Back to Journals » Clinical Ophthalmology » Volume 13
Half-dose photodynamic therapy for serous non-neovascular retinal pigment epithelial detachment
Authors Inoda S , Takahashi H , Inoue Y , Tampo H, Sakamoto S, Arai Y, Kawashima H, Yanagi Y
Received 3 December 2018
Accepted for publication 26 April 2019
Published 6 June 2019 Volume 2019:13 Pages 959—968
DOI https://doi.org/10.2147/OPTH.S196901
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Satoru Inoda,1 Hidenori Takahashi,1 Yuji Inoue,1 Hironobu Tampo,1 Shinichi Sakamoto,1 Yusuke Arai,1 Hidetoshi Kawashima,1 Yasuo Yanagi2
1Ophthalmology, Jichi Medical University, Shimotsuke-Shi, Tochigi, 329-0431, Japan; 2Ophthalmology, Asahikawa Medical University, Asahikawa-Shi, Hokkaido, 078-8510, Japan
Purpose: To investigate the efficacy of half-dose photodynamic therapy (PDT) for treating symptomatic serous non-neovascular pigment epithelial detachment (PED) and to identify factors associated with treatment outcome.
Patients and methods: This is a single-institute retrospective case series involving 12 eyes of 12 consecutive patients with serous non-neovascular PED who received half-dose PDT. We investigated the temporal change in best-corrected visual acuity (BCVA), the anatomic outcomes of PED, including recurrence, central choroidal thickness (CCT), and also choroidal thickness as a possible factor associated with treatment outcome.
Results: At baseline, mean logMAR BCVA was 0.06 (range Snellen equivalent 6/20 to 24/20); 24 months later, it was 0.01 (Snellen equivalent 6/20 to 24/20; P=0.3). At 3 months, PED had completely flattened in 8, decreased in height in 2, and remained unchanged in 2. The CCT in eyes with PED was 410 μm and thicker than that in fellow eyes (290 μm, P<0.0001). Recurrence was noted in 2 within 12 months after treatment. Eyes in which the choroidal thickness with a baseline CCT >350μm or a 100 μm difference in CCT responded favorably to half-dose PDT (P=0.02).
Conclusion: Half-dose PDT is effective in patients with non-neovascular PED. Thicker choroid and some difference in CCT between affected and fellow eye might be a good index for predicting the efficacy of this treatment.
Keywords: epithelial detachment, serous pigment epithelial detachment, non-neovascular pigment photodynamic therapy, photodynamic therapy
Introduction
Retinal pigment epithelial detachment (PED) may occur in patients with central serous chorioretinopathy or age-related macular degeneration (AMD). PED causes loss of central vision due to degeneration of the neural retina and retinal pigment epithelium (RPE). Non-neovascular serous PED is often observed in both young and elderly patients. Although it shares similarities with central serous chorioretinopathy, serous non-neovascular PED might progress to neovascular AMD; a previous report documented that 45 of 140 eyes (32%) developed choroidal neovascularization (CNV) within an average of 19.6 months.1 Therefore, serous non-neovascular PED without CNV can be categorized as a precursor lesion of neovascular AMD.2 In a study of the natural history of the disease, visual acuity in eyes with serous non-neovascular PED was shown to decrease during 12 months of follow-up.3 Intravitreal antivascular endothelial growth factor (anti-VEGF) might temporarily decrease the volume of serous non-neovascular PED. However, in another study, this effect was not maintained over 1 year and the treatment was ineffective in terms of improving retinal function.4 In contrast, another study reported that reduced-fluence photodynamic therapy (PDT) was beneficial for resolving non-neovascular PED.5
However, two important questions remain regarding reduced-dose PDT and serous non-neovascular PED. The first question is whether reduced-dose PDT is an effective treatment for non-neovascular PED. Although it is likely that reduced-dose PDT is as effective as reduced-fluence PDT, there are no reports of the effects of reduced-dose PDT for serous non-neovascular PED. The second question is what factors, if any, contribute to the outcome of reduced-dose PDT? To address these gaps, we evaluated the effects of reduced-dose PDT and investigated factors that could be associated with the anatomic results.
Subjects and methods
Study design
This retrospective case series was conducted in accordance with the tenets of the Declaration of Helsinki and was approved by the Institutional Review Board at Jichi Medical University (A15-184). Written informed consent was obtained from all patients after they had been informed in detail of the potential benefits and risks of treatment with reduced-dose PDT.
