Back to Journals » International Medical Case Reports Journal » Volume 7
Herpes simplex virus type 2-associated recurrent aseptic (Mollaret's) meningitis in genitourinary medicine clinic: a case report
Authors Abou-Foul AK, Buhary T, Gayed S
Received 27 November 2013
Accepted for publication 24 December 2013
Published 3 March 2014 Volume 2014:7 Pages 31—33
DOI https://doi.org/10.2147/IMCRJ.S58377
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Ahmad K Abou-Foul, Thajunisha M Buhary, Sedki L Gayed
Department of Genitourinary Medicine, Royal Blackburn Hospital, East Lancashire Hospitals NHS Trust, Blackburn, UK
Introduction: Cases of idiopathic recurrent benign aseptic meningitis were first described by Mollaret. Today, herpes simplex virus (HSV) is considered the cause of most cases of Mollaret's meningitis.
Case report: A 40-year-old male was referred to our genitourinary medicine clinic with recurrent genital herpetic lesions. He had HSV-2-positive genital ulcers 8 years earlier. One year after the first infection, he developed severe recurrent attacks of headache associated with meningitis symptoms. The results of all radiological and biochemical tests were normal, but the patient reported a correlation between his attacks and genital herpes flare-ups. We diagnosed the patient with Mollaret's meningitis and started him on continuous suppressive acyclovir therapy, which resulted in marked clinical improvement.
Discussion: Mollaret's meningitis is a rare form of idiopathic recurrent aseptic meningitis that has a sudden onset, short duration, and spontaneous remission with unpredictable recurrence. We believe that the presence of concurrent or recurrent mucocutaneous herpetic lesions can aid its diagnosis, prior to which, affected patients usually have many unnecessary investigations and treatments. Therefore, detailed sexual history should be sought in all patients with aseptic meningitis, and clinicians should also ask about history of recurrent headaches in all patients with recurrent herpetic anogenital lesions. Continuous suppressive acyclovir therapy may reduce the frequency and severity of attacks and can dramatically improve lifestyle.
Keywords: HSV-2 virus, acyclovir, Mollaret's meningitis, recurrent aseptic meningitis, HSV-2 virus, viral meningitis, acyclovir
Introduction
Mollaret was the first to describe a rare form of meningitis that presented as recurrent and brief self-limiting attacks of fever, headache, vomiting, and meningeal irritation with no obvious etiology.1 Subsequently, all cases of idiopathic recurrent aseptic meningitis (RAM) were collectively called Mollaret’s meningitis.2 Today, herpes simplex virus (HSV) type 2 (HSV-2) is thought to be the commonest cause of Mollaret’s meningitis, underlying up to 84% of cases,3 although other infectious and noninfectious causes have also been described.2 However, its diagnosis remains challenging despite the use of new sophisticated diagnostic tools, and some authors have advised restricting the term Mollaret’s meningitis to cases with no confirmed etiology.2 The frequency and severity of attacks are variable, and severe forms can be incapacitating if appropriate antiviral therapies are not promptly used. Here, we describe a rare case of a patient with RAM who presented with recurrent and severe self-limiting bouts of headache and responded well to suppressive antiviral therapy.
Case presentation
A 40-year-old man was referred to our genitourinary medicine clinic with chronic recurrent herpetic genital ulcers. His first attack occurred 8 years earlier following unprotected vaginal sexual intercourse. Swabs taken from these initial lesions tested positive for HSV-2 and negative for HSV type 1 (HSV-1). Treponema enzyme immunoassay (EIA) and Neisseria gonorrhea culture were also negative. The patient had a past medical history of migraines that started in adulthood as unilateral headaches and were controlled by triptans and simple analgesics.
One year after his first genital infection, the patient started to develop severe holocranial headaches with distinct characteristics, which failed to respond to usual migraine therapy. The headaches were associated with neck stiffness, mild photophobia, and bilateral pedal paresthesia, but there was no fever, vomiting, or other cranial or radicular neuropathy symptoms. The patient went to the emergency department many times, but his biochemical markers and a computed tomography brain scan were normal. The patient repeatedly refused diagnostic lumbar puncture, but serologic assays (HSV-1, CMV [cytomegalovirus], VZV [varicella-zoster virus], and human immunodeficiency virus) were negative apart from a positive HSV-2 immunoglobulin G titer.
He was clinically diagnosed with meningitis and commenced on empirical parenteral ceftriaxone and acyclovir, to which he responded very well. His attacks often recurred two to three times per month and were usually self-limiting in 3–5 days, regardless of their severity, with no residual functional impairment. The attacks were often triggered by emotional stress or heat and were sometimes preceded by genital herpetic eruptions, for which he was referred to our clinic. The distinctive correlation between his meningitic recurrent headaches and recurrent genital lesions as well as his normal previous diagnostic tests raised our suspicion of a possible Mollaret’s meningitis diagnosis. We placed the patient on a continuous suppressive oral acyclovir therapy regimen (400 mg twice daily), which reduced the frequency of his headaches to one to two attacks every 2–3 months, each lasting 1–3 days. He has been on this regimen for 9 years. Attempts to reduce the total daily dose to 400 mg or 600 mg have led to a remarkable increase in the severity and frequency of his acute relapses, so the total daily dose of 800 mg is being sustained indefinitely with regular follow-up in our clinic.
