Back to Journals » Clinical Ophthalmology » Volume 13
Effectiveness of prophylactic intravitreal bevacizumab injection to proliferative diabetic retinopathy patients with elevated preoperative intraocular VEGF in preventing complications after vitrectomy
Authors Tsubota K , Usui Y , Wakabayashi Y, Suzuki J, Ueda S, Goto H
Received 2 February 2019
Accepted for publication 12 June 2019
Published 28 June 2019 Volume 2019:13 Pages 1063—1070
DOI https://doi.org/10.2147/OPTH.S203921
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Kinya Tsubota, Yoshihiko Usui, Yoshihiro Wakabayashi, Jun Suzuki, Shunichiro Ueda, Hiroshi Goto
Department of Ophthalmology, Tokyo Medical University Hospital, Tokyo, Japan
Purpose: This study aimed to elucidate the effects of intravitreal bevacizumab (IVB) injections for the prevention of post-vitrectomy complications in proliferative diabetic retinopathy (PDR) patients with elevated vitreous vascular endothelial growth factor (VEGF) concentration.
Design: Prospective case series.
Methods: Thirty-three patients (42 eyes) with PDR who underwent primary vitrectomy in the Department of Ophthalmology, Tokyo Medical University Hospital were studied. We measured VEGF concentrations in vitreous humor collected at the time of vitrectomy using ELISA. IVB injections were performed after vitrectomy in patients with vitreous VEGF levels exceeding 1,000 pg/mL. New bleeding occurring within 1 month of vitrectomy was defined as early vitreous hemorrhage (VH).
Main outcome measure: The incidence of complications after vitrectomy including postoperative early VH.
Results: IVB injections were administered to 11 eyes (26%) with vitreous VEGF concentrations exceeding 1,000 pg/mL. None of the 11 eyes that received an IVB injection developed early VH. Among 31 eyes (74%) with vitreous VEGF concentrations lower than 1,000 pg/mL, two eyes (6%) developed early VH after vitrectomy.
Conclusions: Prophylactic IVB injections administered to patients with elevated preoperative intraocular VEGF concentrations were effective in preventing post-vitrectomy early VH.
Keywords: diabetic retinopathy, VEGF, vitreous hemorrhage, intravitreal bevacizumab injection
Introduction
Diabetic retinopathy (DR) is the most common diabetic eye disease and the leading cause of vision loss in diabetes patients.1 With the increasing prevalence of diabetes globally, the number of patients with diabetic retinopathy is rising in many countries. International Diabetes Federation indicated that an estimated 415 million people had diabetes worldwide in 2015, diabetes patients will be increased to 642 million.2 Around 25–50% of diabetic patients will have DR in life, and 10% of patients with DR will have severe visual disorder while 2% will be blind after 15 years from DR onset.1–6 Vitreous hemorrhage (VH) occurs in some DR patients. Pars plana vitrectomy is the standard and effective surgical treatment for VH and traction retinal detachment secondary to proliferative diabetic retinopathy (PDR).7 Although vitrectomy for PDR achieves good anatomical success, the procedure is sometimes associated with postoperative complications such as recurrent VH, neovascular glaucoma (NVG) and traction retinal detachment. VH is one of the most common adverse events after vitrectomy for PDR. Postoperative VH may occur even after anatomically successful vitrectomy for PDR, with rates ranging between 17% and 63%.8–11
Vascular endothelial growth factor (VEGF) is a potent angiogenic factor expressed at high levels in the retina of diabetic patients, resulting in a marked increase in vitreous concentration.11–13 This growth factor promotes migration of leukocytes and adhesion of leukocytes to vascular endothelial cells and increases intraocular vascular permeability and angiogenesis, which may lead to onset and progression of retinopathy and rubeosis in diabetic patients.14–16 A previous study identified a high vitreous humor level of VEGF at the time of primary vitrectomy to be a significant risk factor for early postoperative VH and NVG in patients with PDR.11 Numerous reports have focused on the adjunctive use of bevacizumab, a full-length humanized monoclonal antibody that binds all VEGF isoforms, to reduce active neovascularization in PDR. Several recent clinical studies reported that intravitreal bevacizumab (IVB) injection before or during vitrectomy for PDR was useful to reduce the incidence of early postoperative VH.9,10 This clinical evidence may suggest that high VEGF level at the time of vitrectomy for PDR contributes to the development of postoperative VH.
In this study, we examined the effectiveness of IVB injection in patients undergoing vitrectomy for PDR, who have elevated preoperative vitreous VEGF concentrations, for the prevention of postoperative complications.
