Back to Journals » Clinical Interventions in Aging » Volume 15
Hindi Version of Addenbrooke’s Cognitive Examination III: Distinguishing Cognitive Impairment Among Older Indians at the Lower Cut-Offs
Authors Bajpai S, Upadhyay A, Sati H, Pandey RM, Chaterjee P , Dey AB
Received 3 January 2020
Accepted for publication 13 February 2020
Published 5 March 2020 Volume 2020:15 Pages 329—339
DOI https://doi.org/10.2147/CIA.S244707
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Richard Walker
Swati Bajpai, 1 Ashish Upadhyay, 2 Hemchand Sati, 2 RM Pandey, 2 Prasun Chaterjee, 1 AB Dey 1
1Department of Geriatric Medicine, All India Institute of Medical Sciences, New Delhi, India; 2Biostatistics, All India Institute of Medical Sciences, New Delhi, India
Correspondence: AB Dey
Department of Geriatric Medicine, All India Institute of Medical Sciences, New Delhi 110029, India
Tel +91 11 2659 3639
Email [email protected]
Background: Screening and diagnostic tests provide an objective measure of cognitive performance and also aid in distinguishing mild cognitive impairment (MCI) from major neurocognitive disorder (MNCD). Further, when such tests are culturally and educationally unbiased, it strengthens their diagnostic utility. This study aimed to validate the Hindi version of Addenbrooke’s Cognitive Examination III (ACE-III) in Indian older adults and compare its validity with the Hindi Mini-Mental State Examination (HMSE).
Methods: A sample of 412 consenting older adults visiting a memory clinic was recruited into the study. They were categorized into three groups: healthy controls (n=222), MCI (n=70), and MNCD (n=120). The complete clinical protocol was followed. Hindi ACE-III and HMSE were administered and were statistically analyzed.
Results: The optimal cut-off values to detect MCI and MNCD with ACE-III were 71 and 62 (AUC: 0.849 and 0.884), respectively, which were slightly higher than with HMSE (AUC: 0.822, 0.861). Education- and age-stratified cut-offs were also computed.
Conclusion: Hindi ACE-III has good discriminating power at lower cut-offs than the standard scores in differentiating between MCI and MNCD.
Keywords: ACE-III, Hindi, India, dementia, mild cognitive impairment
Introduction
As the population ages, cognitive disorders, including major neurocognitive disorder (MNCD) or dementia, are becoming more common. According to a WHO report,1 it is one of the major conditions leading to disability and dependency among older people worldwide. It contributes 11.9% of years lived with disability.2 Owing to improved life expectancy globally, the number of cases is expected to escalate further.1 Cognitive disability impacts the person with dementia, as well as their caregivers, family, community, and society.
A diagnosis of dementia, particularly in the preclinical stages, is neither straightforward nor simple. To establish the diagnosis, an assessment will focus on an overall clinical syndrome, which includes a person’s performance on cognitive tests, their behavior, and changes in everyday functioning.3,4 Cognitive evaluation plays a crucial role. In clinical diagnostic criteria5 and research practice, cognitive change is considered the key symptom that is observed in people with dementia.6,7 In addition, the cognitive evaluation includes assessment of a cognitive profile, which includes information about educational attainment, premorbid functioning, language knowledge, sensory impairments (especially visual impairment and deafness), and physical disabilities, which reflect a person’s relative cognitive performance and their brain’s functional integrity.8 Therefore, cognitive assessment aids in the differentiation of mild cognitive impairment (MCI) from dementia, as well as subtyping early in the course of the disease.
With the anticipated increase in research on anti-dementia drugs, the existence of updated and sensitive diagnostic tools becomes a necessity for early diagnosis.9 The most widely used cognitive tools include the Mini-Mental State Examination (MMSE),10 Montreal Cognitive Assessment (MoCA),11 and Clinical Dementia Rating Scale (CDR).12 However, these tests have their limitations. For example, MMSE gives more weight to orientation, and is barely sensitive in detecting MCI.13 In contrast, MoCA evaluates a wide spectrum of cognitive abilities, and hence, is more sensitive to detecting MCI;14 however, it has poor predictive accuracy in identifying domain-specific cognitive difficulty.15 Slightly more elaborate and sensitive comprehensive tools are also available, such as Addenbrooke’s Cognitive Examination (ACE)16 and the Clinical Dementia Rating (CDR) scale.12 These tests are particularly advantageous in differential diagnosis, as they include additional cognitive and behavioral parameters.
