Back to Journals » Clinical Ophthalmology » Volume 8
Long-term visual outcomes after Crystalens® HD intraocular lens implantation
Authors Karavitaki A, Pallikaris I, Panagopoulou S, Kounis G, Kontadakis G , Kymionis G
Received 3 November 2013
Accepted for publication 13 December 2013
Published 22 May 2014 Volume 2014:8 Pages 937—943
DOI https://doi.org/10.2147/OPTH.S57009
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Alexandra E Karavitaki, Ioannis G Pallikaris, Sophia I Panagopoulou, George A Kounis, George Kontadakis, George D Kymionis
Institute of Vision and Optics, University of Crete, Crete, Greece
Purpose: Our purpose was to evaluate the long-term visual outcomes after bilateral implantation of Crystalens® HD (Bausch and Lomb, Inc.) accommodative intraocular lenses.
Methods: In this study, 25 patients (50 eyes) who underwent cataract surgery and bilateral Crystalens HD accommodative intraocular lens implantation were included. The Crystalens HD lens was implanted in the bag in both eyes, without any intra- or postoperative complications. The visual parameters measured before and after surgery were: uncorrected distance visual acuity, corrected distance visual acuity (CDVA) (using the Early Diabetic Retinopathy Study [EDTRS] logMAR charts), uncorrected intermediate visual acuity, and uncorrected near visual acuity using a Birkhäuser reading chart, at a distance of 66 cm and 33 cm for intermediate and near vision, respectively, directly illuminated by a lamp of 60 watts. After surgery, the patients were evaluated at 1, 3, 6, 12, 24, and 36 months.
Results: The mean age was 68.70±7.1 (range: 54 to 83) years. The mean follow-up was 42.72±0.49 (range: 42.33 to 43.27) months. The uncorrected distance visual acuity improved from 0.56±0.41 (range: 0 to 2) preoperatively to 0.19±0.13 (range: 0 to 0.38) (logMAR scale) at the last follow-up visit, and the CDVA improved from 0.17±0.18 (range: 0 to 0.7) preoperatively to 0.05±0.05 (range: −0.02 to 0.22) at the last follow-up visit. No eyes lost lines of CDVA during the follow-up period, while 54% of patients gained one or more lines of CDVA. The uncorrected intermediate and near visual acuity was J2 or better in 71% and 69% of our patients, respectively.
Conclusion: Crystalens HD implantation seems to provide an improvement in visual acuity for far, intermediate, and near distances.
Keywords: presbyopia, accommodation
Introduction
The evolution in phacoemulsification technologies and intraocular lenses implantation expertise has turned cataract surgery into an extremely safe and refined procedure.1 An increase in patients’ expectations, in terms of visual rehabilitation and independency from spectacles for both far and near vision, has shifted efforts toward optimizing the refractive component.
Different approaches have been proposed in order to overcome the accommodative incompetence of presbyopic patients. Remodeling of the sclera,2–4 presbyopic laser in situ keratomileusis (presbyLASIK),5–7 corneal inlays,8,9 monovision techniques,10 and crystalline lens substitution with multifocal11,12 or accommodative intraocular lens (IOL) typify some of the most popular proposals.
Pseudoaccommodation is the result of an increase in the depth of focus of the eye’s optical system and depends on different phenomenon, such as pupil size, myopic astigmatism, and corneal multifocality.13 Hinge optic IOLs attempt to induce pseudophakic accommodation by shifting the anterior direction of the IOL during the contraction of the ciliary muscle and increasing the vitreous pressure, resulting in an amplification of effective lens power.14
Dogru et al presented a study with the 1-year results of a 1 CU accommodating IOL (HumanOptics AG, Erlangen, Germany). During this study, 77.2% of the eyes implanted with the IOL had an uncorrected distance visual acuity (UDVA) of 20/40 or better and a corrected distance visual acuity (CDVA) of 20/25 or better at the 1-year follow-up visit. Regarding the amplitude of accommodation and near visual acuity, the eyes with the 1 CU IOL showed an improvement starting from the first week after surgery and maximal values at 3 and 6 months after surgery, which declined during the 12-month period. In the study, the eyes implanted with the 1 CU IOL showed an increasing rate of anterior and posterior capsule opacity, which achieved 34% at 6 months and increased to 86% at 12 months. According to the authors, this could be attributed to the absence of a square-edged design of the lens or to the displacement of the optic away from the posterior capsule during the accommodating effort.15
In this study, we prospectively evaluated the visual outcomes for far, intermediate, and near distances, after bilateral implantation of an accommodative IOL (Crystalens® HD; Bausch and Lomb, Inc., Rochester, NY, USA).