Materials
The subjects were 12 patients who received treatment for symptomatic subfoveal PED between October 2012 and November 2013 at Jichi Medical University.
The inclusion criteria were as follows: presence of visual symptoms, such as visual disturbance or metamorphopsia, and patient’s treatment desired; presence of PED with little or no subretinal fluid as confirmed on optical coherence tomography (OCT); no evidence of CNV on fluorescein angiography or indocyanine green angiography (ICGA) (Heidelberg Retina Angiograph 2, Heidelberg Engineering GmbH, Heidelberg, Germany); no drusen near the macular as confirmed on OCT and fundus color photograph; and no leakage at the level of the RPE on fluorescein angiography. The exclusion criteria were previous PDT for any macular disease and PED associated with other inflammatory, infectious, neoplastic, or iatrogenic disease.
The protocol for half-dose PDT with verteporfin has been reported previously.6 In the current case–control study, all eyes showed choroidal vascular hyperpermeability, as characterized by an area of hyperfluorescence with blurred margins in the late frame of ICGA. The area of irradiation was set to cover the hyperfluorescent area during the late phase of ICGA by adding a diameter of 1,000 µm, that is, the area of greatest linear diameter over 6,000 µm was set to 7,000 µm. All patients received a 3 mg/m2 (half-dose) infusion of verteporfin over 10 mins followed by delivery of laser light at 689 nm 15 mins after the start of the infusion using a standard light intensity of 600 mW/cm2. The irradiation time was 83 s.
Assessments
At all visits, each patient underwent routine BCVA and OCT examinations using swept-source OCT (DRI OCT-1 Atlantis, Topcon Medical Laser Systems Inc., Tokyo, Japan) or spectral-domain OCT (RS-3000 Lite, Nidek Co., Ltd., Aichi, Japan). BCVA was measured as decimal visual acuity and converted to logarithm of the minimum angle of resolution (logMAR) units for the statistical analysis. The height of the PED was defined as the distance between the upper surface of the detached RPE and the inner surface of the choroid. Central macular thickness (CMT) was the distance from the inner limiting membrane to Bruch’s membrane on OCT. The central choroidal thickness (CCT) was the distance from the inner surface of the choroid to the chorioscleral interface. The measurements were taken manually using the software built into each OCT device.
The above parameters were independently measured by two of the authors (SI, HT). The interobserver agreement was at least 95%, and the average of each set of measurements was recorded.
The following criteria were used to examine the efficacy of half-dose PDT for non-neovascular PED. Complete resolution of PED was defined as a complete response, a decrease in PED height or diameter of ≥30% as a partial response, a decrease in the PED height or diameter of <30% as no response, and an increase in PED height or diameter as a recurrence.
Statistical analysis
Receiver operating characteristic (ROC) curve analysis was used to determine the predictive value of baseline factors associated with resolution of PED. The baseline distribution of CCT in the affected eye, the affected-fellow CCT ratio, and the difference in CCT between the affected eye and the fellow eye were evaluated for fit to a normal distribution or a lognormal distribution. A logistic model was calculated for these three factors to determine the cutoff values for their ability to predict resolution of PED with half-dose PDT. Complete flattening of the PED was set as the event. The area under the ROC curve (AUC) was calculated for each of the three parameters, and the best value of the positive likelihood ratio (PLR: sensitivity – [1 – specificity]) was sought. The P-value of Fisher’s exact test for each potential cutoff threshold was calculated. Students’ t-test was used for continuous variables. BCVA was evaluated 2 years after half-dose PDT. P<0.05 was considered to be statistically significant.
Results
The patient demographic and clinical characteristics are summarized in Table 1.
 | Table 1 Demographic and clinical data for patients with non-neovascular pigment epithelial detachment |
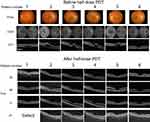
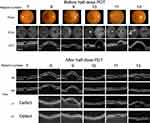
All cases of fundus photography, ICGA before RD-PDT procedure, and OCT before and after RD-PDT procedure are shown in Figures 1 and 2.
