Back to Journals » Clinical Ophthalmology » Volume 14
Emulsification of Different Viscosity Silicone Oil in Complicated Retinal Detachment Surgery: A Randomized Double-Blinded Clinical Trial
Authors Ratanapakorn T, Thongmee W ![]() , Meethongkam K
, Meethongkam K ![]() , Sinawat S, Sanguansak T
, Sinawat S, Sanguansak T ![]() , Bhoomibunchoo C, Laovirojjanakul W
, Bhoomibunchoo C, Laovirojjanakul W ![]() , Yospaiboon Y
, Yospaiboon Y ![]()
Received 18 December 2019
Accepted for publication 23 January 2020
Published 7 February 2020 Volume 2020:14 Pages 359—367
DOI https://doi.org/10.2147/OPTH.S242804
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Tanapat Ratanapakorn, Watcharaporn Thongmee, Kidakarn Meethongkam, Suthasinee Sinawat, Thuss Sanguansak, Chavakij Bhoomibunchoo, Wipada Laovirojjanakul, Yosanan Yospaiboon
KKU Eye Center, Department of Ophthalmology, Faculty of Medicine, Khon Kaen University, Khon Kaen, Thailand
Correspondence: Yosanan Yospaiboon
KKU Eye Center, Department of Ophthalmology, Faculty of Medicine, Khon Kaen University, 123 Mitraparb Highway, Khon Kaen 40002, Thailand
Tel +66 43 348383
Email [email protected]
Objective: To study silicone oil (SO) emulsification, anatomic and visual outcome after complicated retinal detachment surgery by pars plana vitrectomy (PPV) with intraocular SO tamponade, comparing between low and high viscosity SO.
Design: Randomized, double-blinded, controlled trial.
Patients and Methods: Patients with complicated retinal detachment who had been surgically treated by PPV and intraocular SO tamponade were randomly divided into low viscosity (Group 1) or high viscosity (Group 2). Main Outcome Measures were rate of silicone oil emulsification, anatomic retinal reattachment and visual outcome, assessed at 1,3,6,9 and 12 months.
Results: One hundred patients were divided into 50 patients in each group. The rate of silicone oil emulsification in Group 1 (63.64%) was higher than Group 2 (40%), but the difference was not statistically significant (p = 0.08). Rates of anatomic retinal reattachment were 88% in Group 1 and 90% in Group 2 (p = 0.76). For functional visual outcome, final best-corrected visual acuity (BCVA) at Month 12 compared with the baseline BCVA was improved vision 52% in Group 1 and 58% in Group 2 (p = 0.82).
Conclusion: SO emulsification rate in low viscosity group is non-significantly higher than high viscosity group, but results in a comparable reattachment rate and final visual recovery. Emulsification in both groups is detected at as early as 1 month and mostly within 3 months. We recommend using either low or high viscosity SO in complicated retinal detachment surgery and removing it as early as possible to prevent the consequent serious complications.
Clinical Trial Registration: ClinicalTrials.gov Identifier: NCT02988583.
Keywords: silicone oil, viscosity, emulsification
Introduction
Surgical procedures for rhegmatogenous retinal detachment include scleral buckling, pneumatic retinopexy and pars plana vitrectomy. In case of complicated retinal detachment such as proliferative vitreoretinopathy, proliferative diabetic retinopathy, cytomegalovirus retinitis and traumatic retinal detachment, pars plana vitrectomy (PPV) combined with intraocular silicone oil (SO) tamponade usually improves the surgical results.1–3 SO is a hydrophobic polymer and inert to human tissue. The chemical structure consists of silicon-oxygen core structure and hydrogen-carbon side chain and end terminal. The SO differs by viscosity, degree of purification and chemical composition.4 The viscosities are between the ranges of 1000 and 10,000 centistokes (cs.). The length of the polymeric compound determines the viscosity of the SO. Clinically most frequently used SO is highly purified polydimethylsiloxanes with two different viscosities, i.e. low viscosity (1000 or 1300 cs.) and high viscosity (5000 or 5700 cs.). Intraocular SO not only flattens the retina for a longer period of time but also allows the clear visualization of the fundus post-operatively due to its transparent property.
