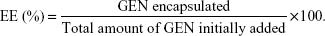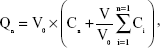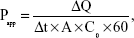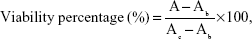Back to Journals » International Journal of Nanomedicine » Volume 9 » Issue 1
Nanostructured lipid carrier surface modified with Eudragit RS 100 and its potential ophthalmic functions
Authors Zhang W, Li X, Ye T, Chen F, Yu S, Chen J, Yang X, Yang N, Zhang J, Liu J, Pan W, Kong J
Received 3 March 2014
Accepted for publication 25 May 2014
Published 11 September 2014 Volume 2014:9(1) Pages 4305—4315
DOI https://doi.org/10.2147/IJN.S63414
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Wenji Zhang,1,3,* Xuedong Li,2,* Tiantian Ye,3 Fen Chen,3 Shihui Yu,3 Jianting Chen,3 Xinggang Yang,3 Na Yang,2 Jinsong Zhang,2 Jinlu Liu,2 Weisan Pan,3 Jun Kong2
1Department of Pharmaceutics, School of Traditional Chinese Medicine, Shenyang Pharmaceutical University, Shenyang, People’s Republic of China; 2Department of Ophthalmology, The Fourth Affiliated Hospital of China Medical University, Eye Hospital of China Medical University, Key Lens Research Laboratory of Liaoning Province, Shenyang, People’s Republic of China; 3Department of Pharmaceutics, School of Pharmacy, Shenyang Pharmaceutical University, Shenyang, People’s Republic of China
*These authors contributed equally to this work
Abstract: This study was carried out to evaluate the ocular performance of a cationic Eudragit (EDU) RS 100-coated nanostructured lipid carrier (NLC). The genistein encapsulated NLC (GEN-NLC) was produced using the melt-emulsification technique followed by surface absorption of EDU RS 100. The EDU RS 100 increased the surface zeta potential from −7.46 mV to +13.60 mV, by uniformly forming a spherical coating outside the NLC surface, as shown by transmission electron microscopy images. The EDU RS 100 on the NLC surface effectively improved the NLC stability by inhibiting particle size growth. The obtained EDU RS 100-GEN-NLC showed extended precorneal clearance and a 1.22-fold increase in AUC (area under the curve) compared with the bare NLC in a Gamma scintigraphic evaluation. The EDU RS 100 modification also significantly increased corneal penetration producing a 3.3-fold increase in apparent permeability coefficients (Papp) compared with references. Draize and cytotoxicity testing confirmed that the developed EDU RS 100-GEN-NLC was nonirritant to ocular tissues and nontoxic to corneal cells. These results indicate that the NLC surface modified by EDU RS 100 significantly improves the NLC properties and exhibits many advantages for ocular use.
Keywords: genistein, precorneal retention, cytotoxicity
Introduction
Posterior capsular opacification (PCO) remains the most frequent complication following extracapsular cataract surgery and can cause secondary blurred vision.1 Currently, surgical treatments are suffering from a variety of inevitable complications and are expensive to perform,2 whereas chemical prevention by selectively killing the lens epithelial cells using cytotoxic drugs or other pharmacological agents has shown much promise.3–6 In most clinical cases, PCO is treated by physical surgery of laser capsulectomy. The attempts of treating PCO by eye drops were hindered by the intrinsic poor precorneal penetration ability and limited retention time on the cornea.
Recently, a number of colloid systems have been used for ophthalmic drug delivery, aiming to increase precorneal retention and enhance drug absorption. A nanostructured lipid carrier (NLC), derived from lipid nanoparticles, is composed of a solid lipid matrix with spatially incompatible liquid lipids, and could offer many advantages for drug delivery such as controlled drug release, higher drug loading, and good bioavailability.7 More importantly, the structural affinity for cell membrane means that NLC shows excellent cellular uptake and absorption. The advantages above make NLC an attractive candidate for the topical administration of eye drops.8–10 However, the precorneal retention time of NLC is limited due to the rapid removal by tears and the minimal permeability across the corneal epithelium,11 which hinders the absorption of drugs through the cornea.
The Eudragit (EUD) series of polymers have been used for a long time as the coating film for solid dosage forms providing targeted or controlled drug delivery.12,13 EUD RS 30D is the 30% aqueous dispersion of EDU RS 100, which is a copolymer of ethyl acrylate and methyl methacrylate and has a low content of a methacrylic acid ester, with between 4.5% and 6.8% of quaternary ammonium groups.14 Recently, investigations were conducted to develop EDU RS 100 as nanoparticle matrix for the sustained delivery of drugs with a high bioavailability along with good tissue tolerance as shown by the Draiz test in ocular tissues.15,16 In spite of the good compatibility, few studies have shown that EDU RS 100, a mixture of methacrylate polymers, can be degraded into smaller fragments for absorbance in the eye, and the production process of EDU RS 100 nanoparticles usually requires organic solvents for polymer and drug dissolution and, therefore, it is hard to predict its safety during long-time use. In our study, attempts were made to reduce the amount of EDU RS 100 in the formulation by distributing EDU RS 100 on the surface of the controlled-release and highly biocompatible drug-delivery system-NLC without the use of organic solvents to further reduce any potential toxicity. In addition, we also wished to prove that such surface modification with low amount of EDU RS 100 could still provide excellent ocular properties including a high bioavailability in the eye.
