Back to Journals » Journal of Blood Medicine » Volume 6
Emerging and future therapies for hemophilia
Authors Carr ME, Tortella B
Received 22 April 2015
Accepted for publication 3 June 2015
Published 3 September 2015 Volume 2015:6 Pages 245—255
DOI https://doi.org/10.2147/JBM.S42669
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Martin Bluth
Marcus E Carr,1,2 Bartholomew J Tortella3,4
1Robert Wood Johnson Medical School, Rutgers University, New Brunswick, NJ, 2Worldwide Research and Development, Pfizer, Inc., Cambridge, MA, 3Drexel University College of Medicine, Philadelphia, PA, 4Global Innovative Pharma, Pfizer, Inc., Collegeville, PA, USA
Abstract: The evolution of care in hemophilia is a remarkable story. Over the last 60 years, advances in protein purification, protein chemistry, donor screening, viral inactivation, gene sequencing, gene cloning, and recombinant protein production have dramatically enhanced the treatment and lives of patients with hemophilia. Recent efforts have produced enhanced half-life (EHL) clotting factors to better support prophylaxis and decrease the frequency of infusions. Medical needs remain in the areas of alternate modes of administration to decrease the need for venous access, better treatment, and prophylaxis for patients who form antibodies to clotting factors, and ultimately a cure of the underlying genetic defect. In this brief review, the authors summarize data on EHL clotting factors, introduce agents whose mode of action is not clotting factor replacement, and list current gene therapy efforts.
Keywords: extended half-life clotting factors, gene therapy, anti-tissue factor pathway inhibitor antibodies, clotting factor analogs, nonfactor replacement therapy
Introduction
Hemophilia is an inherited bleeding disorder. The most common forms, hemophilia A (one in every 5,000 live male births) and hemophilia B (one in every 30,000 live male births), are caused by the inheritance of abnormal forms of Factor VIII (FVIII) and Factor IX (FIX), respectively.1,2 Since the genes for both FVIII and FIX are located on the X chromosome, the disease displays the characteristic sex-linked pattern of inheritance. Since males have only one X chromosome, while females have two, males are most often affected. When a normal male and a female heterozygous for one abnormal FVIII (or IX) gene have children, male offspring have a 50% chance of inheriting hemophilia, and female offspring will have a 50% chance of carrying one abnormal gene copy. If a male with hemophilia has offspring with a female with two normal genes, all the males will be normal, and all the females will possess one copy of the abnormal gene. Due to the random shutdown of one of the pair of X chromosomes (the lyonization process), females may, if a higher percentage of the normal gene is silenced, have FVIII levels low enough to have increased bleeding. Recognized from biblical times, bleeding in affected males can occur virtually from birth. Thus, circumcision and heel sticks should be avoided in infants known to be at risk, and unsuspected occurrences may be diagnosed early due to excessive bleeding secondary to one of these procedures.
While greater than 300 unique mutations have been described for the FVIII gene, 40% of FVIII deficiency results from an inversion mutation of the short arm of the X chromosome, and a significant percentage are due to mutations of Xq28. While FIX deficiencies are also due to multiple mutations, the majority are due to mutations of Xq27.1-q27.2. The consequence of these mutations is a reduction in clotting factor activity. This might be due to production of a protein with decreased intrinsic activity, abnormal binding characteristics, or decreased plasma half-life. In this case, the antigenic amount of factor exceeds clotting factor activity. In other cases, very little of the abnormal protein is produced, and both factor antigen and activity are very low. In either case, the resultant decrease in activity puts the patient at risk of bleeding. Clotting factors are typically measured and reported as the number of units per given volume of plasma (1 unit/mL or 100 units/dL) or as a percent activity (100% = 100 units/dL). Since the amount of clotting factor varies significantly between individuals, a unit is defined as the amount of clotting factor activity in 1 mL of normal pooled plasma. Normal pooled plasma is prepared by pooling plasma from at least 20 normal volunteers. Given the marked variability of FVIII and FIX levels in normal individuals, a patient is considered to have a normal value if his level is greater than 50% but less than 150%. In hemophilia, the level of clotting factor activity is a primary determinant of the severity of the disease. Individuals with <1% activity have “severe” hemophilia with spontaneous bleeding.3 Patients with factor levels between 1% and 5% have moderate disease with occasional spontaneous bleeding and severe bleeding with surgery or trauma. Individuals with factor levels >5% but <40% rarely have spontaneous bleeding but can bleed excessively bleed if challenged major surgery or trauma. While bleeding can occur at any location, hemorrhage into muscles or joints with little or no obvious trauma is the hallmark of hemophilia.
Evolution of modern hemophilia treatment
Prior to the 1960s, treatment of bleeding in hemophilia involved whole blood or plasma transfusions. In 1964, Judith Pool discovered that cryoprecipitate from plasma contains large amounts of FVIII.4 This discovery led to treatment with cryoprecipitate followed by the development of lyophilized plasma concentrates of coagulation factors. These concentrates required donations from approximately 1,000 volunteers per vial. Their routine application in the 1970s improved the quality of life and extended the life expectancy of patients with hemophilia. Elective (even orthopedic) surgery became possible, and the availability of stable products enabled adoption of home treatment, facilitating early control of hemorrhage. While early treatment was good, it was demonstrated in Sweden that routine infusion of clotting factor in the non-bleeding state could actually prevent spontaneous bleeding and the severe joint damage that was caused by repetitive bleeding into joints.5 This process of “primary prophylaxis” was rapidly adopted for younger patients with remarkable reductions in secondary arthropathy due to hemarthrosis.6 Specialized hemophilia treatment centers came into existence, further enhancing and standardizing the care of hemophilia and providing routine access to orthopedic surgeons, physiotherapists, dentists, dieticians, and social workers. The discovery that 1-Desamino-8d-Arginine Vasopressin (DDAVP Desmopression Acetate) could be used to raise the level of FVIII in most patients with mild hemophilia A allowed the total avoidance of blood product exposure in these patients.7 Within 15 years, the care of hemophilia patients dramatically improved.