Emulsification is a clinically significant complication of intraocular SO injection. Consequently, fine SO droplets can cause a secondary glaucoma by blocking aqueous outflow, keratopathy by interfering with metabolic exchange of the corneal endothelium, cataract by interfering with lens metabolism and also retinopathy.5–7 The rate of emulsification depends on the physicochemical properties of the SO. In an experimental in vitro study, high viscosity SO was distinctly more stable and less emulsification rate than the low viscosity SO.4 It is therefore hypothesized that using high viscosity SO tends to have a lower rate of emulsification and delayed time to emulsification than low viscosity SO. Some surgeons, however, preferred using low viscosity SO because it is easier and faster in injection and removal than high viscosity SO. To the best of our knowledge, no clinical trial has compared the emulsification rate of low viscosity and high viscosity SO. Therefore, we conducted a randomized, double-blinded, controlled study to confirm this hypothesis. The objectives were to study SO emulsification rate, anatomic retinal attachment rate and visual outcome after complicated retinal detachment surgery by PPV with intraocular SO tamponade, comparing between low and high viscosity SO.
Patients and Methods
This study followed the tenets of Declaration of Helsinki and was approved by the Khon Kaen University Ethics Committee for Human Research. Patients with diagnosis of complicated retinal detachment who had been surgically treated by pars plana vitrectomy combined with intraocular SO tamponade at the KKU Eye Center, Department of Ophthalmology, Faculty of Medicine, Khon Kaen University, Khon Kaen, Thailand from 2016 to 2018 were recruited into the study. The eligibility criteria were complicated retinal detachment patients, whose age ≥18 years and had signed an informed consent form. The patients who had Inflammatory eye diseases, i.e. uveitis, corneal scar, history of scleral buckling procedure and history of using surfactant drugs were excluded from the study.
All patients in this study had been surgically treated by pars plana vitrectomy and intraocular SO tamponade. SO was fully filled in the vitreous cavity in case of phakic eyes and filled up to the iris level in aphakic eyes. Inferior peripheral iridectomy was performed in all cases to prevent pupillary block. Recruited patients were allocated into two groups by block randomization with varying block size. Patients were surgically treated by PPV using low viscosity or high viscosity SO (Figure 1). We used 1300 and 5700 cs. (Oxane, Bausch & Lomb Inc., Ireland) because they were commonly used for complicated retinal detachment surgery at our hospital. One surgical nurse prepared the different viscosity silicone oils for intravitreal injection. The silicone oil vials were coded A or B for use in the complicated retinal detachment surgery. The allocation sequence was generated using computer-generated random numbers with randomly varying block size (STATA version 10.1; StataCorp, College Station, TX 77845 USA). The random allocation sequence was concealed in a sealed opaque envelope, numbered sequentially, and kept by this surgical nurse, who was the only one who knew which code was for low or high viscosity silicone oils. The researchers and patients did not know the code, and unblinding was done at the end of the study.
 |
Figure 1 Flow diagram of patients in the study. |
The primary outcome in this study was the SO emulsification rate, assessed at 1, 3, 6, 9 and 12 months. Each visit, the patients were examined to search for clinical evidence of small emulsified SO droplets using slit-lamp biomicroscopy, gonioscopy and indirect ophthalmoscopy. The emulsification rate was calculated from the number of patients who had clinical evidence of emulsification divided by the total number of patients with attached retina after surgery throughout the study period. The patients with re-detached retina were re-operated and withdrawn from the study.
The secondary outcomes included retinal reattachment rate and the best-corrected visual acuity, assessed at 1, 3, 6, 9 and 12 months. The retinal reattachment rate was calculated from the number of patients who had an attached retina after surgery divided by the total number of patients operated. The final visual recovery was the difference between the preoperative and postoperative best-corrected visual acuity. Postoperative best-corrected visual acuity that improved by one or more lines on a Snellen acuity chart was classified as “improved”; postoperative best-corrected visual acuity that decreased by one or more lines was classified as “worse.” The patients with emulsified silicone oil were immediately scheduled to have silicone oil removal to prevent serious consequent complications.
Since there had been no such clinical trial before, we estimated from our pilot study that SO emulsification rate would have 50% and 20% for low and high viscosity groups, respectively. At a 95% confidence level, a power of 80%, and a withdrawal of 10%, the required number of patients per group was calculated to be 43.