The ACE and its subsequent iterations23 have good psychometric properties for diagnosing Alzheimer’s disease and other types of dementia.17,18 The most recently developed version is ACE-III, which has been initially validated in English and in Spanish.19–21 Its good diagnostic utility has prompted many translations into other languages. It has been found to be versatile as it helps in establishing a cognitive profile; it is also reported to have good potential to detect cognitive impairment in the early stages, which may aid in differential diagnosis.21 Moreover, ACE-III has also been validated in other conditions, such as brain injury, stroke, and Parkinsonism,18,22 yielding excellent diagnostic utility and helping clinicians to make more precise decisions about cognitive impairment and dementia. Several studies24,25 have shown that the cut-off values of ACE-III are influenced by socio-demographic variables, such as age,26 education,20 and language.27 Such factors play an important role when interpreting the suggested cut-off values as they affect the diagnostic accuracy in diverse populations.
Hence, to avoid any type I and II errors,28 the present study aimed to investigate the validity of the Hindi ACE-III in detecting MCI and MNCD in a memory clinic setting, establish optimal cut-off values, and explore the impact of age and educational attainment on the performance of the ACE-III.
Methods
A prospective cross-sectional study was conducted in which the Hindi ACE-III was validated against the Hindi MMSE. It was hypothesized that the Hindi ACE-III29 would be more sensitive than the conventional MMSE30 in detecting early cognitive decline (MCI) and MNCD in an Indian population. Our aim, therefore, was to find the optimal cut-off scores for the Hindi ACE-III to distinguish between MCI and MNCD, and compare it with that of the Hindi Version of the MMSE (HMSE). The sample was also stratified on age and educational attainment (years of education) to study the correlations of these factors with cognitive performance (Table 1). The present study was approved by the All India Institute of Medical Sciences, New Delhi, India ethical committee (IEC-667/06.09.2019). The study was conducted in accordance with the Declaration of Helsinki.
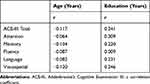 |
Table 1 Correlation Coefficients of ACE-III Test and Its Subdomains by Age and Education |
Participants
In total, 412 participants were included in this study (Table 2). Written informed consent was obtained from all participants before enrolling them in the study. This study was carried out over a span of 2 years (September 2017 to October 2019). There was a total of 438 eligible participants; of these, 26 were excluded: 18 cases either refused to participate or did not attend with a reliable informant, and eight healthy controls did not consent to participate. A reliable informant is defined as a person who has known the participant closely for at least 10 years. All participants underwent clinical testing, laboratory testing, and cognitive evaluation at the dedicated Memory Clinic of the Department of Geriatric Medicine, All India Institute of Medical Sciences, New Delhi, India. All diagnoses were made as per the existing diagnostic criteria. All participants with MCI met Petersen’s 2011 criteria31 of 1) concern about their cognitive deficit in comparison to their previous level; 2) performance that is lower than their age- and education-matched normative; 3) no or minimal impairment in their basic and instrumental activities of daily living (ADL); and 4) not meeting DSM-V-2013 criteria32 for dementia. All vascular dementia diagnoses met the NINDS-AIREN33 criteria for probable vascular dementia, and Alzheimer’s disease diagnosis was based on NIA-AA 2011 criteria.34 Individuals above 60 years of age, with no reported major systemic or psychiatric illness, and with a score of ≥25 on the HMSE were recruited as healthy controls (HC). They were consenting volunteers and informants who accompanied the patients to the geriatric department.
 |
Table 2 Comparison of Baseline Socio-Demographic and Clinical Data, ACE-III Total and Subscores in Three Groups: HC, MCI, and MNCD |
The exclusion criteria for all groups were 1) delirium, 2) the presence of diagnosed major psychiatric diseases, 3) an inability to comprehend the Hindi language, 4) absence of a reliable informant, for participants in the MCI and MNCD groups only, or 5) being unable to complete the assessment. To prevent biases, the cognitive protocol was usually performed by different neuropsychologists and geriatricians, who were blinded to grouping. Moreover, to avoid any practice effect, the cognitive tests (HMSE and ACE-III) were administered sparsely within the clinical and laboratory testing schedule, which were completed within a week.