Patients and methods
Twenty-five patients (50 eyes), 12 males and 13 females, who underwent cataract surgery were enrolled. The exclusion criteria were: corneal astigmatism more than 1.25 D, corneal dystrophies, glaucoma, retinal conditions or predisposition to retinal conditions, and trauma. None of the patients were using any medication as topical therapy before surgery.
Crystalens HD characteristics
The Crystalens HD is a multipiece posterior chamber intraocular lens developed for the treatment of cataract. The optic plate, consisting of a third generation silicone material (Biosil), has a 5.0 mm diameter and biconvex squared edge. The overall diameter is between 11.5−12.0 mm. The IOL has a 1.5 mm diameter modification in the center, resulting in increased negative spherical aberration. Moving toward the periphery of the lens, there is a gradual increase in positive spherical aberration. The haptics of the lens are designed to allow movement or changes in the position and shape of the optic.16,17
Preoperative examination
The preoperative data obtained from the case records included patient age, sex, slit-lamp biomicroscopy, intraocular pressure, fundoscopy, and endothelial cell count. The IOLMaster® 500 (Carl Zeiss Meditec, Jena, Germany) was used for IOL power calculations. The visual parameters measured were: UDVA and CDVA, using the Early Diabetic Retinopathy Study (EDTRS) logMAR charts (Precision Vision, La Salle, IL, USA), and uncorrected intermediate visual acuity (UIVA) and uncorrected near visual acuity (UNVA), using a Birkhäuser reading chart, at a distance of 66 cm and 33 cm for intermediate and near vision, respectively, directly illuminated by a lamp of 60 watts.
A wavefront analyzing system (WASCA Analyzer; Carl Zeiss Meditec) was used in conjunction with a Badal optometer, to allow the measurement of total higher-order eye aberrations, for far and near (30 cm).18
All patients were informed of the risks and benefits prior to cataract surgery, and they provided a written informed consent in accordance with the Declaration of Helsinki and the institutional guidelines. The study was approved by the ethics committee (protocol number 71/71/24-06-2009) of the University Hospital (University Hospital of Heraklion, Crete, Greece).
Surgical technique
All surgeries were performed under sterile conditions by the same experienced surgeon (IGP), using a standard phacoemulsification technique. Local anesthetic drops were used. A clear corneal incision of 2.8 mm was made, and an anterior curvilinear capsulorhexis of 5.5 mm was performed with extreme care, for an exact central position of the lens. Phacoemulsification was performed, with thorough cortical removing and meticulous cleaning of the posterior capsule and anterior capsular leaflets. The Crystalens HD IOL was implanted using the specific single use injector device (Crystalsert Crystalens Delivery System) that allows the haptics to fold inwards toward the optic during folding and insertion.
After surgery all the patients received antibiotic/corticosteroid (tobramycin/dexamethasone) drops (TobraDex®; Alcon Laboratories, Inc., Fort Worth, TX, USA) four times daily, with a tapering regime of a week. All eyes also received cyclopentolate 1% (Cyclogyl®; Alcon Laboratories, Inc.) twice daily for 5 days in order to assure the posterior lens position.19
Postoperative examination
Postoperatively, the patients were evaluated at 1, 3, 6, 12, 24, and 36 months. The same experienced investigator (AEK) performed all the postoperative examinations, using the same investigation protocol that was used for the preoperative evaluation.
