Back to Journals » Clinical Ophthalmology » Volume 9
Efficiency, safety, and patient preference of switching from dorzolamide 1%/timolol 0.5% to brinzolamide 1%/timolol 0.5% while maintaining the prostaglandin F2α analog
Authors Shimizu Y, Nakakura S, Nishiyama M, Tabuchi H , Kiuchi Y
Received 21 December 2014
Accepted for publication 28 January 2015
Published 12 March 2015 Volume 2015:9 Pages 475—482
DOI https://doi.org/10.2147/OPTH.S79680
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Scott Fraser
Yoshie Shimizu,1 Shunsuke Nakakura,1 Makiko Nishiyama,1 Hitoshi Tabuchi,1 Yoshiaki Kiuchi2
1Department of Ophthalmology, Saneikai Tsukazaki Hospital, Himeji, 2Department of Ophthalmology and Visual Sciences, Graduate School of Biomedical Sciences, Hiroshima University, Hiroshima, Japan
Background: We investigated the efficiency, safety and patient preference of switching from dorzolamide 1%/timolol 0.5% to brinzolamide 1%/timolol 0.5% while maintaining the prostaglandin F2α analog.
Methods: We initially enrolled 44 eyes from 44 primary open angle glaucoma patients, and a total of 42 patients completed the study. All patients were under treatment with various prostaglandin F2α analogs and dorzolamide 1%/timolol 0.5%. While maintaining the prostaglandin F2α analog, dorzolamide 1%/timolol 0.5% was switched to brinzolamide 1%/timolol 0.5%. Conjunctival hyperemia, superficial punctate keratopathy, and intraocular pressure (IOP) were evaluated at baseline and at 4, 12, and 24 weeks. Adverse events and patient preferences, measured using a questionnaire at study initiation and at 24 weeks, were also noted.
Results: The IOP was 17.7±1.7, 16.8±2.6, 16.7±2.2, and 16.7±2.4 mmHg at baseline and at 4, 12, and 24 weeks, respectively, with no significant differences in IOP values at any time point (P=0.117, one-way analysis of variance). In addition, no significant differences were found in the incidence of conjunctival hyperemia or SPK score at any time point (all P>0.5, by Kruskal–Wallis test). Based on the evaluation of side effects using the questionnaire, stinging/burning was less common (P=0.042), while blurred vision was more common (P=0.003), after switching to brinzolamide 1%/timolol 0.5%. Regarding patient preferences, 13 patients (31%) preferred dorzolamide 1%/timolol 0.5%, 12 patients (29%) preferred brinzolamide 1%/timolol 0.5%, and 17 patients (40%) preferred neither.
Conclusion: When switching from dorzolamide 1%/timolol 0.5% to brinzolamide 1%/timolol 0.5%, the IOP values and incidence of superficial punctate keratopathy and conjunctival hyperemia were sustained throughout the 24-week observation period, and the patient preferences were similar for the two regimens. However, differences were observed in the ocular sensations of stinging/burning with dorzolamide 1%/timolol 0.5% and blurred vision with brinzolamide 1%/timolol 0.5%.
Keywords: glaucoma, prostaglandin F2α, brinzolamide, dorzolamide, fixed combination, timolol
Introduction
Fixed combination therapy has an advantage with respect to patient adherence in subjects with glaucoma by decreasing the number of eye bottles required.1,2 The effectiveness of this treatment has been shown to be equal to or greater than that of combination therapy.3–8 Carbonic anhydrase inhibitor and beta-blocker fixed combination therapies are now available in two types: dorzolamide 2% or 1%/timolol 0.5% and brinzolamide 1%/timolol 0.5%. According to a dose-response study of dorzolamide at 0.5%, 1%, and 2% in Japanese patients;9 1% dorzolamide is used in Japan and applied as a fixed combination with timolol dorzolamide 1%/timolol 0.5% (Cosopt®, Santen Pharmaceutical Co Ltd, Osaka, Japan).
