Back to Journals » International Medical Case Reports Journal » Volume 13
Macular Hole Formation in Eye After Cryotherapy and Intravitreal Bevacizumab Treatment for Vasoproliferative Tumor
Authors Sato A, Fukui E, Ohta K
Received 9 June 2020
Accepted for publication 31 July 2020
Published 14 September 2020 Volume 2020:13 Pages 419—423
DOI https://doi.org/10.2147/IMCRJ.S266986
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Scott Fraser
Atsuko Sato, Emi Fukui, Kouichi Ohta
Department of Ophthalmology, Matsumoto Dental University, Shiojiri, Nagano, Japan
Correspondence: Atsuko Sato
Department of Ophthalmology, Matsumoto Dental University, 1780 Gobara, Hirooka, Shiojiri, Nagano 399-0781, Japan
Tel/Fax +81-263-51-2210
Email [email protected]
Purpose: To report a case of a full-thickness macular hole (MH) that developed after cryotherapy and intravitreal bevacizumab injection (IVB) to treat a retinal vasoproliferative tumor (VPT).
Methods: Case report of a man with a retinal VPT.
Results: A 64-year-old Japanese man complained of blurred vision in his right eye. At the initial examination, his best-corrected visual acuity (BCVA) was 20/25 in the right eye and 20/20 in the left eye. Ophthalmoscopy showed a VPT in the lower peripheral retina of the right eye. An exudative retinal detachment and hard exudates were seen around the tumor. Cryotherapy and intravitreal injections of bevacizumab (IVB) were performed. Although the exudative changes were reduced, a MH developed two months after the initial IVB treatment. He underwent 25-gauge pars plana vitrectomy, and the MH was closed. His postoperative BCVA was 20/32 and the VPT was inactive. The reduced BCVA was due to damage of the outer retinal layers.
Conclusion: Our findings indicate that cryotherapy and IVB are effective treatments for VPT although the possibility of developing a MH should be considered.
Keywords: intravitreal bevacizumab injection, macular hole, optical coherence tomography, vasoproliferative tumor, vitrectomy
Introduction
A vasoproliferative tumor (VPT) is a benign retinal vascular tumor.1–3 The tumor is characterized by yellowish-pink lesions and can be classified as a primary or a secondary tumor. Shields reported that primary VPTs occurred in 80% and secondary in 20% of the patients.2 Secondary VPTs occur in patients with retinitis pigmentosa, pars planitis, Coats’ disease, and other retinal disorders.4,5 Many complications are associated with VPT including intraocular hemorrhages, macular edema, macular exudation, epiretinal membrane (ERM), and exudative retinal detachment, but macular hole (MH) formation is rare.6 The purpose of this report is to present our findings in a case of VPT that developed a MH after treatment.
Case Presentation
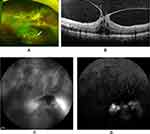
A 64-year-old Japanese man complained of blurred vision in his right eye in January 2015. At his initial examination, his best-corrected visual acuity (BCVA) was 20/25 in the right eye and 20/20 in the left eye. Ophthalmoscopy showed an approximately 3-disc diameter (DD) yellowish-pink elevated lesion with retinal hemorrhages in the lower peripheral retina. There was vitreomacular traction in his right eye (Figure 1A and B). A shallow exudative retinal detachment was also present in the inferior quadrant, and hard exudates were seen around the tumor. In addition, hard exudates had also accumulated around the inferior vascular arcade and optic disc.
The differential diagnosis of retinal tumors includes secondary tumor such as von Hippel-Lindau (VHL) syndrome and other systemic hemangiomas. In VHL syndrome, the tumors are usually multiple and bilateral which were not present in our case. Fluorescein angiography (FA) and indocyanine green angiography (ICGA) were performed. A dilated feeder vessel was not seen as is usually seen in eyes with VHL syndrome. FA showed early leakage from the tumor (Figure 1C), and ICGA showed no hypofluorescence or late granular hyperfluorescence which are characteristics of metastatic choroidal tumors (Figure 1D). The patient did not have any family history for VHL syndrome or other systemic hamartomas. General systemic screening by MRI and PET-CT showed the presence of renal cancer and thyroid cancer. In addition, no other tumors such as malignant melanomas and metastatic tumors were detected.