Subjects
This prospective case series complied with the tenets of the Declaration of Helsinki and was approved by the Review Ethics Committee of the Tokyo Medical University. Written informed consent was obtained from all participants before enrollment. This study was conducted with the approval of the Research Ethics Committee of the University of Tokyo and registered in the UMIN Registry in April 2013 (registration number: UMIN000009801; http://www.umin.ac.jp/ctr/index.htm,.registration date: April 2013). We enrolled patients with PDR who underwent primary pars plana vitrectomy for VH at the Department of Ophthalmology, Tokyo Medical University Hospital between April 2013 and March 2014. The following cases were excluded from analysis: 1) drop-out from follow-up within 4 weeks after primary vitrectomy, 2) intraoperative use of silicone oil, 3) deterioration in general condition precluding IVB injection, and 4) presence of retinal disease other than PDR, and (5) IVB injection not performed to avoid after trabeculectomy.
Materials and methods
Clinical data analysis
The primary outcome of this study was incidence of complications after vitrectomy for PDR, including early (<4 weeks) and late (>4 weeks) VH and NVG after vitrectomy. We analyzed the effect of selective IVB injection for the prevention of postoperative complications. Postoperative VH was evaluated according to the Diabetic Retinopathy Vitrectomy Study grading system and was defined as a new episode of VH of grade 1 or above occurring later than 3 days after primary surgery.11 Both early (<4 weeks) and late (>4 weeks) VH were recorded. VH detected on the first day after surgery was classified as early VH if the VH progressed to grade 2 or above at postoperative day 3. NVG was defined as stromal and chamber angle neovascularization, with intraoperative pressure (IOP) elevated to 25 mmHg or higher. In the case of gas-injected eye, complications were assessed in the region without the gas bubble.
Preoperative, intraoperative, and postoperative data were collected for each patient. Preoperative data at the time of primary surgery included age; sex; status of diabetes mellitus (HbA1C); other systemic diseases such as hypertension and dyslipidemia; renal function (serum creatinine); medications such as anticoagulant for systemic disease; and ophthalmic parameters including best-corrected visual acuity (BCVA), lens status, and IOP. Intraoperative data included phacoemulsification and aspiration (PEA) and intraocular lens (IOL) procedures; SF6 or air tamponade; previous pan-retinal photocoagulation (PRP); and the presence or absence of fibrovascular proliferation (FVP), neovascularization of optic disc (NVD), and traction retinal detachment. Postoperative data included BCVA at the final visit and number of episodes of complications.
Decimal visual acuity was measured using Landolt C visual acuity chart and converted to logarithm of minimal angle of resolution (logMAR) scale. Counting fingers and hand movement were assigned as visual acuity of 0.01 (2.0 logMAR units) and 0.001 (3.0 logMAR units), respectively. Visual improvement was defined as an increase of at least 0.3 logMAR units.
Surgical technique
We conducted standard pars plana vitrectomy using a 25-gauge three-port system and high-speed vitreous cutter (2,500 cycle/min, ACCURUS®; Alcon, Fort Worth, TX, USA) under local anesthesia in all patients. During vitrectomy, intravitreal triamcinolone acetonide was used to visualize the vitreous gel and vitreoretinal adhesions in all patients. We performed PEA simultaneously in patients with cataract and placed an acrylic foldable IOL in the capsular bag as needed. Fibrovascular membrane dissection, segmentation and delamination were performed mainly with vitreoretinal scissors or forceps in 25G vitrectomy. Endolaser was applied to complete PRP up to the ora serrate in all patients. Hemostasis was achieved by increasing intraocular pressure, coagulation with endodiathermy, or applying pressure with vitreous surgical instrument. We ensured no rebleeding from vascular membrane or fragile vessels by controlling intraocular pressure at 2–5 mmHg until the end of vitrectomy, as reported previously.11 Some patients who had traction retinal detachment were given 0.5–0.8 mL of 100% sulfur hexafluoride (SF6) tamponade at the end of surgery. Patients who were taking an anticoagulant for general systemic diseases discontinued anticoagulant 1 week before surgery and resumed within 1 week after surgery. All surgeries were performed by three surgeons who have a rich experience of vitrectomy (Yoshihiko Usui, MD, PhD; Yoshihiro Wakabayashi, MD, PhD; Jun Suzuki, MD, PhD).