Unfortunately, the promise of the 1970s suddenly darkened with the realization that the early plasma-derived concentrates were contaminated with life-threatening viral pathogens. Since bleeding episodes might require several vials of concentrates per event, patients with hemophilia were being exposed to blood from thousands of people with each treatment. While the risk of hepatitis from hepatitis B virus and the less well-characterized “Non-A Non-B” was known, the true depth of the danger was not appreciated until the emergence of the human immunodeficiency virus (HIV).8 HIV and hepatitis C virus (HCV) were transmitted to patients with hemophilia. Virtually, all patients would develop hepatitis C with resultant risk of cirrhosis and hepatocarcinoma, and thousands would die in the 1980s and 1990s of AIDS.
This unforeseen disaster and the tragic consequences resulted in rapid technological and therapeutic advances. New ways and tests for screening donors were put into place. New methods to screen blood for viruses (nucleic acid amplification testing) were developed and implemented. Viral inactivation techniques (heat, solvent–detergent, nanofiltration, etc) were developed and incorporated as critical steps in the production of factor concentrates.9 The genes for FVIII (1982) and FIX (1984) were cloned opening the way for the production of recombinant FVIII and FIX in the late 1980s and early 1990s.10 Perhaps most importantly, the first effective antiviral treatments were developed allowing effective treatment of HIV and HCV. These advances set the stage for new and future treatments.
Current status
Clotting factor concentrates, both plasma derived and recombinant, have reached a very high level of purity. Nucleic acid testing testing (HIV 1-1, hepatitis B virus, HCV, hepatitis A virus, B19) and viral inactivation (dry heat, pasteurization, vapor, and solvent–detergent) for plasma-derived factor preparations have resulted in no blood-borne transmission of hepatitis or HIV in the last 25 years.9 While having reached a high degree of safety, one documented case of parvovirus B19 transmission,11 and the postmortem detection of variant Creutzfeldt–Jacob disease (vCJD) in a patient with hemophilia treated with plasma concentrates known to contain donations from a vCJD-infected donor, indicates that our guard cannot be dropped.12 Therefore, removal of animal and human proteins from recombinant products has continued to advance with production of third-generation products devoid of albumin during production or formulation.
While the risk of infection has been minimized, the risk of antibody production against administered clotting factor continues to complicate the treatment or prevention of bleeding in patients who develop these “inhibitors”.13 Inhibitors continue to be a major problem, and while treatment of acute bleeding in inhibitor patients is now possible in most cases, prophylaxis is poor, and immune tolerance induction is not uniformly effective.14 Less immunogenic treatments and better ways to eradicate these antibodies are needed.
As a consequence of the advances outlined earlier, the life span of people with hemophilia is now similar to people without hemophilia, and we must pay increasing attention to the disorders of aging.
Remaining unmet medical needs
With the current hemophilia treatment paradigm, significant unmet need persists. While on-demand therapy for hemophilia A and B is very good with a variety of plasma-derived and recombinant factor concentrates, on-demand therapy for patients with inhibitors is not as reliable. While 80%–90% of these patients will respond to either recombinant activated Factor VIIa (rFVIIa) or Factor VIII inhibitor bypassing agent (FEIBA),14 not all patients respond to both equally. Some patients may require a combination of rFVIIa and FEIBA which may be associated with an increased risk of thrombosis.15,16 However, several recent successful case reports and a small series of three patients treated safely with concomitant low doses of rFVIIa and FEIBA call into question the magnitude of the thrombotic risk.17–19
Prophylactic treatment for hemophilia B is good with the majority of patients having virtual elimination of spontaneous bleeding with one or two infusions per week. With the introduction of extended half-life FIX molecules, prophylactic therapy for hemophilia B promises to be excellent with even further reductions in dosing frequency. Prophylaxis in patients with hemophilia A generally requires two to three infusions per week, and the enhanced half-life (EHL) FVIII agents may not adequately support once-per-week prophylaxis in all patients. Prophylaxis in inhibitor patients is especially problematic with only one agent, FEIBA, having the approved indication and therapy requiring rather large volume (75 mL) intravenous (IV) infusions of product every other day to provide a 60% reduction in spontaneous bleeding.20 Alternate modes of administration of treatment (subcutaneous [SC] or even oral) are needed, and innovative nonfactor replacement therapies may offer broader spectrum treatments. At the present time, cures for hemophilia A and B are not available. While it is clear that stopping bleeding is good and preventing bleeding is better, cure remains the ultimate goal in the quest to normalize the lives of patients with hemophilia.
In the following sections, we briefly review the new extended half-life clotting factor products, newer agents being developed for patients with inhibitors, and new nonfactor replacement therapies for hemophilia. We conclude with a brief summary of current gene therapy efforts.
Extended half-life clotting factor products
rFIX:Fc
Recent advances in hemophilia treatment have centered on linkage of native, recombinant FIX (rFIX) to various molecules designed to prolong the intravascular residence of the molecule. These efforts have utilized the common, current strategies employed to extend the half-life of protein therapeutics: linkage to the fragment crystallizable (Fc) region of human antibodies, to polyethylene glycols (PEGs) of various sizes (PEGylation), and to recombinant albumin (Table 1).