With time, the hard exudates extended to macula leading to an increase in the vitreomacular traction. The BCVA was reduced to 20/63. Before the surgery for renal cancer in April 2015, he was diagnosed with primary VPT, and transscleral cryotherapy and an intravitreal injection of bevacizumab (IVB) were performed in March 2015. However, the hard exudates remained and the subretinal fluid (SRF) increased after these treatments. A secondary IVB was performed one month later prior to the surgery for renal cancer at another university hospital. One month after the second IVB, the vitreomacular traction was released, and the level of subfoveal fluid was decreased.
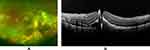
However, a full-thickness MH developed with exudates in May 2015, but he did not complain of a further decrease in his vision (Figure 2A and B). Although pars plana vitrectomy for the MH was considered, his highest priority was surgery for another thyroid tumor which was scheduled for August 2015 in another hospital.
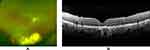
The hard exudates and subretinal fluid were resolved after the eighth IVB injection in November 2015. (Figure 3A and B). The size of the VPT was also decreased after the IVB injection. The BCVA remained 20/63. The ellipsoid zone (EZ) and external limiting membrane (ELM) were not detected over the MH. After successful resections of the 2 cancerous lesions, he decided to undergo 25-gauge pars plana vitrectomy combined with cataract surgery and intraocular lens implantation in February 2016. Internal limiting membrane peeling and gas tamponade with 25% sulfur hexafluoride were performed. After the surgery, the MH was completely closed (Figure 4A and B). His BCVA was still reduced to 20/63 at 1 year after the vitrectomy. The reduced BCVA was probably related to the 8 months during which the MH remained open. A disruption of the ellipsoid zone (EZ) and thinning of the outer nuclear layer (ONL) were observed beyond the edge of the original MH. However, his BCVA gradually improved, and it was 20/32 at 4 years after the MH surgery. Both the renal cancer and thyroid tumor were successfully excised without metastasis. Both surgeons for the renal and the thyroid carcinomas stated that there was no association between the renal and thyroid tumor and the VPT. Unfortunately, genetic testing was not performed.
Discussion and Conclusions
It has been reported that VPTs can lead to severe vision reduction because of ocular complications such as intraocular hemorrhages (35%), macular edema (32%), macular exudation (23%), and ERM formation (20%).2 However, the formation of a MH is a rare complication in eyes with a VPT.6
The various treatments for VPT include laser photocoagulation,7 cryotherapy,3,8 photodynamic therapy,9 and intravitreal injections of anti-vascular endothelial growth factor (VEGF) including bevacizumab.10 Cryotherapy is also known to be an effective treatment for VPT, and it has been reported that the tension created by a VPT-related epiretinal membrane (ERM) was released in 63% of the cases after cryotherapy.8 In our case, the cryotherapy released the vitreoretinal traction but a MH still developed.
The formation of a MH has been reported in eyes with VPT6 and also eyes with von VHL syndrome.11,12 In our case, a MH developed after cryotherapy and two IVB injections. The IVB could have been the cause of the MH because there have been several reports of MH formation after anti-VEGF therapy.13–16 Other possible mechanisms for the MH formation include an increase in the anterior-posterior transvitreal traction, vitreous incarceration at the injection site, and anti-VEGF-induced contraction of the target of the disease. In our case, the exudative changes were reduced after the combined therapy which could then increase the degree of vitreoretinal traction. It has also been reported that MH can form after an air injection and cryotherapy for a retinal detachment.17,18 We suggest that the MH formation in our case was related to the cryotherapy and/or anti-VEGF therapy rather than the VPT.
The MH was successfully closed by the initial vitrectomy with ILM peeling. The postoperative poorer BCVA may be related to the delayed MH surgery and also the disruption of the EZ and thinning of the ONL beyond the original MH edge by the exudates from the VPT. Although subretinal exudations can extend rapidly to the macula, they have been absorbed after IVB treatment. The outer retinal layer was damaged by these exudates in our case. In the end, the original VPT was completely scarred without any recurrence for 4 years after the treatments.
In conclusion, a MH developed after cryotherapy and IVB injections in a case of VPT. Our findings indicate that cryotherapy with IVB is effective in treating VPT, although ophthalmologists should be aware of the risk of MH formation.
Ethics Approval and Consent to Participate
Ethics approval was provided by the Institutional Review Board of the Matsumoto Dental University, Shiojiri, Japan. The patient provided a written informed consent for the case details and images to be published.