Several previous reports have compared the efficiency and safety of fixed combination therapy, including dorzolamide 2%/timolol 0.5% and brinzolamide 1%/timolol 0.5%,10–17 with many authors reporting similar intraocular pressure (IOP)-lowering efficacy for these treatments.11,15,16
Regarding patient preferences, some reports have shown that patients prefer brinzolamide 1%/timolol 0.5% when asked an alternative question.10,13,16,17 However, in clinical practice, we often hesitate over which eye drop to choose because some patients cannot tolerate brinzolamide 1%/timolol 0.5% but can tolerate dorzolamide 1%/timolol 0.5%. We suspect that patients’ real preferences cannot be judged by two selections, and in actuality some patients may be tolerant to both. Conversely, whether dorzolamide 1%/timolol 0.5% has a similar IOP-lowing efficacy to that of dorzolamide 2%/timolol 0.5% is uncertain.
To our best knowledge, no previous reports have compared the IOP values or real patient preferences between dorzolamide 1%/timolol 0.5% and brinzolamide 1%/timolol 0.5% and/or combination therapy with prostaglandin F2α, ie, the major full medication regime for medical treatment of glaucoma.
The primary purpose of this study was to investigate the efficiency and safety of switching from dorzolamide 1%/timolol 0.5% ophthalmic solution to brinzolamide 1%/timolol 0.5% ophthalmic suspension (Azorga®, Alcon Laboratories Inc, Fort Worth, TX, USA) for a 24-week period. The second purpose was to investigate the incidence of side effects and real patient preferences using a questionnaire administered before and after switching therapy.
Patients and methods
We conducted an open-label, prospective, single-center study. The study protocol received approval from the institutional review board at Saneikai Tsukazaki Hospital and was conducted according to the tenets of the Declaration of Helsinki. Written informed consent was obtained from each participant prior to enrollment in the study.
The patients were enrolled between December 2013 and March 2014 at Saneikai Tsukazaki Hospital. All patients had received a diagnosis of primary open angle glaucoma and had attended our clinic for more than one year and been prescribed the same medication regimen (prostaglandin F2α analog + dorzolamide 1%/timolol 0.5%) for more than 6 months. The diagnosis of primary open angle glaucoma was made based on the results of a gonioscopic examination showing an open angle and the presence of visual field defects in the Humphrey 30-2 SITA program (Humphrey Field Analyzer; Carl Zeiss Meditec, Dublin, CA).
In at least one of the eyes whose location corresponded to glaucomatous disc excavation, namely the presence of a focal or diffuse defect of the optic disc rim with or without retinal nerve fiber layer defects. Additional criteria included a stable IOP for more than 3 months, as measured using a Goldmann applanation tonometer, with values greater than 14 mmHg and treatment with various preparations of prostaglandin F2α analog + dorzolamide 1%/timolol 0.5%. If both eyes met the criteria, both eyes were switched to brinzolamide 1%/timolol 0.5% and the data for the right eye was included in the statistical analysis.
The exclusion criteria were as follows: congenital or narrow-angle glaucoma; ocular surgery, including laser surgery, within the previous 6 months; ocular inflammation, neovascular glaucoma, or steroid-induced glaucoma; inability to perform measurements for IOP by a Goldmann applanation tonometer; risk of worsening of visual acuity or field during the study; and allergy to preservatives.
The baseline visit assessments involved measurements of IOP using a Goldmann applanation tonometer on two occasions (the average value was used for the subsequent analysis), slit-lamp examinations, including funduscopy (evaluated by SN), visual field tests (Humphrey 30-2 SITA program) conducted within 3 months, and age, sex, medical history, history of cataracts and/or glaucoma surgery, and current anti-glaucoma drugs (evaluated by MN). Photographs of conjunctival hyperemia and superficial punctate keratopathy (SPK, area-density [AD] classification)18 were obtained by SN using slit-lamp photography prior to the IOP measurements. Conjunctival hyperemia and SPK scores were judged independently by YS. The conjunctival hyperemia score was determined using a four-grade photographic scale (slight, 0; mild, +1; moderate, +2; severe, +3) and the SPK score was assessed on fluorescein staining observed using a blue-free filter and evaluated according to the AD classification.18 The AD score was calculated by adding the area score (0–3) and density score (0–3).
