Back to Journals » Clinical Ophthalmology » Volume 11
Neovascular age-related macular degeneration without drusen in the fellow eye: clinical spectrum and therapeutic outcome
Authors Chung WH, van Dijk EHC, Mohabati D , Dijkman G, Yzer S, de Jong EK, Fauser S, Schlingemann RO, Hoyng CB, Boon CJF
Received 17 September 2016
Accepted for publication 3 October 2016
Published 21 December 2016 Volume 2017:11 Pages 63—70
DOI https://doi.org/10.2147/OPTH.S122568
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Wing H Chung,1 Elon H C van Dijk,1 Danial Mohabati,1 Greet Dijkman,1 Suzanne Yzer,2 Eiko K de Jong,3 Sascha Fauser,4 Reinier O Schlingemann,5–7 Carel B Hoyng,3 Camiel J F Boon1,5
1Department of Ophthalmology, Leiden University Medical Center, Leiden, 2Rotterdam Eye Hospital, Rotterdam, 3Department of Ophthalmology, Donders Institute for Brain, Cognition and Behaviour, Radboud University Medical Center, Nijmegen, the Netherlands; 4Department of Ophthalmology, University Hospital of Cologne, Cologne, Germany; 5Department of Ophthalmology, 6Ocular Angiogenesis Group, Departments of Ophthalmology and Cell Biology and Histology, Academic Medical Center, 7Netherlands Institute for Neuroscience, Amsterdam, the Netherlands
Purpose: To investigate the clinical characteristics and therapeutic outcome of patients with neovascular age-related macular degeneration (nAMD) in 1 eye, without drusen in the fellow eye.
Patients and methods: Medical records of 381 patients were analyzed to identify the cases. The main outcomes included Early Treatment Diabetic Retinopathy Study (ETDRS) best-corrected visual acuity (BCVA) and change in central retinal thickness (CRT). These parameters were reviewed at baseline, first follow-up visit, and after 6, 12, and 24 months.
Results: Out of 381 patients, 29 cases (8%) were included (of whom 3 had polypoidal choroidal vasculopathy [PCV]) who were treated with anti-vascular endothelial growth factor (anti-VEGF) therapy which was supplemented by photodynamic therapy (PDT) in the PCV patients. Overall, no statistically significant change in mean BCVA was observed during follow-up. BCVA improved or remained stable (defined as a gain in BCVA, a stable BCVA, or a loss of <5 ETDRS letters) in 22 patients (76%), and 7 patients (23%) had lost ≥5 ETDRS letters at final follow-up. A gain of ≥15 ETDRS letters at final follow-up was seen in 5 patients (17%). Mean CRT had decreased significantly with 99 µm (P<0.001) at 24 months after the initial visit.
Conclusion: There is a clinical spectrum of nAMD that is not associated with drusen in the fellow eye. Patients with nAMD without drusen in the fellow eye respond to anti-VEGF treatment and, in cases of PCV, to supplemental PDT. The pathophysiology of this spectrum of nAMD may be different from drusen-associated age-related macular degeneration.
Keywords: anti-vascular endothelial growth factor, unilateral age-related macular degeneration, non-neovascular age-related macular degeneration, polypoidal choroidal vasculopathy, precursor lesion, best-corrected visual acuity
Introduction
Age-related macular degeneration (AMD) is the most common cause of irreversible vision loss among elderly in the Western world. Marked vision loss can occur in late AMD, as a result of choroidal neovascularization (CNV) and/or profound chorioretinal atrophy (geographic atrophy).1
Traditionally, macular drusen are characteristic for early and intermediate stages of this disease. However, the definition of AMD has varied among authors.2–4 Although most classifications consider drusen mandatory lesions in AMD, 1 or more small hard drusen with a diameter <63 μm can be detected in 85%–99% of the overall population aged ≥50 years.5–8
In cases of unilateral neovascular AMD (nAMD), the fellow eye is at high risk of developing CNV, with cumulative incidence rates of 10%, 22%, 28%, 37%, and 42% after 1, 2, 3, 4, and 5 years, respectively.9,10 Factors that contribute to these rates are the presence, number, location, and the individual and cumulative size of drusen in the fellow eye.11 However, there is a clinical observation that some patients with nAMD in 1 eye with no drusen in the fellow eye. Little is known about the clinical characteristics and therapeutic outcome of this subgroup of patients with unilateral nAMD without drusen in the fellow eye. As drusen are often considered pivotal precursor lesions for advanced AMD such as nAMD, the question arises whether nAMD without drusen in the fellow eye is clinically distinct from drusen-associated nAMD. Therefore, this study analyzed the clinical spectrum, characteristics, and treatment outcome of a subgroup of patients with nAMD in 1 eye, without drusen in the fellow eye.
