtaining cerebrospinal fluid (CSF) through the spinal needle acts an additional confirmation that the epidural space has been reached.26,27 Furthermore, detection of an inadequate epidural catheter was not delayed with a CSE technique compared with a straight epidural technique in one study.28
An alternative technique to CSE in the obese parturient is the dural puncture epidural (DPE) technique. This technique is similar to a needle through needle CSE technique, except that after puncturing the dura with the spinal needle, no intrathecal medications are given, and all medications are given through the epidural catheter. A study by Chau and colleagues29 reported less asymmetric block with the DPE compared with the epidural technique, as well as less pruritus, hypotension, tachysystole and category II fetal heart tracing with the DPE compared with the CSE technique. Furthermore, return of CSF through the spinal needle confirms correct placement of the epidural needle, which is valuable in the obese parturient. Further studies confirming those findings in the obese parturient are warranted.
Continuous spinal labor analgesia may be considered: either unplanned following an inadvertent dural puncture; or electively. Elective use of this technique for labor analgesia is limited by the risk of post-dural puncture headache (PDPH) since it is usually performed using a 17 or 18 gauge Tuohy needle, given that smaller gauge spinal catheter over the needle kits are not widely available. Some authors do argue that obesity may actually have a protective effect against the development of PDPH.30 This may be related to increased intraabdominal pressure and the presence of engorged epidural veins and fat that may tamponade CSF leak.31 However, studies that have investigated the risk of PDPH in obese parturients have reported inconsistent results. A retrospective study by Miu et al32 did not show a difference in the incidence of PDPH between patients with BMI less than or greater than 30kg/m2. In contrast, a larger study by Peralta and colleagues suggested that obese patients (defined as BMI > 31.5kg/m2) were less likely to develop PDPH following inadvertent dural puncture with a Tuohy needle compared to those with lower BMI.33 A separate retrospective analysis concluded that the risk of PDPH was lower in parturients with BMI ≥ 50 kg/m2 following dural puncture compared to those with BMI < 50 kg/m2, but the risk was not reduced in those with BMI 30–39.9 kg/m2 or 40–49.9 kg/m2 compared to those with BMI <30 kg/m2.34 It is important to note, however, that in the studies that reported a reduced risk of PDPH with obesity,33,34 between 40–45% of obese patients still suffered PDPH. Continuous spinal anesthesia can also be achieved using a catheter over needle technique where a 22–24 gauge catheter is advanced over a 27- or 29-gauge Quincke needle, respectively, into the intrathecal space. This particular method has been reported to have a failure rate ranging from 9% to 24%,35,36 and has not been specifically evaluated in morbidly obese parturients. Another continuous spinal set available is the Wiley spinal catheter over the needle system, which consists of a flexible 23-gauge intrathecal cannula over a 27-gauge pencil-point spinal needle. Experience with this system is limited, and not specific to the obese patient population.37,38
Anesthetic Considerations For Cesarean Delivery
Neuraxial anesthesia is the preferred anesthetic for cesarean delivery. Surgical anesthesia for cesarean delivery may be accomplished using several neuraxial techniques: single-shot subarachnoid block (spinal); CSE; epidural; continuous spinal; or a double catheter technique (lumbar and thoracic catheters). General anesthesia is associated with more complications in the obese parturient, but may be necessary in emergencies or where a neuraxial technique is not feasible or contraindicated.
Single-Shot Spinal Anesthesia
There are many benefits to a single-shot spinal anesthetic technique for cesarean delivery. It produces a block that is dense, quick in onset, reliable, yields optimal surgical conditions with high level of patient comfort, and reduces the need for sedating analgesic adjuncts.39 Despite the benefits of this anesthetic technique, it does have limitations for morbidly-obese parturients. The primary consideration for this technique is the time-limited nature of the block. In the morbidly obese parturient, more time is required to position the patient as well as perform a cesarean delivery relative to a non-obese patient,40 therefore a continuous neuraxial technique is preferred. Another important concern when using this technique is the difficulty in block placement, particularly when using a 25–27 gauge pencil point spinal needle in a parturient with excessive truncal adipose tissue. The identification of the epidural space with the larger Tuohy needle is technically easier in an obese patient as it provides greater tactile feedback to the practitioner. The Tuohy needle then acts as an introducer for the spinal needle and facilitates identification of the intrathecal space using a needle through needle CSE technique. Ross et al41 reported that use of a CSE technique resulted in fewer attempts to identify the intrathecal space compared to the standard single-shot spinal technique in obese women undergoing cesarean delivery.
Combined-Spinal Epidural Anesthesia
A CSE technique combines the desirable features of a single-shot spinal technique with the ability to extend the block duration through the epidural catheter.40 Because operative times are often longer for obese parturients and CSE can be easier to place, CSE is the technique of choice for elective cesarean delivery in this population. It also allows for the administration of a lower spinal dose and titration of the anesthetic level using the epidural catheter, when a lower dose might be warranted such as in super obese parturients or those with cardiac disease. Extending the block can be accomplished using epidural saline (the epidural volume expansion technique) or additional local anesthetic. The epidural volume expansion technique, however, has not been specifically evaluated in the morbidly obese parturient and its effects may be less predictable.42
Continuous Spinal Anesthesia
A continuous spinal technique has several advantages: it allows for the gradual titration of a dense block to a desired cephalad level while monitoring hemodynamics and respiratory status; it minimizes the risk of a total spinal anesthetic; and it allows extension of block duration.43 The use of this technique has been advocated by some for emergency cesarean delivery in the obese parturient as it may be a more efficient way to locate the intrathecal space when compared to a smaller spinal needle or a combined spinal epidural technique.44 Routine use of this technique is however limited by the increased risk of PDPH as previously described.
Double Catheter Technique
Due to technical challenges when using a Pfannenstiel incision in patients with a large pannus, surgeons will occasionally perform a cesarean delivery using a supraumbilical vertical midline incision. This surgical technique can increase the risk of postoperative respiratory compromise as a result of pain and diaphragmatic splinting. The use of the double catheter technique has been described for super obese parturients who underwent cesarean delivery using a high vertical midline incision. The first report using this double catheter technique described a parturient with a BMI of 76 kg/m2 who had a lumbar CSE placed for intraoperative anesthesia and a low thoracic epidural catheter used for postoperative pain management.45 A 2015 case series reported the use of a low thoracic epidural catheter combined with a continuous lumbar spinal catheter in three super obese parturients (BMI 73–95 kg/m2) who had high vertical midline incisions for cesarean delivery.43 The lumbar spinal catheter was used to titrate the surgical block, and the thoracic epidural catheter was used for postoperative analgesia.43 Of note, in one of the three parturients, adequate surgical anesthesia at the upper end of the incision could only be obtained after bolusing the thoracic epidural catheter. This suggests that the thoracic epidural catheter could be useful for intraoperative anesthesia as well as postoperative analgesia with this technique.