A written questionnaire to assess current treatment with eye drops was administered in order to record adverse effects. Adverse effects were evaluated on the written questionnaire based on six questions regarding the patient’s experience of stinging/burning, foreign body sensations, blurred vision, conjunctival hyperemia, frequency of forgetting to apply eye drops, and comfort while administering the drops (Figure 1). Stinging/burning, foreign body sensations, blurred vision, and conjunctival hyperemia were judged on a three-point response scale, ie, yes, no, or neither. The frequency of forgetting to take the drops was judged as never, once, or twice per week or more than three times per week. The patient’s level of comfort was judged as comfortable, neither comfortable nor uncomfortable, or uncomfortable.
  | Figure 1 Questionnaire. |
The patient preference between the two eye drops was judged on a three-point scale: Cosopt (dorzolamide 1%/timolol 0.5%), Azorga (brinzolamide 1%/timolol 0.5%), or neither.
Patients deemed eligible for the study were enrolled and switched from dorzolamide 1%/timolol 0.5% (two times per day) to brinzolamide 1%/timolol 0.5% (two times per day) with the maintenance of a single prostaglandin F2α analog without a washout period.
Each patient was advised to keep the same method of instillation before and after switching the therapy. In order not to wash out the previous eye drop he two eye drops (prostaglandin F2α analog and dorzolamide 1%/timolol 0.5%) should be applied more than 5 minutes apart (two times a day).
Follow-up visits were made at 4, 12, and 24 weeks. Assessments of the IOP (two measurements), conjunctival hyperemia score, and SPK score were conducted at each visit, and all examinations were performed between 9 am and noon in order to avoid the effects of diurnal IOP fluctuations. The written questionnaire to monitor adverse effects and patient preferences regarding the two fixed combination therapies was administered again at 24 weeks.
Statistical analysis
The statistical analysis was performed using JMP software program version 10.0.0 (SAS Institute Inc, Cary, NC, USA) and the Statcel 3 software program (OMS Publishing Ltd, Tokyo, Japan). The values are shown as the mean ± standard deviation. Differences in IOP between the visits were evaluated using one-way analysis of variance to analyze the differences at individual time points after switching treatment. With respect to SPK (AD score) and conjunctival hyperemia, the Kruskal–Wallis test was used to compare differences between scores at individual time points. All questions on the questionnaire were evaluated according to the χ2 test. P-values less than 0.05 were considered to be statistically significant. The sample size to detect a 1.5 or 1.0 mmHg difference between visits with a significance level of 5% and power of 80%, based on a standard deviation of 1.79 mmHg in baseline IOP, was estimated to be 24 and 52 patients, respectively.
Results
Patients
Based on our criteria, 44 eyes from 44 patients were enrolled during the study period. Two patients dropped out of the study at 4 weeks due to stinging and blurred vision caused by brinzolamide 1%/timolol 0.5%. Finally, a total of 42 patients completed the study, and their data were used for the subsequent analysis. Patient demographic data are shown in Table 1. The mean patient age was 66.7 years and 22 patients were males. Although the combination of prostaglandin analogs prescribed varied, travoprost without preservative was the most common (57.1%).
  | Table 1 Patient demographics |
Intraocular pressure
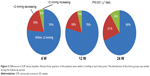
The IOP was 17.7±1.7 (range 14–22) mmHg at baseline, 16.8±2.6 (range 11–24) mmHg at 4 weeks, 16.7±2.2 (range 12–25) mmHg at 12 weeks, and 16.7±2.4 (range 11–23) mmHg at 24 weeks. There were no significant differences between the time points (P=0.117, one-way analysis of variance, Figure 2). Further, we evaluated the differences in IOP between the baseline and follow-up visits. The IOP differences were classified into three groups, ie, within 2 mmHg, a <2 mmHg increase, and a <2 mmHg decrease. The distribution of IOP differences compared with baseline is shown in Figure 3. Almost three quarters of the patients were within 2 mmHg at each time point. The distribution of the three groups was similar during the follow-up period (P=0.821 by χ2 test).