A single molecule of rFIX is fused covalently and directly without linkers to the dimeric Fc of IgG1 (rFIX:Fc) and has been named “eftrenonacog alfa”. It is currently approved to use in the USA as Coagulation Factor IX (Recombinant), Fc Fusion Protein (ALPROIX, Biogen Idec, Cambridge, MA, USA). The molecule is expressed in HEK-293H cells, a human cell line in a serum-free medium.21 Linkage to the Fc prolongs the intravascular residence and hence biologic action of FIX by utilizing the Fc intracellular trafficking process. There may also be a component of reduced renal clearance.22
The Fc trafficking process utilizes the body’s pathway whereby proteins are endocytosed and sorted. Non-bound endosome proteins are sorted to intracellular degradation in lysosomes, while bound proteins (eg, Fc bound to the neonatal Fc-receptor) are routed for exocytosis.23,24 While trafficking through the endosome sorting/exocytosis processes, the FIX is shielded from intravascular degradation proteases.25 Initial trials yielded favorable safety results.26,27 FIX:Fc is denominated in FIX international units (IU) where 1 IU is defined as the amount of FIX in 1 mL of normal pooled plasma, and dosing performed on the basis of a patient’s weight in kilogram. Fc linkage does not affect coagulation factor activity but does confer an extended half-life (geometric mean =82.1 hours) compared to native FIX (nonacog alfa, geometric mean =33.8 hours) using a 96-hour sampling schedule. The incremental recovery of FIX:Fc (geometric mean =0.92, 95% confidence interval [CI] 0.77–1.10) was comparable to standard FIX (nonacog alfa, geometric mean =0.95, 95% CI 0.81–1.01).28 The model-predicted percentage of patients with a trough level >1 IU/dL and >3 IU/dL after weekly dosing was higher with FIX:Fc compared to native FIX (nonacog alfa; Table 2).29
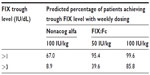  | Table 2 Predicted proportion of patients with FIX trough levels above 1 IU/dL and 3 IU/dL (steady state after six doses) with weekly dosing |
In the pivotal trial, annualized bleeding rates (ABRs) of prophylaxis regimens once weekly (Group 1) or initially once every 10 days (Group 2, interval adjusted as needed) were compared with on-demand therapy (Group 3, episodic). Low ABRs were achieved in both prophylaxis regimens (Table 3), and overall, 23% of patients receiving weekly dosing and 42.3% of patients receiving adjusted interval dosing experienced no bleedings during the trial.28 Efficacy of acute bleed treatment was 90.4% with a single dose and 97.3% with one or two doses. No inhibitors or vascular thrombotic events were detected. Adverse events were noted in 73.9% of patients, and 10.9% had at least one serious adverse event with only one event judged possibly related to rFIX:Fc, an obstructive ureteral clot which resolved with medical management.30
  | Table 3 Annualized bleeding rates by treatment regimen, rFIX:Fc |
When rFIX:Fc was used during surgery, only a single dose was needed to maintain hemostasis in 85.7% of patients (presurgery to the end of the operation). Two to three infusions were required on postoperative days 1–3, and no patient required dosing every day during the perioperative interval (days 0–14). One dose was required in most minor surgery cases. All major and 83% of minor surgeries reported excellent/good hemostasis. Two out of 12 patients with major surgeries required transfusions (one patient with three packed red blood cell units and one patient with seven total units of packed red blood cells and fresh frozen plasma, individual breakdown not reported). Adverse events were reported in 83.3% of major surgeries (six serious adverse events), and none of the adverse events were related to rFIX:Fc. No patient developed inhibitors, had an anaphylactic reaction, or experienced a vascular thrombotic event.30
FIX:PEG
Conjugation of proteins to PEG has been employed to prolong their duration of action. Controlled PEGylation is employed, since random PEGylation tended to increase protein immunogenicity.31,32 Targeted PEGylation is important so as to preserve the catalytic activity of the protein while achieving a prolonged duration of action.
GlycoPEGylated rFIX (rFIX:PEG), nonacog beta pegol, is currently in clinical trials. rFIX is expressed in Chinese hamster ovary cells, and glycoPEGylation (40 kDa) is performed at the activation peptide, so the PEG is jettisoned upon FIX activation.33 When FIX is activated, two internal peptide bonds are broken (specific arginine–alanine and arginine–valine bonds) releasing a small “activation peptide” with the resulting larger protein now termed “activated FIX (FIXa)”. Since the PEG moiety is linked to the activation peptide, the PEG is jettisoned along with the activation peptide when FIX is activated. PEGylation within the activation peptide domain was shown to maintain catalytic activity and prolong half-life approximately fivefold.34,35 The Phase I trial showed IV tolerability with one hypersensitivity reaction attributed to the molecule.35
A single IV dose and steady-state pharmacokinetic (PK) study showed a mean half-life of 85 hours (coefficient of variation percent [CV%] =21.8) in the 10 IU/kg group and 111 hours (CV% =11.8) in the 40 IU/kg group. The estimated mean 7-day rFIX trough levels after IV injection were 8.5 IU/dL (95% CI 7.7–9.3) for the 10 IU/kg group and 27.3 IU/dL (95% CI 24.8–30.0) for the 40 IU/kg group.36
In the Phase III pivotal trial, rFIX:PEG weekly IV prophylaxis at 10 IU/kg or 40 IU/kg was compared with IV on-demand dosing. Both prophylaxis arms had lower ABRs than on-demand with the 40 IU/kg having the lowest ABR (Table 4). During the prophylaxis period, 45% of patients in the 40 IU/kg group and 17% of those in the 10 IU/kg group experienced no treatment-requiring bleeding episodes. Target joint bleeding resolution was achieved in 66.7% in the 40 IU/kg group and in 7.7% in the 10 IU/kg group. When treating bleeding episodes, in the 40 IU/kg group, 99% resolved with one dose, while in the 10 IU/kg and on-demand groups, 84% resolved with one dose. No inhibitors developed, and no deaths or thromboembolic events were noted. Adverse events were experienced by 81% of patients (3.33 adverse events per patient-year), and the serious adverse event rate was 5.4% with all being unrelated to rFIX:PEG.36