General Anesthesia For Cesarean Delivery
While neuraxial anesthesia is preferable for a cesarean delivery, general anesthesia is sometimes necessary when maternal or fetal condition iscritical, or technical difficulty or patient refusal prohibit placement of a neuraxial anesthetic.
Following induction of general anesthesia, rapid desaturation occurs during the apneic period secondary to decreased functional residual capacity combined with increased oxygen consumption in obese parturients. Adequate preoxygenation is critical for these patients. This can be achieved using 3 mins of tidal volume breathing or 8 deep breaths in 1 min with 100% oxygen.46 The anesthesia provider should also use a nasal cannula insufflating oxygen at 5L/min during intubation to prolong the time to desaturation.46
Proper positioning in order to optimize the laryngoscopic view can assist in securing the often-difficult airway in the parturient. The ramped position improves the laryngoscopic view compared to the traditional sniffing position,47 and can be achieved by elevating the head of an obese patient above the shoulders by reconfiguring the operating room table or by using blankets under the upper body.48 Despite the most careful positioning, some patients remain difficult to intubate or mask ventilate. Multiple providers should therefore be available since it may require two hands to perform an adequate jaw thrust maneuver, and the additional personnel can assist with positive-pressure ventilation and cricoid pressure. A video-laryngoscope should be immediately available, with strong consideration to using it for all intubations in obese parturients49 Practitioners should be prepared to deal with a challenging airway in this patient population as guided by the guidelines developed by the Obstetric Anesthetists Association and the Difficult Airway Society for the management of difficult and failed intubations in the obstetric patient.49
The dosing of induction agents in the obese parturient should be based on ideal body weight rather than actual body weight.50 Thoughtful attention regarding the dose of neuromuscular blocking agents is also warranted. A 2006 study sought to identify the dose of succinylcholine that provided ideal intubating conditions in non-pregnant obese patients comparing doses of 1 mg/kg of ideal body weight, lean body weight, and total body weight, concluding that the third regimen was superior for providing optimum intubating conditions.51 Alternatively, rocuronium may be given to provide adequate intubating conditions at a dose of 1–1.2 mg/kg of ideal body weight.52 Sugammadex, a cyclodextrin, can be used to reverse neuromuscular blockade induced with rocuronium or vecuronium in as little as two minutes.53 However, at this time it has not been studied in parturients though its use has been documented with no ill effect to mother or fetus54 but the ideal dose has yet to be established.
Emergence and tracheal extubation after general anesthesia represent a period where undivided attention of the anesthesiologist is required. Deaths have been reported during this vulnerable time in the obese parturient attributed to hypoventilation and airway obstruction.16 It is critical to ensure the patient is fully awake with complete neuromuscular blockade reversal prior to tracheal extubation. Keeping the head of the bed elevated during this period will serve to optimize respiratory dynamics.
Technical Considerations
General Operating Room Protocols
Operating room tables come with maximum weight standards. The team should be aware of the weight limits of these tables and gurneys and have plans in place to obtain equipment which will appropriately hold the patient. In addition, some surgical tables have weight variance based on orientation. The placement of table extenders can assist in supporting some redundant tissue and may be necessary to prevent skin or tissue injury of the parturient.
When a laboring parturient presents to the operating room for cesarean delivery, assistance is often required to move the patient from the laboring bed to the operating table. The difficulty in safely getting an obese parturient to the operating room table may be compounded in the presence of a neuraxial blockade. This movement represents a potential risk of injury to both the patient and the staff assisting in this transition. Some of this risk can be mitigated with the use of an air-inflated mat to assist in moving the patient as well as adequate help available to move the patient carefully.
Finally, when actually performing the cesarean delivery, the abdominal girth and panniculus in the obese may result in suboptimal surgical visualization. Therefore, retraction of the panniculus is often performed to optimize surgical exposure. This could be achieved using cephalad retraction and fixation of the panniculus using tape or commercially available retraction tape devices. It is crucial to carefully monitor the hemodynamics and respiratory status following cephalad retraction, as there has been a report of fetal death associated with protracted hypotension following powerful cephalad retraction of a panniculus for cesarean delivery.55 Alternately, angled suspension has been described to minimize the hemodynamic and respiratory consequences of cephalad retraction.56
Monitoring Equipment
The American Heart Association recommends that the width of the noninvasive blood pressure cuff’s compression bladder should not exceed 40% of the upper arm circumference or 80% of its length.57 In obese women, the upper arm is often conical in shape, which prevents noninvasive blood pressure cuffs from fitting correctly. When ill-fitting blood pressure cuffs are used, the result is not only patient discomfort and possible skin or soft tissue damage, but also inaccurate readings.58 Therefore, blood pressure cuffs are sometimes placed on the forearm to avoid this issue. Forearm measurement correlates well with upper arm measurement, but exceeds it by 10 mmHg on average.59 This led to the development and validation of a conically-shaped noninvasive blood pressure cuff for obese patients designed for the forearm, and it has been reported to correlate with arterial blood pressure measurements.60
In some morbidly obese parturients at high risk for cardiac or bleeding complications, practitioners may prefer to monitor blood pressure continuously. Multiple devices are available that provide continuous, non-invasive blood pressure readings, though their accuracy in patients with obesity is not well characterized.58 Invasive arterial blood pressure monitoring remains an attractive alternative when non-invasive devices are not available or not reliable. A recent study examined the use of invasive arterial blood pressure monitoring compared to a noninvasive method of continuous blood pressure monitoring and found that the accuracy and precision of the noninvasive methods was good for mean and diastolic pressure measurements, but only moderate for systolic blood pressure.61
Spinal And Epidural Needles
In the majority of obese parturients, neuraxial anesthesia can be successfully performed using epidural and spinal needles of standard length.44 However, the distance from the skin to the epidural space is increased with a greater BMI due to a thicker subcutaneous layer.62 Because of this, longer epidural and spinal needles may be required and should be available. However, control of those long needles might be more difficult than standard length needles. Therefore, even in the morbidly obese parturient, our practice is to begin neuraxial placement using the standard-length needles and only transition to the longer set when assured the length of the needle is the only barrier to reaching the epidural space. Another approach is to use neuraxial ultrasound to estimate the distance from the skin to the epidural space to inform needle choice. Estimates of the epidural space depth with neuraxial ultrasound are often useful, but should consider the compression of the subcutaneous skin layer with the ultrasound probe to obtain clear images at greater depths.
