Back to Journals » Clinical Interventions in Aging » Volume 16
Incidence, Predictors and Outcomes of Delirium in Complicated Type B Aortic Dissection Patients After Thoracic Endovascular Aortic Repair
Authors Liu J, Yang F, Luo S, Li C, Liu W , Liu Y, Huang W, Xie E, Chen L, Su S, Yang X, Geng Q , Luo J
Received 9 July 2021
Accepted for publication 16 August 2021
Published 26 August 2021 Volume 2021:16 Pages 1581—1589
DOI https://doi.org/10.2147/CIA.S328657
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Zhi-Ying Wu
Jitao Liu,1,* Fan Yang,2,* Songyuan Luo,1,* Chenxi Li,3 Weijie Liu,4 Yuan Liu,1 Wenhui Huang,1 Enmin Xie,5 Lyufan Chen,6 Sheng Su,5 Xinyue Yang,6 Qingshan Geng,1 Jianfang Luo1,5
1Department of Cardiology, Guangdong Cardiovascular Institute, Guangdong Provincial Key Laboratory of Coronary Heart Disease Prevention, Guangdong Provincial People’s Hospital, Guangdong Academy of Medical Sciences, Guangzhou, People’s Republic of China; 2Department of Emergency and Critical Care Medicine, Guangdong Provincial People’s Hospital, Guangdong Academy of Medical Sciences, Guangzhou, People’s Republic of China; 3Department of Cardiac Surgery Intensive Care Unit, Guangdong Cardiovascular Institute, Guangdong Provincial People’s Hospital, Guangdong Academy of Medical Sciences, Guangzhou, People’s Republic of China; 4Center for Information Technology and Statistics, The First Affiliated Hospital of Sun Yat-sen University, Guangzhou, People’s Republic of China; 5The Second School of Clinical Medicine, Southern Medical University, Guangzhou, People’s Republic of China; 6School of Medicine, South China University of Technology, Guangzhou, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Jianfang Luo; Qingshan Geng
Department of Cardiology, Guangdong Cardiovascular Institute, Guangdong Provincial Key Laboratory of Coronary Heart Disease Prevention, Guangdong Provincial People’s Hospital, Guangdong Academy of Medical Sciences, #106 Zhongshan Second Road, Yuexiu District, Guangzhou, Guangdong, 510080, People’s Republic of China
Tel +86-20-83827812
Fax + 86-20-83875453
Email [email protected]; [email protected]
Purpose: The present study aimed to investigate the incidence and predictors of post-operative delirium (POD) in patients with complicated type B aortic dissection (TBAD) undergoing TEVAR with/without concomitant procedures and to assess the association of POD with early and follow-up outcomes.
Methods: A retrospective single-center cross-sectional analysis was conducted using a prospectively maintained database from 2010 to 2017. Outcomes were postoperative clinical outcomes, early and follow-up survival.
Results: A total of 517 complicated TBAD patients were enrolled. POD was observed in 13.3% (69/517) patients and was associated with increased hospital length of stay (LOS) and hospital costs (P< 0.001 for both). Besides, POD was found to be an independent risk factor for prolonged ICU stay (odds ratio [OR] 4.39, 95% confidence interval [CI] 2.40– 8.01, P< 0.001) and early death (OR 4.42, 95% CI 1.26– 15.54, P= 0.020). Predictors of POD were hybrid procedure (OR 2.17, 95% CI 1.20– 3.92, P= 0.010), the use of benzodiazepine (OR 1.86, 95% CI 1.07– 3.23, P= 0.027) or quinolone (OR 2.35, 95% CI 1.26– 4.38, P= 0.007), creatinine > 2 mg/dL (OR 3.25, 95% CI 1.57– 6.72, P= 0.001) and preoperative blood transfusion (OR 3.31, 95% CI 1.76– 6.21, P< 0.001). After a median follow-up of 73.6 months, POD remained as an independent indicator for follow-up mortality (hazard ratio [HR] 2.40, 95% CI 1.31– 4.38, P= 0.005) after adjusting potential confounders.
