Back to Journals » Clinical and Experimental Gastroenterology » Volume 7
From depolarization-dependent contractions in gastrointestinal smooth muscle to aortic pulse-synchronized contractions
Received 10 January 2014
Accepted for publication 18 February 2014
Published 28 March 2014 Volume 2014:7 Pages 61—66
DOI https://doi.org/10.2147/CEG.S60448
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Sarah B Marion, Allen W Mangel
RTI Health Solutions, Research Triangle Park, NC, USA
Abstract: For decades, it was believed that the diameter of gastrointestinal smooth muscle cells is sufficiently narrow, and that the diffusion of calcium across the plasma membrane is sufficient, to support contractile activity. Thus, depolarization-triggered release of intracellular calcium was not believed to be operative in gastrointestinal smooth muscle. However, after the incubation of muscle segments in solutions devoid of calcium and containing the calcium chelator ethylene glycol tetraacetic acid, an alternative electrical event occurred that was distinct from normal slow waves and spikes. Subsequently, it was demonstrated in gastrointestinal smooth muscle segments that membrane depolarization associated with this alternative electrical event triggered rhythmic contractions by release of intracellular calcium. Although this concept of depolarization-triggered calcium release was iconoclastic, it has now been demonstrated in multiple gastrointestinal smooth muscle preparations. On the basis of these observations, we investigated whether a rhythmic electrical and mechanical event would occur in aortic smooth muscle under the same calcium-free conditions. The incubation of aortic segments in a solution with no added calcium plus ethylene glycol tetraacetic acid induced a fast electrical event without corresponding tension changes. On the basis of the frequency of these fast electrical events, we pursued, contrary to what has been established dogma for more than three centuries, the question of whether the smooth muscle wall of the aorta undergoes rhythmic activation during the cardiac cycle. As with depolarization-triggered contractile activity in gastrointestinal smooth muscle, it was “well known” that rhythmic activation of the aorta does not occur in synchrony with the heartbeat. In a series of experiments, however, it was demonstrated that rhythmic contractions occur in the aortic wall in synchrony with the heartbeat and share a common pacemaker with the heart. We conclude that important observations in the vascular system became derivative from those in the gastrointestinal system. The challenging of scientific dogma potentially leads to the expansion of our fundamental knowledge base.
Keywords: gastrointestinal smooth muscle, aortic smooth muscle, contractions, intracellular calcium pools, Windkessel hypothesis, pulse-synchronized contractions
Introduction
Recordings of electrical and mechanical activity in vitro from gastrointestinal smooth muscle segments show rhythmic mechanical activity that is triggered by corresponding rhythmic changes in membrane potential.1–3 The underlying membrane potential changes are usually composed of two events, starting with a slow rhythmic change (often denoted as slow waves), which serves to depolarize the membrane potential to a sufficient level to elicit a second event, which is the spike or action potential (Figure 1). Although in some gastrointestinal smooth muscles slow waves themselves may trigger contractions,4 the usual physiologic trigger for contractions is considered the spike. The depolarizing phase of the spike is predominantly a result of an influx of calcium.2,5 Considering the narrow diameter of smooth muscle cells, it had been concluded that activator calcium for contractions in gastrointestinal smooth muscle originated from an influx of calcium across the plasma membrane associated with spikes, rather than a release of calcium from intracellular pools.1,5
  | Figure 1 Voltage profiles of electrical activity in cat small intestinal smooth muscle. |
After the incubation of gastrointestinal smooth muscle segments in solutions devoid of calcium and containing the chelating agent ethylene glycol tetraacetic acid (EGTA), normal slow waves and spikes were eliminated, and an alternative rhythmic activity (prolonged potentials) develops6–8 (Figure 1). Prolonged potentials may persist for hours in calcium-free solutions and have a voltage excursion similar to the spike3,8 (Figure 1). In several gastrointestinal and visceral smooth muscle preparations, prolonged potentials trigger rhythmic contractile activity7–10 (Figure 2). Considering that preparations can be incubated for hours in solutions containing no added calcium plus EGTA, it was concluded that gastrointestinal smooth muscle contains an intracellular calcium pool that is released after membrane depolarization.
