Back to Journals » Hepatic Medicine: Evidence and Research » Volume 12
Elbasvir/Grazoprevir for HCV Infection in Russia: A Randomized Trial
Authors Zhdanov K, Isakov V , Burnevich E, Kizhlo S, Bakulin I , Pokrovsky V, Liang L, Hwang P, Talwani R, Haber BA , Robertson MN
Received 7 December 2019
Accepted for publication 2 February 2020
Published 21 April 2020 Volume 2020:12 Pages 61—68
DOI https://doi.org/10.2147/HMER.S241418
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Gerry Lake-Bakaar
Konstantin Zhdanov,1 Vasily Isakov,2 Eduard Burnevich,3 Svetlana Kizhlo,4 Igor Bakulin,5 Vadim Pokrovsky,6 Liwen Liang,7 Peggy Hwang,7 Rohit Talwani,7 Barbara A Haber,7 Michael N Robertson7
1Military Medical Academy n.a. S.M. Kirov, St. Petersburg, Russia; 2Department of Gastroenterology & Hepatology, Federal Research Centre of Nutrition, Biotechnology and Food Safety, Moscow, Russia; 3I.M. Sechenov First Moscow State Medical University, Moscow, Russia; 4City Center for AIDS and Infectious Diseases Treatment and Prophylaxis, St. Petersburg, Russia; 5I.I. Mechnikov North-Western State Medical University of the Ministry of Health of the Russian Federation, St. Petersburg, Russia; 6Department of AIDS, Central Research Institute of Epidemiology, Moscow, Russia; 7Merck & Co., Inc., Kenilworth, NJ, USA
Correspondence: Barbara A Haber
Merck & Co., Inc., 2000 Galloping Hill Road, Kenilworth, NJ 07033, USA
Tel +1 267-305-3729
Email [email protected]
Purpose: Hepatitis C virus (HCV) infection is a major healthcare concern in Russia, where almost 5 million individuals are viremic. Elbasvir/grazoprevir is a fixed-dose combination therapy for the treatment of HCV genotype 1 and genotype 4 infection. The present analysis aimed to assess the safety and efficacy of elbasvir/grazoprevir in individuals with HCV infection enrolled at Russian study sites in the C-CORAL study.
Patients and Methods: C-CORAL (Protocol PN-5172-067; NCT02251990) was a Phase 3, placebo-controlled, double-blind study conducted throughout Asia and Russia. Treatment-naive participants with chronic HCV infection were randomly assigned to receive immediate or deferred treatment with elbasvir 50 mg/grazoprevir 100 mg once daily for 12 weeks. Participants in the immediate-treatment group received elbasvir/grazoprevir for 12 weeks, and those in the deferred-treatment group received placebo for 12 weeks, followed by open-label elbasvir/grazoprevir for 12 weeks. The primary endpoint was sustained virologic response at 12 weeks after completion of therapy (SVR12).
Results: One hundred and nineteen Russian participants were randomized (immediate-treatment group, n=88; deferred-treatment group, n=31). Most participants were white (99%) with HCV genotype 1b infection (97%) and mild-to-moderate (F0–F2) fibrosis (70%). SVR12 was achieved by 98.9% participants in the immediate-treatment group and by 100% of those receiving deferred elbasvir/grazoprevir in the deferred-treatment group. One participant relapsed with nonstructural protein 5A (NS5A) L28M and Y93H resistance-associated substitutions at baseline and at time of failure. Drug-related adverse events were reported by 19% of participants receiving elbasvir/grazoprevir in the immediate-treatment group and by 16% of those receiving placebo in the deferred-treatment group. No serious adverse event or deaths occurred, and no participant discontinued treatment owing to an adverse event.
Conclusion: Elbasvir/grazoprevir for 12 weeks was highly effective in treatment-naive Russian individuals with HCV genotype 1b infection.
Keywords: hepatitis C, therapy, placebo, Russia, viral drug resistance
Plain Language Summary
Hepatitis C virus (HCV) infection is a major healthcare concern in Russia. Elbasvir/grazoprevir (EBR/GZR) is a two-drug treatment for HCV genotypes 1 and 4 infection. In this study, 117 of 118 (99%) Russian participants with HCV infection who received EBR/GZR for 12 weeks were cured of their infection. Most had genotype 1b HCV infection. Adverse event rates were similar between the EBR/GZR and placebo groups. Among participants receiving EBR/GZR, no serious safety events occurred and none discontinued treatment because of a safety event. In conclusion, EBR/GZR for 12 weeks was highly effective in Russian individuals with HCV genotype 1b infection.