Conclusion: POD after TEVAR has an incidence of around 13% and could profoundly increase the in-hospital LOS, hospital costs, as well as the early and follow-up mortality. A series of risk factors, including hybrid procedure, the use of benzodiazepine or quinolone, creatinine > 2 mg/dL and preoperative blood transfusion, were identified as independent risk factors for POD. Effective risk-stratification and patient-tailored management strategy should be developed to reduce the incidence of POD.
Keywords: type B aortic dissection, thoracic endovascular aortic repair, hybrid procedure, postoperative delirium, predictors, outcomes
Introduction
Type B aortic dissection (TBAD) is a devastating vascular emergency with high morbidity and mortality rates.1 When it is complicated, thoracic endovascular aortic repair (TEVAR) has been suggested as the first-line strategy.1,2 Hybrid or chimney technique is recommended for thoracic aortic pathologies that involve the aortic arch, and could significantly improve the outcomes.2 However, the incidence of postoperative complications remain relatively high, diminishing the quality of life and conferring a poor prognosis.3 Early identification and prevention of risk factors of these complications are of critical importance.
Postoperative delirium (POD) is a neuropsychiatric disorder with features of an acute onset and fluctuating course of inattention and confusion, and is one of the most common postoperative complications in vascular surgery procedures with an incidence ranging from 4.8% to 39%.4–6 Predisposing factors, such as increasing age, multiple comorbidities, prior cognitive impairment, alcohol or drug abuse, could make patients more prone to develop delirium postoperatively. Besides, perioperative factors, such as blood transfusion, glycemic control and medication expose, could also extremely influence the incidence of POD.5 It is well-established that POD could result in longer hospital length of stay (LOS), increased use of medical resources and higher mortality in cardiac procedure, abdominal aortic aneurysm repair and critical limb surgery.7–9 Patients with aortic surgery or amputation surgery are frequently deemed to be in a frail condition and more prone to POD compared to other vascular surgical procedures.10 However, studies that report the incidence and impact of POD after endovascular treatment for TBAD are lacking.
Obtaining more information on the risk factors for POD could assist clinicians in preventing and early recognizing POD, thus improving the decision-making process and diminishing morbidity and mortality. Therefore, the objectives of the present study were to investigate the incidence, predictors, and outcomes of POD in patients with complicated TBAD undergoing TEVAR.
Patients and Methods
Study Population
From January 2010 to December 2017, 576 consecutive patients with acute or subacute complicated TBAD received TEVAR with/without concomitant procedures at out center. The diagnosis of TBAD was confirmed by enhanced computed tomography angiography (CTA). The exclusion criteria were as follows: (1) blunt traumatic thoracic aortic injury, (2) malignant tumor, (3) connective tissue disease, (4) previous aortic intervention, (5) preexisting dementia or cognitive impairment, (6) missing baseline or operative information. The remaining 517 patients were included for the final analysis (Figure 1).
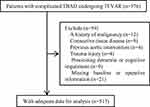 |
Figure 1 Study flow-chart. Abbreviations: TBAD, type B aortic dissection; TEVAR, thoracic endovascular aortic repair. |
Ethics
The present study was a single-center, retrospective observational design using the prospectively maintained database at Guangdong Provincial People’s Hospital (Guangdong, China). The Health Informatics Center anonymize all related data and oversee the study protocol. In the process of the study, researchers covering all data confidentiality and compliance with the Declaration of Helsinki. Therefore, patient consent to review their medical records was not required in this retrospective study. The Ethics Committee of the Guangdong Provincial People’s Hospital approved this study (No. 201807).
Definitions and Data Collection
Acute TBAD was defined as a type B aortic dissection occurring less than 14 days after the onset of symptoms, whereas subacute TBAD was defined by an elapsed time between 15 and 90 days.1 Complicated TBAD was described as persistent or recurrent pain, uncontrolled hypertension despite full medication, early aortic expansion, malperfusion, and signs of rupture (haemothorax, increasing periaortic and mediastinal haematoma).1 The POD was assessed per routine for 14 days after the operation by nurses who were trained to use the Confusion Assessment Method-Intensive Care Unit (CAM-ICU) tool: (1) an acute change in mental status or fluctuation in the level of consciousness over the prior 24 h, (2) inattention, (3) disorganized thinking, and (4) an altered level of consciousness.11 The CAM-ICU defines delirium if at least two features are present (Feature 1 and 2 OR 3 and 4). If delirium was present or suspected, two independent clinicians were responsible for the final diagnostic validation.