A complete baseline demographic and clinical profile was completed. A complete protocol was completed, which included the HMSE, ACE-III-Hindi, Instrumental Activities of Daily Living – Elderly (IADL-E),35 Abe’s Behavioral and Psychological Symptoms of Dementia (BPSD),36 Geriatric Depression Scale (GDS),37 and Kingston Standardized Behaviour Assessment (KBSA).38 Since the MMSE is most frequently studied and well known, we selected this test as most suitable for our algorithm. When all examinations had been completed, the clinical diagnosis was established by multidisciplinary consensus between the geriatricians and a neuropsychologist, after reviewing the clinical and cognitive profile. The clinical diagnosis was established independently, without relying on performance on the Hindi ACE-III. In total, 412 participants were finally categorized into three groups: MNCD group (n=120), MCI group (n=70), and HC group (n=222).
Instruments
The ACE-III is a simple, brief, paper-and-pencil-based measure of global cognitive function. It covers five cognitive domains. Each cognitive domain measures specific cognitive ability, contributing to the overall score. The scale is 100 points. Each domain carries different points, as follows: 18 points are allocated to the attention and orientation domain, 26 points to the memory domain, 14 points to the verbal fluency domain, 26 points to the language domain, and 16 points to the visuospatial domain. A higher score is interpreted as better cognitive ability. The ACE-III is available in many languages, including Tamil, Hindi, Indian English, Kannada, Telugu, Malay, Urdu, and Marathi.39,40 The cultural adaptation of ACE-III in Urdu and Hindi language was used in the present study. This test is freely available at the following website: https://sydney.edu.au/brain-mind/resources-for-clinicians/dementia-test.html.29
There are no published validation studies that used the adapted version of ACE-III in the Hindi language. A few items on the original ACE-III were modified and adapted for better suitability among the Indian population. In anterograde memory, recall, and recognition items, the address and name were replaced as per the Indian context. In the retrograde memory item, the last question was replaced as per Indian historical events, where we asked “who is the father of the nation?” In the verbal fluency item, the participants were asked to name as many words as possible starting with the Hindi alphabet letter “प” during 1 minute. In the repetition item, Hindi phrases were used. The object naming part of the domain “language” has 12 pictures, of which eight pictures were replaced considering their cultural appropriateness; these were goat, giraffe, sickle, drum, pig, umbrella, candle, and flag. Following this, the four questions on the comprehension part of the language domain were revised: “Point to the one which is used in rain”, “Point to the one which emits light”, “Point to the one which is associated with agricultural farming”, and, “Point to the one which is found in the desert”. Finally, in the reading item, participants were asked to read aloud five Hindi alphabets instead of English words.
Clinical Protocol
The first component comprised a thorough clinical examination. This included a detailed patient history-taking (socio-demographics and educational background), physical examination, and evaluation of the presence of comorbidities (diabetes mellitus, hypertension, coronary artery disease, thyroid function, vitamin B12 deficiency, and vitamin D deficiency), if any. The second component comprised a cognitive evaluation. All patients underwent cognitive testing using the HMSE.30 This assesses all of the cognitive functions that are usually found to be impaired in neurocognitive disorders (orientation, memory, language, visuospatial ability, and attention). Besides the cognitive evaluation, the patients were assessed for any orbitofrontal signs, such as depression and behavioral abnormalities. These were assessed using the GDS37 and KSBA.38 Any participant with GDS score ≥8and KSBA ≥26 was considered for detailed neuropsychiatric assessment. The third component was functional evaluation, which included the assessment of everyday functionality using the IADL-E.35 Any participant with a score of ≥16 on Cognitive disability index (CDI) was considered to have impaired ADL functioning. After all the componential assessments, the reports were collated and discussed thoroughly by the team of the Memory Clinic to make a diagnosis. The cognitive parameter was taken as the primary variable for clinical and statistical analysis.
Statistical Analysis
Data were managed on Excel spreadsheets. All statistical analysis was performed using Stata 14.0 software. Comparison of the three groups was done using one-way analysis of variance (ANOVA) and Bonferroni correction. The chi-squared test was administered to determine the statistical significance in categorical data. Skewed data were compared using the Kruskal–Wallis test followed by multiple comparisons using Dunn's test with Bonferroni correction. Multiple regression analysis was conducted to examine possible relationships of the socio-demographic variables and clinical variables with the total ACE-III score and subdomain scores. A p-value <0.05 was accepted as significant. To determine the sensitivity and specificity at different cut-offs, receiver operating characteristics (ROC) curve analysis was applied. The area under the curve (AUC) was also estimated with sensitivity and specificity, followed by positive predictive values (PPVs) and negative predictive values (NPVs) for each optimal cut-off score. We selected the optimal cut-off, which corresponds to higher specificity and provides maximal accuracy in distinguishing dementia from HC and MCI. This method was chosen considering the aim of the study, in that the adapted tool should be able to rule in cognitive impairment, ie, the test should have higher specificity. Missing a case of cognitive impairment would be more serious than the false-positive case. Moreover, further intervention would have no adverse effects. Therefore, we selected the cut-off which corresponds to higher specificity. In addition to this, the Youden index was calculated (Table 3).