Introduction
Hepatitis C virus (HCV) infection is a major healthcare concern in Russia, representing the third leading cause of death from infections after AIDS and tuberculosis.1 The prevalence of HCV infection is 3.3% of the national Russian population, amounting to almost 5 million viremic individuals.2 Unlike many other countries in which the prevalence of HCV infection is declining, Russia faces an increasing prevalence driven by new infections among people who inject drugs.2 In 2013, most new acute HCV infections in Russia were among people aged 20–40 years.3 The consequences of HCV infection in Russia are also widespread, as Russian migrants make up an important proportion of people with HCV infection in other European countries. An estimated 49,000 cases of chronic hepatitis C infection have been found among Russian migrants throughout Europe, primarily in Germany, Latvia, Estonia, Italy, Lithuania, and Spain.4 Approximately 70%–80% of HCV infections in Russia are thought to be attributable to HCV genotype (GT) 1b infection.5,6 Despite the high prevalence and clear unmet healthcare need, the proportion of HCV-infected Russian individuals accessing treatment for HCV infection remains very low.7 Oral direct-acting antiviral therapies currently available in Russia are simeprevir, sofosbuvir, glecaprevir/pibrentasvir, paritaprevir/ombitasvir/ritonavir with or without dasabuvir, and elbasvir/grazoprevir.8 However, of the ~5 million HCV-infected individuals in Russia, it is estimated that only 35,000 have currently received direct-acting antiviral therapy, possibly owing to a lack of widespread screening programs and poor access to treatment.8
Elbasvir/grazoprevir (EBR/GZR) is a two-drug fixed-dose combination treatment for HCV GT1 and GT4 infection. The constituent components, both individually and in combination, have been shown to potently inhibit HCV replication in vitro, and phase 3 clinical trials have confirmed the efficacy and safety of this combination across a wide spectrum of individuals with HCV infection.9–15 This combination is approved for the treatment of HCV GT1 and GT4 infection in Europe, the United States, and numerous other countries worldwide. The clinical profile of EBR/GZR in people with HCV infection from Asia and Russia was established in the phase 3 C-CORAL study.16,17 In a study population of 489 participants with HCV GT1, GT4, or GT6 infection receiving EBR/GZR for 12 weeks, 459 (94.4%) achieved a sustained virologic response at 12 weeks after completion of treatment (SVR12). Of the 25 participants with virologic failure in this study, 17 had HCV GT6 infection and were primarily enrolled in Thailand and Vietnam. SVR12 was 98.2% (382/389) in the subpopulation with HCV GT1b infection. In this manuscript, we present the safety and efficacy of EBR/GZR for 12 weeks in the population of 118 participants with HCV infection enrolled at Russian study sites in the C-CORAL study. The aim of this analysis is to inform the clinical use of EBR/GZR for the treatment of HCV infection in Russia.
Methods
C-CORAL (Protocol PN-5172-067; NCT02251990) was a randomized, phase 3, placebo-controlled, double-blind study conducted at 49 sites in mainland China (13), Australia (2), South Korea (6), Taiwan (7), Thailand (3), Vietnam (3), and Russia (15). The study was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines. Independent institutional review boards or ethics committees reviewed and approved the protocol and applicable amendments for each institution, and all participants gave written informed consent. The trial was funded by Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA. The primary outcomes from this study and data from a prespecified interim analysis of participants enrolled at study centers outside China have been published previously.16,17
Participants
Adult treatment-naive participants with chronic HCV GT1, GT4, or GT6 infection and HCV RNA ≥10,000 IU/mL at baseline were enrolled at 15 study sites in Russia. Individuals with and without cirrhosis were eligible, with cirrhosis defined as METAVIR F4 on liver biopsy within 24 months of enrollment, FibroScan® >12.5 kPa within 12 months of enrollment, or a combination of FibroTest® score >0.75 and aspartate aminotransferase (AST):platelet ratio index >2. Individuals with evidence of decompensated liver disease, human immunodeficiency virus (HIV) or hepatitis B virus coinfection, clinically relevant drug or alcohol abuse, or hepatocellular carcinoma or who had previously received any treatment for HCV infection were excluded.
