Back to Journals » International Medical Case Reports Journal » Volume 10
Identifying features of primary fallopian tube carcinoma using magnetic resonance imaging
Authors Winarto H ![]() , Fernando D
, Fernando D ![]()
Received 28 February 2017
Accepted for publication 5 May 2017
Published 26 June 2017 Volume 2017:10 Pages 213—217
DOI https://doi.org/10.2147/IMCRJ.S135836
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Ronald Prineas
Hariyono Winarto,1 Darrell Fernando2
1Gynecology Oncology Division, Department of Obstetrics and Gynecology, Faculty of Medicine, University of Indonesia, Dr. Cipto Mangunkusumo National Referral Hospital, 2Department of Obstetrics and Gynecology, Faculty of Medicine, University of Indonesia, Jakarta, Indonesia
Abstract: Definitively diagnosing primary fallopian tube carcinoma (PFTC) prior to surgery is difficult. In the first working diagnosis, PFTC is often misdiagnosed as ovarian cancer. Pre-operative workups using magnetic resonance imaging (MRI) are capable of differentiating PFTC from epithelial ovarian carcinomas (EOCs). Both the sensitivity and the specificity of MRI for identifying PFTC are high. The presence of a hydrosalpinx is a hallmark of PFTC. On MRI, hydrosalpinges have characteristic sausage-shaped appearances due to intrauterine fluid accumulation and fallopian tube distention. Additionally, MRI scans are superior to computed tomography (CT) scans or ultrasound images in detecting tumor infiltration into surrounding organs. Here, we report a case in which PFTC was pre-operatively misdiagnosed as EOC.
Keywords: primary fallopian tube carcinoma, epithelial ovarian carcinoma, magnetic resonance imaging, pre-operative diagnosis
Introduction
Primary fallopian tube carcinoma (PFTC) is a rare female genital tract malignancy, accounting for just 0.1–1.8% of all gynecological carcinomas.1,2 Because of the rarity of this disease, and the similar histologic and clinical findings with ovarian carcinoma, it is difficult to pre-operatively diagnose PFTC.1,3 Rates of correct pre-operative diagnoses are low, ranging from 0% to 10%.2 Typically, conclusive PFTC diagnoses are made peri- or post-operatively, when PFTC is found incidentally during surgery. Pre-operative PFTC workups include physical examinations, pelvic ultrasounds, Pap smears, measurements of serum CA125 levels, and either computed tomography (CT) scans or magnetic resonance imaging (MRI).1,2 Given that the definitive step of the pre-operative workup is usually CT or MRI scanning, this evidence-based case report aimed to compare the characteristic features of these imaging results.
Case report
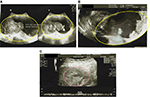
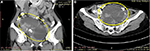
A 47-year-old woman with a parity of three presented to the oncology clinic with a lower abdominal mass that had persisted for 1 year. The woman did not have abdominal pain, vaginal bleeding, or difficulty with micturition or defecation. The patient had no prior use of any type of contraception. Her vital signs were stable, and her Eastern Cooperative Oncology Group (ECOG) performance status was zero. Pelvic examination revealed a non-tender adnexal mass that was 16 cm across and mobile; the uterus appeared normal. Upon ultra-sonographic examination, a cystic mass was identified that contained a solid region. The mass was septated, and gross measurements indicated that it was 10.0 × 5.9 × 13.8 cm in size. The mass had low-resistance blood flow into the solid region (resistance index 0.19), supporting the suspected diagnosis of a malignant bilateral cystic ovarian neoplasm (Figure 1). Abdominal CT scans revealed a lobulated cystic mass with septation and papillary projections on the left adnexum, with gross measurements of 13.5 × 11.8 × 7.6 cm. The mass was suspected to have adhesions to the rectum, bladder, and uterus, suggestive of malignancy. However, the uterus was within normal size limits. Neither lymphadenopathy nor intra-abdominal metastases were found (Figure 2). CA 125 levels, an ovarian tumor biomarker, were measured at 360.5 U/mL; the normal range is 0–35 U/mL.
Next, the patient was assessed for a suspected malignant cystic ovarian neoplasm. She underwent laparotomy to sample the mass, which was preserved in frozen tissue sections. During the procedure, a cystic/solid mass was found with a smooth surface and no adhesions. The mass measured a distance of 20 × 10 × 10 cm from the right fallopian tube and 10 × 2 × 2 cm from the left fallopian tube. No ascites were found. The uterus and both ovaries were within normal size parameters (Figure 3). Bilateral salpingectomy was performed, and specimens were sent for frozen section examination. Pathological analyses of frozen sections determined that the patient had right fallopian tube carcinoma with atypical cells present in the left fallopian tube. The operative procedure progressed to complete surgical staging (including total hysterectomy, bilateral oophorectomy, pelvic and para-aortic lymphadenectomy, and omentectomy). Tubal specimens are shown in Figure 4. The patient was discharged in good physical condition.