Statistical Analyses
Descriptive statistics were used for the patients’ demographic characteristics and clinical findings. Categorical data including gender, affected eyes, underlying diseases, lens status, causes of retinal detachment and surgical procedures were recorded as number and percentages. Continuous data such as age were recorded as mean and standard deviation or median and ranges. Kaplan–Meier survival analysis was used to compare the emulsification and visual recovery at different periods of time. Hazard ratio by Cox regression and 95% confidence interval were used to determine the factor associated with emulsification. A p value of <0.05 was considered as statistical significance.
Results
A total of 100 patients with complicated retinal detachment were recruited into the trial. We allocated 50 patients to the low viscosity group and another 50 patients to the high viscosity group (Figure 1). The demographic characteristics of the patients in each group, including age and gender and lens status, were similar at study entry. The clinical findings in each group, including causes of retinal detachment and baseline best-corrected visual acuity were also similar in both groups (Table 1).
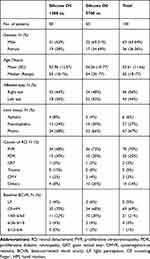 |
Table 1 Demographic Data and Clinical Characteristics of Patients in the Study |
For primary outcome, SO emulsification rate of the low viscosity group (63.64%) was higher than the high viscosity group (40%), but the difference was not statistically significant (p = 0.088, Hazard ratio 1.66, 95% confident interval 0.92–3.01) (Figures 1 and 2). For secondary outcomes, anatomic retinal reattachment rate of the low viscosity group (88%) was comparable to the high viscosity group (90%) (p = 0.761, Hazard ratio 1.20, 95% confident interval 0.37–3.94) (Figure 1 and Table 2).
 |
Table 2 Anatomic and Functional Visual Outcome of Patients in the Study |
 |
Figure 2 Kaplan–Meier survival analysis comparing emulsification of silicone oil 1300 and 5700 cs. |
Regarding the visual recovery after the surgery, we compared the best-corrected visual acuity at 1, 3, 6, 9, and 12 months after the surgery to the baseline BCVA. There was no statistically significant difference in proportion of patients with visual improvement, stable vision and worse vision among the low and high viscosity groups (p = 0.824, Hazard Ratio 0.95, 95% CI 0.60–1.50) (Figure 3 and Table 2)
 |
Figure 3 Kaplan–Meier survival analysis comparing final visual outcomes of patients using silicone oil 1300 and 5700 cs. |
Time to detect clinical evidence of SO emulsification is demonstrated in Figure 4. It was noted that most of SO emulsification occurred within 3 months (21/46, 45.65%) and 6 months (36/46, 78.26%). Furthermore, it occurred as early as 1 month (6/46, 13.04%) after the surgery and high viscosity SO did not have more delayed time to emulsification than low viscosity.
 |
Figure 4 Numbers of patients with silicone oil emulsification at the time of detection. |
Factors that may affect the SO emulsification were also studied. Table 3 analyses the demographic characteristics that may affect the emulsification. The study showed negative result. There were no demographic characteristics that might affect the rate of emulsification. Table 4 analyses the surgical factors that may be associated with the SO emulsification. SO viscosity tended to be the only surgical factor that affected the emulsification, but without statistical significance. The factors that should influence the rate of emulsification, i.e. scleral buckling, post-operative inflammation and post-operative hemorrhage demonstrated negative results.
 |
Table 3 Demographic Factors Affecting Silicone Oil Emulsification in the Study |
 |
Table 4 Surgical Factors Affecting Silicone Oil Emulsification in the Study |
Discussion
The results of this study demonstrate that after complicated retinal detachment surgery, the emulsification rate of low viscosity SO was higher than high viscosity silicone oil. This finding agrees well with a previous experimental study.4 Heidenkummer et al evaluated in vitro stability of SO in viscosity ranges from 1000 to 5000 cs. with active emulsifiers and found that SO at 5000 cs. was more stable and less emulsification than the SO with a viscosity up to 4000 cs.4 However, the difference in this study was not statistically significant. The explanation for this discrepancy might lie in some additional in vivo factors that were not involved in vitro study.
Due to the physicochemical properties, high viscosity SO should be more stable and expected to have delayed time to emulsification than low viscosity SO.4,8 The result of this study, however, showed that both groups had similar SO emulsification at as early as 1 month and the occurrence was peak in frequency at months 3 and 6.