Patients and methods
Data collection
A retrospective review of charts was performed at the Department of Ophthalmology of Leiden University Medical Center, the Netherlands. This study adhered to the tenets of the Declaration of Helsinki, and approval for this study was obtained from the Medical Ethical Committee of Leiden University Medical Center. All participants gave their written informed consent.
All data were obtained from patients who were diagnosed with unilateral nAMD between January 2010 and July 2015. These cases were retrospectively analyzed to identify cases of unilateral nAMD without drusen in the macular region of the fellow eye at initial diagnosis. The grading of drusen was performed according to the International Classification for age-related maculopathy and AMD,2 and the location of the lesions was determined using the Early Treatment Diabetic Retinopathy Study (ETDRS) grid. Fellow eyes with <5 small hard drusen with a diameter <63 μm in the macular region were not considered to have AMD, but rather a consequence of normal aging.3 All images were obtained from routine eye examinations and were assessed by an experienced retina specialist (CJFB).
Patients had to meet all of the following inclusion criteria: 1) age ≥50 years; 2) AMD with signs of CNV confirmed on optical coherence tomography (OCT) (on which either subretinal or intraretinal fluid or both could be detected) and fluorescein angiography (FA) in 1 eye; 3) <5 small hard drusen in the macular region in the fellow eye as evidenced by fundus photography, FA, and OCT; 4) at least 1 follow-up visit after initial diagnosis. Exclusion criteria were any of the following: 1) multimodal imaging which was considered to be of insufficient quality; 2) a confirmed diagnosis of AMD with signs of CNV or fovea-involving atrophy in the fellow eye; 3) history of another diagnosis as a primary underlying cause/background of neovascularization, such as pseudoxanthoma elasticum, high myopia (ie, ≥6 diopters), central serous chorioretinopathy (CSC), macular telangiectasia, or multifocal choroiditis.
Image acquisition
All patients included in this study underwent color fundus photography (Topcon Corp., Tokyo, Japan), OCT with either the Cirrus OCT (Carl Zeiss Meditec, Dublin, CA, USA) or spectral-domain OCT device (Spectralis HRA + OCT; Heidelberg Engineering, Heidelberg, Germany), and FA was performed with either a Topcon fundus camera (Topcon Corp.) or the Spectralis HRA + OCT camera.
Study measurements
After patient selection, the following clinical parameters were collected at baseline: age at onset, best-corrected visual acuity (BCVA), the anatomical level of fluid accumulation on OCT (intraretinal, subretinal, or both), performed treatments (anti-vascular endothelial growth factor [anti-VEGF] treatment [bevacizumab, ranibizumab, and aflibercept] and/or photodynamic therapy [PDT]), central retinal thickness (CRT), and the type of CNV. The type of CNV was either “type 1” (sub-retinal pigment epithelium [RPE] neovascularization [occult neovascularization]), “type 2” (subretinal neovascularization [classic neovascularization]), or “type 3” (retinal angiomatous proliferation).12,13 In case of a mixed neovascularization, the CNV was considered to be “minimally classic” if the majority of the CNV was type 1. If the mixed neovascularization was predominantly type 2, it was referred to as “predominantly classic,”13 The first choice of anti-VEGF treatment in treatment-naive patients was 3 monthly intravitreal injections of bevacizumab. Switching to another type of anti-VEGF treatment (either ranibizumab or aflibercept) was considered when insufficient reduction in subretinal and/or intraretinal fluid was observed on OCT at a control examination within 4 weeks after the third bevacizumab injection. Indocyanine green angiography was performed either in case of a suspicion of polypoidal choroidal vasculopathy (PCV) or when patients did not respond sufficiently to anti-VEGF treatment. Treatments were applied according to the pro re nata regimen, where patients had a follow-up every 4 weeks after initial treatment. Retreatment of 3 injections every 4 weeks was performed upon reoccurrence of subretinal and/or intraretinal fluid on OCT and/or on FA, or when hemorrhage was seen during fundoscopy.14
Information regarding BCVA and CRT was collected at the first visit before initial treatment (baseline), at the first visit after initial treatment, which was usually scheduled after 3 intravitreal injections, and 6, 12, and 24 months after initial treatment. For patients for whom ETDRS BCVA was not available, a previously published formula was used to convert Snellen BCVA to ETDRS values.15 The number and mode of anti-VEGF injections during follow-up, and changes in subretinal and/or intraretinal fluid on OCT were recorded. The used dosages of bevacizumab, ranibizumab, and aflibercept were 1.25, 0.5, and 2 mg, respectively. In order to quantify the number of anti-VEGF injections required to achieve a total resolution of subretinal and/or intraretinal fluid for ≥3 months without treatment, OCT images were used.