Superficial punctate keratopathy and conjunctival hyperemia
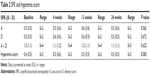
The conjunctival hyperemia scores and the degree of SPK assessed using the AD classification are shown in Table 2. There were no significant differences at any of the time points (all P>0.5, by Kruskal–Wallis test).
  | Table 2 SPK and hyperemia score |
Adverse effects evaluated on written questionnaire
Apparent decreases (ten patients) were observed in response to the question regarding experiences with “stinging/burning” (P=0.042). Apparent increases (14 patients) were observed in response to the question regarding experiences with “blurred vision” (P=0.003). No significant differences were observed in the responses to the other questions (Table 3).
  | Table 3 Adverse effects evaluated by written questionnaire |
Patient preferences on questionnaire
At 24 weeks, 12 patients (29%) preferred Azorga (brinzolamide 1%/timolol 0.5%), 13 patients (31%) preferred Cosopt (dorzolamide 1%/timolol 0.5%), and 17 patients (40%) preferred neither.
Discussion
Intraocular pressure
The present study demonstrated that the IOP-lowering efficacy of brinzolamide 1%/timolol 0.5% in combination with prostaglandin F2α is similar to that of dorzolamide 1%/timolol 0.5% in combination with prostaglandin F2α in Japanese patients treated for 24 weeks (P=0.117). Changing the eye drop prescription itself may affect patient adherence and result in decreases in IOP within a short time period. However, the follow-up period in this study was relatively long, and would be expected to provide more accurate information as to whether the drug is truly effective and safe.
Sezgin et al demonstrated that the IOP-lowering efficacy of brinzolamide 1%/timolol 0.5% is similar to that of the dorzolamide 2%/timolol 0.5% fixed combination.16 The IOP reductions achieved with brinzolamide1%/timolol 0.5% fixed therapy ranged from 6.42 to 9.74 mmHg (26%–37%), whereas treatment with dorzolamide 2%/timolol 0.5% produced a mean IOP reduction ranging from 8.16 to 12.41 mmHg (31%–41%; P>0.05) in a 3-month, randomized, double-blinded, active-controlled, parallel-group trial.16 In contrast, Lanzl and Raber reported that their patients who transitioned from dorzolamide 2%/timolol 0.5% to brinzolamide 1%/timolol 0.5% (n=2,937) demonstrated a significant decrease in mean IOP (P<0.001).13
Dorzolamide is used at a 1% concentration in Japan, whereas 2% solutions are used in European countries, and in South and north America. Hence, our data may not directly compare with those of previous reports or future studies from these areas. Nevertheless, Kitazawa et al reported in their dose-response study that the percentage reduction from baseline was greater after treatment with 0.5%, 1%, or 2% dorzolamide than after treatment with 0.2% dorzolamide. The authors concluded that the IOP-lowering activity dose-response curve for dorzolamide may reach a plateau at concentrations ≥0.5%.9
Superficial punctate keratopathy
It is well known that anti-glaucoma medications affect the ocular surface and tear balance.12,19–28 In the current study, there was no significant difference in conjunctival hyperemia scores or degree of SPK, as assessed using the AD classification score, at any of the time points (P>0.5).
