Back to Journals » Clinical Interventions in Aging » Volume 10
Is oxycodone/naloxone effective and safe in managing chronic pain of a fragile elderly patient with multiple skin ulcers of the lower limbs? A case report
Authors Guerriero F, Maurizi N, Francis M, Sgarlata C, Ricevuti G, Rondanelli M, Perna S, Rollone M
Received 16 March 2015
Accepted for publication 10 May 2015
Published 10 August 2015 Volume 2015:10 Pages 1283—1287
DOI https://doi.org/10.2147/CIA.S84711
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Richard Walker
Fabio Guerriero,1,2 Niccolo Maurizi,1 Matthew Francis,1 Carmelo Sgarlata,1 Giovanni Ricevuti,1,2 Mariangela Rondanelli,2,3 Simone Perna,2,3 Marco Rollone2
1Department of Internal Medicine and Medical Therapy, Section of Geriatrics, University of Pavia, 2Azienda di Servizi alla Persona, Istituto di Cura Santa Margherita of Pavia, 3Department of Public Health, Experimental and Forensic Medicine, Section of Human Nutrition, University of Pavia, Pavia, Italy
Abstract: Skin ulcers are a common issue in the elderly, as physiological loss of skin elasticity, alterations in microcirculation, and concomitant chronic diseases typically occur in advanced age, thereby predisposing to these painful lesions. Wound-related pain is often associated with skin ulcers and negatively impacts both the patient’s quality of life and, indirectly, wound healing. Pain management is an ongoing issue in the elderly, and remains underestimated and undertreated in this fragile population. Recent guidelines suggest the use of opioids as the frontline treatment of moderate and severe pain in nononcological pain in the elderly. However, due to the concerns of adverse reactions, drug interactions, and addiction, clinicians frequently hesitate to prescribe opioids. This case report describes an elderly diabetic patient with multiple ulcers of the lower limbs suffering wound-related pain. In our report, oxycodone/naloxone has proved to be an effective and safe drug, providing pain relief as well as increased compliance when redressing wounds and faster healing compared to that in similar patients. Our case provides anecdotal evidence, supported by other studies, to justify future, larger studies on chronic pain using this therapy.
Keywords: chronic pain, skin ulcers, elderly, opioids, oxycodone, naloxone
Introduction
Skin ulcers are secondary lesions characterized by chronic damage to the dermis, and subsequently the underlying fascia, with little or no tendency to spontaneous healing. Skin ulcers may represent the clinical manifestation of the interaction between numerous systemic and local factors, with various pathophysiological mechanisms present contemporarily. With increased age, the prevalence of chronic skin ulcers tends to increase, involving especially the geriatric population. Several pathophysiological mechanisms related to aging underlie this phenomenon, such as the physiological loss of trophic dermo-epidermal elasticity and concomitant alterations in skin microcirculation in addition to the frequent presence of comorbidities that could adversely affect the development of skin ulcers.1
Pain associated with chronic skin ulcers is a very significant factor that should be highlighted. Negative outcomes in terms of quality of life and wound healing are associated with diminished cooperation of the patient during wound redressing. Wound pain can be explained by a variety of mechanisms: inflammatory processes increasing the sensitivity of pain receptors, tissue damage causing stimulation of pain receptors, damage to terminal end of the axon and/or presynaptic terminal, and possibly local infections aggravating sensory nerves.2 Therefore, pain management represents an essential side of wound healing as a satisfactory analgesic control is undoubtedly the first step on the path of healing itself.