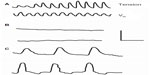  | Figure 2 Recording from cat small intestinal muscle segment of mechanical (upper) and electrical (lower) activity in normal solution (A), 7 minutes after change to calcium-free solution (B), and 50 minutes after change to calcium-free solution (C). As shown in (C), prolonged potentials triggered contractions. Electrical recordings were made with pressure electrodes. Calibration bar: A/B, 0.4 mV, 0.8 g, 16 seconds; C, 0.27 mV, 0.13 g, 10 seconds. Reproduced with permission from Mangel AW, Nelson DO, Rabovsky JL, Prosser CL, Connor JA. Depolarization-induced contractile activity of smooth muscle in calcium-free solution. Am J Physiol. 1982;242(1):C36–C40.8 |
These observations were considered iconoclastic, as it was “well known” that there was no depolarization-sensitive release of intracellular calcium in gastrointestinal smooth muscle. However, this mechanical and electrical activity has now been well described in multiple gastrointestinal and other visceral smooth muscle preparations. Possible explanations for the lack of observing contractile activity in calcium-free solutions by others include observation during the period immediately after changing from normal to calcium-free solutions, as it may take time for plateau potentials to develop, and use of single cells, as there appears to be a minimal geometry needed to observe the activity.9,10
These and other explanations for the failure of some to note contractions in gastrointestinal smooth muscle during incubation in calcium-free solution have recently been described.3
Electrical activity of aortic smooth muscle
In contrast to gastrointestinal smooth muscle, segments of aortic smooth muscle are generally quiescent both electrically and mechanically in organ baths or show spontaneous contractions at a frequency significantly slower than the cardiac cycle.11,12 When aortic segments are incubated in a similar calcium-free solution with EGTA, a fast rhythmic electrical activity, compared with prolonged potentials, develops13 (Figure 3). No changes in mechanical activity were associated with these fast electrical events. Thus, experiments conducted with aortic muscle segments in vitro do not support the notion that phasic contractile activity may occur in the large conduit arteries. However, we remained intrigued that the aortic smooth muscle wall has the machinery to generate a rapid electrical event and, therefore, investigated whether the aortic wall in vivo undergoes rhythmic activation in synchrony with the heartbeat.
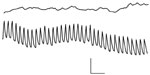  | Figure 3 Recording of electrical activity from a segment of rabbit aorta. In the upper trace, recordings are made in normal solutions containing calcium. In the lower trace, fast rhythmic potentials become apparent after a change to calcium-free solution. Recordings are made with a pressure electrode. Calibration bar: 1 mV, 4 seconds. Reproduced with permission from Mangel A, van Breemen C. Rhythmic electrical activity in rabbit aorta induced by EGTA. J Exp Biol. 1981;90:339–342.13 |
Arterial smooth muscle activity in vivo
The Windkessel hypothesis states that the smooth muscle walls of large arteries behave as passive elastic tubes, which represents the framework for understanding the behavior of the large arterial vasculature. This notion is perpetuated in current textbooks and a well-done recent review article.14 If this fundamental understanding is incorrect, then the paradigm in which the large conduit arteries is understood is incorrect, potentially opening new avenues to develop additional classes of therapeutics for the treatment of cardiovascular diseases.
In 1733, Stephen Hales laid the foundation for the current understanding of arterial blood pressure and of the smooth muscle wall acting as a passive elastic tube.14 In 1899, Otto Frank further expanded this theory, denoting it Windkessel.14 Over the years, many of the underlying assumptions of the model have not changed: large arteries are considered passive elastic tubes, serving as reservoirs for blood pumped from the heart.14 The smooth muscle wall of these vessels is assumed to be incapable of quick, rhythmic contractions, especially in light of the fast frequency of the heart rate.