Discussion
In 1982, HSV was linked to Mollaret’s meningitis when HSV-1 was detected in the cerebrospinal fluid of an affected patient.4 Today, HSV-2 (and less often HSV-1) is believed to be a leading cause of RAM. Its pathogenesis is not fully understood and may be due to retrograde seeding reactivation of the latent virus nested in the sensory ganglia.5 Aseptic meningitis is known to complicate primary genital herpes infection in 36% of women and 11% of men, but meningitis following subclinical infections has also been reported.6,7 Confirmed HSV-2 acute meningitis can recur in 19%–27% of cases, especially in HSV-1 seronegative patients similar to our case.3,8 HSV-1 seroprevalence is declining in many countries, including the UK, and the incidence of recurrent HSV-2-associated meningitis may be increasing.3,9
Practical diagnostic criteria for Mollaret’s meningitis historically included absence of any detectable etiological agents.10 However, occurrence of concurrent or separate episodes of recurring mucocutaneous herpetic lesions, or detection of HSV DNA in the cerebrospinal fluid – which can be positive in up to 82% of samples taken 2–5 days after the onset of symptoms3 – can aid diagnosis. Characteristics of RAM are summarized in Table 1. In 30% of cases, recurrent attacks can be preceded by nonspecific self-limiting constitutional symptoms as paresthesias, neuropathic pain, and arthralgias.3,6 Mollaret’s meningitis usually resolves after a period of 3–5 years, but cases with longer periods, similar to our patient, have also been reported.2,11
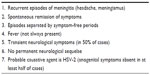  | Table 1 Characteristics of recurrent aseptic (Mollaret’s) meningitis |
The use of long-term suppressive rather than episodic anti-HSV therapy has shown satisfactory results in controlling HSV-2-associated Mollaret’s meningitis, but this practice is not yet supported by high quality evidence.2,7 In our patient and other reported cases, daily acyclovir or valacyclovir has been generally well tolerated with good bioavailability, and has reduced the severity and frequency of attacks.7,11 Other medications such as colchicine and indomethacin have also been tried, but firm evidence of their efficacy is still lacking.12,13
Summary
Incidence of HSV-2-associated recurrent meningitis is suspected to be increasing. However, most patients have unnecessary investigations and treatments prior to diagnosis, and some patients remain undiagnosed. History of herpetic anogenital infections should be sought in all patients with recurrent meningitic headaches and vice versa. Continuous antiviral therapy appears to reduce the frequency of attacks and can dramatically improve lifestyle.
Author contributions
AKAF contributed to the literature review and writing of the article. TMB contributed to the data collection and correction of the manuscript. SLG contributed to the conception of project and review/correction of article before submission.
Disclosure
The authors report no conflict of interests in this work.
References
Mollaret P. Méningite endothélio-leucocytaire multirécurrente bénigne. Syndrome nouveau ou maladie nouvelle? (Documents cliniques) [Recurrent benign endothelio-leukocytic meningitis. A new syndrome or a new disease? (Case reports)]. Rev neurol. 1944;76:657–658. French. | |
Pearce JM. Mollaret’s meningitis. Eur Neurol. 2008;60:316–317. | |
Kallio-Laine K, Seppänen M, Kautiainen H, et al. Recurrent lymphocytic meningitis positive for herpes simplex virus type 2. Emerg Infect Dis. 2009;15(7):1119–1122. | |
Steel JG, Dix RD, Baringer JR. Isolation of herpes simplex virus type 1 in recurrent (Mollaret) meningitis. Ann Neurol. 1982;11(1):17–21. | |
Piskin IE, Yarimay G. Mollaret meningitis: a case report. Turk J Pediatr. 2010;52(3):306–308. | |
Farazmand P, Woolley PD, Kinghorn GR. Mollaret’s meningitis and herpes simplex virus type 2 infections. Int J STD AIDS. 2011;22(6):306–307. | |
Ellerin TB, Walsh SR, Hooper DC. Recurrent meningitis of unknown aetiology. Lancet. 2004;363(9423):1772. | |
Omland LH, Vestergaard BF, Wandall JH. Herpes simplex virus type 2 infections of the central nervous system: A retrospective study of 49 patients. Scand J Infect Dis. 2008;40(1):59–62. | |
Davies N, Tang J, Ward KN. Herpes simplex virus type 2 and recurrent meningitis. Lancet. 2004;364(9433):501–502. | |
Bruyn GW, Straathof LJ, Raymakers GM. Mollaret’s meningitis. Differential diagnosis and diagnostic pitfalls. Neurology. 1962;12:745–753. | |
Mirakhur B, McKenna M. Recurrent herpes simplex type 2 virus (Mollaret) meningitis. J Am Board Fam Pract. 2004;17(4):303–305. | |
Mora JS, Gimeno A. Mollaret’s meningitis: report of a case with recovery after colchicine. Ann Neurol. 1980;8:631–633. | |
Wynants H, Taelman H, Martin JJ, Van den Ende J. Recurring aseptic meningitis after travel to the tropics: a case of Mollaret’s meningitis? Case report with review of the literature. Clin Neurol Neurosurg. 2000;102(2):113–115. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