Chronic skin ulcers, particularly those affecting the lower limbs, are major complications in diabetes and often lead to radical surgery.3 Type 2 diabetes mellitus is one of the main causes of chronic ulcers in the elderly. The treatment of diabetic ulcers in this fragile population is a major challenge due to a significant reduction in the skin’s regenerative capacity. These negative factors are exacerbated by several critical issues in frail geriatric patients such as multiple comorbidities (eg, arterial insufficiency, neuropathy, and diabetic skin microangiopathy), increased risk of infection, polypharmacotherapy, cognitive impairment, and poor compliance to dressing procedures.4,5
Despite the high prevalence of chronic pain in the elderly,6 major barriers to the effective identification and management of pain in the older population have been identified.7 As a matter of fact, pain is often underestimated and undertreated among elderly people.8 A recent study on 29 patients with scleroderma has proved oxycodone to be an effective and safe treatment of pain caused by severe scleroderma skin ulcers; in addition, it markedly improved the patient’s compliance to local wound care procedures.9
Opioids are the established treatment for moderate and severe chronic oncological pain, as recommended by the World Health Organization (WHO);10 furthermore, recent guidelines suggest opioids as the mainstay of treatment for chronic nononcological pain even in elderly patients.11,12
One of the main limitations to the use of opioids in the elderly is the fear of adverse gastrointestinal events (straining, incomplete evacuation, bloating, abdominal distension, and gastric reflux) due to the binding of opioids to receptors in the gastrointestinal tract. This is known to cause a condition known as opioid-induced constipation (OIC),13 which is potentially even more harmful in elderly patients. Oral naloxone, due to its very low systemic bioavailability, predominantly antagonizes opioid receptors in the gastrointestinal tract and is effective in the prevention of OIC.14–17 Oral prolonged-release oxycodone/naloxone combination (OXN-PR) has also been shown to be equally effective compared to oxycodone alone in patients with moderate-to-severe nonmalignant pain.18–21 OXN-PR has been shown to be effective and safe in the treatment of neuropathic pain also.22
Recent studies have shown oxycodone/naloxone association to be safe and effective in the elderly.23–25 To our knowledge, there are no significant systematic data on the use of this combination in the treatment of pain associated with skin ulcers in the elderly.
Here, we describe the case of an elderly diabetic patient suffering from years of multiple chronic ulcers of the lower limbs associated with significant chronic pain, in which the combination of oxycodone/naloxone has provided satisfactory analgesic control, and subsequently, complete healing and (long-term) pain relief.
Written informed consent was obtained from the patient for the publication of this case report and accompanying images. Ethics approval is not needed for the present case report, as it involves current clinical practices.
Case report
The patient, CA, an 82-year-old female, suffering from type II diabetes mellitus for over 20 years complicated by peripheral neuropathy and arteriopathy, came to our attention presenting painful skin ulcers of the lower extremities (Figure 1, day 0 on admission). She was under basal–bolus scheme insulin, and on admission, glycemic values were somewhat uncontrolled (HbA1c 55 mmol/mol, fasting glucose 180 mg/dL). Despite the ongoing analgesic treatment (acetaminophen 3,000 mg daily), pain control was poor and wound treatment was strongly limited by the intense pain. She had also been prescribed tramadol by her general practitioner, stopped early due to nausea and vomiting. On admission, OXN-PR 5/2.5 mg bid (twice a day) was started, and acetaminophen was stopped.
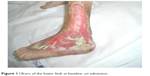  | Figure 1 Ulcers of the lower limb at baseline, on admission. |
In order to evaluate the effectiveness and tolerability of the OXN-PR association, the following parameters were assessed during the time of observation: pain intensity (on a verbally administered 0–10-point Numeric Rating Scale [NRS], expressed as the mean of assessments at rest, on movement, daytime, and nocturnal of the last 24 hours), and the impact of pain on daily activities according to the Brief Pain Inventory (BPI) of both severity and interference domains, cognitive function using the Mini Mental State Examination (MMSE), and functional autonomy through the Barthel Index score. Bowel function was evaluated using the Bowel Function Index (BFI, a validated reproducible measure that detects clinically meaningful changes in constipation, with scores ranging from 0, free from symptoms, to 100, most severe symptoms; normal bowel function is defined as a BFI value of ≤29). Common opioid-related adverse events (nausea, vomiting, dizziness, drowsiness, dry mouth, and itching) were also monitored.
At baseline, pain was severe and had a markedly negative impact on daily activities. Functional autonomy was moderately impaired, with preserved cognitive status and bowel function (Table 1).
Subsequent pain assessment showed a clear reduction in the NRS score, which progressively decreased to 5 after 1 week in the absence of opioid-related adverse events. The dose of OXN-PR was increased to 10/5 mg bid. Residual pain was reported especially in the early hours of the morning and during ulcer dressing procedures. The evaluation after 2 weeks and 4 weeks showed a further reduction in NRS score, which decreased to 2 points during both assessments.