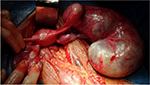  | Figure 3 Intra-operative findings of a normal uterus and both ovaries, with enlargement of both fallopian tubes. |
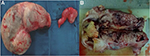  | Figure 4 (A) Macroscopic appearance of both fallopian tubes, (B) solid tubal mass after bisection. |
Pathological analyses performed on paraffin-embedded tissues revealed high-grade serous carcinoma of the right fallopian tube and a left para-tubal cyst. There were no tumor masses present on the omentum, peritoneum, uterus, or any pelvic or para-aortic lymph nodes. Thus, the patient was diagnosed with stage IA high-grade PFTC. Follow-up adjuvant chemotherapy with carboplatin and paclitaxel was planned.
Clinical question and PICO generation
In patients presenting with an adnexal mass, what MRI or CT scan features can definitively identify PFTC? PICO generation is outlined in Table 1.
  | Table 1 PICO generation Abbreviations: MRI, magnetic resonance imaging; CT, computed tomography. |
Methods
Literature search strategies
A secondary review of salient literature was conducted using the PubMed and ScienceDirect search engines on February 5, 2017. The review used the search tool provided on each site, querying the keywords “(CT-scan OR MRI) AND (fallopian AND tube) AND (Cancer OR Carcinoma) NOT ovarian” (Table 2). Search results were filtered by the search engines according to the following criteria: articles published in the past 5 years, human species, and English language. Search strategy, results, and the inclusion and exclusion criteria for further evaluation are outlined in the flowchart in Figure 5.
  | Table 2 Search strategy used in PubMed and Science Direct, conducted on February 5, 2017 Abbreviations: CT, computed tomography; MRI, magnetic resonance imaging. |
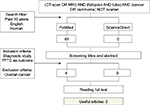  | Figure 5 Flowchart of the secondary literature search strategy. Abbreviations: CT, computed tomography; MRI, magnetic resonance imaging; PFTC, primary fallopian tube carcinoma. |
Selection processes
The titles of the filtered results from PubMed and ScienceDirect queries were screened using the filtering inclusion criteria, discussed earlier. Given the rarity of published cases, the inclusion criteria were expanded to encompass articles published with the past 10 years. Four articles passed preliminary screens. A second screening step was conducted by reading the full text of each article. Finally, two articles were included and reviewed (Table 3).
  | Table 3 Critical appraisal of search results* Note: *Critical appraisal forms are available at http://www.cebm.net/critical-appraisal/. |
Written informed consent was given by the patient for the publication of the case report and images.
Results
Ma et al4 compared potential discriminatory MRI features of PFTC and primary epithelial ovarian carcinoma (EOC). The study was a retrospective design that included 27 PFTC cases and 42 EOC cases. MRI features of PFTC that were significantly different from EOC included maximum diameter (p<0.001), sausage-like shape (p<0.001), solid configuration (p=0.015), homogeneity on T2-weighted images (T2WI; p=0.001), mild or moderate enhancement (p<0.001), presence of hydrosalpinges (p<0.001), and intrauterine fluid accumulation (p=0.001). Laterality, peritoneal implants, lymphadenopathy, and presence of ascites were not statistically different between PFTC and EOC groups. The diagnostic values of the statistically differentiated features are given in Table 4.4
  | Table 4 Accuracy of MRI in characterizing adnexal masses as PFTC Notes: Data in parentheses are the number of masses (n/N). *Homogenous signal on T2-weighted images. Copyright © 2015. Reproduced from John Wiley and Sons. Ma FH, Cai SQ, Qiang JW, Zhao SH, Zhang GF, Rao YM. MRI for differentiating primary fallopian tube carcinoma from epithelial ovarian cancer. J Magn Reson Imaging. 2015;42(1):42–47.4 Abbreviations: MRI, magnetic resonance imaging; PFTC, primary fallopian tube carcinoma; PPV, positive predictive value; NPV, negative predictive value. |
Mao et al5 performed a retrospective analysis of MRI features in nine cases of PFTC. The results were presented as both number of cases and displayed as percentages; diagnostic values were not generated. Sausage-like shapes were present in three cases (33%), well-defined margins were found in six cases (67%), hyper-intensities on T2WI and iso- or hypo-intensities on T1WI were found in all nine cases (100%), and internal papillary projections were seen in four cases (44%).5
Discussion
PFTC is a rare gynecologic malignancy.1,2 Serous carcinoma is the most frequent histological subtype of PFTC (70–90% of tumors), with 50–65% presenting as poorly differentiated (grade 3) tumors.2,6 The patient described here fit these characteristics, presenting with a high-grade serous tubal carcinoma. Interestingly, the classical Latzko’s triad of PFTC symptoms [1) intermittent profuse serosanguineous vaginal discharge, 2) colicky pain relieved by discharge, and 3) an abdominal or pelvic mass] has been found in only 15% of cases.7 Another pathognomonic symptom of PFTC is hydrops tubae profluens, in which shrinkage of an adnexal mass and pain relief occurs after discharge of clear or blood-tinged fluid. However, this only occurs in 5% of cases. Using imaging techniques (ultrasound), PFTC often resembles a mixed solid and cystic mass; therefore, the usual pre-operative diagnosis is an ovarian tumor.4 These combined factors make it difficult to achieve accurate pre-operative diagnoses of PFTC.