 |
Table 3 Cut-Off Values, Sensitivity, Specificity, AUC, and Youden Index of the ACE-III for Identifying MCI and MNCD |
Results
At baseline, on demographic variables, no significant differences were seen between the MCI, MNCD, and HC groups except on age (p≤0.023) (Table 2). On average, participants were older in the MNCD group than in the MCI or HC group. Among the three groups, which were matched for education and gender, the total scores and subscores of the Hindi ACE-III was ranked as follows: healthy control group > MCI group > MNCD group. All the differences were found to be statistically significant (p<0.001).
The total score and the subscores of Hindi ACE-III were found to be significantly higher in the HC group than in the MCI or MNCD group (p<0.0001). Statistical analyses showed significant differences between all groups in the domains of attention and orientation (p≤0.001), memory (p<0.0001), fluency (p<0.0001), language (p≤0.001), and visuospatial ability (p≤0.001) (Table 2).
The ROC curve analysis suggested a cut-off score of 71 (sensitivity=76.13%, specificity=78.42%; AUC: 0.849) as the optimum to differentiate MCI from HC. On the other hand, 62 was found to be optimum cut-off for identifying cases with MNCD, with a sensitivity of 81.59% and a specificity of 80.00%, with the area under the curve (AUC: 0.884) (Table 3). The Hindi ACE-III was found to have slightly more discriminating power than HMSE among both the group comparisons (HC vs MCI: 0.822; MCI vs MNCD: 0.861) (Figure 1).
Age- and education-stratified norms were also computed (Tables 4 and 5). Both variables had a significant impact on the total score and domain scores of the Hindi ACE-III (Table 1). As the age bracket increased (60–69, 70–79, and ≥80 years), the Hindi ACE-III total scores decreased with constant sensitivity and specificity in distinguishing between HC and MCI [72/100 (75.63%, 77.91%; AUC 0.843); 70/100 (75.90%, 76.92%; AUC 0.8371); 68/100 (75.00%, 82.05%; AUC 0.866)] and between MCI and MNCD [64/100 (79.87%, 80.39; AUC 0.896); 63/100 (79.61%, 80.00%); 55/100 (77.14%, 75.00%; AUC 0.883)], respectively (Table 4).
 |
Table 4 Cut-Offs, Sensitivity, and Specificity of the ACE-III Test and Its Subdomains for Different Age Bands for MCI and MNCD Groups |
 |
Table 5 Cut-Offs, Sensitivity, and Specificity of the ACE-III Test and Its Subdomains for Different Education Bands for MCI and MNCD Groups |
For the education variable (Tables 1 and 5), there were four educational categories (0, 1–5, 6–9, and ≥10 years), and a significant difference in Hindi ACE-III score was seen with increasing years of education. For HC vs MCI, the optimal score increased from 53 to 74 following the educational categories, while for MCI vs MNCD, the optimal cut-off for 0 years of education category was 43, followed by 56, 63, and 68 for 1–5, 6–9, and ≥10 years, respectively.
 |
Figure 1 ROC curve of the Addenbrooke’s Cognitive Examination III (Hindi version) and Hindi Mental State Examination (HMSE): (A) mild cognitive impariment; (B) major neurocognitive disorder. |
Discussion
Timely and early diagnosis of MNCD or dementia has been recognized as being key in the improvement of dementia services, and is supported by clinical guidelines and national dementia strategies around the globe. Early identification of such cognitive changes facilitates timely psychosocial interventions, which have the potential to improve the quality of life of a person with dementia and their caregivers.41
As per the reported literature,39 cognition assessment plays a crucial role in screening and detecting MNCD. It adds further evidence to the clinical assessment and investigations. Therefore, brief and precise cognitive tests that can reliably tap cognitive changes over time are essential for use in cognitive and dementia clinics. A number of validated cognitive scales and tests is used in general practice. One of the widely used scales is the MMSE, which has established ability to distinguish dementia from cognitive healthy controls.10 However, the MMSE has drawbacks in that it lacks the potential to detect early cognitive changes (MCI) and to screen multiple cognitive domains.13 Other rating scales, such as the CDR and the Rowland Universal Dementia Assessment Scale (RUDAS), have failed to be used widely for detecting early changes (MCI) because of their complexity, lack of sensitivity/specificity in comparison to the ACE-III, and/or the effects of other demographic causes such as educational ability, language, hearing ability, and culture.42
Therefore, the aims of the present study were to evaluate the validity of the Hindi version of the ACE-III scale and to study the effects of age and education on identifying the MCI and dementia groups.