The 12 patients comprised 11 males and 1 female of mean age 59 (range 41–69) years. At baseline, the mean PED height was 420 (range 110–920) µm and mean PED diameter was 1,920 (range 610–3,600) µm. The fovea was involved in all cases. There was a significant correlation between PED height and diameter (R =0.86). Mean CMT was 313 (range 230–400) µm and mean CCT was 410 (range 230–540) µm. All eyes were approximately emmetropic and within ±1.75 diopters. Mean duration of symptoms was 13 (range 2–61) months. One eye had a history of central serous chorioretinopathy. Three patients had received intravitreal anti-VEGF injections before half-dose PDT, with no change in the size of PED before half-dose PDT.
Half-dose PDT was applied to cover the entire area showing hyperfluorescence in late-phase ICGA, including the subfoveal PED. Mean greatest linear diameter was 5,270 (range 2,900–7,000) µm. Mean logMAR BCVA was 0.06 (range 0.52 to −0.08) (Snellen equivalent 6/20 to 24/20) at baseline and 0.01 (range 0.52 to −0.08) (Snellen equivalent 6/20 to 24/20) at 24 months, with no significant change from baseline.
As shown in Table 2, 1 month after application of half-dose PDT, 7 (58%) of the 12 eyes showed a complete response and 1 eye (8%) showed a partial response. At 3 months, 8 (67%) of the 12 eyes showed a complete response, 2 (17%) showed a partial response, and 2 (17%) showed no response. In 1 of the patients with a partial response at 1 month, there was a continued decrease in the size of the PED over 12 months. Recurrence was documented in 1 of the 12 eyes at 5 months and another at 9 months after half-dose PDT. No eye developed CNV or an RPE tear.
 | Table 2 Patients’ ocular characteristics |
At 3 and 24 months after half-dose PDT, the mean PED height was 390 (range 0–710) µm (P=0.006) and 23.6 (range 0–169) µm (P<0.001 vs baseline), mean CMT was 230 (range 121–365) µm (P=0.009 vs baseline) and 198 (range 112–262) µm (P=0.002 vs baseline), and mean CCT was 289 (range 197–352) µm (P=0.0003 vs baseline) and 276 (range 166–374) µm (P<0.001 vs baseline), respectively.
Three patients who received intravitreal anti-VEGF injections before treatment subsequently received 3-monthly anti-VEGF injections after half-dose PDT.
There was no correlation between efficacy of treatment (as assessed by flattening of PED without recurrence) and age (P=0.02), sex (P=0.5), greatest linear diameter (P=1), anti-VEGF treatment before half-dose PDT (P=1), PED height (P=0.4), or PED diameter (P=0.2). Therefore, we focused on CCT. For this analysis, parameters were calculated in three different ways: 1) the baseline CCT in the affected eye, 2) the affected-fellow CCT ratio, and 3) the difference in CCT between the affected eye and the fellow eye. These parameters were used as an explanatory variable to calculate the predictive accuracy. The results demonstrated that the AUCs for the CCT in the affected eye, affected-fellow CCT ratio, and difference in CCT between the affected eye and the fellow eye were 0.84, 0.91, and 0.95, respectively (with respective P-values of 0.05, 0.006, and 0.001, Figure 3 A–C). There was no significant difference between the 3 models (P=0.4). The best PLR for the affected-fellow CCT ratio was 0.75 at a ratio of 1.37 (P-values at ratios of 1.30 and 1.40 were 0.08 and 0.07, respectively [Fisher’s exact test]) and that for the difference in CCT between the affected eye and the fellow eye was 0.75 at 107 and 112 µm (P-values at 100 and 110 µm were 0.02 and 0.06, respectively [Fisher’s exact test]). With this arbitrary cutoff value of 100 µm, the sensitivity and specificity were 100% and 75%, respectively. Regarding the baseline CCT of the affected eye, the best PLR was 0.75 at 360 µm, and a threshold of 350 µm was chosen as a convenient cutoff value (Table 3). The sensitivity and specificity for the threshold were 100% and 75%, respectively, with a P-value of 0.02 (Fisher’s exact test).
 | Table 3 Receiver operating characteristic data |
Discussion
In this study, half-dose PDT was confirmed to be effective for non-neovascular PED. And we revealed the baseline CCT of the affected eye and the difference CCT between affected and fellow eyes, quantitative measure of anatomic abnormalities, might be a better predictive factor to anatomic results.
