Back to Journals » Therapeutics and Clinical Risk Management » Volume 14
Intraoperative anterior optical coherence tomography-guided synechiolysis in a post-penetrating keratoplasty patient with peripheral corneal opacification
Authors Petrovic A, Gianniou C, Hashemi K, Kymionis G
Received 1 March 2018
Accepted for publication 14 May 2018
Published 7 August 2018 Volume 2018:14 Pages 1387—1390
DOI https://doi.org/10.2147/TCRM.S167025
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Garry Walsh
Aleksandra Petrovic,1,2 Christina Gianniou,1,2 Kattayoon Hashemi,1,2 Georgios Kymionis1,2
1Department of Ophthalmology, University of Lausanne, Jules-Gonin Eye Hospital, Lausanne, Switzerland; 2Cornea and Refractive Surgery Department, Lausanne, Switzerland
Purpose: The aim of this study was to describe intraoperative optical coherence tomography (OCT) as an adjunctive device for peripheral synechiolysis in a post-penetrating keratoplasty patient.
Case report: A 75-year-old female patient presented for follow-up 9 months after a penetrating keratoplasty in the left eye. She presented with anterior peripheral synechiae involving the graft for three clock hours. Peripheral native cornea appeared totally opaque. Synechiolysis was planned, and intraoperative OCT allowed us to detect nonclinically visible synechiae and to confirm complete synechiolysis immediately after surgery. No postoperative complication was recorded. Two months after surgery, the graft was clear and anterior segment OCT did not reveal any residual synechiae or recurrence.
Conclusion: Intraoperative OCT is useful to overcome the difficulties in visualization through clinically opaque corneas, as it gives real-time feedback upon the anatomy, the extension of the remaining lesions, and the success of the surgery.
Keywords: penetrating keratoplasty, peripheral anterior synechiae, intraoperative optical coherence tomography
Introduction
Optical coherence tomography (OCT) is a high-resolution imaging modality used to image the anterior segment, first described in 1994.1 High-speed OCT provides in vivo cross-sectional images of the anterior eye, similar to histological sections, and could be a potential tool for noninvasive evaluation of the anterior chamber angle,2 especially in cases of corneal opacity.
Intraoperative real-time microscope-integrated OCT (iOCT) is an emerging modality with the potential to revolutionize the surgical techniques by providing a real-time dynamic feedback of the tissue alterations during surgery.3,4 In 2014, the PIONEER study described the feasibility, utility, and safety of iOCT in ophthalmic surgery and showed that the tool impacted surgical decision in >40% of the cases.3 iOCT has already been described in DMEK and DSAEK surgeries with advanced bullous keratopathy.5–7
Anterior synechiae after penetrating keratoplasty (PK) is a major risk factor for developing glaucoma and endothelial failure by rejection.8,9 Glaucoma, in turn, is a major risk factor for graft failure, and furthermore, glaucoma surgery is associated with shorter time to graft rejection.9
Although management of peripheral anterior synechiae (PAS) after PK is not clearly defined in the literature, clinicians often tend to surgically treat this condition, in order to avoid complications such as glaucoma and graft rejection.
We describe here the first case of iOCT-guided synechiolysis in a patient with peripheral corneal opacity after a PK.
Case report
A 75-year-old female patient presented for follow-up 9 months after a PK in the left eye.
She had undergone a deep sclerectomy for open-angle glaucoma 12 years prior to the keratoplasty followed by cataract surgery 3 years later and a Baerveldt tube implantation, due to medically uncontrolled intraocular pressure, 5 years later. Implantation of the Baerveldt tube was performed in the sulcus to avoid any corneal touch. She had also been treated for recurrent anterior uveitis. Uveitic workup was negative for sarcoidosis, syphilis, lyme, and tuberculosis. As herpetic origin could not be excluded, patient was treated with prophylactic systemic valaciclovir to avoid recurrence.
One year after the last surgery, patient presented with endothelial decompensation and stromal edema. A DMEK was performed under local anesthesia, without any intraoperative complication. Postoperative treatment consisted of dexamethasone and ofloxacin drops four times a day. Valaciclovir was continued. One month after surgery, the patient presented with total corneal opacification, stromal edema, and graft failure. After 2 months, she developed anterior stromal fibrosis. A PK was planned. A 7.75 mm graft was sutured after a 7.5 mm trephination. The postoperative treatment included dexamethasone and tobramycine drops (Tobradex®; Alcon, Fort Worth, TX, USA) six times a day. Valaciclovir was continued. No postoperative complication was recorded for the first 9 months.
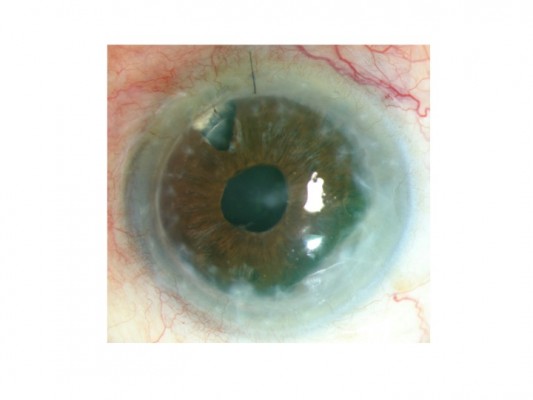
Nine months after the surgery, the patient presented with anterior peripheral synechiae involving the graft for three clock hours. Peripheral native cornea appeared totally opaque (Figure 1). Graft was clear with no sign of rejection. Anterior chamber was calm, with no sign of recurrent uveitis. Intraocular pressure was 15 mmHg.