Demographics, comorbidities, laboratory and imaging findings, and preoperative medications use were recorded by two researchers. Abnormal serum electrolyte was considered present when serum potassium levels were less than 3.0 mmol/L or greater than 6 mmol/L, serum sodium was less than 130 mmol/L or greater than 150 mmol/L, or blood glucose values were less than 60 mg/dL or greater than 300 mg/dL.12 All patients underwent preoperative CTA of the entire aorta. The maximum outer-to-outer diameter of the aorta, the extent of the dissection and false lumen status were evaluated using commercial software (Terarecon, San Mateo, CA, USA).
Procedure
TEVAR for complicated TBAD patients was aligned with the European Society of Cardiology (ESC) 2014 guidelines.1 The details of the procedures at our center have been previously described.13 Briefly, the procedures were performed with suitable anatomy in a cardiac catheterization room under local anaesthesia. The diameters of aortic stent-grafts were generally oversized by 5% to 15% according to the aortic morphology. All stent-grafts were deployed retrogradely via percutaneous femoral artery access to obliterate the proximal entry tear. The left subclavian arteries (LSA) and left common carotid artery (LCCA) were covered when necessary to obtain at least a 2-cm proximal landing zone. The technique of reconstruction of the arch vessels (chimney or hybrid [TEVAR concomitant with supra-arch bypass]) was at the discretion of the operating surgeon according to the characteristics of aortic pathologies. Notably, the hybrid procedure was conducted in a staged fashion and required general anaesthesia in our center. The majority of patients were treated with a single stent graft, and additional pieces were placed only when the initial graft did not produce the desired result of coverage of the entry tear and expansion of the true lumen as determined by angiography.
Outcomes
The primary outcomes were the incidence of POD and follow-up all-cause mortality. Early outcomes occurring in-hospital or 30 days after TEVAR, including death and prolonged ICU stay, were considered as the secondary outcomes. Prolonged ICU stay was defined as a treatment in ICU longer than 72 hours after the procedure.
Follow-Up Protocol
All in-hospital survival patients underwent clinical, and imaging follow-up at 1, 3, 6, 12 months, and annually thereafter. Each patient was suggested to return to the hospital for a review at corresponding intervals. Data including symptoms, medication, imaging findings and other relevant conditions were collected during follow-up. Assessment of patients’ condition was mainly completed by an outpatient clinic visit or telephone interview.
Statistical Analysis
Categorical variables were analyzed using the Chi-square test or Fisher exact test and presented as numbers and percentages. Continuous variables were tested using the Student’s t-test or Mann–Whitney U-test according to the distribution and presented as mean ± standard deviation or median and interquartile range (IQR). The D-dimer level was log-transformed to account for the skewed distribution.
The cumulative rate of follow-up mortality was presented as Kaplan-Meier curves and compared using the Log rank test. Factors associated with POD were analyzed with multivariable logistic regression. Receiver operating characteristic (ROC) curves were plotted, and the area under the ROC curve (AUC) values were calculated to describe the model’s prediction accuracy for the incidence of POD. Logistic and Cox regression analyses were carried out to investigate the association between POD and outcomes. Variables with a P < 0.10 in the univariate analysis were included in the multivariable model to identify independent predictors using a forward stepwise selection procedure. Proportional hazards (PH) assumption was evaluated by calculating weighted Schoenfeld residuals, and no violations of the PH assumptions were found. A 2-tailed P < 0.05 was considered significant in all tests. All statistical analyses were performed using R software (version 3.5.1).