Besides viscosity, there are several factors that can promote or prevent SO emulsification after retinal detachment surgery, including protein surfactants, contaminants, and shear forces.7 Hemorrhages or inflammation after the surgery may be contaminants and play some roles in the development of emulsification.8,9 Our study, however, did not demonstrate this association. There were no statistically significant differences whether the patients had hemorrhages and inflammation or not. This might be attributed to that biologically active emulsifiers found in hemorrhages or inflammatory situations might be lowered in vivo by hemostasis and sufficient postoperative anti-inflammatory therapy.8
In the case of SO under-filling, the shear force between SO bubble and aqueous or residual vitreous during eye movements may be related to the development of SO emulsification. Previous studies determined factors that might affect such shear force induced by eye movements, and found that the presence of indentation within the eye such as scleral buckling and encircling bands significantly decreased the velocity of SO movements and reduced the emulsification.10–12 Our study, however, did not agree with this finding. There were no statistically significant differences between emulsification and no emulsification in patients who had scleral buckling procedure combined with PPV and SO tamponade. This might be attributed to too small sample size for patients with scleral buckling in the subgroup analysis. Furthermore, long axial length, combined choroidal detachment and re-detached retina might also be associated with early development of SO emulsification. In eyes with complicated retinal detachment, the refractive status and axial length could not be accurately measured. Due to ethical concern, patients who had post-operative complications such as choroidal detachment or retinal re-detachment were withdrawn from the study for re-operation. Therefore, these conditions were not studied in the clinical trial.
Odrobina et al found hyper-reflective round-shaped droplets at 3 months after SO tamponade by spectral-domain optical coherence tomography and hypothesised that emulsification of SO began within 3 months after surgery.13 In another study, Toklu et al investigated the natural course of emulsification after SO tamponade in retinal detachment surgery and found that emulsification time ranged from 5 months to 24 months. Since most cases had SO emulsification within the first year, they proposed to extend the silicone oil removal time up to 1 year to achieve a stable retina status in cases with a high risk of redetachment.14 Our study, however, detected emulsified SO droplets as early as 1 month and the emulsification was most commonly found at 3 and 6 months after surgery.
In this study, we used clinical evidence of SO emulsification by examination using slit-lamp biomicroscopy, gonioscopy and indirect ophthalmoscopy. Ultra-wide field fundus photography may facilitate the detection of emulsified SO in the retinal periphery. However, the clinical emulsification rate in this study might be underestimated when compared to that quantitatively assessed using the Coulter counter and laser light scattering.15,16 This sophisticated technique could detect tiny emulsified droplets that were too small in size to be seen on clinical examination. Once emulsification was detected clinically in the anterior chamber, extensive emulsification would have already occurred in the eye. Perhaps emulsification might occur earlier than those detected in this study.
After emulsification has occurred, these small droplets might account for the consequent complications such as glaucoma, keratopathy, cataract, and retinopathy. Cataract is the only complication that can be simply treated by phacoemulsification later or prevented by combined PPV and phacoemulsification. Since other complications secondary to emulsified SO are difficult to treat and usually result in poor visual prognosis, limiting the amount of time the silicone oil remained in the eye is the most important factor in reducing these serious complications.7 Therefore, silicone oils should be used as a surgical tool to enhance the reattachment of the retina and should be removed as early as possible to prevent these complications.
Our study is the first, to our knowledge, to prospectively compare the emulsification rate of the low and high viscosity SO in a randomized, double-blinded, controlled trial. Furthermore, the number of patients who have been withdrawn after randomization was minimal. The one limitation of this study is that clinical detection of emulsified silicone oils may be underestimated.
In summary, PPV using low viscosity SO is associated with a non-significantly higher emulsification rate than those using high viscosity SO, but results in a comparable reattachment rate and final visual recovery. We recommend using either low or high viscosity SO in complicated retinal detachment surgery and removing it as early as possible to prevent the consequent serious complications.
Data Sharing
The authors do not intend to share individual participant data. During the informed consent process, the investigators clearly informed the participants that the collecting data were used for the clinical trial only and could not be shared.