Identification Of Neuraxis: Conventional Methods And Ultrasonography
Positioning obese parturients for neuraxial anesthetic placement can be challenging due to body habitus, but good positioning is crucial to optimize block placement. Adequate personnel and positioning devices, where available, are important to optimally and safely position the obese parturient while the neuraxial anesthetic is being placed.
While neuraxial blocks can be placed in both the sitting and lateral positions, in the obese parturient the sitting position confers the highest probability of success. The sitting flexed position minimizes the distance between the skin and epidural space relative to the lateral position.62 Additionally, obese parturients may have lateral fat pads that could obscure the midline in the lateral position. Also, the 7th cervical vertebra prominence and the gluteal cleft landmarks are easier to identify in the seated position. Despite this, subcutaneous adipose tissue can make palpation of traditional landmarks difficult.63 One study in non-pregnant patients reported difficulty identifying spinous processes by palpation in 68% of obese nonpregnant patients versus only 5% of those with a normal BMI.64
Neuraxial ultrasound can be used to facilitate identification of the midline, as well as to estimate the depth from the skin to the epidural space. Balki et al65 reported a strong correlation between the predicted depth of the epidural space with ultrasound and the physical distance as measured by the Tuohy needle during epidural placement. However, they noted a tendency of the ultrasound to underestimate epidural space depth as that distance increased, possibly due to the compression of subcutaneous tissue by the ultrasound probe. Ultrasound use was shown to reduce the number of attempts required to successfully place a spinal anesthetic for obese parturients undergoing cesarean delivery.66 The use of ultrasound was also shown to reduce epidural needle redirections, as well as the risk of a failed epidural catheter.65 However, as BMI increases, optimal image quality may be difficult to obtain due to increased thickness of the subcutaneous tissue.67 While most studies utilize ultrasound to facilitate a midline neuraxial approach,65–67 a separate study found that a pre-procedure ultrasound may produce better image acquisition in the paramedian sagittal oblique plane when compared to the midline plane in obese parturient.68
Dosing Of Neuraxial Local Anesthetics
Compared to their nonpregnant counterparts, pregnant women require a reduced dose of intrathecal bupivacaine.69 This dose reduction is due to increased spread of intrathecal local anesthetics as a result of epidural venous engorgement and alteration in neural tissue permeability to local anesthetics secondary to the hormonal changes in pregnancy.70 Obese parturients may require even lower local anesthetic doses, although this is the subject of ongoing debate. Studies using magnetic resonance imaging have demonstrated a reduced lumbar CSF volume in the obese as well as an inverse correlation between lumbar CSF volume and the cephalad extension of the block.71,72 The gravid uterus and abdominal panniculus are thought to cause caval compression resulting in epidural vein engorgement and increased abdominal pressures displacing soft tissues through the intervertebral foramina ultimately decreasing CSF volume and possibly increasing CSF pressure. The theoretical concerns that local anesthetics administered in the CSF may spread higher in obese women and increase the risk of high spinal block in an already high risk patient have led some to advocate for a reduction in spinal local anesthetic doses in the obese parturient.44,73,74 However, a number of studies do not support these concerns. In fact, two separate studies found no correlation between height, weight, or BMI and the height of block achieved with spinal anesthesia when using a standard 12 mg dose of bupivacaine for cesarean delivery (BMI 22–36 kg/m2).75,76 In these studies, morbidly obese patients were not specifically evaluated. Lee et al69 conducted a study examining the dose requirements for bupivacaine in obese and non-obese parturients and reported the 95% effective dose (ED95) to be similar between the two groups. Two studies from the same institution and utilizing the same methodology also reported no difference in the ED50 and ED95 of hyperbaric spinal bupivacaine for cesarean delivery in morbidly obese parturients when compared to their non-obese counterparts.77,78 It should be noted however, that data on the actual cephalad spread of a spinal block in the super obese are limited. A retrospective study by Lamon et al suggested no risk of increased cephalad spread of a standard spinal local anesthetic dose (10.5–12 mg) until the BMI exceeded 50 kg/m2.79 Those findings were confirmed in a prospective study reporting higher block level by an average of two dermatomes, and longer time to block regression, in obese parturients with a mean BMI of 51 kg/m2 compared to non-obese parturients following spinal anesthesia with 10 mg hyperbaric bupivacaine.80 There were however no significant differences in vasopressor requirements, hand grip strength or peak expiratory flow rate between the groups in this small study with 25 patients per group.
Data regarding dosing of epidural catheters for labor analgesia in the obese parturients are limited. An up-down sequential allocation study reported that obese parturients required a significantly lower dose of bupivacaine for initiating labor epidural analgesia compared to non-obese parturients.81 On the other hand, other studies suggested minimal or no impact of body weight on the dosing requirements for labor analgesia,82,83 although obese patients were not specifically studied.
Postpartum Care
Postpartum Complications
Obese parturients are at increased risk for a number of postpartum complications: wound complications; urinary tract infection; peripheral nerve injury; venous thromboembolism; respiratory complications; sepsis, myocardial infarction, and death.44
Respiratory depression is of particular concern in patients with morbid obesity. Mhyre et al16 reported obesity as a risk factor for the development of hypoventilation and airway obstruction during anesthesia emergence and in the post-operative phase of care. As discussed previously, obesity is a well-known risk factor for OSA. Providers should consider the post-operative monitoring needs of a patient with OSA and triage patients to the appropriate setting.84
Analgesia
Because early mobilization reduces the risk of deep venous thrombosis, pulmonary embolism, and respiratory complications, adequate post-delivery analgesia in the obese parturient is crucial. After cesarean delivery, neuraxial morphine has a superior analgesic profile when compared to either oral or intravenous opioids although it does carry an increased risk of pruritus and nausea.85,86 Concern remains as to whether obese parturients may be at increased risk of opioid-induced respiratory depression from neuraxial morphine, but limited data exist. A large systematic review conducted in 2018 found the lowest and highest range of clinically significant respiratory depression to be 1.08 (95% CI, 0.24–7.22) and 1.63 (95% CI, 0.62–8.77)per 10,000, respectively.87 A retrospective review of over 5,000 parturients who received neuraxial morphine for post-cesarean analgesia did not show any need for naloxone administration or the involvement of the rapid response team for the management of respiratory depression. The study population included 2,283 obese (class I and II) and 886 morbidly obese (class III) parturients.88 Based on these findings and in accordance with guidelines from the ASA, the Society of Obstetric Anesthesia and Perinatology, and the American Society of Regional Anesthesia and Pain Medicine, we continue to use neuraxial morphine in this patient population in conjunction with appropriate monitoring.89,90 When neuraxial morphine is contraindicated due to allergy or intolerance, intravenous patient-controlled analgesia may be used cautiously in a setting where patients can be monitored closely for sedation and respiratory depression. The concurrent use of other sedating medications should be avoided.