Our focus in this paper is to improve the precorneal retention time and cornea penetration of the developed drug-loaded NLC to achieve a better ophthalmic performance. Usually, the surface of the cornea and conjunctiva is covered with a mucus film, a thin fluid layer mostly composed of high molecular weight glycoprotein with a negative charge at physiological pH.17 Taking this into account, our approach is based on the electrostatic modification of cationic EDU RS 100 on the NLC surface, so that the resultant cationic polymer coated surface could interact with cornea cells leading to a longer retention and an improved penetration. Genistein (GEN) was selected as the model drug; it is poorly soluble in water and can be effectively used for the treatment of PCO.18 In this study, attempts were made to evaluate EDU RS 100 as an efficient ophthalmic delivery system and to investigate its merits.
Materials and methods
Reagents, cell lines, and animals
GEN was purchased from Huike Pharmaceutical Company (Xian, People’s Republic of China); Compritol 888 ATO and Gelucire 44/14 were kindly donated by Gattefosse (Paris, France); Miglyol 812 N was obtained from Sasol (Witten, Germany); Cremphor® EL was provided by (Ludwigshafen, Germany). Egg phosphatidylcholine (EPC) was obtained from Shanghai Taiwei Pharmaceutical Co., Ltd, and EDU RS 30D was supplied by Evonic Industry (Germany). Purified water was used after deionization and filtration.
The human cornea epithelial (HCE) cell line was provided by China Medical University; Dulbecco’s Modified Eagle’s Medium (DMEM), fetal bovine serum (FBS), and trypsin were all obtained from Gibco Technology (Shanghai, People’s Republic of China); Cell Counting Kit-8 (CCK-8) was obtained from Dojindo Molecular Technology Inc. (Shanghai, People’s Republic of China); Trypan blue and penicillin–streptomycin for cell culture were both obtained from Beyotime Technology (Jiangsu, People’s Republic of China). TrypLE™ Express was obtained from Invitrogen (Carlsbad, CA, USA). All other reagents and solvents used were of analytical grade.
New Zealand albino rabbits free of any ocular damage were obtained from Shenyang Pharmaceutical University Animal Center (Shenyang, People’s Republic of China). All animal studies were conducted in accordance with the Principles of Laboratory Animal Care and approved by Shenyang Pharmaceutical University Animal Ethical Committee.
Methods
Preparation of GEN-loaded NLC (GEN-NLC)
GEN-NLC was produced using the melt emulsification technique combined with ultra-sonication as described in our previous study with minor modification.19–22 Briefly, selected amounts of GEN (0.05%, 10 mg), Compritol 888 ATO (1.10%, 220 mg), Miglyol 812 N (0.38%, 75 mg), and Gelucire 44/14 (0.50%, 100 mg) were blended and heated under moderate stirring at 85°C to form a transparent and uniform oil phase. Then, Cremphor® EL (1.13%, 225 mg) and EPC (0.38%, 750 mg) were dissolved in 20 mL purified water before being heated to 85°C and added dropwise to the oil phase, with magnetic stirring at 600 rpm over a period of 10 minutes. After the coarse emulsion was formed, it was homogenized using a probe-ultrasonic cell disruptor (JY-92-II; Xinzhi, Ningbo, People’s Republic of China) for different time periods. The obtained nanoemulsions were rapidly cooled in an ice bath (0°C–4°C) to allow them to solidify and form GEN-NLC.
Surface modification with EDU RS 100
To obtain NLC surface-modified with EDU RS 100, appropriate amounts of EDU RS 30D were diluted with purified water to form EDU RS 100 solutions (0.2%, 0.6%, 1.0%, and 2.0%, w/w). In each case, the GEN-NLC was added slowly to the EDU RS 100 solutions freshly diluted to the same volume, then subjected to magnetic agitation at 20°C for 10 minutes, to give final EDU RS 100-NLC concentrations of 0.1%, 0.3%, 0.5%, and 1.0%.