Study Design
Participants were randomized 3:1 to receive immediate or deferred treatment with the fixed-dose combination of EBR 50 mg/GZR 100 mg once daily for 12 weeks. Participants in the immediate-treatment group (ITG) received EBR/GZR for 12 weeks and those in the deferred-treatment group (DTG) received a matched placebo once daily for 12 weeks followed by a 4-week follow-up period and then open-label treatment with EBR/GZR for 12 weeks. Across the study, enrollment was managed to result in populations having ~20% of participants with compensated cirrhosis and ~15% with HCV GT4 or GT6 infection.
Randomization was stratified according to the presence of cirrhosis and country of enrollment (China vs South Korea vs Taiwan vs Russia vs other) using an interactive voice response system/integrated web response system. Participants, site personnel, and the sponsor remained blinded through week 16, after which the treatments were unmasked and participants in the DTG received active treatment.
Procedures
Plasma HCV RNA levels were measured by the COBAS® AmpliPrep/COBAS® TaqMan® HCV test, version 2.0 (Roche Molecular Diagnostics, Branchburg, NJ, USA), with a lower limit of quantitation of 15 IU/mL. HCV genotype was determined using Abbott HCV Real Time Genotype II assay (Abbott Diagnostics, Chicago, IL, USA).
Endpoints
The primary efficacy endpoint was the proportion of participants with a sustained virologic response 12 weeks after completion of therapy (undetectable HCV RNA at follow-up week 12; SVR12). Virologic relapse was defined as HCV RNA >lower limit of quantitation during follow-up after having undetectable HCV RNA at end of treatment. SVR24 was evaluated as a secondary efficacy endpoint. The primary safety outcome was a comparison between the blinded treatment arms (EBR/GZR versus placebo) during the initial 12-week treatment period and up to 14 days after the end of treatment. Safety was assessed by monitoring adverse events (AEs), vital signs, and laboratory evaluations. Events of clinical interest were the first instance of: alanine aminotransferase (ALT) or AST level >500 IU/mL; ALT/AST level >3× baseline and >100 IU/mL; and alkaline phosphatase >3× upper limit of normal (ULN). Late elevations in ALT/AST were also evaluated and defined as ALT/AST elevations >5× ULN after treatment week (TW) 4 in participants who had ALT/AST ≤ULN between weeks 2 and 4. Finally, the presence of resistance-associated substitutions (RASs) was assessed in viral samples from participants who met the criteria for virologic failure and who had HCV RNA >1000 IU/mL.
Statistical Analysis
The target enrollment for the Asia-Pacific region and Russia was 453 participants, with an interim analysis of the ITG from the ex-China cohort prespecified in the protocol. This is a subgroup analysis of the C-CORAL study, and hence, the study was not powered for an assessment of efficacy in the Russia cohort. The full analysis set population was used for the primary efficacy analysis (all randomized participants who received at least one dose of study treatment). Two-sided 95% asymptotic confidence intervals (CI) were calculated for SVR12 results. Safety analyses were conducted in all participants who received at least one dose of study medication.
Results
Participants
A total of 129 participants were screened in Russia, of whom ten failed screening; the remaining 119 participants were randomly assigned (ITG, n=88; DTG, n=31). The first participant received the first dose of study medication on August 5, 2015, and the last participant in the ITG completed 12 weeks of follow-up on February 25, 2016. All participants in the ITG completed 12 weeks of treatment and 12 weeks of follow-up. One participant in the DTG was found to have undetectable HCV RNA during the placebo treatment phase and was discontinued from the study prior to starting deferred active treatment. As a result, 31 participants in the DTG were eligible for inclusion in the primary safety comparison but only 30 received deferred active treatment and were eligible for evaluation of SVR12.
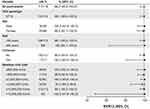
Most participants in the Russian cohort were white (n=117, 99.2%), with a slightly higher proportion of females compared with males (57.6% vs 42.4%) (Table 1). Most had HCV GT1b infection (n=114, 96.6%), and 39% had baseline viral load ≤800,000 IU/mL. The majority of participants had mild-to-moderate (F0–F2) fibrosis, but 17 participants with cirrhosis were also included in this study cohort. Within the Russian cohort, demographic characteristics were well-balanced between the immediate- and deferred-treatment groups.