Arterial walls behave as passive elastic tubes
In 1939, Wiggers and Wegria15 recorded changes in thoracic aortic diameter and aortic pressure in anesthetized dogs. The pressure–diameter curves obtained revealed that the diameter was smaller on aortic deflation than it had been at the same pressure during aortic dilation. In other words, after passive distension of the vessel by the pulse wave, an active reduction in diameter occurs. The interpretation of these results was that during the cardiac cycle, “some active change in the vessel wall … more probably a muscular action”15 is occurring. In 1955, Rushmer16 measured pressure–circumference relationships in the dog aorta in vivo and observed “consistent differences in the shape of the pressure and circumference curves during the cardiac cycle(s)”.16 Rushmer noted that if passive distension by the pulse wave were responsible for circumference changes, then simultaneous and consistent patterns would be observed. However, because of the observed hysteresis, energy was being exchanged, and the direction of hysteresis was such that energy was being imparted by the aorta to the blood (ie, a contraction).
Nevertheless, Rushmer discounted the possibility that active phasic contractions occur in the smooth muscle wall during the cardiac cycle; instead, he suggested that an inadequate quantity of smooth muscle exists in the vessel wall and that the responsiveness of the vascular smooth muscle is too slow to participate in the cardiac cycle. He concluded that technical artifacts or changes in vessel length during the cardiac cycle erroneously suggest a phasic aortic contraction. Peterson et al,17 in 1960, reported on the mechanical properties of the walls of major arteries in dogs in vivo and concluded that from the analysis of 5,000 pulse cycles, “no evidence has been found to suggest that the arterial wall [undergoes] active contraction synchronous with the cardiac cycle”.17 Furthermore, they conclude that the mechanical properties of vascular smooth muscle are too slow to synchronize with the cardiac cycle.
In a more recent study, Gentile and Gross18 found no evidence of active diameter changes in bypassed aortic segments in synchrony with pulsatile pressure changes in a bypassed tube. The methodology used to record diameter changes in this study was inferior and may account for the lack of diameter changes being observed.
Arterial wall shows rhythmic contractility
In contrast to the studies discussed earlier, on the basis of the fast rhythmic electrical activity observed in aortic muscle segments during incubation in calcium-free solution, Mangel et al19–21 directly measured tension changes in dog and rabbit aorta, femoral artery, and coronary artery in vivo. The initial experimental design involved bypassing the blood flow and recording tension changes in the bypassed segment. In the bypass tube, changes in arterial pressure were recorded. These experiments demonstrated a 1:1 coupling between pulse pressure changes and aortic, coronary, or femoral artery smooth muscle contractions. On the basis of the phasing of the pulse wave with the arterial contractions, these contractions were described as pulse-synchronized contractions (PSCs)21 (Figure 4).
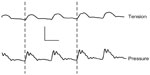  | Figure 4 Simultaneous recording of tension (upper) and blood pressure (lower) from the rabbit aorta in vivo. |
As this direct demonstration of smooth muscle contractions in large conduit arteries in vivo was considered heretical, significant effort was focused on ensuring that these events were not caused by either a movement artifact related to the pulse wave or cardiac contractility. After the local application of the neural blocker tetrodotoxin or the local anesthetic xylocaine, either to the bypassed segment or proximal to it, a block of aortic contractions, without affecting the pulse wave, occurred.20 In addition to the pharmacologic dissociation with tetrodotoxin and xylocaine, several other manipulations were performed. The definitive experiment to eliminate the pulse wave as a source of a movement artifact that could be producing PSCs was to bleed the animals while recording cardiac and aortic contractility.21 After animals were bled, both cardiac muscle contractions and aortic PSCs persisted for some time. Under these circumstances, there was no pulsatile pressure wave, and therefore, distension from the pulse wave could not be responsible for the PSC.
Nevertheless, questions persisted: Could movement associated with cardiac contractility lead to an artifact by tugging on the aorta? And, therefore, could PSCs result from a movement artifact from the heartbeat? In the next series of experiments,21 the effects of excision of the left atrial appendage on PSCs were evaluated. This maneuver had no effect on PSCs. However, excision of the right atrial appendage caused irreversible abolishment of the PSC. PSCs were eliminated even if cardiac contractions were maintained at a greater-than-baseline amplitude level by direct electrical stimulation. Thus, this series of experiments showed both that PSCs do not result as a consequence of an artifact caused by cardiac contractions and that the pacemaker for PSCs is similar to that for the heart, in the right atrium.