Results
Clinical Characteristics
A total of 517 acute or subacute complicated TBAD patients (453 males, 87.6%) with an average age of 53.2 ± 10.9 years were enrolled in the current study. Hypertension was confirmed in 445 (86.1%) patients and was the most common comorbidity. One hundred-twenty (23.2%) patients underwent hybrid procedures, 82 (15.9%) patients underwent TEVAR concomitant with chimney procedure. POD was confirmed in 69 (13.3%) patients. Detailed baseline characteristics were summarized in Table 1.
 |
Table 1 Baseline and Operative Characteristics of Patients with and without Delirium |
Predictors of POD
In the univariate logistic analysis, age, hybrid procedure, the use of benzodiazepine or quinolone, albumin <30 g/L, creatinine >2 mg/dL, maximum aortic diameter ≥40 mm, preoperative blood transfusion, abnormal serum electrolyte, and insertion of ≥2 aortic stents were significant predictors for POD (P< 0.05 for each, Supplementary table S1). After adjusting for confounding factors, hybrid procedure (odds ratio [OR] 2.17, 95% confidence interval [CI] 1.20–3.92, P= 0.010), the use of benzodiazepine (OR 1.86, 95% CI 1.07–3.23, P= 0.027) or quinolone (OR 2.35, 95% CI 1.26–4.38, P = 0.007), creatinine >2 mg/dL (OR 3.25, 95% CI 1.57–6.72, P= 0.001) and preoperative blood transfusion (OR 3.31, 95% CI 1.76–6.21, P< 0.001) remained as the independent risk factors for POD (Supplementary table S1 and Figure 2A). The Receiver operating characteristic curve analysis revealed that the final model had a moderate predictive value for POD (AUC 0.75, 95% CI 0.68–0.81, Supplementary figure 1).
 |
Figure 2 Forest plot of POD predictors (A), and multivariable analysis results of follow-up mortality (B). Abbreviations: POD, postoperative delirium; BMI, body mass index. |
POD and Early Outcomes
As presented in Table 2, patients with POD had significantly higher rates of prolonged ICU stay, 30-day death, stroke, spinal cord ischemia and re-intervention, as well as higher hospital cost (P< 0.05 for each), but had the similar rates of postoperative acute kidney injury (AKI) and limb ischemia compared with those without POD (P> 0.05 for both). Multivariable logistic analysis showed that POD was an independent predictor for prolonged ICU stay (OR 4.39, 95% CI 2.40–8.01, P< 0.001) and 30-day death (OR 4.42, 95% CI 1.26–15.54, P= 0.020) (Table 3).
 |
Table 2 Post-Operative Outcomes of Patients with and without Delirium |
 |
Table 3 Results of Multivariate Regression Analysis for Early Outcomes |
POD and Follow-Up Survival
The median duration of follow-up was 73.6 months (IQR, 47.7–92.3 months). In this period, 58 (11.6%) patients died after the procedure, with 19 (30.2%) and 39 (8.9%) patients for delirium and non-delirium group (P< 0.001), respectively (Table 2). The cumulative survival rate at 1, 3, and 5 years was 92.1%, 78.6%, and 58.5% in patients with delirium and 99.3%, 98.6%, and 97.6% in those without delirium. Compared with patients without POD, the cumulative follow-up mortality was much prominent in those with POD (log-rank=19.38; P< 0.001) (Figure 3). Additionally, the incidence of stroke, re-intervention and re-admission rates were similar between patients with/without delirium.
 |
Figure 3 The Kaplan-Meier survival curve of the association between postoperative delirium and follow-up survival. |
In univariate Cox regression analysis, delirium was significantly related to follow-up mortality (Supplementary table S2). Other significant parameters included age, BMI, the use of analgesics, coronary artery disease, albumin <30 g/L, creatinine >2 mg/dL and maximum aortic diameter ≥40 mm. In the multivariable analysis, delirium (HR 2.40, 95% CI 1.31–4.38, P=0.005), BMI (HR, 0.89; 95% CI 0.81–0.97; P= 0.010), hybrid procedure (HR 1.93, 95% CI 1.06–3.54, P= 0.033), the use of analgesics (HR 0.57, 95% CI 0.33–0.98, P= 0.042), albumin <30 g/L (HR 2.10, 95% CI 1.24–3.57, P= 0.006), creatinine >2 mg/dL (HR 3.14, 95% CI 1.66–5.97, P< 0.001) and maximum aortic diameter ≥40 mm (HR 1.92, 95% CI 1.12–3.29, P= 0.018) remained as the independent indicators for follow-up mortality (Supplementary table S2; Figure 2B).