Applying a combination of anti-glaucoma eye drops can cause corneal damage19 due to the effects of preservatives and substrates.19,21–24 Switching from an eye drop with preservatives to one without preservatives among beta-blockers has been shown to result in a statistically significant improvement for break-up time and Schirmer’s test, as well as patient feelings of dryness and foreign body sensations.23
In the current study, the patients cornea had already been exposed to the preservatives of dorzolamide 1%/timolol 0.5%, and the eye drops were switched to brinzolamide 1%/timolol 0.5% without a washout period, which is supposedly why no critical increases in the incidence of SPK were observed. However, Rossi et al reported that the number of patients without positive findings on corneal fluorescein staining increased significantly after substituting brinzolamide 1%/timolol 0.5% for dorzolamide 2%/timolol 0.5% for 6 months.12
A possible explanation for this finding may be the formulation of brinzolamide/timolol, which (being isotonic and having a pH of approximately 7.2) may be less irritating than dorzolamide 2%/timolol 0.5%, which has a pH of 5.6. However, this observation requires assessment in further specific studies.12 Firat et al reported the short-term effects of a fixed combination of brinzolamide 1%/timolol 0.5% on the ocular surface in patients with newly diagnosed glaucoma; namely, the Schirmer test, break-up time test, and central corneal thickness values decreased, although the only statistically significant decrease was seen on the break-up time test (P=0.03).20
Arita et al demonstrated that anti-glaucoma medication causes meibomian gland dysfunction.27 These authors reported that eyes treated with anti-glaucoma therapy exhibited significantly greater lid margin abnormalities, with more cases of SPK, a shorter tear break-up time, and reduced Schirmer values than observed in the control eyes.27
Adverse effects
Our results are consistent with findings showing that dorzolamide 1%/timolol 0.5% causes more stinging/burning and less frequent blurred vision compared with brinzolamide 1%/timolol 0.5%. It is well known that dorzolamide causes stinging/burning due to its lower pH of 5.6 and use of sodium citrate as a buffer.14 Brinzolamide 1%/timolol 0.5% also causes stinging/burning; however, the frequency of this complication is lower and shorter in duration compared with that observed with dorzolamide 2%/timolol 0.5%.14 Another group reported that brinzolamide 1%/timolol 0.5% causes significantly less ocular irritation (2.7% versus 10.6%; P<0.0001) than dorzolamide 2%/timolol 0.5%.11 On the other hand, it is also well known that brinzolamide induces blurred vision due to the use of a white suspension.14,26
In contrast, one report showed no statistically significant difference in “blurred vision” between brinzolamide 1%/timolol 0.5% and dorzolamide 2%/timolol 0.5%.11 This may be due to the fact that the absorption time is relatively short for brinzolamide. This observation is supported by Hiraoka et al who demonstrated that administering brinzolamide or timolol gel formation significantly decreases contrast sensitivity for at least 5 minutes after instillation, although this parameter recovers to the normal level within 10 minutes.26 Therefore, the personal experience differs for each patient, and treated patients may become accustomed to the therapy with long-term use.11
Meanwhile, the Ocular Surface Disease Index29 may be useful to determine the degree of ocular discomfort, not only with dry eye disease but also in vision-related functioning with glaucoma. Nebbioso et al compared Ocular Surface Disease Index scores between a group receiving brinzolamide 1%/timolol 0.5% and a group receiving dorzolamide 2%/timolol 0.5%.21 They reported that the score was significantly lower in the brinzolamide 1%/timolol 0.5% group, and showed that brinzolamide 1%/timolol 0.5% was better tolerated than dorzolamide 2%/timolol 0.5%.21
Patient preference
The application of a fixed combination is generally preferable and actually provides better adherence. The present study indicated that patient preference rates were similar, ie, 29% preferred brinzolamide 1%/timolol 0.5%, 31% preferred dorzolamide 2%/timolol 0.5%, and 40% had no preference. We believe the finding that 40% patients could tolerate both is informative for clinicians in selecting eye drops. Conversely, Saneau et al reported more one-sided preference rates for brinzolamide 1%/timolol 0.5% versus dorzolamide 2%/timolol 0.5% (67.0% versus 30.4%, respectively),17 which was confirmed in a study by Lanzl and Raber, who similarly reported skewed preference rates (82.0% versus 8.8%, respectively).13 The deviation in the preference rates between the two fixed combination therapies may be the result of the alternative question.
The limitations of this study include the small number of patients, its open-label nature, and the lack of a crossover design. Further large-scale, randomized trials are thus needed. The second limitation is that, in the statistical analysis, we excluded two patients who dropped out at 4 weeks due to side effects of brinzolamide 1%/timolol 0.5%. Therefore, the patient preferences may not be exactly correct.