  | Table 4 Annualized bleeding rates by treatment regimen, rFIX:PEG |
rFIX:Albumin
Albumin is the most plentiful plasma protein with a half-life of 20 days. Fusion to albumin prolongs drug half-life via the intracellular processing/trafficking process as well as reduced renal clearance for large molecules.22 Recombinant albumin is also well characterized. rFIX:Albumin is produced in Chinese hamster ovary cells with albumin linked to the C-terminus of rFIX with a cleavable linker derived from region Ser136 or Val137 to Val153 covering the cleavage site composed of the C-terminus of the FIX light chain which was designed to allow albumin removal in parallel with FIX activation.37 This linkage differs from the FIX:PEG construct (activation peptide linkage) but accomplishes the same goal which is to release the albumin from FIXa so as to neither affect the activity of FIX once activated nor prolong the activity of FIXa once formed.37
A Phase I dose escalation study showed that rFIX:albumin was well tolerated with no hypersensitivity reactions noted. The mean (standard deviation) half-life was assessed over three doses and was 104.71 (55.08) hours for 25 IU/kg, 91.57 (20.74) hours for 50 IU/kg, and 98.82 (17.48) hours for 75 IU/kg. The mean (standard deviation) FIX activity on 168 hours (7 days) after a single dose of 25 IU/kg was 7.41 (3.87) IU/dL, for 50 IU/kg was 13.41 (2.91) IU/dL, and for 75 IU/kg was 17.39 (4.46) IU/dL.38,39
New agents for patients with inhibitors
OBIZUR
Due to variations in the amino acid sequence between human and porcine FVIII, there is reduced reactivity of human FVIII antibodies to porcine FVIII. As a consequence, porcine FVIII can be used to treat bleeding episodes in patients with human FVIII antibodies with no or low cross reactivity with porcine FVIII. Plasma-derived porcine FVIII was utilized for the treatment of bleeding in such patients, until it was withdrawn from the market in 2004. Withdrawal of Hyate C was due to the detection of low levels of parvovirus in the drug product. Subsequently, a recombinant B-domain truncated porcine FVIII (OBI-1) containing a 24-amino acid linker comprising the first and last 12 amino acids of the B-domain was developed. First-in-human trials of this molecule revealed higher bioavailability than Hyate C in hemophilia A patients without antibodies to porcine FVIII, and the preparation was found to be well tolerated.40 Subsequent development and clinical trials led, in 2014, to the approval of OBI-1 (OBIZUR) for treatment of bleeding episodes in patients with acquired hemophilia A.41 The 29 enrolled patients who spontaneously developed antibodies to FVIII had a variety of underlying conditions including malignancies and autoimmune disorders. Ten out of the 29 patients had baseline anti-porcine FVIII cross reactivity, and one failed treatment inclusion criteria. Control of qualifying bleed was ultimately achieved in 24 out of 28 patients. Approval for treatment in hemophilia A patients who develop inhibitors has not been sought to this point (Table 5).
Alb-rFVIIa-FP
Alb-rFVIIa-FP is a fusion protein (FP) linking human coagulation Factor VIIa (FVIIa) and human albumin. A single recombinant gene construct is expressed in Chinese hamster ovary cells yielding a recombinant human albumin fused to the C-terminus of rFVIIa via a flexible glycine–serine linker.42–44 Preclinical studies in hemophilia A mice, rats, rabbits, and cynomolgus monkeys confirmed variable but consistent prolongations of the PK effects of Alb-rFVIIa-FP versus rFVIIa.38 PK half-life was prolonged four to five times that of rFVIIa in the various animals. The potency of rFVIIa was noted to be 2.7-fold higher than rFVIIa-FP. This molecule is now in clinical trials, and with a half-life of 8.5 hours, holds promise for on-demand and prophylaxis treatment in hemophilia patients with inhibitors.
EHL-rFVIIa-CTP
FVIIa-CTP (mod-5014), a novel long-acting coagulation factor, displays a prolonged hemostatic effect following IV and SC administration in hemophilic animal models.45 When PK parameters for rFVIIa and FVIIa-CTP were compared in animal models, the half-life and area under the curve of FVIIa-CTP were five- and threefold higher. The carboxyl terminal peptide addition is small and does not significantly increase the solution viscosity. IV and SC administration in animals confirmed the potential for SC administration. Development of the molecule continues with human trials planned to evaluate safety and efficacy of both IV and SC administration in hemophilia patients with inhibitors.
TheraPEG-Factor VIIa
TheraPEG™ is a conjugation technology for site-specific PEGylation at solvent-accessible disulfide bonds.46 PolyTherics Ltd is applying this technique to conjugate PEG to recombinant human clotting factors at a site remote from the active site in an effort to cover epitopes responsible for eliciting an immune response. The goal is to retain clotting activity while reducing immunogenicity, prolonging the plasma half-life and allowing small-volume SC administration of clotting factors. TheraPEG-F VIIa has been administered to hemophilic dogs by both IV and SC routes. When given intravenously, the time over which the whole blood clotting time remained less than 12 minutes (96 hours) was 12 times longer than that seen with rFVIIa (8 hours). When administered via the SC route, TheraPEG-F VIIa had 89% bioavailability and maintained the whole blood clotting time to less than 12 minutes for 72 hours. In the same dog model, rFVIIa given subcutaneously at an equivalent dose did not correct the whole blood clotting time and continues in development.
rFVIIaFc
Recombinant FVIIaFc (rFVIIaFc) is a recombinant Fc FP which uses the neonatal Fc receptor-mediated recycling pathway to protect the FP from catabolism.47 Based on activity, rFVIIaFc has a 5.5 times longer terminal half-life than rFVIIa in hemophilic mice. In a thrombin generation assay and in hemophilia A mouse tail clip model, the potency of rFVIIaFc was comparable to rFVIIa at comparable molar doses. The potential of rFVIIaFc to enable prolonged efficacy awaits additional investigational confirmation.