Unless contraindicated, a multimodal analgesic regimen that includes scheduled dosing of acetaminophen and nonsteroidal anti-inflammatory drugs can optimize post-delivery analgesia and decrease opioid consumption. Additionally, there may be a role for local anesthetic techniques. The transversus abdominis plane (TAP) block has been shown to reduce pain scores and analgesic consumption in patients who have not received neuraxial morphine.91 It does not however confer additional, if any, benefit to those who have received neuraxial morphine. It is important to note however that all previous studies used standard preparations of local anesthetics, and it is unclear if liposomal preparations might confer additional benefit in patients receiving neuraxial morphine. Furthermore, while this technique can benefit select patients with post-cesarean pain, it provides coverage for incisional rather than visceral pain and is therefore inferior when compared to neuraxial morphine. Other truncal blocks such as quadratus lumborum and erector spinae plane blocks might provide some visceral analgesia but data are limited in general with no studies specifically conducted in obese parturients. The placement of those truncal blocks might be technically challenging in the morbidly obese parturient who could benefit from their use.92 The TAP block has also been useful as a rescue for parturients reporting continued incisional pain despite multimodal analgesic therapy.93 Another option is single infiltration or continuous infusion of local anesthetics in the wound, which could be an attractive option in the obese parturient in whom a truncal block might be challenging. A recent meta-analysis suggested a benefit of those techniques in reducing postoperative analgesic consumption following cesarean delivery, though this was not specifically examined in the morbidly obese parturient.94
Thromboprophylaxis
Venous thromboembolism (VTE) is one of the leading causes of maternal mortality.95,96 In the 2000s in the United Kingdom, maternal mortality from VTE declined following better recognition of patient risk factors and increased use of thromboprophylaxis.13 Obesity is a known risk factor for the development of VTE in both the antepartum and postpartum periods. A 2018 study by Butwick et al demonstrated increased odds of VTE as a function of increasing BMI. They reported an antepartum adjusted odds ratio (95% CI) for VTE of 2.9 (2.2–3.8) and a postpartum odds ratio (95% CI) of 3.6 (2.9–4.6) in obese parturients relative to their non-obese counterparts.97 Despite the increased risk of VTE in the obese parturient, significant variability exists in the recommendations for prophylaxis between the RCOG, ACOG, and American College of Chest Physicians as seen in Table 3. Recommendations were also published by the National Partnership for Maternal Safety in 2016, supporting routine VTE risk assessment, with appropriate use of pharmacologic and mechanical thromboprophylaxis.98
 |
Table 3 Thromboprophylaxis Guidelines For The Parturient |
Due to the variability in recommendations from multiple bodies, most institutions have developed their own protocols to provide VTE prophylaxis in this vulnerable patient population.
Summary
- The prevalence of obesity and its associated complications continue to increase
- Antepartum anesthetic consultation should be performed to assess comorbidities, counsel patients, plan for care, and optimize patients
- Neuraxial anesthesia is recommended for all obese parturients, and in the event of cesarean delivery, a continuous technique is preferred
- Postoperative analgesia and thromboprophylaxis are important considerations for the post-delivery period
Disclosure
Ashraf S Habib reports grants from Pacira Biosciences, BioQ Pharma, and Avanos Inc, outside the submitted work. The authors report no other conflicts of interest in this work.
References
1. Ogden CL, Carroll MD, Kit BK, Flegal KM. Prevalence of obesity among adults: United States, 2011–2012. NCHS Data Brief. 2013;131:1–8.
2. Obesity: Preventing and Managing the Global Epidemic. Report of a WHO Consultation. Vol. 894. World Health Organization Tech Rep Ser; 2000:
3. Sinha A, Jayaraman L, Punhani D, Chowbey P. Enhanced recovery after bariatric surgery in the severely obese, morbidly obese, super-morbidly obese and super-super morbidly obese using evidence-based clinical pathways: a comparative study. Obes Surg. 2017;27(3):560–568. doi:10.1007/s11695-016-2366-y
4. Dennis AT, Lamb KE, Story D, et al. Associations between maternal size and health outcomes for women undergoing caesarean section: a multicentre prospective observational study (The MUM SIZE Study). BMJ Open. 2017;7(6):e015630. doi:10.1136/bmjopen-2016-015630
5. Kominiarek MA, Chauhan SP. Obesity before, during, and after pregnancy: a review and comparison of five national guidelines. Am J Perinatol. 2016;33(5):433–441. doi:10.1055/s-0035-1567856
6. Tabet M, Flick LH, Tuuli MG, Macones GA, Chang JJ. Prepregnancy body mass index in a first uncomplicated pregnancy and outcomes of a second pregnancy. Am J Obstet Gynecol. 2015;213(4):
7. Dempsey JC, Ashiny Z, Qiu CF, Miller RS, Sorensen TK, Williams MA. Maternal pre-pregnancy overweight status and obesity as risk factors for cesarean delivery. J Matern Fetal Neonatal Med. 2005;17(3):179–185. doi:10.1080/14767050500073456
8. Dietz PM, Callaghan WM, Morrow B, Cogswell ME. Population-based assessment of the risk of primary cesarean delivery due to excess prepregnancy weight among nulliparous women delivering term infants. Maternal and Child Health Journal. 2005;9(3):237–244. doi:10.1007/s10995-005-0003-9
9. Hood DD, Dewan DM. Anesthetic and obstetric outcome in morbidly obese parturients. Anesthesiology. 1993;79(6):1210–1218. doi:10.1097/00000542-199312000-00011
10. Tonidandel A, Booth J, D’Angelo R, Harris L, Tonidandel S. Anesthetic and obstetric outcomes in morbidly obese parturients: a 20-year follow-up retrospective cohort study. Int J Obstet Anesth. 2014;23(4):357–364. doi:10.1016/j.ijoa.2014.05.004
11. Schellpfeffer MA, Gillespie KH, Rohan AM, Blackwell SP. A review of pregnancy-related maternal mortality in Wisconsin, 2006–2010. WMJ. 2015;114(5):202–207.