Discussion
In the present study, we found that the incidence rate of POD was approximately 13.3% in patients with acute or subacute complicated TBAD who underwent TEVAR. POD could result in significant prolongation of hospital LOS and ICU stay, as well as increased use of hospital resources. Furthermore, POD could profoundly affect the early and follow-up survival among these patients. The hybrid procedure, the use of benzodiazepine or quinolone, creatinine >2 mg/dL and preoperative blood transfusion were independent predictors of POD.
Data on the incidence of POD in TBAD patients are lacking. A meta-analysis reported that the incidence of POD in vascular surgery ranged from 4.8%-39%,6 which was in line with the 13.3% in our result. Nevertheless, the rate was lower than 24.25% which was reported by Cai et al in patients with type A aortic dissection.14 This discrepancy might attribute to more arch vessels involvement and more severe ischemic brain injury in type A aortic dissection. Despite the frequency of delirium in this postoperative setting, POD is often considered a less serious event than other major postoperative complications. Gottesman et al pointed out that the impact of POD on both in-hospital and long-term outcomes might be underestimated, and demonstrated that POD after cardiac surgery was a strong independent indicator of mortality up to 10 years postoperatively.9 This relationship was also observed in other non-cardiac operations including transcatheter aortic valve implantation, abdominal aortic aneurysm repair, and critical limb ischemia surgery.6,14,15 Our results based on relatively large sample size and long follow-up duration further corroborated this conclusion and enriched the literature.
Consistent with previous studies,5,16 patients with POD were older and a greater comorbidities burden in the present study. These might partly explain the high mortality in patients who developed POD. Rather than being causally associated with poor outcomes, POD may reflect a vulnerable status with diminished reserve capacity.15,16 However, considering increasing evidence confirmed the association between POD and poor prognosis and 30% to 40% of POD might be prevented,17 it seems imperative to develop effective management strategies to identify patients at high risk for POD and prevent this entity arising.
The risk of developing POD could be considered as the result of predisposing and precipitating factors. Advanced age, malnutrition (eg lower BMI or albumin level) and a high burden of coexisting conditions are common predisposing risk factors.5,17 However, we did not find any association between older age and POD in our study. This might be attributable to the relatively young mean age (53.2 ± 10.9 years) in our sample. It might be the same reason that the difference between patients with/without POD was not significant (P= 0.723). Although the univariate analysis showed hypoalbuminemia (albumin <30 g/L) was a significant risk factor for POD (HR 1.74, P= 0.040, Supplementary table S1), the statistical significance disappeared after adjusting for other confounders. Nonetheless, there should be little doubt that nutritional assistance and physical training are important and useful to reduce the incidence of post-operative complications including the POD.5
In our analysis, the hybrid procedure, consisting of supra-arch bypass and endovascular treatment, was identified as an independent risk factor for POD in our data. A distinguishing feature of patients undergoing hybrid procedure is the extensive aortic arch involvement, which might induce severe brain ischemia.18 Additionally, hybrid technique involves a strong noxious stimulus, due to the requirement for general anesthesia, prolonged ICU stay, severe postoperative pain and subsequent use of pain medication, and the disorienting effect of the frequent change of environment, thus being more prone to develop POD. Preventive strategies, such as the proactive geriatric consultation, effective pain management, early mobilization and orienting communication, have displayed positive effect on reducing POD, ideally when implemented before and continued after intervention. Notably, total endovascular aortic arch repair using branch stent-graft or fenestration techniques have been developed as less invasive alternatives to hybrid technique, which could avoid general anesthesia, minimize the ICU stay and diminish incision-related pain, thus has the potential to significantly reduce the incidence of POD.2 Improvement on stent-graft design and material, and operation technology, might facilitate the implementation of these novel techniques.