  | Figure 1 Color photography showing penetrating keratoplasty with peripheral opacification. |
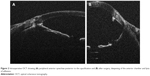
Because of the history of uveitis, synechiolysis was planned. Surgery was performed under local anesthesia and was assisted by Leica enfocus Ultra deep OCT (Leica Microsystems, Wetzlar, Germany), which delivers high definition real-time images of the anterior segment. Due to peripheral corneal opacification, anterior synechiae could not be properly seen with slit lamp microscopy or operative microscope. Intraoperative OCT allowed us to detect nonclinically visible synechiae and to confirm complete synechiolysis immediately after surgery (Figure 2). Synechiolysis was performed using a viscoelastic cannula (Viscoat®, Alcon, Fort Worth, USA) and an ophthalmic dispersive device (Viscoat).
No postoperative complication was recorded. Postoperative treatment included tobramycine and dexamethasone drops six times a day for 3 months with progressive tapering. Valaciclovir was continued 1 g three times a day for 1 month and then tapered weekly until 500 mg per day. Two months after the surgery, vision was 0.2 limited by advanced glaucomatous neuropathy. The graft was clear and anterior segment OCT (Spectralis, Heidelberg, Germany) did not reveal any residual synechiae or recurrence.
Ethics
Written informed consent has been provided by the patient for the publication of her case details and images.
Discussion
We describe a case of synechiolysis intraoperatively guided by OCT. The device allowed good quality images of the anterior segment, even in the presence of corneal opacification, as infrared illumination used by the device scatters less than light used by microscopes.7 iOCT helped localize the synechiae, facilitated the synechiolysis, and confirmed complete synechiolysis at the end of the procedure. Without the device, operative strategy would be planned differently. Anterior segment OCT would be performed preoperatively, and synechiae position would be marked.
iOCT has already been described in anterior segment surgery, especially for lamellar keratoplasty such as DMEK and DSAEK.3,5–7 In DMEK, iOCT has been reported to help in graft rotation without touching the graft as well as graft orientation.5,6 In DSAEK surgery, iOCT has been reported to help in graft–host interactions and to verify the presence or absence of fluid interface.7 For both surgeries, iOCT may expand their indications and diminish the need for PK, which is a more invasive surgery with a higher comorbidity rate. The actual impact on overall surgical time has been studied and appears to be generally minimal.3
However, this device has still some limitations: distortion of the images during eye movements and restriction of the scanning zone necessitate frequent adjustment of the viewing field during surgery. Automated tracking would be a welcome addition to facilitate rapid imaging of the region of interest and to minimize surgeon burden of manual adjustment.10 Moreover, metallic instruments create difficulties for intraoperative OCT systems due to the light scattering and shadowing characteristics of metal, which render underlying tissue invisible.10
Conclusion
Despite these limitations, it seems that iOCT is a valuable device in order to overcome the difficulties in visualization through clinically opaque corneas.
To the best of our knowledge, this is the first report of intraoperative OCT-guided anterior synechiolysis.
Disclosure
The authors report no conflicts of interest in this work.
References
Izatt JA, Hee MR, Swanson EA, et al. Micrometer-scale resolution imaging of the anterior eye in vivo with optical coherence tomography. Arch Ophthalmol. 1994;112(12):1584–1589. | ||
Radhakrishnan S, Rollins AM, Roth JE, et al. Real-time optical coherence tomography of the anterior segment at 1310 nm. Arch Ophthalmol. 2001;119(8):1179–1185. | ||
Ehlers JP, Dupps WJ, Kaiser PK, et al. The prospective intraoperative and perioperative ophthalmic ImagiNg with optical CoherEncE TomogRaphy (PIONEER) study: 2-year results. Am J Ophthalmol. 2014;158(5):999–1007. | ||
Titiyal JS, Kaur M, Falera R. Intraoperative optical coherence tomography in anterior segment surgeries. Indian J Ophthalmol. 2017;65(2):116–121. | ||
Cost B, Goshe JM, Srivastava S, Ehlers JP. Intraoperative optical coherence tomography-assisted descemet membrane endothelial keratoplasty in the DISCOVER study. Am J Ophthalmol. 2015;160(3):430–437. | ||
Saad A, Guilbert E, Grise-Dulac A, Sabatier P, Gatinel D. Intraoperative OCT-assisted DMEK: 14 consecutive cases. Cornea. 2015;34(7):802–807. | ||
Pasricha ND, Shieh C, Carrasco-Zevallos OM, et al. Real-time microscope-integrated OCT to improve visualization in DSAEK for advanced bullous keratopathy. Cornea. 2015;34(12):1606–1610. | ||
Price MO, Thompson RW Jr, Price FW Jr. Risk factors for various causes of failure in initial corneal grafts. Arch Ophthalmol. 2003;121(8):1087–1092. | ||
Baltaziak M, Chew HF, Podbielski DW, Ahmed IIK. Glaucoma after corneal replacement. Surv Ophthalmol. 2018;63(2):135–148. | ||
Ehlers JP. Intraoperative optical coherence tomography: past, present, and future. Eye. 2016;30(2):193–201. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.