12. Main EK, McCain CL, Morton CH, Holtby S, Lawton ES. Pregnancy-related mortality in California: causes, characteristics, and improvement opportunities. Obstet Gynecol. 2015;125(4):938–947. doi:10.1097/AOG.0000000000000746
13. Cantwell R, Clutton-Brock T, Cooper G, et al. Saving Mothers’ Lives: reviewing maternal deaths to make motherhood safer: 2006–2008. The eighth report of the confidential enquiries into maternal deaths in the United Kingdom. BJOG. 2011;118 Suppl 1:1–203. doi:10.1111/j.1471-0528.2010.02847.x
14. Lewis G. Saving Mothers’ Lives: the continuing benefits for maternal health from the United Kingdom (UK) Confidential Enquires into Maternal Deaths. Semin Perinatol. 2012;36(1):19–26. doi:10.1053/j.semperi.2011.09.005
15. Lewis G. The Confidential Enquiry into Maternal and Child Health (CEMACH). Saving mother’s lives: reviewing maternal deaths to make motherhood safer — 2003–2005. The seventh report on confidential enquiries into maternal deaths in the United Kingdom. CEMACH. 2007.
16. Mhyre JM, Riesner MN, Polley LS, Naughton NN. A series of anesthesia-related maternal deaths in Michigan, 1985–2003. Anesthesiology. 2007;106(6):1096–1104. doi:10.1097/01.anes.0000267592.34626.6b
17. Denison FC, Aedla NR, Keag O, et al. Care of women with obesity in pregnancy: green-top guideline No. 72. BJOG. 2019;126(3):e62–e106. doi:10.1111/1471-0528.15386
18. ACOG practice bulletin No 156: obesity in pregnancy. Obstet Gynecol. 2015;126(6):e112–e126. doi:10.1097/AOG.0000000000001211
19. Hawkins JL, Chang J, Palmer SK, Gibbs CP, Callaghan WM. Anesthesia-related maternal mortality in the United States: 1979–2002. Obstet Gynecol. 2011;117(1):69–74. doi:10.1097/AOG.0b013e31820093a9
20. Dominguez JE, Grotegut CA, Cooter M, Krystal AD, Habib AS. Screening extremely obese pregnant women for obstructive sleep apnea. Am J Obstet Gynecol. 2018;219(6):
21. Facco FL, Parker CB, Reddy UM, et al. Association between sleep-disordered breathing and hypertensive disorders of pregnancy and gestational diabetes mellitus. Obstet Gynecol. 2017;129(1):31–41. doi:10.1097/AOG.0000000000001805
22. Bourjeily G, Danilack VA, Bublitz MH, et al. Obstructive sleep apnea in pregnancy is associated with adverse maternal outcomes: a national cohort. Sleep Med. 2017;38:50–57. doi:10.1016/j.sleep.2017.06.035
23. Facco FL, Ouyang DW, Zee PC, Grobman WA. Development of a pregnancy-specific screening tool for sleep apnea. J Clin Sleep Med. 2012;8(4):389–394. doi:10.5664/jcsm.2030
24. Louis JM, Koch MA, Reddy UM, et al. Predictors of sleep-disordered breathing in pregnancy. Am J Obstet Gynecol. 2018;218(5):521 e521–521 e512. doi:10.1016/j.ajog.2018.01.031
25. Bauer ME, Kountanis JA, Tsen LC, Greenfield ML, Mhyre JM. Risk factors for failed conversion of labor epidural analgesia to cesarean delivery anesthesia: a systematic review and meta-analysis of observational trials. Int J Obstet Anesth. 2012;21(4):294–309. doi:10.1016/j.ijoa.2012.05.007
26. Miro M, Guasch E, Gilsanz F. Comparison of epidural analgesia with combined spinal-epidural analgesia for labor: a retrospective study of 6497 cases. Int J Obstet Anesth. 2008;17(1):15–19. doi:10.1016/j.ijoa.2007.07.003
27. Pan PH, Bogard TD, Owen MD. Incidence and characteristics of failures in obstetric neuraxial analgesia and anesthesia: a retrospective analysis of 19,259 deliveries. Int J Obstet Anesth. 2004;13(4):227–233. doi:10.1016/j.ijoa.2004.04.008
28. Booth JM, Pan JC, Ross VH, Russell GB, Harris LC, Pan PH. Combined spinal epidural technique for labor analgesia does not delay recognition of epidural catheter failures: a single-center retrospective cohort survival analysis. Anesthesiology. 2016;125(3):516–524. doi:10.1097/ALN.0000000000001222
29. Chau A, Bibbo C, Huang CC, et al. Dural puncture epidural technique improves labor analgesia quality with fewer side effects compared with epidural and combined spinal epidural techniques: a randomized clinical trial. Anesth Analg. 2017;124(2):560–569. doi:10.1213/ANE.0000000000001798
30. Faure E, Moreno R, Thisted R. Incidence of postdural puncture headache in morbidly obese parturients. Reg Anesth. 1994;19(5):361–363.