In an elegant series of experiments, Ravi and Fahim22 stimulated the sinoatrial nodal region while recording PSCs from cat pulmonary arteries. When stimuli were applied during times when the heart was mechanically refractory, an “ectopic” PSC was produced without a corresponding pulse wave. Thus, in this configuration, PSCs could not be caused by a mechanical artifact from either the pulse wave or cardiac contractions. Furthermore, these experiments also confirmed the pacemaker region for the PSC to be in the right atrial region, helping to ensure coordination with the heartbeat.
A further review of the literature does show significant information in support of the concept of large arteries undergoing rhythmic contractions in phase with the cardiac cycle. Heyman, in a series of papers published from 1955 to 1961,23–26 demonstrated in both human and dog that active participation of the arterial wall occurs during the cardiac cycle. Unfortunately, these papers have previously been largely ignored. Boucek et al27 also made cineangiographic recordings of the ascending aorta in dogs during the cardiac cycle. Their results suggested that the ascending aorta, during the cardiac cycle, undergoes active diameter changes with the heartbeat.
Conclusion
Challenging well-accepted notions in science has led to important discoveries. In gastroenterology, recognition that acid secretion was physiologic versus pharmacologic was not an easy concept to establish. In a similar vein, establishment of the significance of Helicobacter pylori in ulcer foundation was a high hill to climb.
Although depolarization-activated release of calcium in gastrointestinal smooth muscle was not easy to establish, it has now been demonstrated in multiple tissue types. This observation led to experimentation in aortic smooth muscle.
The observations made in vascular smooth muscle should be of interest to those studying the properties and behaviors of gastrointestinal smooth muscle, as smooth muscle is often segmented into visceral (including gastrointestinal) and vascular smooth muscle and studied separately. Findings such as those described here remind the gastrointestinal physiologist that important lessons for the gastrointestinal tract may be learned from vascular smooth muscle, and vice versa.
The hypothesis that the arterial walls of the conduit arteries behave as passive elastic tubes has been well engrained as scientific dogma for more than a century. The bias in favor of this notion was predicated on the belief that the quantity of smooth muscle in the arterial wall was too small to be significant and that the dynamics of vascular smooth muscle contraction were too slow to coincide with the cardiac cycle. It is now clear that the smooth muscle wall of the large arteries appears to undergo a contraction–relaxation cycle in synchrony with the pulse wave. The timing of the events is such that the upstroke of the contraction occurs during systole; hence the term pulse-synchronized contractions. Because of the phasing between the two events, the rhythmic contractions may help to reduce arterial wall stress from pulse pressure waves by limiting vessel wall distension. Understanding of PSCs may lead to the development of new therapeutic interventions in the field of cardiology that never would have been observed had it not been for experimentation in the gastrointestinal tract evaluating depolarization-mediated release of intracellular calcium.
Acknowledgments
Funds for conduct of this work were provided by RTI Health Solutions, of which Ms Marion and Dr Mangel are employees.
Disclosures
The authors report no conflicts of interest in this work.