Acknowledgments
The authors thank Dr Kaewjai Thepsuthammarat, Clinical Epidemiology Unit, Faculty of Medicine, Khon Kaen University, for statistical analysis. This study was supported by an invitation research grant (IN59161) from the Faculty of Medicine, Khon Kaen University.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Lucke KH, Foerster MH, Laqua H. Long-term results of vitrectomy and silicone oil in 500 cases of complicated retinal detachments. Am J Ophthalmol. 1987;104:624–633. doi:10.1016/0002-9394(87)90176-0
2. Yeo JH, Glaser BM, Michels RG. Silicone oil in the treatment of complicated retinal detachments. Ophthalmology. 1987;94:1109–1113. doi:10.1016/S0161-6420(87)33328-7
3. The Silicone Study Group. Vitrectomy with silicone oil or sulfur hexafluoride gas in eyes with severe proliferative vitreoretinopathy: results of a randomised clinical trial. Silicone study report 1. Arch Ophthalmol. 1992;110:770–779. doi:10.1001/archopht.1992.01080180042027
4. Heiddenkummer HP, Kampik A, Thierfelder S. Experimental evaluation of in vitro stability of purified polydimethylsiloxanes (silicone oil) in viscosity ranges from 1000 to 5000 centistokes. Retina. 1992;12:828–832. doi:10.1097/00006982-199212031-00007
5. Ichhpujani P, Jindal A, Katz LJ. Silicone oil induced glaucoma: a review. Graefes Arch Clin Exp Ophthalmol. 2009;247(12):1585–1593. doi:10.1007/s00417-009-1155-x
6. Lai W, Wong D, Li K, Leow P. Emulsification and inverted hypopyon formation of oxane HD in the anterior chamber. Graefes Arch Clin Exp Ophthalmol. 2008;246(11):1633–1635. doi:10.1007/s00417-008-0851-2
7. Miller JB, Papakostas TD, Vavvas DG. Complications of emulsified silicone oil after retinal detachment repair. Semin Ophthalmol. 2014;29(5–6):312–318. doi:10.3109/08820538.2014.962181
8. Heiddenkummer HP, Kampik A, Thierfelder S. Emulsification of silicone oil with specific physicochemical characteristics. Graefes Arch Clin Exp Ophthalmol. 1991;229(1):88–94. doi:10.1007/BF00172269
9. Savion N, Alhalel A, Treister G, Bartov E. Role of blood components in ocular silicone oil emulsification. Study on an in vitro model. Invest Ophthalmol Vis Sci. 1996;37(13):2694–2699.
10. Chan YK, Cheung N, Wong D. Factors influencing the shear rate acting on silicone oil to cause silicone oil emulsification. Invest Ophthalmol Vis Sci. 2014;55(11):7451–7456. doi:10.1167/iovs.14-14572
11. De Silva DJ, Lim KS, Schulenburg WE. An experimental study on the effect of encircling band procedures on silicone oil emulsification. Br J Ophthalmol. 2005;89(10):1348–1350. doi:10.1136/bjo.2004.063768
12. Chan YK, Ng CO, Knox PC, Garvey MJ, Williams RL, Wong D. Emulsification of silicone oil and eye movement. Invest Ophthalmol Vis Sci. 2011;52(13):9721–9727. doi:10.1167/iovs.11-8586
13. Odrobina D, Laudanska-Olszewska I. Analysis of the time and location of the silicone oil emulsification by spectral-domain optical coherence tomography after silicone oil tamponade. Biomed Res Int. 2014;2014:372045. doi:10.1155/2014/372045
14. Toklu Y, Cakmak HB, Ergun SB, Yorgun MA, Simsek S. Time course of silicone oil emulsification. Retina. 2012;32(10):2039–2044. doi:10.1097/IAE.0b013e3182561f98
15. Chan YK, Czanner G, Shum HC, Williams RL, Cheung N, Wong D. Towards better characterization and quantification of emulsification of silicone oil in vitro. Acta Ophthalmol. 2017;95(5):e385–e392. doi:10.1111/aos.2017.95.issue-5
16. Chan YK, Cheung N, Chan WS, Wong D. Quantifying silicone oil emulsification in patients: are we only seeing the tip of the iceberg? Graefes Arch Clin Exp Ophthalmol. 2015;253(10):1671–1675. doi:10.1007/s00417-014-2866-1
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