31. Vallejo MC. Anesthetic management of the morbidly obese parturient. Curr Opin Anaesthesiol. 2007;20(3):175–180. doi:10.1097/ACO.0b013e328014646b
32. Miu M, Paech MJ, Nathan E. The relationship between body mass index and post-dural puncture headache in obstetric patients. Int J Obstet Anesth. 2014;23(4):371–375. doi:10.1016/j.ijoa.2014.06.005
33. Peralta F, Higgins N, Lange E, Wong CA, McCarthy RJ. The relationship of body mass index with the incidence of postdural puncture headache in parturients. Anesth Analg. 2015;121(2):451–456. doi:10.1213/ANE.0000000000000802
34. Franz AM, Jia SY, Bahnson HT, Goel A, Habib AS. The effect of second-stage pushing and body mass index on postdural puncture headache. J Clin Anesth. 2017;37:77–81. doi:10.1016/j.jclinane.2016.10.037
35. Alonso E, Gilsanz F, Gredilla E, Martinez B, Canser E, Alsina E. Observational study of continuous spinal anesthesia with the catheter-over-needle technique for cesarean delivery. Int J Obstet Anesth. 2009;18(2):137–141. doi:10.1016/j.ijoa.2008.11.001
36. Dresner M, Pinder A. Anaesthesia for caesarean section in women with complex cardiac disease: 34 cases using the Braun Spinocath spinal catheter. Int J Obstet Anesth. 2009;18(2):131–136. doi:10.1016/j.ijoa.2008.09.009
37. McKenzie CP, Carvalho B, Riley ET. The wiley spinal catheter-over-needle system for continuous spinal anesthesia: a case series of 5 cesarean deliveries complicated by paresthesias and headaches. Reg Anesth Pain Med. 2016;41(3):405–410. doi:10.1097/AAP.0000000000000367
38. Tao W, Grant EN, Craig MG, McIntire DD, Leveno KJ. Continuous spinal analgesia for labor and delivery: an observational study with a 23-gauge spinal catheter. Anesth Analg. 2015;121(5):1290–1294. doi:10.1213/ANE.0000000000000903
39. Riley ET, Cohen SE, Macario A, Desai JB, Ratner EF. Spinal versus epidural anesthesia for cesarean section: a comparison of time efficiency, costs, charges, and complications. Anesth Analg. 1995;80(4):709–712. doi:10.1097/00000539-199504000-00010
40. Butwick A, Carvalho B, Danial C, Riley E. Retrospective analysis of anesthetic interventions for obese patients undergoing elective cesarean delivery. J Clin Anesth. 2010;22(7):519–526. doi:10.1016/j.jclinane.2010.01.005
41. Ross VH, Dean LS, Thomas JA, Harris LC, Pan PH. A randomized controlled comparison between combined spinal-epidural and single-shot spinal techniques in morbidly obese parturients undergoing cesarean delivery: time for initiation of anesthesia. Anesth Analg. 2014;118(1):168–172. doi:10.1213/ANE.0000000000000022
42. McNaught AF, Stocks GM. Epidural volume extension and low-dose sequential combined spinal-epidural blockade: two ways to reduce spinal dose requirement for caesarean section. Int J Obstet Anesth. 2007;16(4):346–353. doi:10.1016/j.ijoa.2007.03.013
43. Polin CM, Hale B, Mauritz AA, et al. Anesthetic management of super-morbidly obese parturients for cesarean delivery with a double neuraxial catheter technique: a case series. Int J Obstet Anesth. 2015;24(3):276–280. doi:10.1016/j.ijoa.2015.04.001
44. Soens MA, Birnbach DJ, Ranasinghe JS, van Zundert A. Obstetric anesthesia for the obese and morbidly obese patient: an ounce of prevention is worth more than a pound of treatment. Acta Anaesthesiol Scand. 2008;52(1):6–19. doi:10.1111/j.1399-6576.2007.01483.x
45. McDonnell NJ, Paech MJ. The management of a super morbidly obese parturient delivering twins by caesarean section. Anaesth Intensive Care. 2007;35(6):979–983. doi:10.1177/0310057X0703500622
46. Chiron B, Laffon M, Ferrandiere M, Pittet JF, Marret H, Mercier C. Standard preoxygenation technique versus two rapid techniques in pregnant patients. Int J Obstet Anesth. 2004;13(1):11–14. doi:10.1016/S0959-289X(03)00095-5
47. Collins JS, Lemmens HJ, Brodsky JB, Brock-Utne JG, Levitan RM. Laryngoscopy and morbid obesity: a comparison of the “sniff” and “ramped” positions. Obes Surg. 2004;14(9):1171–1175. doi:10.1381/0960892042386869
48. Rao SL, Kunselman AR, Schuler HG, DesHarnais S. Laryngoscopy and tracheal intubation in the head-elevated position in obese patients: a randomized, controlled, equivalence trial. Anesth Analg. 2008;107(6):1912–1918. doi:10.1213/ane.0b013e31818556ed
49. Mushambi MC, Kinsella SM, Popat M, et al. Obstetric Anaesthetists’ Association and Difficult Airway Society guidelines for the management of difficult and failed tracheal intubation in obstetrics. Anaesthesia. 2015;70(11):1286–1306. doi:10.1111/anae.13260
50. Ingrande J, Lemmens HJ. Dose adjustment of anaesthetics in the morbidly obese. Br J Anaesth. 2010;105(Suppl 1):i16–i23. doi:10.1093/bja/aeq312
51. Lemmens HJ, Brodsky JB. The dose of succinylcholine in morbid obesity. Anesth Analg. 2006;102(2):438–442. doi:10.1213/01.ane.0000194876.00551.0e
52. Meyhoff CS, Lund J, Jenstrup MT, et al. Should dosing of rocuronium in obese patients be based on ideal or corrected body weight? Anesth Analg. 2009;109(3):787–792. doi:10.1213/ane.0b013e3181b0826a
53. Puhringer FK, Rex C, Sielenkamper AW, et al. Reversal of profound, high-dose rocuronium-induced neuromuscular blockade by sugammadex at two different time points: an international, multicenter, randomized, dose-finding, safety assessor-blinded, phase II trial. Anesthesiology. 2008;109(2):188–197. doi:10.1097/ALN.0b013e31817f5bc7
54. Stourac P, Adamus M, Seidlova D, et al. Low-dose or high-dose rocuronium reversed with neostigmine or sugammadex for cesarean delivery anesthesia: a randomized controlled noninferiority trial of time to tracheal intubation and extubation. Anesth Analg. 2016;122(5):1536–1545. doi:10.1213/ANE.0000000000001197
55. Hodgkinson R, Husain FJ. Obesity and the cephalad spread of analgesia following epidural administration of bupivacaine for Cesarean section. Anesth Analg. 1980;59(2):89–92. doi:10.1213/00000539-198002000-00002