Statistical analysis
Statistical analysis was performed using IBM SPSS Statistics, version 23 (IBM Corporation, Armonk, NY, USA). The clinical parameters at baseline and during follow-up were analyzed in an autoregressive model within a linear mixed model with time as a factor. A P-value of <0.05 was considered to be statistically significant.
Results
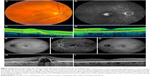
Fundus photography, FA, and OCT of 381 patients were analyzed to identify cases within the spectrum of unilateral nAMD without drusen in the macular region of the fellow eye at initial diagnosis. Twenty-nine patients (8%) (16 male and 13 female patients) met the inclusion criteria. The mean age at onset of disease of these 29 patients was 75 years (range: 53–89 years). In 1 patient (3%), a few small hard drusen were found in the eye with nAMD (Table 1). A spectrum of neovascular abnormalities was observed (Figure 1): 18 patients were diagnosed with type 1 neovascularization, 7 patients with type 2 neovascularization, and 4 patients with a mixed neovascularization. Three of these 29 patients (10%) were also diagnosed with PCV (of whom in 1 patient the lesion was an isolated polyp, and in 2 patients the lesions occurred together with a neovascularization/branching vascular network, confirmed on additional indocyanine green angiography) (Table 1). Patients received the first intravitreal injection at a mean of 7 days (range: 0–16 days) after being diagnosed with nAMD. Some patients were also scheduled for either half-dose or full-dose PDT (Table 2). In none of the patients, ophthalmic surgery had been performed in their study eye during follow-up. One patient received cataract surgery in the fellow eye during the study.
Overall, BCVA in all patients increased with a mean of 2.5 ETDRS letters at 24 months, which was not statistically significant as compared to baseline (P=0.456) (Figure 2). The BCVA of 22 patients (76%) improved or remained stable (defined as a gain in BCVA, a stable BCVA, or a loss of <5 ETDRS letters), whereas BCVA decreased (defined as a loss of ≥5 ETDRS letters) in 7 patients (24%). A gain of ≥15 ETDRS letters from baseline was seen in 5 patients (17%). The BCVA of the 3 PCV patients during follow-up and the treatments performed in these patients are shown separately (Table 3).
Compared to baseline, the mean CRT had significantly decreased with 99 μm (P<0.001) at final follow-up (Figure 3). At all follow-up visits, the decrease in CRT was statistically significant (Figure 3). Based on the type of neovascularization, no significant difference in CRT decrease between the 2 groups at several time points was found (Figure 4).
  | Figure 3 Mean change in CRT during follow-up, compared to baseline. |
In 6 of 29 treated patients (21%), there were no signs of CNV activity on OCT for ≥3 months (Table 4) after baseline. However, 1 patient needed full-dose PDT after 8 intravitreal bevacizumab injections and 3 ranibizumab injections before a complete resolution of subretinal and/or intraretinal fluid was achieved for these 3 months.
Discussion
This study examined the clinical spectrum and therapeutic outcome of patients with unilateral nAMD without drusen in the macular region of their fellow eye. In these patients, no statistically significant improvement in BCVA occurred after a follow-up of 24 months after first treatment for nAMD. Moreover, in 7 patients (24%), the BCVA declined with ≥5 ETDRS letters. Overall, the mean decrease in CRT was statistically significant at the final follow-up visit. In all patients, the mean increase in BCVA in this study was 2.5 ETDRS letters at the last follow-up visit at 24 months after baseline, which is relatively low in comparison with other studies. In these studies, a statistically significant increase in BCVA of 14 and 6–7.9 ETDRS letters was found from baseline until the follow-up visits at 12 and 24 months, respectively, after using intravitreal injections according to the pro re nata regimen.16,17 However, the mean number of intravitreal injections used in our study over the period of 24 months, and the significant decrease in CRT at 24 months after baseline, is comparable to another study.17 This may suggest that subgroups of AMD could have a different prognosis and that different treatment strategies may be needed to achieve a significant improvement in BCVA.