The Hindi ACE-III was found to be a slightly more sensitive (76.13%) and specific (78.42%) test to distinguish between HC and MCI (AUC: 0.849) and between MCI and MNCD (81.59%, 80.00%; AUC: 0.884) in this Hindi-speaking sample than the HMSE (AUC: 0.822; 0.861) (Table 3). This result is consistent with previous studies.16,24 The optimal cut-off points for identifying MCI from HC, and MNCD from MCI were 71 and 62, respectively, which were found to be lower than the established cut-off values of the original ACE-III19 and ACE-R.16 Similarly, in Spanish20 and Thai,43 an identical lower cut-off values was reported. There are also studies in other languages, such as Chinese,44 Cantonese,45 and Korean,46 where lower cut-offs were reported. Such effects can be attributed to socio-demographic factors, as extensively supported by neuropsychological batteries.47 Similar trends can also be seen in the present study. Our findings (Table 1) showed that age was inversely correlated while years of education was positively correlated with ACE-III score performance. Such a trend was also evident in other studies.20,26,48 Another similar study49 showed that in a younger age group (60–69 years) with higher educational attainment (>10 years of schooling), the majority of the population scored above 80 on the ACE-III. In contrast, a completely opposite trend was seen with the cut-off value below 80.24,26,43
Thus, level of educational attainment has been observed to have a substantial effect on cognitive performance. The presence of items dependent on the level of education or literacy, such as the use of irregular words, phonemic verbal fluency, tasks, and constructional abilities, consequently affect the cognitive report.
In comparison to the HMSE, the Hindi ACE-III was found to be more sensitive in distinguishing MNCD from HC. In the present study, the AUC (0.884) of the Hindi ACE-III was slightly higher than that of the HMSE (0.861). Similar findings were reported in other languages as well.26,43 In addition, the subdomains of ACE-III cover more cognitive abilities than the HMSE, which makes it more advantageous in detecting underlying cognitive difficulties in dementia and its other subtypes, such as frontotemporal dementia. The high sensitivity and specificity obtained in the present study and other studies19,26 further reinforce its usefulness.
Similarly to the Japanese22 and Thai41 versions of ACE-III, this study showed adequate diagnostic accuracies of the Hindi ACE-III in discriminating MCI from HC (AUC: 0.994) compared to other validation studies of ACE-R.44 However, upon further scrutiny, an effect of education was evident. With increasing educational level, the cut-off varies (Table 5). Such a trend can be seen in different studies as well. In Chinese44 and Japanese22 research, the study population had more than 10 years of education, with lower scores on ACE-III evaluation, while in the Korean46 study, the opposite was found. With less than 10 years of education, the AUC (0.697), sensitivity (67%), and specificity (65%) of ACE-R were much lower than in the present study (sensitivity 81.92%, specificity 78.26%; AUC0.890). Such studies show effects of age and years of education on the performance of ACE-III. Nonetheless, the AUC value of ACE-III was still larger than that of the HMSE, indicating its better diagnostic accuracy.
There are some drawbacks in this study. First, the patients with MNCD were not divided into different groups (such as Alzheimer's disease, vascular dementia, or frontotemporal dementia). Such subtyping is only possible, if radiological and detailed neuropsychological investigations of all the MNCD participants have been completed. Owing to extensive patient load, the scarcity of neuropsychologists in India, and the lack of rigorous training, such a protocol was not found to be feasible at the tertiary care hospital. Previous studies reported that ACE-III could discriminate between different causes of dementia.46,50 Thus, the current findings are limited to institutional referrals and may not be pertinent for community-based populations. Third, normative data for different age and educational brackets were used in this study; however, the sample size was too small in each educational and age bracket to make any affirmative claims. Further studies on the Hindi ACE-III should be conducted in other populations with lower educational background. Finally, other than MMSE, the Hindi ACE-III should be validated against other cognitive screening tests, such as the Montreal Cognitive Assessment, CDR, and RUDAS, which are more comprehensive. However, we did not use any of these tests as we found that they either involve subjectivity, or require extensive training, or lack cultural appropriateness to be used in Hindi-speaking populations. Hence, the HMSE was seen as an apt test for use in the present study.