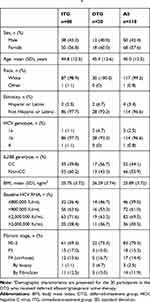 |
Table 1 Participant Demographicsa |
Virologic Response
SVR12 was achieved by 98.9% (87/88) participants in the ITG and 100% (30/30) of those receiving deferred EBR/GZR in the DTG, yielding an overall SVR12 rate of 99% (117/118) (Figure 1). With only one participant failing to achieve SVR12, SVR12 rates were high across all participant subgroups. In the combined ITG and DTG populations, SVR12 was achieved by 113/114 (99.1%) participants with HCV GT1b infection, by all three participants with GT1a infection, and by the single participant with HCV GT4 infection. SVR12 rates were unaffected by sex, age, presence or absence of cirrhosis, or baseline viral load (Figure 2).
Of the 114 participants with HCV GT1b infection, 16 (14%) had one or more baseline nonstructural protein 5A (NS5A) RAS at amino acid positions 28, 30, 31, or 93. Of these 16 participants, 15 achieved SVR12, yielding an SVR12 rate of 94% among participants with baseline NS5A RASs. All participants with no baseline NS5A RAS achieved SVR12.
A noncirrhotic woman aged <65 years with HCV GT1b infection and baseline plasma HCV RNA >2,000,000 IU/mL experienced relapse after having undetectable HCV RNA at end of treatment. This participant had NS5A L28M and Y93H RASs at baseline and at time of failure. Analysis of nonstructural protein 3 (NS3) RASs was not conducted on baseline samples, but a V132I RAS was also detected at the time of failure.
Safety
The primary safety analysis was a comparison of safety events between the randomized ITG and the initial placebo treatment phase of the DTG. AEs were reported in similar proportions of participants receiving EBR/GZR and placebo (35.2% vs 32.3%), with asthenia (5.7% vs 3.2%) and headache (6.8% vs 3.2%) being the only AEs reported at an incidence >5% in either treatment group (Table 2). Drug-related AEs were reported by 17 participants (19.3%) receiving EBR/GZR in the ITG and by five participants (16.1%) receiving placebo in the DTG. No serious AEs or deaths occurred, and no participant discontinued treatment because of an AE in the ITG or when receiving placebo in the DTG.
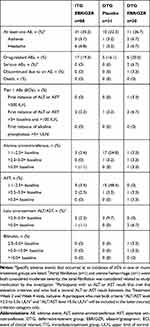 |
Table 2 Adverse Events |
Three events of clinical interest occurred. Two participants in the ITG receiving EBR/GZR and one participant receiving placebo in the DTG had an ALT or AST level >3× baseline and >100 U/L. Three participants from each group also had late ALT/AST elevations >2× baseline after having normalized transaminase levels through treatment weeks 2–4 (only one of these participants receiving EBR/GZR had a late transaminase elevation >5× baseline). In all cases, transaminase elevations were asymptomatic and were not associated with bilirubin elevations.
The safety profile of EBR/GZR administered for 12 weeks was also similar in the active treatment phase of the DTG. Two participants reported serious AEs of paroxysmal atrial fibrillation and uterine bleeding: the AE of paroxysmal atrial fibrillation was considered related to study medication in the opinion of the investigator. Three events of clinical interest occurred (one instance of ALT or AST level >500 IU/L and two participants with ALT or AST level >3× baseline and >100 IU/L), and two participants met the criteria for a late ALT/AST elevation >5× baseline. One participant in the DTG discontinued treatment owing to a grade 2 increase in ALT, which was considered related to study medication. This participant achieved SVR12.
Discussion
Previously reported data from the C-CORAL study indicate that EBR/GZR for 12 weeks is a safe and highly effective treatment option for people with HCV GT1 infection from the Asia-Pacific region and Russia.16,17 In the present analysis, these observations were extended to confirm high rates of SVR12 among participants enrolled in Russia. This was a treatment-naive population with primarily HCV GT1b infection: 70% had mild to moderate (F0–F2) liver fibrosis, and 39% had baseline HCV RNA <800,000 IU/mL. Only one participant from Russia had virologic failure: this participant experienced relapse with L28M and Y93H RASs present at baseline and at time of failure. Testing for baseline NS5A RASs is not required prior to treatment of individuals with HCV GT1b infection with EBR/GZR.18 Safety of EBR/GZR in this Russian population was similar to that in previous reports in similar studies,12 with no difference in safety profile between EBR/GZR and placebo treatment arms.