The correlation between nAMD and clinical characteristics of non-neovascular fellow eyes has been studied previously.18–20 Available studies indicate a correlation between the subtype of neovascularization, based on the anatomical classification, and phenotypic characteristics of the non-neovascular fellow eyes.18,19 Predominantly, intraretinal lesions are more likely to be accompanied by reticular pseudodrusen and/or a decreased subfoveal choroidal thickness compared to supra-RPE lesions.19 Other studies have shown an association between the development of nAMD and reticular pseudodrusen in the fellow eye.18 However, no studies of patients with unilateral nAMD without any drusen in the fellow eye have been performed thus far.
Patients with nAMD without drusen in the fellow eye could be argued to have overlapping phenotypes and/or phenocopies mimicking AMD, which share clinical and possibly pathophysiologic features with drusenoid AMD, such as idiopathic PCV, and neovascular CSC.21,22 At baseline only 1 patient (3%) had a few small hard drusen in the eye with nAMD. The remaining patients (97%) had no drusen in their eye with nAMD. A possible explanation for this finding is that drusen in earlier stages may regress before being converted to advanced nAMD.23 The clinical observation that some patients can have this type of unilateral nAMD raises the question whether these patients truly have AMD in the strict sense, because early AMD is characterized by the appearance of drusen with a size of >63 μm in either eye.3,6 However, this study excluded patients with other diagnoses explaining the occurrence of CNV, such as pseudoxanthoma elasticum, high myopia, CSC, macular telangiectasia, or multifocal choroiditis. The mean age at onset of the nAMD patients in this study was 75 years, and CNV in the affected eye could be easily classified as type 1 or 2 (or mixed type) CNV, sometimes with a component of PCV. Apart from an absence of drusen in the fellow eye in the current patient group, any drusen in the eye affected by nAMD was also not observed although the absence of drusen in the case of neovascularization is not a definitive proof that these were not present prior to the onset of nAMD. It is therefore argued that the patient group that is described here truly has a form of nAMD although they do not have drusen in the fellow eye. This study seems to point out that for a subgroup of nAMD patients the presence of drusen is not a prerequisite to develop nAMD, as evidenced indirectly by an absence of drusen in the fellow eye.
Drusen are located between the RPE and Bruch’s membrane and consist of a broad spectrum of components, such as proteins that are involved in the complement system.24 Drusen are considered to be by-products of chronic, local inflammatory events in Bruch’s membrane and play an important role in the pathogenesis and progression of AMD.24 They also contain esterified and unesterified cholesterol which may be produced by the RPE.25,26 Moreover, strong evidence has been found that genes involved in lipid transport and processing are consistent with the composition of drusen.27 It is possible that the current patient cohort had earlier, clinically invisible underlying pathophysiologic signs such as basal laminar deposits and basal linear deposits, and membranous debris in the fellow eye without visible drusen, which may then still cause a drusenoid environment with pathophysiological consequences such as CNV.28 A better understanding of the development of drusen in nAMD, and the possible prerequisite of drusen or their precursor material to cause CNV, could provide insight in the pathogenesis, prognostic factors, and intervention strategies for CNV in all nAMD patients.