Mean particle size and zeta potential
The mean particle size, polydispersity index (PI), and zeta potential were determined by photon correlation spectroscopy using a Zeta-sizer Nano (Malvern Instruments, Worcestershire, UK) at 25°C.23 The particle size and PI values were obtained at an angle of 90 degrees with respect to the incident beam in 10 mm disposable polystyrene cells. The zeta potential was measured in disposable plain folded capillary zeta cells by determining the electrophoretic mobility on the particle surface using the same instrument. Each measurement was made in triplicate.
Transmission electron microscopy (TEM) analysis
The morphology of the nanoparticles was observed by TEM (JEM-1200EX JEOL, Tokyo, Japan). Samples were prepared by drying a dispersion of the GEN nanoparticles, diluted 50-fold with double-distilled water, on a copper grid coated with an amorphous carbon film and then negatively stained with 1% phosphotungstic acid for observation.
Encapsulation efficiency (EE)
GEN-NLC or EDU RS 100-GEN-NLC was separated from unentrapped drug using a Sephadex G-50 column (2.5 cm ×1.0 cm). Briefly, Sephadex G-50 soaked in 100°C distilled water for a few hours was loaded into a 2.5 mL syringe and then centrifuged at 2,000 rpm for 2 minutes to obtain a dehydrated column. Subsequently, 0.2 mL samples of NLC suspensions were added to the column and subjected to centrifugation (2,000 rpm, 2 minutes) to remove any untrapped drug. Afterwards, the column was washed four times with 0.2 mL distilled water to achieve complete clearance (based on the results of our previous tests) of the GEN-NLCs in the column. The eluted NLCs were collected and destroyed using a mixed solvent of dichloromethane and methanol (1:4, v/v). The amount of encapsulated GEN was determined by high-performance liquid chromatography (HPLC) using a Diamasil® C18 column (200 mm ×4.6 mm, 5 μm, Dikma, People’s Republic of China). The mobile phase was a mixture of methanol −0.05% phosphoric acid aqueous solution (60/40, v/v), and the flow rate was 1.0 mL/min. The wavelength was set at 260 nm. Samples (0.2 mL) of GEN-NLC or CH-GEN-NLC suspension were destroyed with methanol and dichloromethane and determined by HPLC. The EE was calculated using the following equation:
|
|
Physical stability
The physical stability of the prepared bare GEN-NLC and EDU RS 100-GEN-NLC was assessed briefly as follows. NLC samples of 10 mL (1 mg/mL) were kept in a closed glass vial and stored at 25°C over 10 days. At predetermined time intervals, aliquots were taken and subjected to particle size and zeta potential analysis.
Corneal penetration
The influence of EDU RS 100 on drug corneal penetration was evaluated using freshly excised rabbit cornea (approximate available areas 0.70 cm2) on a diffusion apparatus.17,24 The donor and receiver cells were filled with 1 mL sample and 8 mL glutathione bicarbonate Ringer (GBR) buffer, respectively, and maintained at 34°C. At predetermined intervals, 1 mL samples were withdrawn from the receiver compartment and immediately replaced with an equal volume of GBR buffer. The osmotic pressures of the sample and GBR solutions were adjusted with glycerol to 290–300 mOsm/kg, in an osmometer (STY-1, Tianjin, Zhejiang, People’s Republic of China) to obtain a normal corneal morphology.
The cumulative penetration at various intervals was calculated as follows:
|
|
where V0 is the volume of GBR in the receiver cell (8 mL); V is the sampling volume (1 mL); Cn is the drug concentration in the receiver compartment at various intervals; and Ci is the drug concentration in the receiver compartment before determination.
The rate of drug penetration was measured by the apparent permeability coefficient (Papp) and Jss (steady-state flux)17 as follows:
|
|
Jss= C0 ⨯Papp, | (4) |
where ΔQ/Δt refers the gradient of the straight line of Qn versus time; and A is the penetrating region area (0.70 cm2).
In vivo precorneal retention
The precorneal retention potential of the developed EDU RS 100-NLC was evaluated by installation after 99mTc labeling to the precornea area followed by scheduled detection. Equal amounts of 150 μL 99mTc solutions were used to label the NLC by addition to the water phase during the preparation process. The 99mTc-NLC was passed through a 100 nm membrane filter to separate it from the free pertechnetates. The radio intensity of the filtrate was measured and compared with the 99mTc-NLC to assess the radiolabeling efficacy. Non-coated NLC and physiological saline solution were labeled and used as a control. Instillation was carried out by dropping 150 μL samples into the lower conjunctival sac, and recording was started 5 seconds afterwards. Pre-corneal images were recorded using a Gamma camera, and the radiation of 99mTc (140 keV) in the precorneal area was detected over 10 minutes using a 128×128 pixel matrix.