The SVR12 rates achieved in Russian participants are similar to those attained in participants from several other countries that participated in the C-CORAL study. Notably, high rates of SVR12 were achieved in participants from China (146/151, 97%), South Korea (48/50, 96%), Taiwan (83/85, 98%) and Australia (26/28, 93%), but rates were lower in those from Vietnam (27/33, 82%) and Thailand (12/21, 57%), where the majority of virologic failures occurred in participants with HCV GT6 infection. EBR/GZR is not approved for the treatment of individuals with HCV GT6 infection. Overall rates of SVR12 in the C-CORAL population (including participants from both the Asia-Pacific region and Russia) were 94.4% (459/486), including participants who received EBR/GZR for 12 weeks in both the immediate and deferred treatment arms. The present analysis population, which includes only the Russian subgroup of participants from this previously reported dataset, accounts for 24.3% of the total C-CORAL population and is largely representative of the overall study results.17
Several other studies have also examined the safety and efficacy of direct-acting antiviral therapies for the treatment of HCV infection in Russian patients. SVR12 was achieved by 99% (118/119) of participants from Russia (n=103) and Sweden (n=16) receiving sofosbuvir/velpatasvir for 12 weeks.19 In this study, 59% of patients had HCV GT1b infection, 76% were treatment naive, and 18% had cirrhosis. One case of virologic failure occurred in a participant with GT3 infection who had a treatment-emergent Y93H RAS at the time of failure. High rates of SVR have also been reported with an 8-week regimen of ledipasvir/sofosbuvir in treatment-naive, noncirrhotic Russian participants with HCV GT1 infection (86% had HCV GT1b infection).20 SVR rates were 100% (67/67) in persons with HCV mono-infection and 97% (57/59) in those with HCV/HIV coinfection. Two participants had relapse, both with HCV GT1a infection and HIV coinfection, and both were receiving concomitant efavirenz, lamivudine, and azidothymidine antiretroviral therapy; neither had NS5A RASs present at the time of failure. In the TURQUOISE IV study, an SVR12 rate of 100% (36/36) was achieved in Russian patients with HCV GT1b infection and compensated cirrhosis who received ombitasvir/paritaprevir/ritonavir once daily plus dasabuvir and ribavirin twice daily for 12 weeks.21 This study population included 69% pegylated interferon/ribavirin treatment–experienced participants, and most (92%) had a Child-Pugh score of 5. Finally, the efficacy of sofosbuvir plus ribavirin for 16 or 24 weeks was evaluated in treatment-naive Russian individuals with HCV GT1 or GT3 infection.22 Among participants with GT3 infection, SVR12 rates of 87% (26/30) and 90% (28/31) were achieved in those treated for 16 or 24 weeks. In those with HCV GT1 infection, however, the SVR12 rate was lower in the 16-week arm (16/32, 50%) than in the 24-week arm (26/34, 74%), with virologic failure associated with the baseline presence of the NS5B L159F substitution. Among participants with the L159F RAS, SVR12 rates were 25% (3/12) in those treated for 16 weeks and 80% (8/10) in those treated for 24 weeks.
Interpretation of the data from this analysis is subject to several limitations. The study population was a relatively easy-to-cure population comprising treatment-naive individuals with HCV GT1b infection, the majority of whom had mild to moderate liver fibrosis. Caution should be exercised when extrapolating these data to other Russian populations such as treatment-experienced individuals or those with advanced liver fibrosis/cirrhosis, although other studies have confirmed the safety/efficacy profile of EBR/GZR in these populations.13,23 In particular, Kwo and colleagues reported SVR12 rates of 100% (34/34) in peginterferon/ribavirin treatment–experienced participants with HCV GT1b infection receiving EBR/GZR for 12 weeks, with no impact of baseline NS5A RAS.13 As a secondary analysis of the C-CORAL study, these data are also limited by the small size of the study population compared with the original study population and should not be considered as completely original, as this is a subgroup analysis. The number of participants in several of the subgroups assessed in the present analysis was also low, such as those aged >65 years, those with baseline HCV RNA >10,000,000 IU/mL, and those with cirrhosis. Individuals with decompensated liver disease were not enrolled in the C-CORAL study, and the safety profile of EBR 50 mg/GZR 100 mg has not been studied in people with decompensated liver disease.