In concomitance with the striking subjective improvement, the lady became more compliant to dressing procedures, significantly contributing to the complete healing of the ulcers in the following 4 weeks (Figures 2 and 3). Throughout this period, no minor or major adverse effects were observed. No significant changes were also found regarding cognitive status and bowel function; at final functional assessment, optimal analgesic control was associated with a marked functional improvement (Table 1).
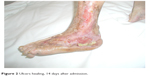  | Figure 2 Ulcers healing, 14 days after admission. |
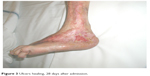  | Figure 3 Ulcers healing, 28 days after admission. |
Discussion
In caring for this patient, the association of OXN-PR demonstrated high efficacy and tolerability in the treatment of pain caused by diabetic lower limb ulcers. At the same time, an increase in the patient’s compliance to ulcer dressing procedures was noted.
In total, 20/10 mg daily dose of OXN-PR proved sufficient in obtaining a more than satisfactory analgesic efficacy. No adverse effects were observed, consistent with the previously reported tolerability of this oral opioid agonist–antagonist combination in the elderly.16,18,19,26 In particular, no changes were observed in the BFI score, confirming the previously reported excellent gastrointestinal tolerability of this drug combination, which is certainly of extreme importance in the geriatric population.27,28 Besides its effectiveness, the additional benefit of OXN-PR is to prevent OIC obviating the need of laxatives in the majority of patients. Avoiding OIC represents a major issue in older and fragile patients.
Topical analgesic applications for ulcerations are available; on the other hand, topical analgesics are not so widely used, and data from clinical trials are limited. In our patient, skin ulcers were too large for a topical analgesic treatment.
This case report emphasizes three main issues in pain management in the elderly: 1) switching opioids (in our case, from tramadol to OXN-PR) is a reasonable approach, and it is always worth considering while managing pain, as current guidelines on pain management for geriatrics suggest;11,12 2) slowly titrating opioids is required in order to avoid adverse events; prescribing opioids to this fragile population should always follow the rule “Start slowly, go slowly”;11,12 and 3) OXN-PR, according to its peculiar pharmacological profile, should be considered as a potentially safe tool for managing pain, preventing OIC in older patients.27,28
In the literature, the OXN-PR combination has previously been demonstrated to be effective in controlling both neuropathic and nociceptive pain.18,22 As wound-related pain is typically both neuropathic and nociceptive, the OXN-PR combination could be reasonably considered a potentially ideal solution to a very complex and common problem: chronic pain due to skin ulcers in geriatric comorbid patients.
OXN-PR combination has been recently found effective and safe in the treatment of chronic nononcological pain in the elderly with a good efficacy profile and a low incidence of adverse effects.22–25
Considering all the current evidence, as well as our experience, we strongly endorse further clinical studies on larger cohorts of geriatric patients, to formally evaluate the effectiveness of this drug combination in the management of skin ulcer-related pain in the elderly.
Acknowledgments
Written informed consent was obtained from the patient for publication of this case report and accompanying images.
Author contributions
All authors contributed toward data analysis, drafting and critically revising the paper and agree to be accountable for all aspects of the work.
Disclosure
The study was completed independently with no funding. The authors report no conflicts of interest in this work.