Nonfactor replacement therapies for hemophilia
Recently, several groups have departed from the normal paradigm of developing new and better versions of replacement clotting factors, to explore the potential and ability of nonclotting factor replacements to correct hemostasis in patients with hemophilia. On the one hand, several approaches involve decreasing the capacity of normal clotting factor inhibitors (tissue factor pathway inhibitor [TFPI], antithrombin, etc) to tamp down clotting activity. Another approach is to engineer a completely new protein to mimic or replace the activity of a protein that is either missing from birth or has been removed by an antibody. While all are relatively early in their development, all have reached human trials and appear to hold promise for treatment across a variety of hemophilia patients. These approaches are summarized in Table 6.
NN-7415 (concizumab/mAB2021) (anti-TFPI)
Concizumab is an anti-TFPI antibody that binds to the K2 domain of TFPI.48 The goal is to reduce the inhibitor activity of TFPI against the tissue factor pathway and thereby enhance the ability of this pathway to support hemostasis. By so doing, the need for intrinsic pathway support of activated Factor X (FXa) production is reduced. As a consequence, the amount of FVIII and FIX needed for normal function is decreased. Since this mechanism should be relevant to FIX deficiency, FVIII deficiency, and to patients with inhibitors to these proteins, this approach may have broader potential applications. The safety and PKs of this molecule have been studied in healthy volunteers and in patients with hemophilia.49 In a first-in-human, Phase I, multicenter, randomized, double-blind, placebo-controlled trial, escalating single IV (0.5–9,000 μg/kg) or SC (50–3,000 μg/kg) doses of concizumab were given to healthy volunteers (n=28) or hemophilia patients (n=24). No serious adverse events occurred, and no antidrug antibodies were seen. Dose-dependent procoagulant effects were noted as increased D-dimers and prothrombin 1 + 2 levels. Noted nonlinear PKs was felt to be due to target-mediated clearance. Further studies are planned to evaluate the potential of SC-administered concizumab in hemophilia treatment.
ALN-AT3 (Alnylam)
ALN-AT3 is an RNAi therapeutic targeting antithrombin 3 (AT3). In a manner analogous to anti-TFPI, ALN-AT3 seeks to reduce the requirement for clotting factors required for the production of FXa by reducing the inhibition of FXa by AT3. Since AT3 is also a primary controller of FXI, FIX, FII, and to some degree FVII, this approach has the potential to modify a variety of clotting factor deficiencies. In animal models, ALN-AT3 yields potent (up to 100%), dose-dependent (1–30 mg/kg), and durable (>30 days) knockdown of AT3.50 This silencing of antithrombin results in up to a fourfold increase in thrombin generation. In a Phase I multiple ascending SC dosing study in normal volunteers (low dose only) and patients with hemophilia A or B, 70% AT3 knockdown with concurrent 334% increase in thrombin generation was noted at a 45 μg/kg dose given weekly for 3 weeks. The AT3 nadir occurred at 28 days. As with the animal models, the effect was durable up to 60 days at the 15 μg/kg dose.51 Further studies are planned to evaluate effects at longer dosing intervals.
ACD-910
ACD-910 is a bispecific antibody with binding specificity for FIX, FIXa, FX, and FXa.52–56 It is a humanized, monoclonal modified antibody designed to replace the cofactor function of FVIII by bringing FIXa and FX into approximate orientation for rapid conversion of FX to FXa. The mode of action is nonclotting factor replacement of FVIII activity, thereby reducing or eliminating the need for FVIII. In a Phase I trial of three dose levels (1 mg/kg loading followed by 0.3 mg/kg weekly, 3 mg/kg loading followed by 1 mg/kg weekly, and 3 mg/kg weekly) given SC, the mean half-life of the molecule was noted to be 28–34 days. Greater than 5% FVIII activity levels were achieved with marked reductions in bleeding even at the lowest dose. At the highest dose, no bleeding occurred. In this 12-week study, anti-ACE-910 antibody developed in one patient. No significant safety concerns have been identified, and additional trials are planned to evaluate the safety and efficacy of this agent for prophylaxis in patients with hemophilia A with and without inhibitors.
Gene therapy for hemophilia
While a review of current activity in gene therapy for hemophilia is beyond the scope of this short review, it is instructive to consider the magnitude of the effort summarized in Table 7. Multiple universities, biotechnology enterprises, and most recently larger pharmaceutical companies have coalesced around the development of gene therapy for hemophilia. After the missteps of the late 1990s, steady progress has led to the current state. Apparently, safe adeno-associated viral (AAV) vectors have been identified for delivery of the FIX gene to hematocicytes in a manner similar to that illustrated for adenovirus in Figure 1. FIX activity levels, sufficient to dramatically reduce spontaneous bleeding, have been obtained in patients with severe disease, and these levels have been maintained for greater than 4 years in some patients.57–59 The use of high-activity variants of FIX may allow levels of activity approaching normal to be achieved without additional improvements in the levels of transduction.60 The importance of preexisting antibodies to vectors and the occurrence of cell-specific T-cell responses capable of eradicating transduced cells have been identified, and approaches to avoidance and treatment are being developed.
  | Table 7 Gene therapy programs for hemophilia |
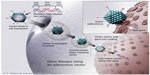  | Figure 1 Gene therapy using an adenovirus vector. |
The progress in gene therapy for hemophilia A has been hampered to a significant degree by the size of the gene. While the FIX gene is accommodated nicely in AAV vectors, the FVIII gene barely fits. While modifications to the FVIII gene cassette to allow accommodation in AAV vectors may offer one avenue to solve this problem, advances in the use of lentiviral vectors that are capable of carrying the FVIII gene may also provide the solution.