Draize test
The ocular irritation study was carried out on New Zealand white albino rabbits, 10–12 months old and weighing 3.92–3.96 kg, obtained from Shenyang Pharmaceutical University Animal Centre (Shenyang, People’s Republic of China) and scored based on the Draize test. Rabbits were checked to ensure they had normal eyes before the test. EDU RS 100 (0.5%, w/v)-GEN-NLC was administered into the lower conjunctival sac of the left eyes five times a day and the right eyes were used as a control. Evaluations were made every 24 hours up to 3 days after administration.25 Ocular changes were scored for signs of lachrymation, irritation, erythema, edema, and any other ocular reactions until the end of the study.26 After these examinations, the rabbits were euthanized, and their cornea, conjunctiva, and iris were removed and fixed in 4% formaldehyde, followed by the preparation of histological sections for viewing.
In vitro cell viability assays
Cell culture
The immortalized human cornea epithelial cell lines were provided by the Fourth Affiliated Hospital of China Medical University (Shenyang, People’s Republic of China) and grown in medium composed of DMEM, 50 μg/mL penicillin–streptomycin, and 20% FBS in an atmosphere of 5% CO2 −95% air at 37°C. Cell feeding was conducted three times a week, and cells were passaged using trypsin (0.125%)–EDTA (0.5 mM) after reaching confluence. For all procedures, cells growing as a monolayer were harvested using TrypLE™ Express, and were maintained in a logarithmic growth phase during the studies. Cell counts were conducted routinely using the trypan blue dye exclusion method.
Viability assays of HCE cells
The viability tests were conducted in HCE cell lines using CCK-8 assay to investigate any cytotoxic effects of EDU RS 100-GEN-NLC. HCE cells were seeded in 96-well plates at a density of 10,000 cells per well in 100 μL growth medium, and allowed to grow for 24 hours. Then, the medium was removed and cells were exposed to 100 μL of various concentrations of GEN formulations or blank NLC (n=5) for further incubation in a 5% CO2 atmosphere at 37°C for 4 hours. After incubation, cells were washed twice with phosphate-buffered saline (PBS) pH 7.4 and incubated with 100 μL 10% CCK-8 solution per well for another 4 hours. The absorbance was then measured spectrophotometrically at 450 nm using a micro-plate reader (Tristar LB 941; Berthold Technology, Hertfordshire, UK). Untreated cells served as a 100% cell viability control, and the media served as a background reference. The survival percentage was calculated in comparison with the control excluding the background reference.
|
|
where A, Ab, and Ac is the absorbance of the sample well, background well and control well, respectively.
Statistical methods
Student’s t-test in this study was performed with a level of significance defined as a P-value <0.05 using the statistical package for social sciences (SPSS, version 17.0).
Results and discussion
Physicochemical properties
The physicochemical properties of EDU RS 100-NLC were evaluated, and the results are shown in Table 1. The NLC was surface-modified with EDU RS 100 dispersions of various concentrations to determine the saturated degree of adsorption. As shown, the particle size of uncoated NLC was 88.34 nm, with a negative charge (−6.98 mV). With an increase in the EDU RS 100 polymer concentration up to 0.5%, the particle size of EDU RS 100-GEN-NLC increased (PI<0.30). When EDU RS 100 reached 1.0%, double peaks appeared in the intensity size distribution, revealing that two groups of different size coexist in the solution system at this EDU RS 100 concentration. The particle size and zeta potential of the EDU RS 100 dispersion (1.0%) were measured for comparison, and this showed that the particle size of the EDU RS 100 dispersion was very close to that of the smaller size in the EDU RS 100 (1.0%)-GEN-NLC system, implying that the smaller particles in the system represent an excess of EDU RS 100 polymers. Similarly, the zeta potential of EDU RS 100-GEN-NLC increased along with the EDU RS 100 concentration, then leveled off at 0.5% and reached a relatively constant value when the concentration rose to 1.0%. Therefore, it can be concluded that the EDU RS 100 absorbed on the NLC surface reached saturation in the presence of 0.5% EDU RS 100, and any excess would not affect the thickness and strength of the coating and surface charges, but would lead to polymer aggregation in the solution instead, as indicated by another sharp increase in particle size.
The cationic EDU RS 100 polymers reduced the electron cloud density surrounding the NLC particle surface, rendering the integral particles positive. In addition, the coating mechanism was also partially attributed to the affinity of the lipophilic lipid NLC for the long chain of the EDU RS 100 molecules. The result of the zeta potential and particle size determinations supported the adsorption of EDU RS 100.