References
Briggs M, Closs SJ. Patients’ perceptions of the impact of treatments and products on their experience of leg ulcer pain. J Wound Care. 2006;15:333–337. | ||
Fleck CA. Managing wound pain: today and in the future. Adv Skin Wound Care. 2007;20(3):138–145. | ||
Cevera JJ, Bolton LL, Kerstein MD. Options for diabetic patients with chronic heel ulcers. J Diabetes Complications. 1997;11:358–366. | ||
Falanga V. Wound healing and its impairment in the diabetic foot. Lancet. 2005;366:1736–1743. | ||
Frank C. Approach to skin ulcers in older patients. Can Fam Physician. 2004;50:1653–1659. | ||
Bergh I, Steen G, Waern M, et al. Pain and its relation to cognitive function and depressive symptoms: a Swedish population study of 70-year-old men and women. J Pain Symptom Manage. 2003;26:903–912. | ||
Allcock N, McGarry C. Management of pain in older people within the nursing home: a preliminary study. Health Soc Care Comm. 2002;10:464–471. | ||
Bernabei R, Gambassi G, Lapane K, et al. Management of pain in elderly patients with cancer. JAMA. 1998;279:1877–1882. | ||
Giuggioli D, Manfredi A, Colaci M, Ferri C. Oxycodone in the long-term treatment of chronic pain related to scleroderma skin ulcers. Pain Med. 2010;10(10):1500–1503. | ||
Ventafridda V, Saita L, Ripamonti C, De Conno F. WHO guidelines for the use of analgesics in cancer pain. Int J Tissue React. 1985;7:93–96. | ||
American Geriatric Society. Pharmacological management of persistent pain in older persons. J Am Geriatr Soc. 2009;57:1331–1346. | ||
British Geriatric Society. Guidance on the management of pain in older people. Age Ageing. 2013;42(1):i1–i57. | ||
Kurz A, Sessler DI. Opioid-induced bowel dysfunction: pathophysiology and potential new therapies. Drugs. 2003;63:649–671. | ||
Mueller-Lissner S. Fixed combination of oxycodone with naloxone: a new way to prevent and treat opioid-induced constipation. Adv Ther. 2010;27:581–590. | ||
Löwenstein O, Leyendecker P, Hopp M, et al. Combined prolonged-release oxycodone and naloxone improves bowel function in patients receiving opioids for moderate-to-severe non-malignant chronic pain: a randomised controlled trial. Expert Opin Pharmacother. 2009;10(4):531–543. | ||
Meissner W, Leyendecker P, Mueller-Lissner S, et al. A randomised controlled trial with prolonged-release oral oxycodone and naloxone to prevent and reverse opioid-induced constipation. Eur J Pain. 2009;13:56–64. | ||
Burness CB, Keating GM. Oxycodone/naloxone prolonged-release: a review of its use in the management of chronic pain while counteracting opioid-induced constipation. Drugs. 2014;74(3):353–375. | ||
Vondrackova D, Leyendecker P, Meissner W, et al. Analgesic efficacy and safety of oxycodone in combination with naloxone as prolonged release tablets in patients with moderate to severe chronic pain. J Pain. 2008;9:1144–1154. | ||
Davis M, Goforth HW, Gamier P. Oxycodone combined with opioid receptor antagonists: efficacy and safety. Expert Opin Drug Saf. 2013;12(3):389–402. | ||
Löwenstein O, Leyendecker P, Lux EA, et al. Efficacy and safety of combined prolonged-release oxycodone and naloxone in the management of moderate/severe chronic non-malignant pain: results of a prospectively designed pooled analysis of two randomised, double-blind clinical trials. Clin Pharmacol. 2010;29(10):12. | ||
Mercadante S, Giarratano A. Combined oral prolonged-release oxycodone and naloxone in chronic pain management. Expert Opin Investig Drugs. 2013;22(1):161–166. | ||
Hermanns K, Junker U, Nolte T. Prolonged-release oxycodone/naloxone in the treatment of neuropathic pain – results from a large observational study. Expert Opin Pharmacother. 2012;13(3):299–311. | ||
Guerriero F, Sgarlata C, Marcassa C, Ricevuti G, Rollone M. Efficacy and tolerability of low-dose oral prolonged-release oxycodone/naloxone for chronic nononcological pain in older patients. Clin Interv Aging. 2015;10:1–11. | ||
Ahmedzai SH, Leppert W, Janecki M, et al. Long-term safety and efficacy of oxycodone/naloxone prolonged-release tablets in patients with moderate-to-severe chronic cancer pain. Support Care Cancer. 2015;23:823–830. | ||
Blagden M, Hafer J, Duerr H, Hopp M, Bosse B. Long-term evaluation of combined prolonged-release oxycodone and naloxone in patients with moderate-to-severe chronic pain: pooled analysis of extension phases of two Phase III trials. Neurogastroenterol Motil. 2014;26(12):1792–1801. | ||
Gatti A, Casali M, Lazzari M, et al. Prolonged-release oxycodone/naloxone in nonmalignant pain: single-center study in patients with constipation. Adv Ther. 2013;30(1):41–59. | ||
Leppert W. Oxycodone/naloxone in the management of patients with pain and opioid-induced bowel dysfunction. Curr Drug Targets. 2014;15(1):124–135. | ||
Cuomo A, Russo G, Esposito G, Forte CA, Connola M, Marcassa C. Efficacy and gastrointestinal tolerability of oral oxycodone/naloxone combination for chronic pain in outpatients with cancer: an observational study. Am J Hosp Palliat Care. 2014;31(8):867–876. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