Conclusion
The Hindi ACE-III is a widely used cognitive test, which has reasonable capacity to detect early cognitive changes. Unlike other screening tests, it provides a brief robust multi-component cognitive profile. Currently, in addition to the English version, this test has been adapted and validated in other Asian languages (Chinese, Thai, Japanese, and Malay). It has been demonstrated that the ACE-III has significant correlations with age and education, which will eventually help in yielding an optimal cut-off for and in detecting true cases. Similar results were found in the current study. Lower cut-offs yielded more sensitive scores in the north Indian Hindi-speaking population, which were found to be of good clinical utility in the memory clinic setting.
Acknowledgments
This study was carried out at the Memory Clinic of the Department of Geriatric Medicine, All India Institute of Medical Sciences, New Delhi. Special acknowledgment goes to the extended team of the Memory Clinic, which includes geriatricians (Dr Vijay, Dr Sunny, Dr Rishav, Dr Manick, Dr Promod, Dr Abhijit, Dr Ananta, Dr Ambica, and Dr Bharti), nurses (Ms Priti and Mr Mohit), and the secretarial staff.
Disclosure
The authors report no conflicts of interest in this work.
References
1. World Health Organization. Global Action Plan on the Public Health Response to Dementia 2017–2025. World Health Organization; March 2017.
2. Prince M, Guerchet M, Prina M. The Epidemiology and Impact of Dementia: Current State and Future Trends. Geneva: World Health Organization; 2015.
3. Maestre GE. Assessing dementia in resource-poor regions. Curr Neurol Neurosci Rep. 2012;12(5):511–519. doi:10.1007/s11910-012-0300-9
4. Dhedhi SA, Swinglehurst D, Russell J. Timely diagnosis of dementia: what does it mean? A narrative analysis of GPs’ accounts. BMJ Open. 2014;4(3):e004439. doi:10.1136/bmjopen-2013-004439
5. Simpson JR. DSM-5 and neurocognitive disorders. J Am Acad Psychiatry Law. 2014;42(2):159–164.
6. Robinson L, Tang E, Taylor JP. Dementia: timely diagnosis and early intervention. BMJ. 2015;16(350):h3029. doi:10.1136/bmj.h3029
7. Sheehan B. Assessment scales in dementia. Ther Adv Neurol Disord. 2012;5(6):349–358. doi:10.1177/1756285612455733
8. Guthrie DM, Davidson JG, Williams N, et al. Combined impairments in vision, hearing and cognition are associated with greater levels of functional and communication difficulties than cognitive impairment alone: analysis of interRAI data for home care and long-term care recipients in Ontario. PLoS One. 2018;13(2):e0192971. doi:10.1371/journal.pone.0192971
9. Tsoi KK, Chan JY, Hirai HW, Wong SY, Kwok TC. Cognitive tests to detect dementia: a systematic review and meta-analysis. JAMA Intern Med. 2015;175(9):1450–1458. doi:10.1001/jamainternmed.2015.2152
10. Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”: a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12(3):189–198. doi:10.1016/0022-3956(75)90026-6
11. Nasreddine ZS, Phillips NA, Bédirian V, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53(4):695–699.
12. Morris JC. Current vision and scoring rules The Clinical Dementia Rating (CDR). Neurology. 1993;43:2412–2414. doi:10.1212/WNL.43.11.2412-a
13. Henneges C, Reed C, Chen YF, Dell’Agnello G, Lebrec J. Describing the sequence of cognitive decline in Alzheimer’s disease patients: results from an observational study. J Alzheimers Dis. 2016;52(3):1065–1080. doi:10.3233/JAD-150852
14. Roalf DR, Moberg PJ, Xie SX, Wolk DA, Moelter ST, Arnold SE. Comparative accuracies of two common screening instruments for classification of Alzheimer’s disease, mild cognitive impairment, and healthy aging. Alzheimers Dement. 2013;9(5):529–537. doi:10.1016/j.jalz.2012.10.001
15. Moafmashhadi P, Koski L. Limitations for interpreting failure on individual subtests of the Montreal Cognitive Assessment. J Geriatr Psychiatry Neurol. 2013;26(1):19–28. doi:10.1177/0891988712473802