References
Bozler E. The relation of the action potentials to mechanical activity in intestinal muscle. Am J Physiol. 1946;146:496–501. | |
Prosser CL, Mangel AW. Mechanisms of spike and slow wave pacemaker activity in smooth muscles. In: Carpenter D, editor. Cellular Pacemakers. New York: John Wiley & Sons; 1982:273–301. | |
Evans ED, Mangel AW. Depolarization-stimulated contractility of gastrointestinal smooth muscle in calcium-free solution: a review. ISRN Gastroenterol. 2011;2011:692528. | |
Sanders KM. Excitation-contraction coupling without Ca2+ action potentials in small intestine. Am J Physiol. 1983;244(5):C356–C361. | |
Prosser CL, Bortoff A. Electrical activity of intestinal muscle under in vitro conditions. In: Handbook of Physiology. Washington, DC: American Physiological Society; 1968:2025–2050. | |
Prosser CL, Kreulen DL, Weigel RJ, Yau W. Prolonged potentials in gastrointestinal muscles induced by calcium chelation. Am J Physiol. 1977;233(1):C19–C24. | |
Mangel AW, Nelson DO, Connor JA, Prosser CL. Contractions of cat small intestinal smooth muscle in calcium-free solution. Nature. 1979;281(5732):582–583. | |
Mangel AW, Nelson DO, Rabovsky JL, Prosser CL, Connor JA. Depolarization-induced contractile activity of smooth muscle in calcium-free solution. Am J Physiol. 1982;242(1):C36–C40. | |
Mangel AW, Mangel CP, Sanders KM. Modulation of intestinal electrical and mechanical activity by calcium. In: INSERM Symposia Series: Ca Regulations in Smooth Muscle. 1984;124:111–118. | |
Mangel AW. Voltage and receptor mediated contractile activity of colonic smooth muscle in calcium-free solution. Eur J Pharmacol. 1984;102(1):165–168. | |
Furchgott RF, Bhadrakom S. Reactions of strips of rabbit aorta to epinephrine, isopropylarterenol, sodium nitrite and other drugs. J Pharmacol Exp Ther. 1953;108(2):129–143. | |
Hayashida N, Okui K, Fukuda Y. Mechanism of spontaneous rhythmic contraction in isolated rat large artery. Jpn J Physiol. 1986;36(4):783–794. | |
Mangel A, van Breemen C. Rhythmic electrical activity in rabbit aorta induced by EGTA. J Exp Biol. 1981;90:339–342. | |
Westerhof N, Lankhaar JW, Westerhof BE. The arterial Windkessel. Med Biol Eng Comput. 2009;47(2):131–141. | |
Wiggers CJ, Wegria R. Active changes in size and distensibility of the aorta during acute hypertension. Am J Physiol. 1938;124(3):603–611. | |
Rushmer RF. Pressure-circumference relations in the aorta. Am J Physiol. 1955;183(3):545–549. | |
Peterson LH, Jensen RE, Parnell J. Mechanical properties of arteries in vivo. Circ Res. 1960;8:622–639. | |
Gentile BJ, Gross DR. Viscoelastic behavior of the thoracic aorta of dogs and rabbits. Circ Res. 1985;56(5):690–695. | |
Mangel A, van Breemen C, Fahim M, Loutzenhiser R. Measurement of in vivo mechanical activity and extracellular Ca45 exchange in arterial smooth muscle. In: Bevan JA, editor. Vascular Neuroeffector Mechanisms. 1983:347–351. | |
Mangel A, Fahim M, van Breemen C. Rhythmic contractile activity of the in vivo rabbit aorta. Nature. 1981;289(5799):692–694. | |
Mangel A, Fahim M, van Breemen C. Control of vascular contractility by the cardiac pacemaker. Science. 1982;215(4540):1627–1629. | |
Ravi K, Fahim M. Rhythmic contractile activity of the pulmonary artery studied in vivo in cats. J Auton Nerv Syst. 1987;18(1):33–37. | |
Heyman F. Movements of the arterial wall connected with auricular systole seen in cases of atrioventricular heart block. Acta Med Scand. 1955;152(2):91–96. | |
Heyman F. Comparison of intraarterially and extra-arterially recorded pulse waves in man and dog. Acta Med Scand. 1957;157(6):503–510. | |
Heyman F. Extra- and intra-arterial records of pulse waves and locally introduced pressure waves. Acta Med Scand. 1959;163(6):473–475. | |
Heyman F. The arterial pulse as recorded longitudinally, radially and intra-arterially on the femoral artery of dogs. Acta Med Scand. 1961;170:77–81. | |
Boucek RJ, Takashita R, Fojaco R. Functional anatomy of the ascending aorta and the coronary ostia (dog). Am J Anat. 1964;114:273–282. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