Table 2 summarizes the preparation and characterization of non- and EDU RS 100-coated GEN-NLC with various particle sizes and zeta potentials (n=3). It can be seen that 1) the sizes of the GEN-NLC did not change significantly from their estimated sizes; 2) the EDU RS 100 coatings increased the NLC diameters, confirming the successful surface modification; 3) the particle sizes of the uncoated NLC were found to be controlled by the sonication time and power, and varied approximately in a range of 100–1,000 nm; 4) the EE of EDU RS 100-NLC was about 90%, which is not far from the value of pre–EDU RS 100 modification, indicating that the EDU RS 100 polymers absorbed on NLC surfaces did not affect the drug encapsulation, and the resultant EE of EDU RS 100-NLC mainly depends on the NLC inside. Since NLC could be successfully prepared with a high EE, this method of EDU RS 100 surface decoration can produce a higher EE than incorporating the drug into EDU RS 100 polymers based on the results of previous studies which reported only ~50% entrapment efficiency.15,27
Morphological studies
We modified the surface of the lipid NLC with EDU RS 100 on the hypothesis that the cationic polymers would become attached to the negative NLC surfaces by electrostatic attraction and hydrophobic interaction. Figure 1 shows the morphology of uncoated GEN-NLC and EDU RS 100-coated NLC taken by TEM. As shown, GEN-NLC exhibited a spherical structure of nanodroplets (Figure 1A), while the EDU RS 100-coated NLC showed spherical-shaped particles covered by an outer-layer structure (Figure 1B). Such typical architecture proved our hypothesis on the absorbance of polymer on particle surfaces, and was consistent with the results obtained from the Malvern particle size analysis.
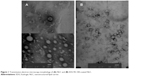  | Figure 1 Transmission electron microscopy morphology of (A) NLC and (B) EDU RS 100-coated NLC. |
Physical stability
Table 3 shows the particle sizes and zeta potentials of developed GEN-NLC and EDU RS 100-GEN-NLC stored over a period of 10 days at room temperature (25°C) for the stability investigation. It was found that the particle sizes of bare NLC increased significantly from 88.34 to 430 nm after 10 days of storage, and visible flocculation could be found, with a PI ranging from 0.25 to 0.46. Clear growth inhibition was observed in the particle sizes of EDU RS 100-coated nanoparticles. The average size was 289.93 nm for EDU RS 100-NLC after 10 days of storage. This can be attributed to the coating of the cationic polymer outer-layer, which reduces the particle collisions by providing a higher strength of electric and steric stabilization effects. This explanation was confirmed by the positive zeta potentials of EDU RS 100-NLC after coating, and the charge was only slightly reduced during the storage period. One interesting finding is that, as we see, a wider particle size distribution was obtained after EDU RS 100 coating, indicating easier particle aggregation based on the theory of Ostwald ripening. However, the stability of EDU RS 100-NLC was actually improved with respect to the particle sizes and zeta potentials. This could possibly be explained by the hypothesis that the marked electrical repulsion overcomes the instability caused by the inhomogeneity of the particle sizes. Therefore, coating the cationic EDU RS 100 polymers is an effective method to improve the storage stability by introducing electric repulsion among nanoparticles, and the sample stability could be further increased if the particles could be made more homogeneous.
In vitro corneal permeation
The corneal penetration study was performed to evaluate the effect of EDU RS 100 on drug transcorneal transportation. Figure 2 shows the corneal penetration curves of GEN-phosphate eye drops (0.1% GEN solubilized by Cremphor EL and EPC in PBS), GEN-NLC, and EDU RS 100-GEN-NLC. The corresponding apparent permeability coefficients (Papp) and R2 (correlation coefficient) are shown in Table 4. As observed, the linear penetration profiles in all cases (R2>0.9968) confirm the cornea integrity throughout the experiments and the constant penetration rate. Compared with the GEN surfactant solution, GEN-NLC showed a significantly higher Papp due to the presence of the lipid matrix, which easily adheres to and is biocompatible with the corneal epithelial cells. When the NLC was surface modified with EDU RS 100, the Papp further increased 3.3-fold and 1.6-fold, compared with the solution and uncoated NLC, respectively. The increase in Papp proved that the EDU RS 100 could improve the cornea permeability. However, the mechanism responsible remains unclear since it is possible that the poly-cationic EDU RS 100 layer increases the penetration either by the bridging electrostatic adherence of NLC to the corneal surface or by serving as a penetration enhancer which facilitates drug absorption into the cornea. To prove our hypothesis on the EDU RS 100 trans-cornea facilitating effect produced by surface modification, NLC coated with two different EDU RS 100 concentrations of 0.3% and 0.5% were investigated. The plots obtained demonstrated that with an increase in the EDU RS 100 amount, the Papp of the samples increased slightly from 5.81 to 6.71 cm·min−1, proving that EDU RS 100 facilitates the trans-cornea passage of GEN-NLC by bridging the electrostatic adherence on the NLC surface and the unattached EDU RS 100 polymers do not exhibit a trans-cornea facilitating effect and, thus, could not act as penetration enhancers.
  | Figure 2 Corneal penetration profiles of GEN in different vehicles (mean ± SD, n=3). |
In vivo precorneal retention
Precorneal retention evaluation using Gamma scintigraphy was performed to investigate the retention ability of developed EDU RS 100-GEN-NLC in the precornea area and also to predict its intraocular bioavailability. The radiolabeling efficacy of EDU RS 100-GEN-NLC and bare GEN-NLC were all above 95% (data not shown).