Lentiviral vectors potentially offer significant advantages. They are able to efficiently integrate into the target genome, even when the cells are not actively dividing.61 If the liver is the target organ, this can lead to transduction of nondividing hepatocytes with the potential of long-term expression. The liver also contains an abundance of antigen-presenting cells (APCs) that can also be transduced. Transduction of APCs might increase the risk of an immune response to FVIII or FIX and thereby inhibit long-term expression.62 The use of hepatocyte-specific promoters and incorporation of hematopoietic-specific microRNA target sequences into the vector many reduce this immune risk.63–66 While the ability to incorporate endows long-term expression, it also raises concerns regarding insertional mutagenesis caused by oncogene activation or inactivation of tumor-suppressor genes.61 This risk may be reduced by partial removal of the long terminal repeat or by the introduction of an inactivating mutation in the integrase to produce an integration-defective lentiviral vector.61,66 Progress in this area continues at a rapid pace and holds promise to solve the problem of FVIII gene delivery.
Gene therapy for hemophilia B is on the horizon, and gene therapy for hemophilia A is becoming feasible. The question could be asked – why is this being pursued when long-acting agents for hemophilia B should reduce the burden of prophylaxis and some of the agents discussed earlier hold promise to do the same for hemophilia A? The answer is probably found in the fact that while all these advances are improving the lives of patients with hemophilia, all will still require recurrent chronic treatment. The majority of these treatments will require a needle either in a vein or in the skin. There is a desire on the part of patients to be free of these treatments and to be free of the risk of bleeding. A desire to be normal drives the quest for a cure. If stopping bleeding is good, and preventing bleeding is better, then curing the disease must be the best treatment goal.
Disclosure
The Authors are employees of Pfizer Inc. This information represents the authors’ views and does not necessarily reflect those of Pfizer. The authors report no other conflicts of interest in this work.
References
Mannucci PM, Tuddenham EGD. The hemophiliac – from royal genes to gene therapy. N Engl J Med. 2001;344:1773–1779. | |
Bolton-Maggs PH, Pasi KJ. Hemophilias A and B. Lancet. 2003;361:1801–1809. | |
White GC 2nd, Rosendaal F, Aledort LM, Lusher JM, Rothschild C, Ingerslev J. Definitions in hemophilia. Recommendation of the scientific subcommittee on factor VIII and factor IX of the scientific and standardization committee of the International Society on Thrombosis and Haemostasis. Thromb Haemost. 2001;85:560. | |
Mannucci PM. Hemophilia and related bleeding disorders: a story of dismay and success. Hematology Am Soc Hematol Educ Program. 2002;1:1–9. | |
Nilsson IM. Experience with prophylaxis in Sweden. Semin Hematol. 1993;30(3 Suppl 2):16–19. | |
Manco-Johnson MJ, Abshire TC, Shapiro AD, et al. Prophylaxis versus episodic treatment to prevent joint disease in boys with severe hemophilia. N Engl J Med. 2007;357:535–544. | |
Mannucci PM. Desmopressin (DDAVP) in the treatment of bleeding disorders: the first twenty years. Haemophilia. 2000;6(Suppl 1):60–67. | |
Mannucci PM. Hemophilia: treatment options on the twenty-first century. J Thromb Haemost. 2005;1:1349–1355. | |
Mannucci PM. Back to the future: a recent history of haemophilia treatment. Haemophilia. 2008;14(Suppl 3):10–18. | |
White GC, McMillan CW, Kingdon HS, Shoemaker CB. Use of recombinant antihemophilic factor in the treatment of two patients with classic hemophilia. N Engl J Med. 1989;320:166–170. | |
Soucie JM, De Staercke C, Monahan PE, et al; US Hemophilia Treatment Center Network. Evidence for the transmission of parvovirus B19 in patients with bleeding disorders treated with plasma-derived factor concentrates in the era of nucleic acid test screening. Transfusion. 2013;53:1217–1225. | |
Llewelyn CA, Hewitt PE, Knight RS, et al. Possible transmission of variant Creutzfeldt-Jakob disease by blood transfusion. Lancet. 2004;363(9407):417–421. | |
Astermark J. Overview of inhibitors. Semin Hematol. 2006;43(2 Suppl 4):S3–S7. | |
Astermark J, Morado M, Rocino A, et al; EHTSB. Current European practice in immune tolerance induction therapy in patients with haemophilia and inhibitors. Haemophilia. 2006;12:363–371. | |
Ng HJ, Loh YSM, Tan DCL, Lee LH. Thrombosis associated with the use of recombinant activated factor VII: profiling two events. Thromb Haemost. 2004;92:1448–1449. | |
Rosenfeld SB, Watkinson KK, Thompson BH, Macfarlan DE, Lentz SR. Pulmonary embolism after sequential use of recombinant factor VIIa and activated prothrombin complex concentrate in a factor VIII inhibitor patient. Thromb Haemost. 2002;87:925–926. | |