The data were gathered, analyzed, and depicted using of an ophthalmic database (G & N Electronic and Medical Solutions, Crete, Greece) (Supplementary material). The statistical analysis included a normality check using the Kolmogorov−Smirnov test and Student’s t-test, to obtain statistically significant differences at the level of 0.05. The Wilcoxon Rank Sum test was used for examining statistical significance in pair tests. The Kruskal−Wallis test was used to examine stability with time, for parameters varying with time.
Results
The mean follow-up after implantation was 42.72±0.49 (42.33 to 43.27) months. The UDVA showed a statistically significant (P<0.0001) improvement, from 0.56±0.41 (range: 0 to 2) to 0.19±0.13 (range: 0 to 0.38) (logMAR scale). The CDVA also showed statistically significant (P=0.006) improvement, from 0.17±0.18 (range: 0 to 0.7) to 0.05±0.05 (range: −0.02 to 0.22). No eye lost lines of CDVA during the follow-up period, while 54% of the patients gained one or more lines of CDVA, confirming the safety of the technique (Figure 1). At the last follow-up visit postoperatively, the UIVA was J2 or better in 71% of our patients, as measured with Birkhäuser reading charts at a distance of 66 cm and under photopic conditions (Figure 2); at the last follow-up postoperatively, the UNVA was J2 or better in 69% of our patients, again, as measured with Birkhäuser reading charts at the distance of 33 cm and under photopic conditions (Figure 3).
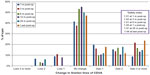  | Figure 1 Change in corrected distance visual acuity (CDVA) bar graph. |
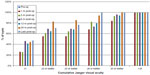  | Figure 2 Cumulative uncorrected intermediate visual acuity (UIVA). |
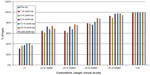  | Figure 3 Cumulative uncorrected near visual acuity (UNVA). |
Zernike coefficients of third and fourth order were obtained (without pharmacologically induced pupil dilation) at 3.5 mm pupil diameter for far and near targets (30 cm), at the last follow-up. All higher-order aberrations were reported according to the OSA notation.20 The third- and fourth-order higher-order aberrations for far and near target are presented in Figure 4. The stability of postoperative UDVA and CDVA are presented in Figure 5.
  | Figure 4 Total eye aberrations of third and fourth order at far and near, at 3.5 mm pupil diameter. |
The mean change in sphere, as measured by the WASCA for far and near targets, was −0.45±1.07 D. The spherical aberration Zernike coefficient mean values ± standard deviation (SD) for far and near respectively changed from 0.06±0.04 μm (range: 0.02 to 0.13) to 0.03±0.05 μm (range: −0.05 to 0.10). The difference in spherical aberration between far and near showed statistical significance in the paired Student’s t-test (P=0.003). There was no correlation between the near visual acuity achieved and the level of aberration.
There were no intra- or postoperative complications, although neodymium-doped yttrium aluminum garnet (Nd:Yag) laser capsulotomy was performed in 12 patients due to posterior capsule opacification, 1 year after surgery.
Discussion
Visual rehabilitation, in terms of the restoration of far vision and near vision, is the goal of modern cataract surgery. In recent decades, different efforts have been made to overcome the challenge of reversing presbyopia.
Accommodative IOL technology is considered to be one of the most promising techniques, providing near vision advantage. The accommodative IOLs have been designed to mimic the physiologic mechanism of accommodation and hence are not subject to the optical side effects of multifocal IOLs.14
The Crystalens HD lens, built on the heritage of Crystalens IOLs, was designed to provide a single focus to the retina. The haptics of the lens are designed to permit changes in the position of the optic.16,17 In this study, the patients presented an improvement in CDVA after the Crystalens IOL implantation, which remained stable during the follow-up period. Furthermore, the patients gained functional intermediate and near vision.”