In conclusion, EBR/GZR for 12 weeks was highly effective in treatment-naive Russian individuals with HCV GT1b infection. The safety observations in this study were similar in participants receiving EBR/GZR and those receiving placebo, suggesting that the safety profile of EBR/GZR does not differ between individuals with HCV infection in Russia and those from other Western countries. The data from this analysis confirm that EBR/GZR is a highly effective treatment option for Russian people with HCV GT1b infection.
Abbreviations
AE, adverse event; ALT, alanine aminotransferase; AST, aspartate aminotransferase; DTG, deferred-treatment group; EBR, elbasvir; GT, genotype; GZR, grazoprevir; HCV, hepatitis C virus; ITG, immediate-treatment group; NS3, nonstructural protein 3; NS5A, nonstructural protein 5A; RAS, resistance-associated substitution; SVR, sustained virologic response; SVR12, sustained virologic response at 12 weeks after completion of treatment; ULN, upper limit of normal.
Ethics Approval
The study was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines. Independent institutional review boards or ethics committees (see supplementary material) reviewed and approved the protocol and applicable amendments for each institution, and all participants gave written informed consent.
Data Sharing Statement
Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA’s data sharing policy, including restrictions, is available at http://engagezone.msd.com/ds_documentation.php. Requests for access to the clinical study data can be submitted through the EngageZone site or via email to [email protected].
Acknowledgments
Medical writing assistance was provided by Tim Ibbotson, PhD, of ApotheCom (Yardley, PA, USA) and funded by Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA.
Author Contributions
All authors contributed to data analysis, drafting and revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Funding
Funding for this study was provided by Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA. The study sponsor designed the protocol, conducted the study, performed the data analyses, and was responsible for the development and submission of the manuscript.
Disclosure
Konstantin Zhdanov, Eduard Burnevich, Svetlana Kizhlo, Igor Bakulin, and Vadim Pokrovsky have no conflicts of interest to disclose. Vasily Isakov reports personal fees from Merck, AbbVie, Gilead, Echosens, and R-pharm. Liwen Liang, Peggy Hwang, Barbara A Haber, and Michael N Robertson are employees of Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA, and hold stock in Merck & Co., Inc., Kenilworth, NJ, USA. Rohit Talwani is an ex-employee of Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA, and holds stock in Merck & Co., Inc., Kenilworth, NJ, USA. The authors report no other conflicts of interest in this work.
References
1. Russian Federal State Statistics Service. Available from: http://www.gks.ru/wps/wcm/connect/rosstat_main/rosstat/ru/statistics/population/demography/#28/05/2019.
2. Polaris Observatory HCVC. Global prevalence and genotype distribution of hepatitis C virus infection in 2015: a modelling study. Lancet Gastroenterol Hepatol. 2017;2(3):161–176. doi:10.1016/S2468-1253(16)30181-9
3. Mukomolov S, Trifonova G, Levakova I, Bolsun D, Krivanogova E. Hepatitis C in the Russian Federation: challenges and future directions. Hepat Med. 2016;8:51–60. doi:10.2147/HMER.S50172
4. Falla AM, Ahmad AA, Duffell E, Noori T, Veldhuijzen IK. Estimating the scale of chronic hepatitis C virus infection in the EU/EEA: a focus on migrants from anti-HCV endemic countries. BMC Infect Dis. 2018;18(1):42. doi:10.1186/s12879-017-2908-5
5. Lvov DK, Samokhvalov EI, Tsuda F, et al. Prevalence of hepatitis C virus and distribution of its genotypes in Northern Eurasia. Arch Virol. 1996;141(9):1613–1622. doi:10.1007/bf01718286
6. Viazov S, Kuzin S, Paladi N, et al. Hepatitis C virus genotypes in different regions of the former Soviet Union (Russia, Belarus, Moldova, and Uzbekistan). J Med Virol. 1997;53(1):36–40.
7. Tsui JI, Ko SC, Krupitsky E, et al. Insights on the Russian HCV care cascade: minimal HCV treatment for HIV/HCV co-infected PWID in St. Petersburg. Hepatol Med Policy. 2016;1:13. doi:10.1186/s41124-016-0020-x