Our results are consistent with the already reported efficacy of PDT and reduced-dose PDT in the treatment of serous non-neovascular PED. Kim and Lee described standard PDT in elderly patients7 and Gomi et al reported the efficacy of reduced-fluence PDT;5 in these reports, they concluded that changes in choroidal permeability play an important role in the development of serous PED and that late-phase hyperfluorescence on ICGA predicts a good response to PDT.
Though the extent (intense or faint) of late-phase hyperfluorescence in ICGA might be a good predictive factor, it should be a qualitative and partly subjective assessment, and difficult to evaluate quantitatively. Indeed, we, the three of authors, evaluated the choroidal permeability on late-phase ICGA before PDT and found that there was poor correspondence in the judgment of the severity of hyperpermeability (Cohen’s kappa 0.06, 0.2, and 0.3; data not shown). Therefore, it would be difficult to generalize the use of hyperfluorescence as a prognostic factor. In contrast, choroidal thickness, the factor associated with the effectivity of half-dose PDT, is a quantitative measure of anatomic abnormalities of the choroid. In patients with central serous chorioretinopathy, the subfoveal choroid is thicker in eyes with choroidal vascular hyperpermeability than in those without.8 In addition to enhanced depth imaging OCT, advances in swept-source OCT have now made it possible to evaluate choroidal thickness more correctly. Thus, choroidal thickness may be used as a surrogate marker for functional abnormalities of the choroid in the setting of central serous chorioretinopathy.
In this study, the efficacy of half-dose PDT for non-neovascular PED varied according to choroidal thickness. The choroidal thickness is affected by age, axial length, and refractometry.9–13 In addition to large individual differences, there is also diurnal variation in choroidal thickness, so it is difficult to define a value that is “normal”. Therefore, we also examined other two indexes, the difference CCT between affected and fellow eyes, and the affected-fellow CCT ratio. As a result, both indexes are useful for predicting the treatment outcome. Moreover, there was no statistically significant difference between the AUCs for these three indexes. In the clinical setting, simple to understand is easier to apply as a predict index, we consider that “CCT in the affected eye” is a better index of efficacy for patients with one eye or those in whom both eyes are affected. For patients with unaffected fellow eyes, “difference CCT” is better index. We have shown that a 100 µm difference in CCT and a choroidal thickness of 350 µm had a P-value of 0.02 and 0.02 (Fisher’s exact test), respectively, and can be useful as convenient cutoff values (Table 3).
There are several reasons why half-dose PDT may be less effective in eyes without increased CCT. A reasonable speculation is that the pathogenesis of PED may be different between eyes. As mentioned by Bird et al,14,15 fluid may accumulate in the sub-RPE space in eyes with choroidal vascular hyperpermeability and be unable to pass through Bruch’s membrane, resulting in elevation of the RPE. In contrast, PED with a relatively thin choroid may be characterized by the presence of an increased hydrophobic barrier to the outward passage of fluid at the level of Bruch’s membrane and the cause is not necessarily increased hydrostatic pressure in the choroid.
There were some limitations to this study. First, it included a small number of patients, all of whom were Japanese and enrolled from a single institution. Further studies are needed to confirm our results. Second, at that time, we had no OCT angiography camera. Some of the cases might have tiny CNV which can be detected only by OCT angiography. Third, the CCT ratio or difference is not useful in patients with 1 eye or those with both eyes affected. Finally, the study included only eyes with non-neovascular PED. We are aware that choroidal thickness and choroidal vascular hyperpermeability reflect different aspects of choroidal abnormalities (the former anatomic and the latter functional) and that choroidal thickness cannot be a surrogate marker for choroidal hyperpermeability, especially in the setting of AMD.
Conclusion
This study investigated CCT data in patients with non-neovascular PED who underwent treatment with half-dose PDT. These data suggest that half-dose PDT is effective for non-neovascular PED and that thicker choroid and some difference in CCT between affected and fellow eye might be a good quantitative index for predicting the efficacy of this treatment.