Economou M, Teli A, Tzantzaroudi A, Tsatra I, Zavitsanakis A, Athanassiou-Metaxa M. Sequentila therapy with activated prothrombin complex concentrate (FEIBA) and recombinant factor VIIa in a patient with severe haemophilia A, inhibitor presence and refractory bleeding. Haemophilia. 2008;14:390–391. | |
Mirand GG, Rodgers GM. Treatment of an acquired factor VIII inhibitor with sequential recombinant factor VIIa and FEIBA. Haemophila. 2009;15:383–385. | |
Martinowitz U, Livnat T, Zivelin A, Kenet G. Concomitant infusion of low doses of rFVIIa and FEIBA in haemophilia patients with inhibitors. Haemophilia. 2009;15:904–910. | |
Antunes SV, Tangada S, Stasyshyn O, et al. Randomized comparison of prophylaxis and on-demand regimens with FEIBA NF in the treatment of haemophilia A and B with inhibitors. Haemophilia. 2014;20:65–72. | |
Ducore JM, Miguelino MG, Powell JS. Alprolix (recombinant factor IX Fc fusion protein): extended half-life product for the prophylaxis and treatment of hemophilia B. Expert Rev Hematol. 2014;7:559–571. | |
Kontermann RE. Strategies for extended serum half-life of protein therapeutics. Curr Opin Biotechnol. 2011;22:868–876. | |
Goebl NA, Babbey CM, Datta-Mannan A, Witcher DR, Wroblewski VJ, Dunn KW. Neonatal Fc receptor mediates internalization of Fc in transfected human endothelial cells. Mol Biol Cell. 2008;19:5490–5505. | |
Rath T, Baker K, Dumont JA, et al. Fc-fusion proteins and FcRn: structural insights for longer-lasting and more effective therapeutics. Crit Rev Biotechnol. 2015;35:235–254. | |
Takaki A, Enfield DL, Thompson AR. Cleavage and inactivation of factor IX by granulocyte elastase. J Clin Invest. 1983;72:1706–1715. | |
Shapiro AD, Ragni MV, Valentino LA. Recombinant factor IX-Fc fusion protein (rFIXFc) demonstrates safety and prolonged activity in a phase 1/2a study in hemophilia B patients. Blood. 2012;119:666–672. | |
Peters RT, Low SC, Kamphaus GD, et al. Prolonged activity of factor IX as a monomeric Fc fusion protein. Blood. 2010;115:2057–2064. | |
Powell JS, Pasi KJ, Ragni MV, et al; B-LONG Investigators. Phase 3 study of recombinant factor IX Fc fusion protein in hemophilia B. N Engl J Med. 2013;369:2313–2323. | |
Powell JS, Shapiro A, Ragni M. Switching to recombinant factor IX Fc fusion protein prophylaxis results in fewer infusions, decreased factor IX consumption and lower bleeding rates. Br J Haematol. 2015;168:113–123. | |
Powell JS, Apte S, Chambost H, et al. Long-acting recombinant factor IX Fc fusion protein (rFIXFc) for perioperative management of subjects with haemophilia B in the phase 3 B-LONG study. Br J Haematol. 2015;168:124–134. | |
Abuchowski A, van Es T, Palczuk NC, Davis FF. Alteration of immunological properties of bovine serum albumin by covalent attachment of polyethylene glycol. J Biol Chem. 1977;252:3578–3581. | |
Mei B, Pan C, Jiang H, et al. Rational design of a fully active, long-acting PEGylated factor VIII for hemophilia A treatment. Blood. 2010;116:270–279. | |
Collins PW, Møss J, Knobe K, Groth A, Colberg T, Watson E. Population pharmacokinetic modeling for dose setting of nonacog beta pegol (N9-GP), a glycoPEGylated recombinant factor IX. J Thromb Haemost. 2012;10:2305–2312. | |
Østergaard H, Bjelke JR, Hansen L, et al. Prolonged half-life and preserved enzymatic properties of factor IX selectively PEGylated on native N-glycans in the activation peptide. Blood. 2011;118:2333–2341. | |
Negrier C, Knobe K, Tiede A, Giangrande P, Moss J. Enhanced pharmacokinetic properties of a glycoPEGylated recombinant factor IX: a first human dose trial in patients with hemophilia B. Blood. 2011;118:2695–2701. | |
Collins PW, Young G, Knobe K, et al; Paradigm 2 Investigators. Recombinant long-acting glycoPEGylated factor IX in hemophilia B: a multinational randomized phase 3 trial. Blood. 2014;124:3880–3886. | |
Metzner HJ, Weimer T, Kronthaler U, Lang W, Schulte S. Genetic fusion to albumin improves the pharmacokinetic properties of factor IX. Thromb Haemost. 2009;102:634–644. | |
Santagostino E, Negrier C, Klamroth R, et al. Safety and pharmacokinetics of a novel recombinant fusion protein linking coagulation factor IX with albumin (rIX-FP) in hemophilia B patients. Blood. 2012; 120:2405–2411. | |
Santagostino E. PROLONG-9FP clinical development program – phase I results of recombinant fusion protein linking coagulation factor IX with recombinant albumin (rIX-FP). Thromb Res. 2013;131(Suppl 2):S7–S10. | |
Kempton CL, Abshire TC, Deveras RA, et al. Pharmacokinetics and safety of OBI-1, a recombinant B domain-deleted porcine factor VIII, in subjects with haemophilia A. Haemophilia. 2012;18:798–804. | |
Jruse-Jarres R, St-Louis J, Greist A, et al. Efficacy and safety of OBI-1, an antihaemophilic factor VIII (recombinant), porcine sequence, in subjects with acquired haemophilia A. Haemophilia. 2015;21:162–170. | |
Zollner S, Schuermann D, Raquet E, et al. Pharmacological characteristics of a novel, recombinant fusion protein linking coagulation factor VIIa with albumin (rFVIIa-FP). J Thromb Haemost. 2014;12:220–228. | |
Schulte S. Use of albumin fusion technology to prolong the half-life of recombinant factor VIIa. Thromb Res. 2008;122(Suppl 4):S14–S19. | |