Figure 3A shows the qualitative distribution image of 99mTc-formulations on the cornea surfaces of rabbits, determined by a HAWKEYE SPECT/CT Gamma camera (VG; GE, USA), and their corresponding quantitative remaining activity is plotted in Figure 4. Regions of interest were divided into four parts: left, reference; right-A, cornea/conjunctiva surface; right-B, inner canthus; and right-C, nasolacrymal duct. As observed, EDU RS 100-coated NLC exhibited good spreading and enhanced retention on the cornea during the investigated time period. Specifically, the radioactive spot in the physiological saline (PS) group transported from the pre-ocular regions (A) to the naso-lacrymal duct (C) during rapid elimination, indicated weak adhesion. Up to 10 minutes, less than 20% of the radioactive 99mTc was found to remain on the corneal surface. Regarding the un-EDU RS 100-coated NLC group, the movement of the radioactive spot was restricted to only the inner canthus (B), and the residual activity on the cornea increased to 50%, illustrating the good affinity of lipid NLC for cornea epithelial cells. When instilled with EDU RS 100-GEN-NLC, a stable intensity of radioactivity was observed mainly on the cornea/conjuctiva surface (A) rather than other regions, which proved the ability of EDU RS 100 to extend the retention time. Also, 80% of the radioactivity on the corneal surface could still be detected after 10 minutes, which was approximately 1.6 and 4 times that of the PS and GEN-NLC group, respectively.
From the results obtained, one possible explanation is the interaction between the EDU RS 100 and cornea cells. As mentioned above, the mucus film covering the surface of the cornea and conjunctiva is negatively charged at physiological pH.28 The cationic polymers of the EDU RS 100 coating layer can therefore bind to the negatively charged sialic acid residues of mucins on the eye surface by electrostatic interactions. The parameters describing the precorneal clearance are summarized in Table 5. Statistical analysis demonstrated that compared with the PS solution and bare NLC, the addition of EDU RS 100 significantly increased the AUC (area under the curve)0–10 min 2.39-fold and 1.22-fold, respectively, indicating that more than 20% of the extra GEN remained on the ocular surface due to the EDU RS 100 polymer coating by charge attractions. Meanwhile, the EDU RS 100-GEN-NLC showed a much slower clearance during the initial phase and a higher t1/2 (half-life) compared with the other two formulations, demonstrating a lower rate of drug elimination. These marked differences further confirmed the retention potential of EDU RS 100 on the cornea.
To further prove the retentive mechanism of NLC on the cornea, GEN-NLC with different particle sizes was subjected to precorneal retention investigation (Figure 3B). It was found that there was no significant difference in retentive radio intensity on the cornea surface between NLCs of diameter 100 and 400 nm. When the particle size increased to 1,000 nm or over, more of the samples were detected in the inner canthus (B) and nasolacrimal duct (C), indicating a lower adherence of particles to the cornea surface. Such results about cornea retention imply that the retention mechanism also includes absorption, along with the above electric attraction. The smaller particles allowed a larger specific surface area to absorb on the cornea surface, and were modified by more cationic EDU RS 100 polymers to allow attachment to the cornea.
Ocular irritation
The Draize score was used to evaluate the influence of EDU RS 100 on ocular tissues. No signs of discomfort, including cornea opacity, conjunctiva redness and discharge, appeared (grade less than 3) during the test after installation of EDU RS 100-GEN-NLC for 3 days, showing no observed difference compared with the control group (data not shown).
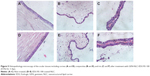
Figure 5 shows the histopathological images of the rabbit eyeballs tested with EDU RS 100-GEN-NLC to visualize its effect on the cell structure and tissue integrity. As shown, no difference in the cell structure and epithelium integrity was observed between the control and EDU RS 100-GEN-NLC-treated eyes. The single-layer basal columnar cells of the cornea were normally packed with tight junction complexes (Figure 5A and D); no abnormality in the size and location of conjunctival lymphoid tissue appeared in all the conjunctivas (Figure 5B and E); and normal levels and integration of polymorphonuclear cells were observed in the iris (Figure 5C and F). These results indicated that there were no signs of inflammation and tissue edema. The histopathological images confirmed that EDU RS 100 produced no ocular irritation compared with untreated eyes. The Draize test scores and the histopathological images demonstrated the excellent ocular tolerance of EDU RS 100-GEN-NLC.