During the literature search, no articles were identified that characterized CT scan features useful for identifying PFTC. Only two articles were identified that explored MRI features of PFTC. Ma et al found that the presence of sausage-shaped masses had 100% specificity for PFTC. Furthermore, identifications of these features were useful in differentiating PFTC from EOC. Other MRI features that differentiated PFTC from EOC were presence of hydrosalpinges and intrauterine fluid accumulations.4 Intrauterine fluid may accumulate due to decompression of tubal fluid through the corneal end of the fallopian tube, a region that would normally open into the uterus.8 MRI is also superior to CT scan or ultrasound in detecting tumor invasion into surrounding organs.2
In this case, ultrasonography and CT scan revealed a cystic mass with solid regions in the adnexum. MRI was not performed. However, from CT scans, a large3 sausage-shaped mass with papillary projections was visualized. Given that neither ovary was easily visualized, the mass was initially diagnosed as a suspected malignant cystic ovarian neoplasm.
Even though the pre-operative PFTC diagnosis was missed, adequate staging was obtained. The management of PFTC is the same as ovarian carcinoma. Consistent with current National Comprehensive Cancer Network (NCCN) guidelines, the patient was ultimately diagnosed with stage IA, high-grade PFTC. Adjuvant chemotherapy with the platinum doublet regimen carboplatin and paclitaxel was planned.9 The expected remission rate for patients with stage IV, high-grade PFTC is ~85%.1
Conclusion
Sausage-shaped features, hydrosalpinges, and intrauterine fluid accumulations, identified by MRI, have high sensitivities and specificities for diagnosing PFTC. Small tumor size, solid configurations, T2WI homogeneities, and mild or moderate enhancements are also useful in differentiating PFTC from EOC, albeit with lower sensitivities and specificities.
Disclosure
The authors report no conflicts of interest in this work.
References
Goswami PK, Kerr-Wilson R, McCarthy K. Carcinoma of the fallopian tube. Obstet Gynecol. 2006;8:147–152. | ||
Pectasides D, Pectasides E, Economopoulos T. Fallopian tube carcinoma: a review. Oncologist. 2006;11(8):902–912. | ||
Lau HY, Chen YJ, Yen MS, Chen RF, Yeh SO, Twu NF. Primary fallopian tube carcinoma: a clinicopathologic analysis and literature review. J Chin Med Assoc. 2013;76(10):583–587. | ||
Ma FH, Cai SQ, Qiang JW, Zhao SH, Zhang GF, Rao YM. MRI for differentiating primary fallopian tube carcinoma from epithelial ovarian cancer. J Magn Reson Imaging. 2015;42(1):42–47. | ||
Mao XF, Hu CH, Hu S, Zhu YM, Zhao YY, Shen J. Magnetic resonance imaging features of fallopian tube carcinoma. Int J Gynecol Obstet. 2015;130(2):204–206. | ||
Stewart SL, Wike JM, Foster SL, Michaud F. The incidence of primary fallopian tube carcinoma in the United States. Gynecol Oncol. 2007;107(3):392–397. | ||
Benedet JL, White GW. Malignant tumors of fallopian tube. In: Coppleson M, editor. Gynecologic Oncology. London: Churchill Livingstone; 1981:621–629. | ||
Shaaban A, Rezvani M. Imaging of primary fallopian tube carcinoma. Abdom Imaging. 2013;38:608–618. | ||
Berek JS, Friedlander M, Hacker NF. Epithelial ovarian, fallopian tube, and peritoneal cancer. In: Berek JS, Hacker NF, editors. Gynecologic Oncology. Philadelphia, PA: Lippincott Williams & Wilkins; 2010:493–495. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.