16. Mioshi E, Dawson K, Mitchell J, Arnold R, Hodges JR. The Addenbrooke’s Cognitive Examination Revised (ACE‐R): a brief cognitive test battery for dementia screening. Int J Geriatr Psychiatry. 2006;21(11):1078–1085. doi:10.1002/(ISSN)1099-1166
17. Mathuranath PS, Nestor PJ, Berrios GE, Rakowicz W, Hodges JR. A brief cognitive test battery to differentiate Alzheimer’s disease and frontotemporal dementia. Neurology. 2000;55(11):1613–1620. doi:10.1212/01.wnl.0000434309.85312.19
18. So M, Foxe D, Kumfor F, et al. Addenbrooke’s cognitive examination III: psychometric characteristics and relations to functional ability in dementia. J Int Neuropsychol Soc. 2018;24(8):854–863. doi:10.1017/S1355617718000541
19. Hsieh S, Schubert S, Hoon C, Mioshi E, Hodges JR. Validation of the Addenbrooke’s Cognitive Examination III in frontotemporal dementia and Alzheimer’s disease. Dement Geriatr Cogn Disord. 2013;36(3–4):242–250. doi:10.1159/000351671
20. Matias-Guiu JA, de Bobadilla RF, Escudero G, et al. Validación de la versiónespañola del test Addenbrooke’s Cognitive Examination III para el diagnóstico de demencia. Neurologia. 2015;30(9):545–551. doi:10.1016/j.nrl.2014.05.004
21. Matias-Guiu JA, Fernández-Bobadilla R, Fernández-Oliveira A, et al. Normative data for the Spanish version of the Addenbrooke’s Cognitive Examination III. Dement Geriatr Cogn Disord. 2016;41(5–6):243–250. doi:10.1159/000445799
22. Takenoshita S, Terada S, Yoshida H, et al. Validation of Addenbrooke’s cognitive examination III for detecting mild cognitive impairment and dementia in Japan. BMC Geriatr. 2019;19(1):123. doi:10.1186/s12877-019-1120-4
23. Amaral-Carvalho V, Caramelli P. Normative data for healthy middle-aged and elderly performance on the Addenbrooke Cognitive Examination-Revised. Cogn Behav Neurol. 2012;25(2):72–76. doi:10.1097/WNN.0b013e318259594b
24. Nieto A, Galtier I, Hernández E, Velasco P, Barroso J. Addenbrooke’s Cognitive Examination-Revised: effects of education and age. Normative data for the Spanish speaking population. Arch Clin Neuropsychol. 2016;31(7):811–818. doi:10.1093/arclin/acw057
25. Mathuranath PS, Cherian JP, Mathew R, George A, Alexander A, Sarma SP. Mini mental state examination and the Addenbrooke’s cognitive examination: effect of education and norms for a multicultural population. Neurol India. 2007;55(2):106. doi:10.4103/0028-3886.32779
26. Wang BR, Ou Z, Gu XH, Wei CS, Xu J, Shi JQ. Validation of the Chinese version of Addenbrooke’s Cognitive Examination III for diagnosing dementia. Int J Geriatr Psychiatry. 2017;32(12):e173–9. doi:10.1002/gps.4680
27. Mirza N, Panagioti M, Waheed MW, Waheed W. Reporting of the translation and cultural adaptation procedures of the Addenbrooke’s Cognitive Examination version III (ACE-III) and its predecessors: a systematic review. BMC Med Res Methodol. 2017;17(1):141. doi:10.1186/s12874-017-0413-6
28. Bruno D, Vignaga SS. Addenbrooke’s cognitive examination iii in the diagnosis of dementia: a critical review. Neuropsychiatr Dis Treat. 2019;15:441. doi:10.2147/NDT.S151253
29. Warsi S. Cultural adaptation of the Addenbrooke’s Cognitive Examination III (ACE-III) in Urdu/Hindi,Bradford District Care NHS foundation trust. Unpublished. Available from: https://sydney.edu.au/brain-mind/resources-for-clinicians/dementia-test.html. Accessed 7 March 2013.
30. Ganguli M, Ratcliff G, Chandra V, et al. A Hindi version of the MMSE: the development of a cognitive screening instrument for a largely illiterate rural elderly population in India. Int J Geriatr Psychiatry. 1995;10(5):367–377. doi:10.1002/(ISSN)1099-1166
31. Albert MS, DeKosky ST, Dickson D, et al. The diagnosis of mild cognitive impairment due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement. 2011;7(3):270–279. doi:10.1016/j.jalz.2011.03.008
32. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM-5). American Psychiatric Pub; May 2013:22
33. Román GC, Tatemichi TK, Erkinjuntti T, et al. Vascular dementia: diagnostic criteria for research studies: report of the NINDS-AIREN International Workshop. Neurology. 1993;43250–43260.