Weimer T, Wormsbächer W, Kronthaler U, Lang W, Liebing U, Schulte S. Prolonged in-vivo half of factor VIIa by fusion to albumin. Thromb Haemost. 2008;99:659–667. | |
Hart G, Hershkovitz O, Lilan AB, Zakar M, Binder L, Fima E. Mod-5014, a novel long-acting FVIIa proposing an improved prophylactic and on demand treatment for hemophilia patients following SC and IV administration comprehensive in-vitro and in-vivo evaluation in preparation for clinical studies. Blood. 2013;122:3578. | |
Balan S, Choi JW, Godwin A, et al. Site-specific PEGylation of protein disulfide bonds using a three-carbon bridge. Bioconjug Chem. 2007;18:61–76. | |
Salas J, Liu T, Lu Q, et al. Enhanced pharmacokinetics of factor VIIa as a monomeric Fc fusion. Thromb Res. 2015;135:970–976. | |
Agersø H, Overgaard RV, Petersen MB, et al. Pharmacokinetics of an anti-TFPI monoclonal antibody (concizumab) blocking the TFPI interaction with the active site of FXa in cynomolgus monkeys after iv and sc administration. Eur J Pharm Sci. 2014;56:65–69. | |
Chowdary P, Lethagen S, Friedrich U, et al; The Explorerâ1 Investigators. Safety and pharmacokinetics of anti-TFPI antibody (concizumab) in healthy volunteers and patients with hemophilia: a randomized first human dose trial. J Thromb Haemost. 2015;13:743–754. | |
Barros SA, Carioto M, Hettinger J, et al. Expanded therapeutic index of antithrombin silencing and correction of APTT in a hemophilia A mouse model. Blood. 2013;122:3585. | |
Sorensen B, Mant T, Akinc A, et al; Aln-AT3 Investigators, International Multicenter Study. A subcutaneously administered RNAi therapeutic (ALN-AT3) targeting antithrombin for treatment of hemophilia: interim phase 1 study results in healthy volunteers and patients with hemophilia A or B. Blood. 2014;124:693. | |
Sampei Z, Igawa T, Soeda T, et al. Non-antigen-contacting region of an asymmetric bispecific antibody to factors IXa/X significantly affects factor VII-mimetic activity. MAbs. 2015;7:120–128. | |
Kitazawa T, Igawa T, Sampei Z, et al. A bispecific antibody to factors IXa and X restores factor VIII hemostatic activity in a hemophilia A model. Nat Med. 2012;18:1570–1574. | |
Sampei Z, Igawa T, Soeda T, et al. Identification and multidimensional optimization of an asymmetric bispecific IgG antibody mimicking the function of factor VIII cofactor activity. PLoS One. 2013;8:e57479. | |
Muto A, Yoshihashi K, Takeda M, et al. Anti-factor IXa/X bispecific antibody (ACE910): hemostatic potency against ongoing bleeds in a hemophilia A model and the possibility of routine supplementation. J Thromb Haemost. 2014;12:206–213. | |
Muto A, Yoshihashi K, Takeda M, et al. The anti-factor IXa/X bispecific antibody ACE910 prevents spontaneous joint bleeds in a long-term primate model of acquired hemophilia A. Blood. 2014;124:3165–3171. | |
High KH, Nathwani A, Spencer T, Lillicrap D. Current status of haemophilia gene therapy. Haemophilia. 2014;20(Suppl 4):43–49. | |
Nathwani AC, Tuddenham EG, Rangarajan S, et al. Adenovirus-associated virus vector-mediated gene transfer in hemophilia B. N Engl J Med. 2011;365:2357–2365. | |
Nathwani AC, Reiss UM, Tuddenham EG, et al. Long-term safety and efficacy of factor IX gene therapy in hemophilia B. N Engl J Med. 2014;371:1994–2004. | |
Crudele JM, Finn JD, Siner JI, et al. AAV liver expression of FIX-Padua prevents and eradicates FIX inhibitor without increasing thrombogenicity in hemophilia B dogs and mice. Blood. 2015;125:1553–1561. | |
Matrai J, Chuah MK, VandenDriessche T. Recent advance in lentiviral vector development and applications. Mol Ther. 2010;18:477–490. | |
VandenDriessche T, Thorrez L, Naldini L, et al. Lentiviral vectors containing the human immunodeficiency virus type-1 central polypurine tract can efficiently transduce nondividing hepatocytes and antigen-presenting cells in vivo. Blood. 2002;100:813–822. | |
Follenzi A, Battaglia M, Lombardo A, Annoni A, Roncarolo MG, Naldini L. Targeting lentiviral vector expression to hepatocytes limits transgene-specific immune response and establishes long-term expression of human antihemophilic factor IX in mice. Blood. 2004;103:3700–3709. | |
VandenDriessche T, Thorrez L, Acosta-Sanchez A, et al. Efficacy and safety of adeno-associated viral vectors based on serotype 8 and 9 vs lentiviral vectors for hemophilia B gene therapy. J Thromb Haemost. 2007;5:16–24. | |
Brown BD, Cantore A, Annoni A, et al. A microRNA-regulated lentiviral vector mediates stable correction of hemophilia B mice. Blood. 2007;110:4144–4152. | |
Matrai J, Cantore A, Bartholomae CC, et al. Hepatocyte-targeted expression by integrase-defective lentivrial vectors induces antigen-specific tolerance in mice with low genotoxic risk. Hepatology. 2011;53:1696–1707. | |
US National Library of Medicine; Gene therapy using an adenovirus vector; Handbook. Available from: http://ghr.nlm.nih.gov/handbook/illustrations/therapyvector. Accessed June 17, 2015. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.