HCE cell cytotoxicity
Based on the results of the ocular irritation test, a further viability assay was carried out to detect any possible adverse effects of lipid materials or EDU RS 100 on cultured human cornea epithelial cell lines. Figure 6 shows the viability of human lens epithelium cells (HEL) cells treated with different GEN-formulations for 4 hours using a gradient concentration. As shown, there was no significant reduction in cell metabolic activity for any of the used blank NLC compared with untreated cells at all concentrations. This result suggests that lipid materials are safe for use as drug carriers. Interestingly, the GEN in solution showed an insignificantly different viability compared with untreated cells, but an observed reduction in viability appeared when cells were incubated with GEN encapsulated in NLC. This suggests that GEN-NLC aggravates the toxic effects of GEN on HCE cells. This is attributed to the affinity of lipid NLC for the cells, which facilitates the entry of drug into cells compared with the drug in solution. When GEN-NLC was further surface-modified with EDU RS 100, the cellular viability was not significantly changed, showing that no toxic effect was produced by the addition of EDU RS 100 to the medium. The EDU RS 100-GEN-NLC showed a viability percentage above 70% at all concentrations ranging from 0 to 100 μg/mL. When EDU RS 100-GEN-NLC was administered directly onto the eyeball, the toxic effect on the epithelial cells could be markedly attenuated compared with the results of the cytotoxicity test, because the cells in the actual ocular tissue environment are more robust than those experimentally incubated ones.
Although investigation of EDU RS 100 polymers as drug carriers has been reported in previous studies, most of these involve EDU RS 100 nanoparticles which were typically produced by incorporating drug into an EDU RS 100 polymer matrix at a preferred drug to polymer ratio (usually 1/10 or less) for drug entrapment.15,27 By modifying EDU RS 100 polymers on the surface of NLC or using other colloid carriers, the polymer used could be reduced to 50 mg in 10 mL samples (EDU RS 100-NLC, 0.1%) in our study (drug/polymer =1/5), together with some biocompatible lipids, which could lead to a lower cytotoxicity and a better ocular tolerance.
Conclusion
The results obtained from our investigations allow us to conclude that after NLCs are surface-modified with EDU RS 100 aqueous suspensions, they exhibit excellent properties as vehicles for ophthalmic application. The EDU RS 100 polymer gives GEN-NLC a cationic charge by uniformly covering the particle surfaces. EDU RS 100-GEN-NLC provides a longer precorneal retention by interacting with the negative mucus, and achieves an improved cornea penetration rate due to the electronic and cell affinitive contribution of EDU RS 100. The modification of EDU RS 100 on the surface of NLC does not produce any irritant effect on ocular tissues and is nontoxic to epithelial cells. Taking these findings into account, we believe that EDU RS 100-coated NLC represents a promising vehicle for ophthalmic applications. Further investigations should focus on the transport of drug carried by improved EDU RS 100-NLC into the aqueous humor through the cornea barrier.
Acknowledgment
This work was financially supported by the National Natural Science Foundation of China (No. 81100654).
Disclosure
The authors report no conflicts of interest in this work.
References
Spalton DJ. Posterior capsular opacification after cataract surgery. Nature. 1999;13:489–492. | ||
Bozukova D, Pagnoulle C, Jérôme R, Jérôme C. Polymers in modern ophthalmic implants – historical background and recent advances. Mater Sci Eng R Rep. 2010;69(6):63–83. | ||
Legler UF, Apple DJ, Assia EI, Bluestein EC, Castaneda VE, Mowbray SL. Inhibition of posterior capsule opacification: the effect of colchicine in a sustained drug delivery system. J Cataract Refract Surg. 1993;19(4):462–470. | ||
Bretton RH, Swearingen A, Kash RL, Cooley R. Use of a polylysine–saporin conjugate to prevent posterior capsule opacification. J Cataract Refract Surg. 1999;25(7):921–929. | ||
Kim SY, Kim JH, Choi JS, Joo CK. Comparison of posterior capsule opacification in rabbits receiving either mitomycin-C or distilled water for sealed-capsule irrigation during cataract surgery. Clin Experiment Ophthalmol. 2007;35(8):755–758. | ||