Conclusion
The present study showed that switching from dorzolamide 1%/timolol 0.5% to brinzolamide 1%/timolol 0.5% results in maintained IOP, with no significant differences in treatment-related adverse events at any of the assessed time points. Further, the results for patient preference were similar, with 40% of patients tolerating both eye drops, whereas differences were observed in the rates of ocular sensation of stinging/burning with dorzolamide 1%/timolol 0.5% and blurred vision with brinzolamide 1%/timolol 0.5%.
Disclosure
The article has not been presented at any meetings. The authors did not receive any financial support from any public or private sources. The authors have no financial or proprietary interests in any products, methods, or materials described herein, and report no conflicts of interest in this work.
References
Djafari F, Lesk MR, Harasymowycz PJ, Desjardins D, Lachaine J. Determinants of adherence to glaucoma medical therapy in a long-term patient population. J Glaucoma. 2009;18(3):238–243. | ||
Kawai-Tsuboi N, Kawai M, Minami Y, Yoshida A. A study of the association between patterns of eye drop prescription and medication usage in glaucoma subjects. J Glaucoma. June 25, 2013. [Epub ahead of print]. | ||
Strohmaier K, Snyder E, DuBiner H, Adamsons I. The efficiency and safety of the dorzalamide-timolol combination versus the concomitant administration of its components. Dorzolamide-Timolol Study Group. Ophthalmology. 1998;105(10):1936–1944. | ||
Higginbotham EJ, Feldman R, Stiles M, Dubiner H; Fixed Combination Investigative Group. Latanoprost and timolol combination therapy vs monotherapy: one-year randomized trial. Arch Ophthalmol. 2002;120(7):915–922. | ||
Barnebey HS, Orengo-Nania S, Flowers BE, et al. Safety and efficiency of travoprost 0.004%/timolol 0.5% fixed combination ophthalmic solution. Am J Ophthalmol. 2005;140(1):1–7. | ||
Diestelhorst M, Larsson LI. A 12-week, randomized double-masked, multicenter study of the fixed combination of latanoprost and timolol in the evening versus the individual components. Ophthalmology. 2006;113(1):70–76. | ||
Hommer A; Ganfort Investigators Group I. A double-masked, randomized, parallel comparison of a fixed combination of bimatoprost 0.03%/timolol 0.5% with non-fixed combination use in patients with glaucoma or ocular hypertension. Eur J Ophthalmol. 2007;17(1):53–62. | ||
Nagayama M, Nakajima T, Ono J. Safety and efficacy of a fixed versus unfixed brinzolamide/timolol combination in Japanese patients with open-angle glaucoma or ocular hypertension. Clin Ophthalmol. 2014;13(8):219–228. | ||
Kitazawa Y, Azuma I, Iwata K, et al. Dorzolamide, a topical carbonic anhydrase inhibitor: a two-week dose-response study in patients with glaucoma or ocular hypertension. J Glaucoma. 1994;3(4):275–279. | ||
Mundorf TK, Rauchman SH, Williams RD, Notivol R; Brinzolamide/Timolol Preference Study Group. A patient preference comparison of Azarga (brinzolamide/timolol fixed combination) vs Cosopt (dorzolamide/timolol fixed combination) in patients with open-angle glaucoma or ocular hypertension. Clin Ophthalmol. 2008;2(3):623–628. | ||
Manni G, Denis P, Chew P, et al. The safety and efficacy of brinzolamide 1%/timolol 0.5% fixed combination versus dorzolamide 2%/timolol 0.5% in patients with open-angle glaucoma or ocular hypertension. J Glaucoma. 2009;18(4):293–230. | ||
Rossi GC, Pasinetti GM, Sandolo F, Bordin M, Bianchi PE. From dorzolamide 2%/timolol 0.5% to brinzolamide 1%/timolol 0.5% fixed combination: a 6-month, multicenter, open-label tolerability switch study. Expert Opin Pharmacother. 2011;12(16):2425–2431. | ||