Although the number of Caucasian PCV patients (10%) involved in this study is too small to draw conclusions about the therapeutic outcome in this specific subgroup, these patients appeared to respond to a treatment combination of PDT and intravitreal anti-VEGF injections. The prevalence of PCV in Caucasians seems to be substantially lower than in Asians,29 and Caucasian PCV patients may differ from PCV patients with an Asian ethnicity in terms of presenting clinical features and angiographic behavior.29–31 Larger studies are necessary to explore the differences in phenotypes, genotypes, and treatment response between the Caucasian and Asian population. It is currently unclear whether PCV can be seen as a specific pathogenetic and/or clinical entity within the spectrum of AMD in Caucasians, or as a variant of CNV.32,33 There is evidence that PCV shares characteristics with nAMD, but different phenotypic subtypes are also possible.34
The findings of this study clearly indicate that there is an entity of unilateral AMD without drusen in the fellow eye. This is the first exploratory study assessing the clinical characteristics and therapeutic outcome of patients with nAMD and no drusen in the fellow eye. The retrospective aspect and non-comparative nature of this study gives its inherent shortcomings. In some cases, different OCT machines were used during follow-up for CRT measurements. Only 17 of the 29 (59%) included patients in the cohort had a follow-up of 24 months. Larger, preferably prospective clinical studies can provide a better understanding of this specific subgroup of AMD. It was demonstrated that therapeutic response in patients with unilateral nAMD without drusen in the fellow eye could differ from typical nAMD patients. These findings indicate that drusen as precursor lesions may not be a prerequisite to develop nAMD.
Acknowledgments
The authors thank MSc Ron Wolterbeek from the Department of Medical Statistics and Bioinformatics of the Leiden University Medical Center, for his statistical support. This research was supported by the Mathilde Grant, Stichting Leids Oogheelkundig Ondersteuningsfonds, Rotterdamse Stichting Blindenbelangen, and the Netherlands Organisation for Scientific Research (NWO). The funding organizations MD Fonds, Landelijke Stichting voor Blinden en Slechtzienden, Retina Netherlands, and BlindenPenning contributed through UitZicht. The aforementioned funding sources provided unrestricted grants and had no role in the design or conduct of this research.
Disclosure
The authors report no conflicts of interest in this work.
References
de Jong PT. Age-related macular degeneration. N Engl J Med. 2006;355:1474–1485. | ||
Bird AC, Bressler NM, Bressler SB, et al. An international classification and grading system for age-related maculopathy and age-related macular degeneration. The International ARM Epidemiological Study Group. Surv Ophthalmol. 1995;39:367–374. | ||
Ferris FL 3rd, Wilkinson CP, Bird A, et al. Clinical classification of age-related macular degeneration. Ophthalmology. 2013;120:844–851. | ||
Freund KB, Zweifel SA, Engelbert M. Do we need a new classification for choroidal neovascularization in age-related macular degeneration? Retina. 2010;30:1333–1349. | ||
Bressler NM, Bressler SB, West SK, Fine SL, Taylor HR. The grading and prevalence of macular degeneration in Chesapeake Bay watermen. Arch Ophthalmol. 1989;107:847–852. | ||
Klein R, Klein BE, Linton KL. Prevalence of age-related maculopathy. The Beaver Dam Eye Study. Ophthalmology. 1992;99:933–943. | ||
Mitchell P, Smith W, Attebo K, Wang JJ. Prevalence of age-related maculopathy in Australia. The Blue Mountains Eye Study. Ophthalmology. 1995;102:1450–1460. | ||
Vingerling JR, Dielemans I, Hofman A, et al. The prevalence of age-related maculopathy in the Rotterdam Study. Ophthalmology. 1995;102:205–210. | ||
Group MPS. Risk factors for choroidal neovascularization in the second eye of patients with juxtafoveal or subfoveal choroidal neovascularization secondary to age-related macular degeneration. Arch Ophthalmol. 1997;115:741–747. | ||
Solomon SD, Jefferys JL, Hawkins BS, Bressler NM. Incident choroidal neovascularization in fellow eyes of patients with unilateral subfoveal choroidal neovascularization secondary to age-related macular degeneration: SST report No 20 from the Submacular Surgery Trials Research Group. Arch Ophthalmol. 2007;125:1323–1330. | ||