In this case series, the spherical aberration changed toward more negative values at the near target, giving a greater depth of focus and providing pseudoaccommodation.21 The change of spherical aberration toward more negative values can be observed in physiological accommodation as well.22 This change in spherical aberration, combined with coma and trefoil-like aberrations, seems to support near vision. The total eye wavefront analysis for far and near accommodative stimulus showed mainly a change in spherical aberration. The mean total spherical aberration at far for a 3.5 mm pupil was 0.06 μm (range: 0.02 to 0.13), while Ramon et al, using a Ray tracing wavefront system, have reported for the HD500, mean values for internal spherical aberration at 5 mm pupil and far object viewing to be 0.13+/−0.63 μm.23
Kim et al in an objective in-vitro evaluation of the lens aberrations have found that the Crystalens HD500 had a spherical aberration of 0.01 microns with a 5.0 mm pupil and approximately −0.04 at a 3 mm pupil.24 Apparently the lens design has the effect of additionally changing the spherical aberration toward negative values when miosis occurs during accommodation. The results from our study cannot easily be compared with other studies that have assessed aberration with the Crystalens as different pupil sizes and wavefront measurement technologies were used. In our study, the in vivo measurements of total eye spherical aberration, calculated at 3.5 mm for both far and near, showed a shift toward more negative values for the near target, and thus supporting near vision. Both effects, the pupil size-related and far/near-related change in spherical aberration, seem to contribute to the satisfactory near vision performance.
Detorakis et al presented that the horizontal angle aperture, in eyes implanted with the Crystalens HD IOL, measured with ultrasound biomicroscopy (UBM) is significantly higher compared with the vertical angle aperture and that may affect the overall quality of visual performance.25 In this study, the anterior chamber angle aperture was measured by UBM and optical coherence tomography (OCT) along the vertical and the horizontal meridian. The difference between the two meridians in the angle aperture was found to be statistically significant. The study may provide evidence on the potential action of IOL haptics in the angle aperture of an IOL implanted in the bag. Regarding Crystalens haptics are posteriorly angulated and in the patients enrolled, the IOL haptics were implanted with an orientation along the horizontal axis. The wider angle along the horizontal meridian measured may be attributed to a mechanical effect induced by the IOL haptics. The wider angle aperture along the horizontal axis could imply that the ciliary body may also be posteriorly inclined along the same axis, and that could potentially cause uneven distribution of generated forces during ciliary muscle contraction between the horizontal and vertical axes. In the same study, there was no statistically significant correlation between far and near vision and angle aperture along horizontal or vertical meridian.
To our knowledge, this is the first study presenting long-term visual outcomes after Crystalens HD lens implantation. No patient cited any discomfort concerning visual function throughout the postoperative period. The Crystalens HD IOL could be considered a solution for patients requiring good far, near, and intermediate vision without spectacle dependency.
However, the presence of posterior capsular opacification in a number of our patients and the need for Nd:Yag capsulotomy could have affected the performance of the IOL. Another limitation of this study could have been the use of the Birkhäuser reading chart for the evaluation of near and intermediate visual acuity. Nevertheless, longer follow-up and a larger series of patients are necessary, in order to confirm our results.
Disclosure
The authors report no conflicts of interest in this work.