All remaining indicators found in the present study, including the use of benzodiazepine or quinolone,19,20 preoperative blood transfusion21,22 and creatinine >2 mg/dL,14,19 have been previously associated with POD. Blood transfusion administration and benzodiazepine use were described as the only two modifiable factors for POD with strong evidence by clinical guidelines.23 A hypothesis is that blood transfusion could activate the release of cytokines and amplify the systemic inflammatory reaction caused by disease or operation, which could permeate through the blood-brain barrier and trigger delirium.24 However, the protective role of avoiding blood transfusion especially in patients with low level of hemoglobin is still inconclusive.24 It seems advisable to transfuse with proper indications and control transfusion complications like volume overload by identifying high-risk patients, slowing down the infusion speed, and carefully monitoring.24 Minimization of POD-related medications (eg, benzodiazepine, quinolone) were effective and recommended by current consensus to decrease the incidence of POD.5 For those receiving hybrid procedures, either propofol or dexmedetomidine over benzodiazepines for sedation might be more appropriate especially in critically ill, mechanically ventilated patients.23 Besides, given that patients with renal dysfunction had higher risk of developing POD, it were preferable to minimize the use of nephrotoxic drugs (eg opioid) and conduct adequate preoperative and intraoperative fluid hydration.23
Limitations
There are several limitations that deserved to be pointed out. First, it was a retrospective, observational single-center design, although we enrolled all TBAD patients undergoing TEVAR continually to minimize the selection bias. Second, the incidence of POD might be underestimated as symptoms could be subtle and unrecognized. The retrospective nature of the study might have also contributed to the underestimation of the prevalence of POD. Third, complete data concerning known risk factors for POD (eg quantification of preoperative cognitive impairment) were not available in our data set. Given this, the variables chosen for the final model are limited, and residual confounding could not be excluded.
Conclusions
POD was one of the most frequent postoperative complications and occurred in 13.3% of patients with TBAD who underwent TEVAR with/without concomitant procedures. It could profoundly increase the in-hospital LOS, ICU stay and hospital costs, as well as the early and follow-up mortality. Besides hybrid procedure, the use of benzodiazepine or quinolone, creatinine >2 mg/dL and preoperative blood transfusion were identified as independent risk factors for POD. More attention should be drawn to identify high-risk patients and development of patient-tailored management strategies to reduce the incidence of POD and improve the prognosis.
Abbreviations
POD, postoperative delirium; TBAD, type B aortic dissection; TEVAR, thoracic endovascular aortic repair; LOS, length of stay; CAM-ICU, Confusion Assessment Method-Intensive Care Unit; LSA, left subclavian arteries; LCCA, left common carotid artery.
Data Sharing Statement
The data used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Acknowledgments
The authors acknowledge the expert language assistance of Dr. Qiuju Chen.
Funding
This research was supported by grants from High-level Hospital Construction Project (grant no DFJH201807 and no DFJH201811), Guangdong Provincial Key Laboratory of Coronary Artery Disease Prevention Fund (grant no Y022017018) and Guangdong Provincial People’s Hospital Clinical Research Fund (grant no Y012018085). The funding bodies did not have any role in the design of the study, data collection, and analysis, nor on the interpretation and dissemination of the results.
Disclosure
The authors declare that there is no conflict of interest.
References
1. Erbel R, Aboyans V, Boileau C, et al. 2014 ESC Guidelines on the diagnosis and treatment of aortic diseases: document covering acute and chronic aortic diseases of the thoracic and abdominal aorta of the adult. The Task Force for the Diagnosis and Treatment of Aortic Diseases of the European Society of Cardiology (ESC). Eur Heart J. 2014;35(41):2873–2926.
2. Czerny M, Schmidli J, Adler S, et al. Current options and recommendations for the treatment of thoracic aortic pathologies involving the aortic arch: an expert consensus document of the European Association for Cardio-Thoracic surgery (EACTS) and the European Society for Vascular Surgery (ESVS). Eur J Cardiothorac Surg. 2019;55(1):133–162.
3. Evangelista A, Isselbacher EM, Bossone E, et al. Insights from the international registry of acute aortic dissection: a 20-year experience of collaborative clinical research. Circulation. 2018;137(17):1846–1860.
4. Galyfos GC, Geropapas GE, Sianou A, et al. Risk factors for postoperative delirium in patients undergoing vascular surgery. J Vasc Surg. 2017;66(3):937–946.