Acknowledgment
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Disclosure
Hidenori Takahashi received lecturer’s fees/personal fees from Kowa Pharmaceutical, Novartis Pharmaceuticals, Bayer Yakuhin, Pfizer, Senju Pharmaceutical and Santen Pharmaceutical, grants from Bayer Yakuhin, Santen Pharmaceutical and Novartis Pharma, outside this work. He is also the founder of DeepEyeVision, LLC. Yuji Inoue received lecturer’s fees/personal fees from Kowa Pharmaceuticals, Novartis Pharmaceuticals, Bayer Yakuhin, Santen Pharmaceuticals, Alcon Pharmaceutical, and Senju Pharmaceutical, outside this work. Hidetoshi Kawashima received lecturer’s fees from Kowa Pharmaceutical, Novartis Pharmaceuticals, Santen Pharmaceutical, Senju Pharmaceutical, Mitsubishi Tanabe Pharma Corporation, Alcon Pharmaceuticals, outside this work. Yasuo Yanagi received lecturer’s fees from Novartis Pharmaceuticals, Santen Pharmaceutical, and Hoya Opticals, grants from Santen Pharmaceutical, and Novartis Pharmaceutlcals, outside this work. Dr Shinichi Sakamoto reports personal fees from Santen Pharmaceutical, Kowa Pharmaceutical, and Novartis Pharmaceutical, outside the submitted work. The authors report no other conflicts of interest in this work.
References
1. Elman MJ, Fine SL, Murphy RP, et al. The natural history of serous retinal pigment epithelium detachment in patients with age-related macular degeneration. Ophthalmology. 1986;93:224–230.
2. Seddon JM, Sharma S, Adelman RA. Evaluation of the clinical age-related maculopathy staging system. Ophthalmology. 2006;113:260–266. doi:10.1016/j.ophtha.2005.11.001
3. Pauleikhoff D, Loffert D, Radermacher M, et al. Pigment epithelial detachment in the elderly. Clinical differentiation, natural course and pathogenetic implications. Graefes Arch Clin Exp Ophthalmol. 2002;240:533–581. doi:10.1007/s00417-002-0505-8
4. Ritter M, Bolz M, Sacu S, et al. Effect of intravitreal ranibizumab in avascular pigment epithelial detachment. Eye. 2010;24:962–968. doi:10.1038/eye.2009.265
5. Goto S, Gomi F, Ueno C, Nishida K. Reduced-fluence photodynamic therapy for subfoveal serous pigment epithelial detachment with choroidal vascular hyperpermeability. Am J Ophthalmol. 2012;154:865–871. doi:10.1016/j.ajo.2012.05.001
6.
7. Kim KE, Lee WK. Photodynamic therapy with verteporfin for avascular serous pigment epithelium detachment in elderly Koreans. Retina. 2010;30:93–99. doi:10.1097/IAE.0b013e3181b094a1
8. Ikuno Y, Kawaguchi K, Nouchi T, Yasuno Y. Choroidal thickness in healthy Japanese subjects. Invest Ophthalmol Vis Sci. 2010;51:21736. doi:10.1167/iovs.09-4383
9. Maruko I, Iida T, Sugano Y, et al. Subfoveal choroidal thickness in fellow eyes of patients with central serous chorioretinopathy. Retina. 2004;31:1603–1608. doi:10.1097/IAE.0b013e31820f4b39
10. Margolis R, Spaide RF. A pilot study of enhanced depth imaging optical coherence tomography of the choroid in normal eye. Am J Ophthalmol. 2009;147:811–815. doi:10.1016/j.ajo.2008.12.008
11. Manjunath V, Taha M, Fujimoto JG, Duker JS. Choroidal thickness in normal eyes measured using Cirrus HD optical coherence tomography. Am J Ophthalmol. 2010;150:325–329. doi:10.1016/j.ajo.2010.04.018
12. Agawa T, Miura M, Ikuno Y, et al. Choroidal thickness measurement in healthy Japanese subjects by three-dimensional high-penetration optical coherence tomography. Graefes Arch Clin Exp Ophthalmol. 2011;249:1485–1492. doi:10.1007/s00417-011-1708-7
13. Hirata M, Tsujikawa A, Matsumoto A, et al. Macular choroidal thickness and volume in normal subjects measured by swept-source optical coherence tomography. Invest Ophthalmol Vis Sci. 2011;52:4971–4978. doi:10.1167/iovs.11-7729
14. Bird AC, Marshall J. Retinal pigment epithelial detachments in the elderly. Trans Ophthalmol Soc. 1986;105:674–682.
15. Pauleikhoff D, Harper CA, Marshall J, Bird AC. Aging changes in Brush’s membrane. A histochemical and morphologic study. Ophthalmology. 1990;97:171–178.
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.