56. Whitty RJ, Maxwell CV, Carvalho JC. Complications of neuraxial anesthesia in an extreme morbidly obese patient for Cesarean section. Int J Obstet Anesth. 2007;16(2):139–144. doi:10.1016/j.ijoa.2006.08.011
57. Pickering TG, Hall JE, Appel LJ, et al. Recommendations for blood pressure measurement in humans and experimental animals: part 1: blood pressure measurement in humans: a statement for professionals from the Subcommittee of Professional and Public Education of the American Heart Association Council on High Blood Pressure Research. Circulation. 2005;111(5):697–716. doi:10.1161/01.CIR.0000154900.76284.F6
58. Eley VA, Christensen R, Guy L, Dodd B. Perioperative blood pressure monitoring in patients with obesity. Anesth Analg. 2019;128(3):484–491. doi:10.1213/ANE.0000000000003647
59. Pierin AM, Alavarce DC, Gusmao JL, Halpern A, Mion D
60. Hersh LT, Sesing JC, Luczyk WJ, Friedman BA, Zhou S, Batchelder PB. Validation of a conical cuff on the forearm for estimating radial artery blood pressure. Blood Press Monit. 2014;19(1):38–45. doi:10.1097/MBP.0000000000000011
61. Rogge DE, Nicklas JY, Schon G, et al. Continuous noninvasive arterial pressure monitoring in obese patients during bariatric surgery: an evaluation of the vascular unloading technique (Clearsight system). Anesth Analg. 2019;128(3):477–483. doi:10.1213/ANE.0000000000003943
62. D’Alonzo RC, White WD, Schultz JR, Jaklitsch PM, Habib AS. Ethnicity and the distance to the epidural space in parturients. Reg Anesth Pain Med. 2008;33(1):24–29. doi:10.1097/00115550-200801000-00005
63. Ellinas EH, Eastwood DC, Patel SN, Maitra-D’Cruze AM, Ebert TJ. The effect of obesity on neuraxial technique difficulty in pregnant patients: a prospective, observational study. Anesth Analg. 2009;109(4):1225–1231. doi:10.1213/ANE.0b013e3181b5a1d2
64. Stiffler KA, Jwayyed S, Wilber ST, Robinson A. The use of ultrasound to identify pertinent landmarks for lumbar puncture. Am J Emerg Med. 2007;25(3):331–334. doi:10.1016/j.ajem.2006.07.010
65. Balki M, Lee Y, Halpern S, Carvalho JC. Ultrasound imaging of the lumbar spine in the transverse plane: the correlation between estimated and actual depth to the epidural space in obese parturients. Anesth Analg. 2009;108(6):1876–1881. doi:10.1213/ane.0b013e3181a323f6
66. Sahin T, Balaban O, Sahin L, Solak M, Toker K. A randomized controlled trial of preinsertion ultrasound guidance for spinal anaesthesia in pregnancy: outcomes among obese and lean parturients: ultrasound for spinal anesthesia in pregnancy. J Anesth. 2014;28(3):413–419. doi:10.1007/s00540-013-1726-1
67. Carvalho JC. Ultrasound-facilitated epidurals and spinals in obstetrics. Anesthesiol Clin. 2008;26(1):
68. Sahota JS, Carvalho JC, Balki M, Fanning N, Arzola C. Ultrasound estimates for midline epidural punctures in the obese parturient: paramedian sagittal oblique is comparable to transverse median plane. Anesth Analg. 2013;116(4):829–835. doi:10.1213/ANE.0b013e31827f55f0
69. Lee Y, Balki M, Parkes R, Carvalho JC. Dose requirement of intrathecal bupivacaine for cesarean delivery is similar in obese and normal weight women. Rev Bras Anestesiol. 2009;59(6):674–683.
70. Saravanakumar K, Rao SG, Cooper GM. Obesity and obstetric anaesthesia. Anaesthesia. 2006;61(1):36–48. doi:10.1111/ana.2006.61.issue-1
71. Carpenter RL, Hogan QH, Liu SS, Crane B, Moore J. Lumbosacral cerebrospinal fluid volume is the primary determinant of sensory block extent and duration during spinal anesthesia. Anesthesiology. 1998;89(1):24–29. doi:10.1097/00000542-199807000-00007
72. Hogan QH, Prost R, Kulier A, Taylor ML, Liu S, Mark L. Magnetic resonance imaging of cerebrospinal fluid volume and the influence of body habitus and abdominal pressure. Anesthesiology. 1996;84(6):1341–1349. doi:10.1097/00000542-199606000-00010
73. Roofthooft E. Anesthesia for the morbidly obese parturient. Curr Opin Anaesthesiol. 2009;22(3):341–346. doi:10.1097/ACO.0b013e328329a5b8
74. Brodsky JB, Lemmens HJ. Regional anesthesia and obesity. Obes Surg. 2007;17(9):1146–1149. doi:10.1007/s11695-007-9207-y
75. Hartwell BL, Aglio LS, Hauch MA, Datta S. Vertebral column length and spread of hyperbaric subarachnoid bupivacaine in the term parturient. Reg Anesth. 1991;16(1):17–19.
76. Norris MC. Height, weight, and the spread of subarachnoid hyperbaric bupivacaine in the term parturient. Anesth Analg. 1988;67(6):555–558. doi:10.1213/00000539-198806000-00010
77. Carvalho B, Collins J, Drover DR, Atkinson RL, Riley ET. ED(50) and ED(95) of intrathecal bupivacaine in morbidly obese patients undergoing cesarean delivery. Anesthesiology. 2011;114(3):529–535. doi:10.1097/ALN.0b013e318209a92d
78. Ginosar Y, Mirikatani E, Drover DR, Cohen SE, Riley ET. ED50 and ED95 of intrathecal hyperbaric bupivacaine coadministered with opioids for cesarean delivery. Anesthesiology. 2004;100(3):676–682. doi:10.1097/00000542-200403000-00031
79. Lamon AM, Einhorn LM, Cooter M, Habib AS. The impact of body mass index on the risk of high spinal block in parturients undergoing cesarean delivery: a retrospective cohort study. J Anesth. 2017;31(4):552–558. doi:10.1007/s00540-017-2352-0
80. Ngaka TC, Coetzee JF, Dyer RA. The influence of body mass index on sensorimotor block and vasopressor requirement during spinal anesthesia for elective cesarean delivery. Anesth Analg. 2016;123(6):1527–1534. doi:10.1213/ANE.0000000000001568
81. Panni MK, Columb MO. Obese parturients have lower epidural local anaesthetic requirements for analgesia in labour. Br J Anaesth. 2006;96(1):106–110. doi:10.1093/bja/aei284
82. Duggan J, Bowler GM, McClure JH, Wildsmith JA. Extradural block with bupivacaine: influence of dose, volume, concentration and patient characteristics. Br J Anaesth. 1988;61(3):324–331. doi:10.1093/bja/61.3.324
83. Milligan KR, Cramp P, Schatz L, Johnston D, Carp H. The effect of patient position and obesity on the spread of epidural analgesia. Int J Obstet Anesth. 1993;2(3):134–136. doi:10.1016/0959-289X(93)90005-3