8. Isakov VA Elimination & microelimination of HCV: Russian perspective.
9. Summa V, Ludmerer SW, McCauley JA, et al. MK-5172, a selective inhibitor of hepatitis C virus NS3/4a protease with broad activity across genotypes and resistant variants. Antimicrob Agents Chemother. 2012;56(8):4161–4167. doi:10.1128/AAC.00324-12
10. Harper S, McCauley JA, Rudd MT, et al. Discovery of MK-5172, a macrocyclic hepatitis C virus NS3/4a protease inhibitor. ACS Med Chem Lett. 2012;3(4):332–336. doi:10.1021/ml300017p
11. Coburn CA, Meinke PT, Chang W, et al. Discovery of MK-8742: an HCV NS5A inhibitor with broad genotype activity. ChemMedChem. 2013;8(12):1930–1940. doi:10.1002/cmdc.v8.12
12. Zeuzem S, Ghalib R, Reddy KR, et al. Grazoprevir-elbasvir combination therapy for treatment-naive cirrhotic and noncirrhotic patients with chronic HCV genotype 1, 4, or 6 infection: a randomized trial. Ann Intern Med. 2015;163(1):1–13. doi:10.7326/M15-0785
13. Kwo P, Gane E, Peng CY, et al. Effectiveness of elbasvir and grazoprevir combination, with or without ribavirin, for treatment-experienced patients with chronic hepatitis C infection. Gastroenterology. 2017;152(1):164–175. doi:10.1053/j.gastro.2016.09.045
14. Rockstroh JK, Nelson M, Katlama C, et al. Efficacy and safety of grazoprevir (MK-5172) and elbasvir (MK-8742) in patients with hepatitis C virus and HIV co-infection (C-EDGE CO-INFECTION): a non-randomised, open-label trial. Lancet HIV. 2015;2(8):e319–e327. doi:10.1016/S2352-3018(15)00114-9
15. Roth D, Nelson DR, Bruchfeld A, et al. Grazoprevir plus elbasvir in treatment-naive and treatment-experienced patients with hepatitis C virus genotype 1 infection and stage 4–5 chronic kidney disease (the C-SURFER study): a combination phase 3 study. Lancet. 2015;386(10003):1537–1545. doi:10.1016/S0140-6736(15)00349-9
16. George J, Burnevich E, Sheen IS, et al. Elbasvir/grazoprevir in Asia-Pacific/Russian participants with chronic hepatitis C virus genotype 1, 4, or 6 infection. Hepatol Commun. 2018;2(5):595–606. doi:10.1002/hep4.1177
17. Wei L, Jia JD, Wang FS, et al. Efficacy and safety of elbasvir/grazoprevir in participants with hepatitis C virus genotype 1, 4, or 6 infection from the Asia-Pacific region and Russia: final results from the randomized C-CORAL study. J Gastroenterol Hepatol. 2019;34(1):12–21. doi:10.1111/jgh.14636
18. Komatsu TE, Boyd S, Sherwat A, et al. Regulatory analysis of effects of hepatitis C virus NS5A polymorphisms on efficacy of elbasvir and grazoprevir. Gastroenterology. 2017;152(3):586–597. doi:10.1053/j.gastro.2016.10.017
19. Isakov V, Chulanov V, Abdurakhmanov D, et al. Sofosbuvir/velpatasvir for the treatment of HCV: excellent results from a phase-3, open-label study in Russia and Sweden. Infect Dis (Lond). 2019;51(2):131–139. doi:10.1080/23744235.2018.1535186
20. Isakov V, Gankina N, Morozov V, et al. Ledipasvir-sofosbuvir for 8 weeks in non-cirrhotic patients with previously untreated genotype 1 HCV infection ± HIV-1 co-infection. Clin Drug Invest. 2018;38(3):239–247. doi:10.1007/s40261-017-0606-0
21. Isakov V, Paduta D, Viani RM, et al. Ombitasvir/paritaprevir/ritonavir+dasabuvir+ribavirin for chronic hepatitis C virus genotype 1b-infected cirrhotics (TURQUOISE-IV). Eur J Gastroenterol Hepatol. 2018;30(9):1073–1076. doi:10.1097/MEG.0000000000001166
22. Isakov V, Zhdanov K, Kersey K, et al. Efficacy of sofosbuvir plus ribavirin in treatment-naive patients with genotype-1 and -3 HCV infection: results from a Russian phase IIIb study. Antivir Ther. 2016;21(8):671–678. doi:10.3851/IMP3065
23. Jacobson IM, Lawitz E, Kwo PY, et al. Safety and efficacy of elbasvir/grazoprevir in patients with hepatitis C virus infection and compensated cirrhosis: an integrated analysis. Gastroenterology. 2017;152(6):1372–1382. doi:10.1053/j.gastro.2017.01.050
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.