34. McKhann GM, Knopman DS, Chertkow H, et al. The diagnosis of dementia due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement. 2011;7(3):263–269. doi:10.1016/j.jalz.2011.03.005
35. Mathuranath PS, George A, Cherian PJ, Mathew R, Sarma PS. Instrumental activities of daily living scale for dementia screening in elderly people. Int Psychogeriatr. 2005;17(3):461–474. doi:10.1017/S1041610205001547
36. Abe K, Yamashita T, Hishikawa N, et al. A new simple score (ABS) for assessing behavioral and psychological symptoms of dementia. J Neurol Sci. 2015;350(1–2):14–17. doi:10.1016/j.jns.2015.01.029
37. Sheikh JI, Yesavage JA, Brink TL. Clinical Gerontology: a guide to Assessment and Intervention. Geriatric Depression Scale (GDS): recent evidence and development of a shorter version. Clin Gerontologist. 1986;5(1–2):165–173.
38. Hopkins RW, Kilik LA, Day DJ, Bradford L, Rows CP. The Kingston standardized behavioural assessment. Am J Alzheimers Dis Other Dement. 2006;21(5):339–346. doi:10.1177/1533317506292576
39. Porrselvi AP, Shankar V. Status of cognitive testing of adults in India. Ann Indian Acad Neurol. 2017;20(4):334. doi:10.4103/aian.AIAN_107_17
40. Kan KC, Subramaniam P, Shahrizaila N, Kamaruzzaman SB, Razali R, Ghazali SE. Validation of the Malay Version of Addenbrooke’s Cognitive Examination III in detecting mild cognitive impairment and dementia. Dement Geriatr Cogn Dis Extra. 2019;9(1):66–76. doi:10.1159/000495107
41. de Vugt ME, Verhey FR. The impact of early dementia diagnosis and intervention on informal caregivers. Prog Neurobiol. 2013;1(110):54–62. doi:10.1016/j.pneurobio.2013.04.005
42. Crawford S, Whitnall L, Robertson J, Evans JJ. A systematic review of the accuracy and clinical utility of the Addenbrooke’s Cognitive Examination and the Addenbrooke’s Cognitive Examination—Revised in the diagnosis of dementia. Int J Geriatr Psychiatry. 2012;27(7):659–669. doi:10.1002/gps.2771
43. Charernboon T, Jaisin K, Lerthattasilp T. The Thai version of the Addenbrooke’s cognitive examination III. Psychiatry Investig. 2016;13(5):571. doi:10.4306/pi.2016.13.5.571
44. Fang R, Wang G, Huang Y, et al. Validation of the Chinese version of Addenbrooke’s cognitive examination-revised for screening mild Alzheimer’s disease and mild cognitive impairment. Dement Geriatr Cogn Disord. 2014;37(3–4):223–231. doi:10.1159/000353541
45. Wong LL, Chan CC, Leung JL, et al. A validation study of the Chinese-Cantonese addenbrooke’s cognitive examination revised (C-ACER). Neuropsychiatr Dis Treat. 2013;9:731.
46. Kwak YT, Yang Y, Kim GW. Korean Addenbrooke’s Cognitive Examination Revised (K‐ACER) for differential diagnosis of Alzheimer’s disease and subcortical ischemic vascular dementia. Geriatr Gerontol Int. 2010;10(4):295–301. doi:10.1111/ggi.2010.10.issue-4
47. Rosli R, Tan MP, Gray WK, Subramanian P, Chin AV. Cognitive assessment tools in Asia: a systematic review. Int Psychogeriatr. 2016;28(2):189–210. doi:10.1017/S1041610215001635
48. Stott J, Scior K, Mandy W, Charlesworth G. Dementia screening accuracy is robust to premorbid IQ variation: evidence from the Addenbrooke’s Cognitive Examination-III and the Test of Premorbid Function. J Alzheimers Dis. 2017;57(4):1293–1302. doi:10.3233/JAD-161218
49. Torralva T, Roca M, Gleichgerrcht E, Bonifacio A, Raimondi C, Manes F. Validation of the Spanish version of the Addenbrooke’s Cognitive Examination-Revised (ACE-R). Neurology. 2011;26(6):351–356. doi:10.1016/j.nrl.2010.10.013
50. Alexopoulos P, Ebert A, Richter-Schmidinger T, et al. Validation of the German revised Addenbrooke’s cognitive examination for detecting mild cognitive impairment, mild dementia in Alzheimer’s disease and frontotemporal lobar degeneration. Dement Geriatr Cogn Disord. 2010;29(5):448–456. doi:10.1159/000312685
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