5. Hughes CG, Boncyk CS, Culley DJ, et al. American society for enhanced recovery and perioperative quality initiative joint consensus statement on postoperative delirium prevention. Anesth Analg. 2020;130(6):1572–1590.
6. Oldroyd C, Scholz AFM, Hinchliffe RJ, et al. A systematic review and meta-analysis of factors for delirium in vascular surgical patients. J Vasc Surg. 2017;66(4):1269–1279.
7. Ziman N, Sands LP, Tang C, et al. Does postoperative delirium following elective noncardiac surgery predict long-term mortality? Age Ageing. 2020;49(6):1020–1027.
8. Gleason LJ, Schmitt EM, Kosar CM, et al. Effect of delirium and other major complications on outcomes after elective surgery in older adults. JAMA Surg. 2015;150(12):1134–1140.
9. Gottesman RF, Grega MA, Bailey MM, et al. Delirium after coronary artery bypass graft surgery and late mortality. Ann Neurol. 2010;67(3):338–344.
10. Visser L, Prent A, van der Laan MJ, et al. Predicting postoperative delirium after vascular surgical procedures. J Vasc Surg. 2015;62(1):183–189. doi:10.1016/j.jvs.2015.01.041
11. Ely EW, Inouye SK, Bernard GR, et al. Delirium in mechanically ventilated patients: validity and reliability of the confusion assessment method for the intensive care unit (CAM-ICU). JAMA. 2001;286(21):2703–2710.
12. Marcantonio ER, Goldman L, Mangione CM, et al. A clinical prediction rule for delirium after elective noncardiac surgery. JAMA. 1994;271(2):134–139.
13. Ding H, Luo S, Liu Y, et al. Outcomes of hybrid procedure for type B aortic dissection with an aberrant right subclavian artery. J Vasc Surg. 2018;67(3):704–711.
14. Cai S, Zhang X, Pan W, et al. Prevalence, predictors, and early outcomes of post-operative delirium in patients with Type A aortic dissection during intensive care unit stay. Front Med (Lausanne). 2020;7:572581.
15. van der Wulp K, van Wely M, van Heijningen L, et al. Delirium after transcatheter aortic valve implantation under general anesthesia: incidence, predictors, and relation to long-term survival. J Am Geriatr Soc. 2019;67(11):2325–2330.
16. Abawi M, Nijhoff F, Agostoni P, et al. Incidence, predictive factors, and effect of delirium after transcatheter aortic valve replacement. JACC Cardiovasc Interv. 2016;9(2):160–168.
17. Inouye SK, Westendorp RGJ, Saczynski JS. Delirium in elderly people. Lancet. 2014;383(9920):911–922.
18. Rudarakanchana N, Jenkins MP. Hybrid and total endovascular repair of the aortic arch. Br J Surg. 2018;105(4):315–327.
19. McPherson JA, Wagner CE, Boehm LM, et al. Delirium in the cardiovascular ICU: exploring modifiable risk factors. Crit Care Med. 2013;41(2):405–413.
20. Sellick J, Mergenhagen K, Morris L, et al. Fluoroquinolone-related neuropsychiatric events in hospitalized veterans. Psychosomatics. 2018;59(3):259–266.
21. Krzych LJ, Wybraniec MT, Krupka-Matuszczyk I, et al. Complex assessment of the incidence and risk factors of delirium in a large cohort of cardiac surgery patients: a single-center 6-year experience. Biomed Res Int. 2013;2013:835850.
22. Stransky M, Schmidt C, Ganslmeier P, et al. Hypoactive delirium after cardiac surgery as an independent risk factor for prolonged mechanical ventilation. J Cardiothorac Vasc Anesth. 2011;25(6):968–974.
23. Devlin JW, Skrobik Y, Gélinas C, et al. Clinical Practice guidelines for the prevention and management of pain, agitation/ sedation, delirium, immobility, and sleep disruption in adult patients in the ICU. Crit Care Med. 2018;46(9):e825–73.
24. van der Zanden V, Beishuizen SJ, Swart LM, et al. The effect of treatment of anemia with blood transfusion on delirium: a systematic review. J Am Geriatr Soc. 2017;65(4):728–737.
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