Totan Y, Yağci R, Erdurmuş M, Bayrak R, Hepşen IF. Cyclosporin effectively inhibits posterior capsule opacification after phacoemulsification in rabbits: a preliminary study. Clin Experiment Ophthalmol. 2008;36(1):62–66. | ||
Müller RH, Radtke M, Wissing SA. Solid lipid nanoparticles (SLN) and nanostructured lipid carriers (NLC) in cosmetic and dermatological preparations. Adv Drug Deliv Rev. 2002;54:131–155. | ||
Liu R, Liu Z, Zhang C, et al. Nanostructured lipid carriers as novel ophthalmic delivery system for mangiferin: improving in vivo ocular bioavailability. J Pharm Sci. 2012;101(10):3833–3844. | ||
Araújo J, Garcia ML, Mallandrich M, et al. Release profile and transscleral permeation of triamcinolone acetonide loaded nanostructured lipid carriers (TA-NLC): in vitro and ex vivo studies. Nanomedicine. 2012;8(6):1034–1041. | ||
Youshia J, Kamel AO, El Shamy A, Mansour S. Design of cationic nanostructured heterolipid matrices for ocular delivery of methazolamide. Int J Nanomedicine. 2012;7:2483–2496. | ||
Zhang W, Prausnitz MR, Edwards A. Model of transient drug diffusion across cornea. J Control Release. 2004;99:241–258. | ||
Zheng W, Sauer D, McGinity JW. Influence of hydroxyethylcellulose on the drug release properties of theophylline pellets coated with Eudragit RS 30D. Eur J Pharm Biopharm. 2005;59:147–154. | ||
Lippold BC, Monells Pagés R. Control and stability of drug release from diffusion pellets coated with the aqueous quaternary polymethacrylate dispersion Eudragit RS 30D. Pharmazie. 2001;56:477–483. | ||
Technical Information Sheets of EUDRAGIT® RL 30D and EUDRAGIT® RS 30D. Available from: http://eudragit.evonik.com/product/eudragit/en/products-services/eudragit-products/sustained-release-formulations/rs-30-d/Pages/default.aspx. Accessed August 19, 2014. | ||
Das S, Suresh PK. Nanosuspension: a new vehicle for the improvement of the delivery of drugs to the ocular surface. Application to amphotericin B. Nanomedicine. 2011;7:242–247. | ||
Pignatello R, Bucolo C, Spedalieri G, Maltese A, Puglisi G. Flurbiprofen loaded acrylate polymer nanosuspensions for ophthalmic application. Biomaterials. 2002;23:3247–3255. | ||
Li N, Zhuang C, Wang M, Sun X, Nie S, Pan W. Liposome coated with low molecular weight chitosan and its potential use in ocular drug delivery. Int J Pharm. 2009;379(1):131–138. | ||
Huang R, Shi F, Lei T, Song Y, Hughes CL, Liu G. Effect of the isoflavone genistein against galactose-induced cataracts in rats. Exp Biol Med (Maywood). 2007;232(1):118–125. | ||
Zhang W, Li X, Ye T, et al. Design, characterization, and in vitro cellular inhibition and uptake of optimized genistein-loaded NLC for the prevention of posterior capsular opacification using response surface methodology. Int J Pharm. 2013;454(1):354–366. | ||
Luo Q, Zhao J, Zhang X, et al. Nanostructured lipid carrier (NLC) coated with Chitosan Oligosaccharides and its potential use in ocular drug delivery system. Int J Pharm. 2011;403(1):185–191. | ||
Tian B, Luo Q, Song S, et al. Novel surface-modified nanostructured lipid carriers with partially deacetylated water-soluble chitosan for efficient ocular delivery. J Pharm Sci. 2012;101(3):1040–1049. | ||
Tian BC, Zhang WJ, Xu HM, et al. Further investigation of nanostructured lipid carriers as an ocular delivery system: in vivo transcorneal mechanism and in vitro release study. Colloids Surf B Biointerfaces. 2013;102:251–256. | ||
Araújo J, Garcia ML, Mallandrich M, Souto EB, Calpena AC. Release profile and transscleral permeation of triamcinolone acetonide loaded nanostructured lipid carriers (TA-NLC): in vitro and ex vivo studies. Nanomedicine. 2012;8(6):1034–1041. | ||
Camber O. An in vitro model for determination of drug permeability through the cornea. Acta Pharm Suec. 1985;22:335–342. | ||
Jain D, Carvalho E, Banthia AK, Banerjee R. Development of polyvinyl alcohol-gelatin membranes for antibiotic delivery in the eye. Drug Dev Ind Pharm. 2011;37(2):167–177. | ||
Jain D, Carvalho E, Banerjee R. Biodegradable hybrid polymeric membranes for ocular drug delivery. Acta Biomaterialia. 2010;6(4): 1370–1379. | ||
Tang J, Xu N, Ji H, Liu H, Wang Z, Wu L. Eudragit nanoparticles containing genistein: formulation, development, and bioavailability assessment. Int J Nanomedicine. 2011;6:2429–2435. | ||
Ludwig A. The use of mucoadhesive polymers in ocular drug delivery. Drug Deliv. 2005;57:1595–1639. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.