Lanzl I, Raber T. Efficacy and tolerability of the fixed combination of brinzolamide 1% and timolol 0.5% in daily practice. Clin Ophthalmol. 2011;5:291–298. | ||
Auger GA, Raynor M, Longstaff S. Patient perspectives when switching from Cosopt® (dorzolamide-timolol) to Azarga™ (brinzolamide-timolol) for glaucoma requiring multiple drug therapy. Clin Ophthalmol. 2012;6:2059–2062. | ||
Cheng JW, Cheng SW, Gao LD, Lu GC, Wei RL. Intraocular pressure-lowering effects of commonly used fixed-combination drugs with timolol: a systematic review and meta-analysis. PLoS One. 2012;7(9):e45079. | ||
Sezgin Akçay BÝ, Güney E, Bozkurt KT, Unlü C, Akçali G. The safety and efficacy of brinzolamide 1%/timolol 0.5% fixed combination versus dorzolamide 2%/timolol 0.5% in patients with open-angle glaucoma or ocular hypertension. J Ocul Pharmacol Ther. 2013;29(10):882–886. | ||
Sanseau A, Sampaolesi J, Suzuki ER Jr, Lopes JF, Borel H. Preference for a fixed combination of brinzolamide/timolol versus dorzolamide/timolol among patients with open-angle glaucoma or ocular hypertension. Clin Ophthalmol. 2013;7:357–362. | ||
Miyata K, Amano S, Sawa M, et al. A novel grading method for superficial punctate keratopathy magnitude and its correlation with corneal epithelial permeability. Arch Ophthalmol. 2003;121(11):1537–1539. | ||
Nagai N, Murao T, Oe K, Ito Y, Okamoto N, Shimomura Y. [An in vitro evaluation for corneal damages by anti-glaucoma combination eye drops using human corneal epithelial cell (HCE-T)]. Yakugaku Zasshi. 2011;131(6):985–991. Japanese. | ||
Firat PG, Samdanci E, Doganary S, Cavdar M, Sahin N, Gunduz A. Short-term effect of topical brinzolamide-timolol fixed combination on ocular surface of glaucoma patients. Int J Ophthalmol. 2012;5(6):714–718. | ||
Nebbioso M, Evangelista M, Librando A, Di Blasio D, Pescosolido N. Fixed topical combinations in glaucomatous patients and ocular discomfort. Expert Opin Pharmacother. 2012;13(13):1829–1835. | ||
Ayaki M, Yaguchi S, Iwasawa A, Koide R. Cytotoxicity of ophthalmic solutions with and without preservatives to human corneal endothelial cells, epithelial cells and conjunctival epithelial cells. Clin Experiment Ophthalmol. 2008;36(6):553–559. | ||
Iester M, Telani S, Frezzotti P, et al; Beta-Blocker Study Group. Ocular surface changes in glaucomatous patients treated with and without preservatives beta-blockers. J Ocul Pharmacol Ther. 2014;30(6):476–481. | ||
Barabino S, Antonelli S, Cimbolini N, Mauro V, Bauzin M. The effect of preservatives and anti-glaucoma treatments on the ocular surface of mice with dry eye. Invest Ophthalmol Vis Sci. 2014;55(10):6499–6504. | ||
Pozarowska D, Pozarowski P, Darzynkiewicz Z. Cytometric assessment of cytostatic and cytotoxic effects of topical glaucoma medications on human epithelial corneal line cells. Cytometry B Clin Cytom. 2010;78(2):130–137. | ||
Hiraoka T, Daito M, Okamoto F, Kiuchi T, Oshika T. Contrast sensitivity and optical quality of the eye after instillation of timolol maleate gel-forming solution and brinzolamide ophthalmic suspension. Ophthalmology. 2010;117(11):2080–2087. | ||
Arita R, Itoh K, Maeda S, et al. Effects of long-term topical anti-glaucoma medications on meibomian glands. Graefes Arch Clin Exp Ophthalmol. 2012;250(8):181–185. | ||
Agnifili L, Fasanella V, Costagliola C, et al. In vivo confocal microscopy of meibomian glands in glaucoma. Br J Ophthalmol. 2013;97(3):343–349. | ||
Schiffman RM, Christianson MD, Jacobsen G, Hirsch JD, Reis BL. Reliability and validity of the Ocular Surface Disease Index. Arch Ophthalmol. 2000;118(5):615–621. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.