Klein R, Myers CE, Lee KE, et al. Small drusen and age-related macular degeneration: The Beaver Dam Eye Study. J Clin Med. 2015;4:425–440. | ||
Agarwal A. In: Saunders, ed. Gass’ Atlas of Macular Diseases. 5th ed. Philadelphia: Elsevier; 2012:92–100. | ||
Yannuzzi L. In: Saunders, ed. The Retinal Atlas. London: Elsevier; 2010:568–577. | ||
Mitchell P, Korobelnik JF, Lanzetta P, et al. Ranibizumab (Lucentis) in neovascular age-related macular degeneration: evidence from clinical trials. Br J Ophthalmol. 2010;94:2–13. | ||
Gregori NZ, Feuer W, Rosenfeld PJ. Novel method for analyzing snellen visual acuity measurements. Retina. 2010;30:1046–1050. | ||
Bohni SC, Bittner M, Howell JP, Bachmann LM, Faes L, Schmid MK. Comparison of Eylea(R) with Lucentis(R) as first-line therapy in patients with treatment-naive neovascular age-related macular degeneration in real-life clinical practice: retrospective case-series analysis. BMC Ophthalmol. 2015;15:109. | ||
Martin DF, Maguire MG, Fine SL, et al. Ranibizumab and bevacizumab for treatment of neovascular age-related macular degeneration: two-year results. Ophthalmology. 2012;119:1388–1398. | ||
Hogg RE, Chakravarthy U. Visual function and dysfunction in early and late age-related maculopathy. Prog Retin Eye Res. 2006;25:249–276. | ||
Marsiglia M, Boddu S, Chen CY, et al. Correlation between neovascular lesion type and clinical characteristics of nonneovascular fellow eyes in patients with unilateral, neovascular age-related macular degeneration. Retina. 2015;35:966–974. | ||
Kang SW, Lee H, Bae K, Shin JY, Kim SJ, Kim JM; Korean Age-related Maculopathy Study (KARMS) Group. Investigation of precursor lesions of polypoidal choroidal vasculopathy using contralateral eye findings. Graefes Arch Clin Exp Ophthalmol. Epub 2016 Sep 5. | ||
Hatz K, Prunte C. Polypoidal choroidal vasculopathy in Caucasian patients with presumed neovascular age-related macular degeneration and poor ranibizumab response. Br J Ophthalmol. 2014;98:188–194. | ||
Schatz H, Madeira D, Johnson RN, McDonald HR. Central serous chorioretinopathy occurring in patients 60 years of age and older. Ophthalmology. 1992;99:63–67. | ||
Schlanitz FG, Baumann B, Kundi M, et al. Drusen volume development over time and its relevance to the course of age-related macular degeneration. Br J Ophthalmol. 2016. | ||
Anderson DH, Radeke MJ, Gallo NB, et al. The pivotal role of the complement system in aging and age-related macular degeneration: hypothesis re-visited. Prog Retin Eye Res. 2010;29:95–112. | ||
Curcio CA, Presley JB, Malek G, Medeiros NE, Avery DV, Kruth HS. Esterified and unesterified cholesterol in drusen and basal deposits of eyes with age-related maculopathy. Exp Eye Res. 2005;81:731–741. | ||
Curcio CA, Presley JB, Millican CL, Medeiros NE. Basal deposits and drusen in eyes with age-related maculopathy: evidence for solid lipid particles. Exp Eye Res. 2005;80:761–775. | ||
Pikuleva IA, Curcio CA. Cholesterol in the retina: the best is yet to come. Prog Retin Eye Res. 2014;41:64–89. | ||
Sarks S, Cherepanoff S, Killingsworth M, Sarks J. Relationship of basal laminar deposit and membranous debris to the clinical presentation of early age-related macular degeneration. Invest Ophthalmol Vis Sci. 2007;48:968–977. | ||
Ciardella AP, Donsoff IM, Huang SJ, Costa DL, Yannuzzi LA. Polypoidal choroidal vasculopathy. Surv Ophthalmol. 2004;49:25–37. | ||
Lafaut BA, Leys AM, Snyers B, Rasquin F, De Laey JJ. Polypoidal choroidal vasculopathy in Caucasians. Graefes Arch Clin Exp Ophthalmol. 2000;238:752–759. | ||
Imamura Y, Engelbert M, Iida T, Freund KB, Yannuzzi LA. Polypoidal choroidal vasculopathy: a review. Surv Ophthalmol. 2010;55:501–515. | ||
Balaratnasingam C, Lee WK, Koizumi H, Dansingani K, Inoue M, Freund KB. Polypoidal choroidal vasculopathy: a distinct disease or manifestation of many? Retina. 2016;36:1–8. | ||
Laude A, Cackett PD, Vithana EN, et al. Polypoidal choroidal vasculopathy and neovascular age-related macular degeneration: same or different disease? Prog Retin Eye Res. 2010;29:19–29. | ||
Coscas G, Lupidi M, Coscas F, et al. Toward a specific classification of polypoidal choroidal vasculopathy: idiopathic disease or subtype of age-related macular degeneration. Invest Ophthalmol Vis Sci. 2015;56:3187–3195. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.