Acknowledgments
We thank Professor Emeritus Duco Hamasaki of the Bascom Palmer Eye Institute for discussions and editing the manuscript.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Shields JA, Decker WL, Sanborn GE, Augsburger JJ, Goldberg RE. Presumed acquired retinal hemangiomas. Ophthalmology. 1983;90(11):1292–1300. doi:10.1016/S0161-6420(83)34389-X
2. Shields CL, Kaliki S, Al-Dahmash S, et al. Retinal vasoproliferative tumors: comparative clinical features of primary vs secondary tumors in 334 cases. JAMA Ophthalmol. 2013;131(3):328–334. doi:10.1001/2013.jamaophthalmol.524
3. Jain K, Berger AR, Yucil YH, McGowan HD. Vasoproliferative tumor of the retina. Eye (Lond). 2003;17(3):364–368. doi:10.1038/sj.eye.6700311
4. Shields CL, Shields JA, Barrett J, De Potter P. Vasoproliferative tumors of the ocular fundus. Classification and clinical manifestations in 103 patients. Arch Ophthalmol. 1995;113(5):615–623. doi:10.1001/archopht.1995.01100050083035
5. Mori K, Ohta K, Murata T. Vasoproliferative tumors of the retina secondary to ocular toxocariasis. Can J Ophthalmol. 2007;42(5):758–759. doi:10.3129/i07-137
6. Clare G, Yorston D, Charteris DH, Aylward GW. Successful treatment of macular holes associated with peripheral retinal vascular tumours. Eye (Lond). 2007;21(3):419–420. doi:10.1038/sj.eye.6702423
7. Krivosic V, Massin P, Desjardins L, Le Hoang P, Tadayoni R, Gaudric A. Management of idiopathic retinal vasoproliferative tumors by slit-lamp laser or endolaser photocoagulation. Am J Ophthalmol. 2014;158(1):154–161. doi:10.1016/j.ajo.2014.03.005
8. Manjandavida FP, Shields CL, Kaliki S, Shields JA. Cryotherapy-induced release of epiretinal membrane associated with retinal vasoproliferative tumor. Retina. 2014;34(8):1644–1650. doi:10.1097/IAE.0000000000000137
9. Blasi MA, Scupola A, Tiberti AC, Sasso P, Balestrazzi E. Photodynamic therapy for vasoproliferative retinal tumors. Retina. 2006;26(4):404–409.
10. Kenawy N, Groenwald C, Damato B. Treatment of a vasoproliferative tumour with intravitreal bevacizumab (Avastin). Eye. 2007;21(6):893–894. doi:10.1038/sj.eye.6702782
11. Loewestein JI. Bilateral macular holes in von Hippel-Lindau disease. Arch Ophthalmol. 1995;113(2):143–144. doi:10.1001/archopht.1995.01100020021016
12. Inoue M, Yamazaki K, Shinoda K, et al. A clinicopathologic case report on macular hole associated with von Hippel-Lindau disease: a novel ultrastructural finding of wormlike, wavy tangles of filaments. Graefes Arch Clin Exp Ophthalmol. 2004;242(10):881–886. doi:10.1007/s00417-004-0908-9
13. Querques G, Souied EH, Soubrane G. Macular hole following intravitreal ranibizumab injection for choroidal neovascular membrane caused by age-related macular degeneration. Acta Ophthalmol. 2009;87(2):235–237. doi:10.1111/j.1755-3768.2008.01226.x
14. Grigoropoulos V, Emfietzoglou J, Nikolaidis P, Theodossiadis G, Theodossiadis P. Full-thickness macular hole after intravitreal injection of ranibizumab in a patient with retinal pigment epithelium detachment and tear. Eur J Ophthalmol. 2010;20(2):469–472. doi:10.1177/112067211002000235
15. Moisseiev E, Goldstein M, Loewenstein A, Moisseiev J. Macular hole following Intravitreal bevacizumab injection in choroidal neovascularization caused by age-related macular degeneration. Case Rep Ophthalmol. 2010;1(1):36–41.
16. Miura M, Iwasaki T, Goto H. Macular hole formation after intravitreal bevacizumab administration in a patient with myopic choroidal neovascularization. Retin Cases Brief Rep. 2011;5(2):149–152. doi:10.1097/ICB.0b013e3181cd1da1
17. Hilton GF, Tornambe PE, Brinton DA, et al. The complication of pneumatic retinopexy. Trans Am Ophthalmol Soc. 1990;88:191–210.
18. Boscia F, Recchimurzo N, Cardascia N, Sborgia L, Furino C, Sborgia C. Macular hole following conventional repair of bullous retinal detachment using air injection (D-ACE procedure). Eur J Ophthalmol. 2004;14(6):572–574. doi:10.1177/112067210401400621
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.