References
Lichtinger A, Rootman DS. Intraocular lenses for presbyopia correction: past, present, and future. Curr Opin Ophthalmol. 2012;23(1):40–46. | |
Schachar RA.Theoretical basis for the scleral expansion band procedure for surgical reversal of presbyopia [SRP]. Compr Ther. 2001;27:39–46. | |
Malecaze FJ, Gazagne CS, Tarroux MC, Gorrand JM. Scleral expansion bands for presbyopia. Ophthalmology. 2001; 108:2165–2171. | |
Qazi MA, Pepose JS, Shuster JJ. Implantation of scleral expansion band segments for the treatment of presbyopia. Am J Ophthalmol. 2002;134:808–815. | |
Epstein RL, Gurgos MA. Presbyopia treatment by monocular peripheral presbyLASIK. J Refract Surg. 2009; 25:516–523. | |
Illueca C, Alio JL, Mas D, et al. Pseudoaccommodation and visual acuity with Technovision presbyLASIK and a theoretical simulated Array multifocal intraocular lens. J Refract Surg. 2008;24:344–349. | |
Alio JL, Chaubard JJ, Caliz A, et al. Correction of presbyopia by technovision central multifocal LASIK (presbyLASIK). J Refract Surg. 2006;22:453–460. | |
Limnopoulou AN, Bouzoukis DI, Kymionis GD, et al. Visual outcomes and safety of a refractive corneal inlay for presbyopia using femtosecond laser. J Refract Surg. 2013;29(1):12–18. | |
Bouzoukis DI, Kymionis GD, Panagopoulou SI, et al. Visual outcomes and safety of a small diameter intrastromal refractive inlay for the corneal compensation of presbyopia. J Refract Surg. 2012;28(3):168–173. | |
Olson RJ, Werner L, Mamalis N, Cionni R. New intraocular lens technology, Am J Ophthalmol. 2005;140;709–716. | |
Versteeg FF. Multifocal IOLs for presbyopia. J Cataract Refract Surg. 2005;31(7):1266. | |
Bellucci R. Multifocal intraocular lenses. Curr Opin Ophthalmol. 2005;16(1):33–37. | |
Dick HB. Accommodative intraocular lenses: current status. Curr Opin Ophthalmol. 2005;16(1):8–26. | |
Cumming JS, Kammann J Experience with an accommodating IOL. Cataract Refract Surg. 1996;22(8):1001. | |
Dogru M, Honda R, Omoto M et al. Early visual results with the 1CU accommodating intraocular lens. J Cataract Refract Surg. 2005;31(5):895–902. | |
Patel S, Alió JL, Feinbaum C. Comparison of Acri. Smart multifocal IOL, crystalens AT-45 accommodative IOL, and Technovision presbyLASIK for correcting presbyopia. J Refract Surg. 2008;24(3):294–299. | |
Pepose JS, Qazi MA, Davies J et al. Visual performance of patients with bilateral vs combination Crystalens, ReZoom, and ReSTOR intraocular lens implants. Am J Ophthalmol. 2007;144(3):347–357. | |
Plainis S, Ginis HS, Pallikaris A. The effect of ocular aberrations on steady-state errors of accommodative response. J Vis. 2005;5(5):466–477. | |
Hantera MM, Hamed AM, Fekry Y, Shoheib EA. Initial experience with an accommodating intraocular lens: controlled prospective study. J Cataract Refract Surg. 2010;36(7):1167–1172. | |
Thibos LN, Applegate RA, Schwiegerling JT, Webb R. Standards for reporting the optical aberrations of eyes; VSIA Standards Taskforce Members. J Refract Surg. 2002; 18:S652–S660. | |
Wang L, Koch DD. Custom optimization of intraocular lens asphericity. J Cataract Refract Surg. 2007; 33(10):1713–1720. | |
Lopez-Gil N, Fernandez- Sanchez V. The change of spherical aberration during accommodation and its effect on the accommodation response. J Vis. 2010;10(13):12. | |
Ramón ML, Piñero DP, Blanes-Mompó FJ, Pérez-Cambrodí RJ. Clinical and quality of life data correlation with a single-optic accommodating intraocular lens. J Optom. 2013;6:25–35 | |
Kim MJ, Zheleznyak L, MacRae S, Tchah H, Yoon G. Objective evaluation of through-focus optical performance of presbyopia-correcting intraocular lenses using an optical bench system. J Cataract Refract Surg. 2011;37:1305–1312 | |
Detorakis ET, Karavitaki A, Stojanovic N, Kontadakis G, Pallikaris IG. Anterior chamber angle evaluation with ultrasound biomicroscopy and optical coherence tomography in eyes implanted with a Crystalens. Int Ophthalmol. October 29, 2013. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