84. Gross JB, Bachenberg KL, Benumof JL, et al. Practice guidelines for the perioperative management of patients with obstructive sleep apnea: a report by the American Society of Anesthesiologists Task Force on Perioperative Management of patients with obstructive sleep apnea. Anesthesiology. 2006;104(5):
85. Bonnet MP, Mignon A, Mazoit JX, Ozier Y, Marret E. Analgesic efficacy and adverse effects of epidural morphine compared to parenteral opioids after elective caesarean section: a systematic review. Eur J Pain. 2010;14(9):
86. McDonnell NJ, Paech MJ, Browning RM, Nathan EA. A randomised comparison of regular oral oxycodone and intrathecal morphine for post-caesarean analgesia. Int J Obstet Anesth. 2010;19(1):16–23. doi:10.1016/j.ijoa.2009.03.004
87. Sharawi N, Carvalho B, Habib AS, Blake L, Mhyre JM, Sultan P. A systematic review evaluating neuraxial morphine and diamorphine-associated respiratory depression after cesarean delivery. Anesth Analg. 2018;127(6):1385–1395. doi:10.1213/ANE.0000000000003636
88. Crowgey TR, Dominguez JE, Peterson-Layne C, Allen TK, Muir HA, Habib AS. A retrospective assessment of the incidence of respiratory depression after neuraxial morphine administration for postcesarean delivery analgesia. Anesth Analg. 2013;117(6):1368–1370. doi:10.1213/ANE.0b013e3182a9b042
89. Apfelbaum JL, Horlocker TT, Agarkar M, et al. Practice guidelines for the prevention, detection, and management of respiratory depression associated with neuraxial opioid administration: an updated report by the american society of anesthesiologists task force on neuraxial opioids and the American Society of Regional Anesthesia and Pain Medicine. Anesthesiology. 2016;124(3):535–552. doi:10.1097/ALN.0000000000000975
90. Bauchat JR, Weiniger CF, Sultan P, et al. Society for obstetric anesthesia and perinatology consensus statement: monitoring recommendations for prevention and detection of respiratory depression associated with administration of neuraxial morphine for cesarean delivery analgesia. Anesth Analg;2019:1. doi:10.1213/ANE.0000000000004195
91. Mishriky BM, George RB, Habib AS. Transversus abdominis plane block for analgesia after Cesarean delivery: a systematic review and meta-analysis. Can J Anaesth. 2012;59(8):766–778. doi:10.1007/s12630-012-9729-1
92. Toshniwal G, Soskin V. Ultrasound-guided transversus abdominis plane block in obese patients. Indian J Anaesth. 2012;56(1):104–105. doi:10.4103/0019-5049.93368
93. Mirza F, Carvalho B. Transversus abdominis plane blocks for rescue analgesia following Cesarean delivery: a case series. Can J Anaesth. 2013;60(3):299–303. doi:10.1007/s12630-012-9866-6
94. Adesope O, Ituk U, Habib AS. Local anaesthetic wound infiltration for postcaesarean section analgesia: a systematic review and meta-analysis. Eur J Anaesthesiol. 2016;33(10):731–742. doi:10.1097/EJA.0000000000000462
95. Khan KS, Wojdyla D, Say L, Gulmezoglu AM, Van Look PF. WHO analysis of causes of maternal death: a systematic review. Lancet. 2006;367(9516):1066–1074. doi:10.1016/S0140-6736(06)68397-9
96. Callaghan WM, Creanga AA, Kuklina EV. Severe maternal morbidity among delivery and postpartum hospitalizations in the United States. Obstet Gynecol. 2012;120(5):1029–1036. doi:10.1097/aog.0b013e31826d60c5
97. Butwick AJ, Bentley J, Leonard SA, et al. Prepregnancy maternal body mass index and venous thromboembolism: a population-based cohort study. BJOG. 2019;126(5):581–588. doi:10.1111/1471-0528.15567
98. D’Alton ME, Friedman AM, Smiley RM, et al. National partnership for maternal safety: consensus bundle on venous thromboembolism. Anesth Analg. 2016;123(4):942–949. doi:10.1213/ANE.0000000000001569
99. Guh DP, Zhang W, Bansback N, Amarsi Z, Birmingham CL, Anis AH. The incidence of co-morbidities related to obesity and overweight: a systematic review and meta-analysis. BMC Public Health. 2009;9:88. doi:10.1186/1471-2458-9-88
100. Bhattacharya S, Campbell DM, Liston WA, Bhattacharya S. Effect of Body Mass Index on pregnancy outcomes in nulliparous women delivering singleton babies. BMC Public Health. 2007;7:168. doi:10.1186/1471-2458-7-168
101. Larsen TB, Sorensen HT, Gislum M, Johnsen SP. Maternal smoking, obesity, and risk of venous thromboembolism during pregnancy and the puerperium: a population-based nested case-control study. Thromb Res. 2007;120(4):505–509. doi:10.1016/j.thromres.2006.12.003
102. Leung TY, Leung TN, Sahota DS, et al. Trends in maternal obesity and associated risks of adverse pregnancy outcomes in a population of Chinese women. BJOG. 2008;115(12):1529–1537. doi:10.1111/bjo.2008.115.issue-12
103. Sebire NJ, Jolly M, Harris JP, et al. Maternal obesity and pregnancy outcome: a study of 287,213 pregnancies in London. Int J Obes Relat Metab Disord. 2001;25(8):1175–1182. doi:10.1038/sj.ijo.0801670
104. Usha Kiran TS, Hemmadi S, Bethel J, Evans J. Outcome of pregnancy in a woman with an increased body mass index. BJOG. 2005;112(6):768–772. doi:10.1111/bjo.2005.112.issue-6
105. Chu SY, Kim SY, Lau J, et al. Maternal obesity and risk of stillbirth: a metaanalysis. Am J Obstet Gynecol. 2007;197(3):223–228. doi:10.1016/j.ajog.2007.03.027
106. Kristensen J, Vestergaard M, Wisborg K, Kesmodel U, Secher NJ. Pre-pregnancy weight and the risk of stillbirth and neonatal death. BJOG. 2005;112(4):403–408. doi:10.1111/bjo.2005.112.issue-4
107. Bates SM, Greer IA, Middeldorp S, Veenstra DL, Prabulos AM, Vandvik PO. VTE, thrombophilia, antithrombotic therapy, and pregnancy: antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 Suppl):e691S–e736S. doi:10.1378/chest.11-2300
108. Royal College of Obstetricians and Gynaecologists. Reducing the Risk of Venous Thromboembolism during Pregnancy and the Puerperium. Green-top Guideline No. 37a. RCOG Press, 2015.
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