Vitreous sample collection and VEGF measurement
A mid-vitreous sample was collected with a vitreous cutter at the start of vitrectomy before intraocular infusion. The VEGF concentration in the vitreous humor sample was assayed at a commercial laboratory (SRL Inc., Tokyo, Japan) using an ELISA. The lowest detectable concentration of this assay was 20 pg/mL. Concentrations below this level were recorded as 20 pg/mL for statistical analysis.11
IVB injection
We performed IVB injection only once after surgery in patients with preoperative vitreous VEGF concentration exceeding 1,000 pg/mL. Our group previously reported that a high vitreous humor level of VEGF (median 532.6 pg/mL) at the time of primary vitrectomy was a significant risk factor for early postoperative VH in patients with PDR.11 When we reanalyzed the data after excluding the outlier, the median vitreous VEGF concentration in the group with early VH group was approximately 1,000 pg/mL. Based on this finding, we selected a vitreous VEGF concentration of 1,000 pg/mL as the cut-off value for prophylactic IVB injection. Patients with preoperative vitreous VEGF concentration exceeding 1,000 pg/mL were treated with an intravitreal injection of 0.05 mL (1.25 mg) bevacizumab (Avastin, Roche, Basel, Switzerland) by three surgeons who have a rich experience of IVB (Yoshihiko Usui, MD, PhD; Yoshihiro Wakabayashi, MD, PhD; Jun Suzuki, MD, PhD). All IVB injections were conducted in our outpatients’ clinic; then, the patients were discharged, because it took several days for the result of vitreous VEGF concentration to be available. Any patients who did not have the injection of IVB before where asked to reveal the VEGF concentration in vitreous.
Statistical analysis
Vitreous concentrations of VEGF and clinical data are expressed as mean or median and range. We compared several clinical data and post-vitrectomy complication in PDR patients with and those without selective IVB injection. Chi-square test and Mann–Whitney test were used to compare VEGF concentrations and other clinical data between patients who received IVB injection and those who did not. A P<0.05 was considered statistically significant. All analyses were performed using commercial statistical analysis software (JMP version 5.01J; SAS Institute, Cary, NC, USA).
Results
Patient demographics and preoperative ocular and clinical findings are summarized in Table 1. A total of 42 eyes of 33 diabetic patients (21 males and 12 females) that underwent vitrectomy for PDR complications were studied. Their median age was 56 years (range, 37–82 years). The median hemoglobin (Hb) A1c level was 7.6% (range, 5.9–11.6%). Twelve patients (36%) had hypertension and 5 patients (15%) had dyslipidemia. The median serum creatinine was 0.78 mg/dL (range, 0.47–11.9 mg/dL). Eight patients (24%) were using oral anticoagulant. The median baseline BCVA was 1.59 logMAR units (range, 0.22–3.00) and median IOP was 14.7 mm Hg (range, 9–26 mm Hg) before vitrectomy. PRP had been performed before surgery in 26 eyes (62%). All eyes had VH, 16 eyes (38%) had traction retinal detachment, 4 eyes (10%) had neovascularization of the iris, 35 eyes (83%) had neovascularization of the disc, and 21 eyes (50%) had FVP.
 |
Table 1 Patient demographics and baseline ocular and clinical findings |
Surgical procedures and outcome are summarized in Table 2. Simultaneous PEA and IOL implantation were performed in 31 eyes (74%), and SF6 tamponade was conducted in 6 eyes (14%). Endolaser was added to complete PRP up to the ora serrate in all 42 eyes (100%, range, 651–3,000 shots). Final anatomic success rate was 100% (all 42 eyes). The median postoperative BCVA was 0.22 logMAR units (range, −1.57 to +2.00), which was improved significantly compared with baseline BCVA. Postoperative BCVA improved by 3 lines or more in 37 eyes (88%), was unchanged in 5 eyes (12%), and decreased in 0 eye (0%). The incidence of major postoperative complications was 17% (7 of 42 eyes). Recurrent VH occurred in 7 eyes (17%) during follow-up periods. Among them, early VH occurred in 2 eyes (5%) and late VH in 5 eyes (12%). The median durations of early and late VH onset after vitrectomy were 1 and 110 days (50–110 days), respectively. Postoperative NVG occurred in 0 eye (0%) and progressive retinal detachment in 0 eye (0%). Six eyes (14%) needed SF6 tamponade at the end of surgery. The median follow-up period after primary surgery was 14.7 months.
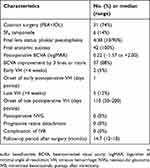 |
Table 2 Surgical procedures and outcome |
Vitreous VEGF concentrations and patients selected for IVB injection are summarized in Table 3. The mean VEGF level in the vitreous was 774.5 pg/mL (range, 20–3,560 pg/mL). The median time until VEGF result was available was 6.4 days (range, 3–14 days). Eleven eyes (26%) had vitreous VEGF levels exceeding 1,000 pg/mL and were given IVB injection after surgery. The median time from vitrectomy to IVB injection was 11 days (range, 7–17 days).
 |
Table 3 VEGF concentrations and patients selected for IVB injection |
Table 4 summarizes the frequencies of postoperative complications in patients who underwent IVB injection and those who did not. Eleven eyes with vitreous VEGF levels exceeding 1,000 pg/mL underwent IVB injection (IVB group) and 31 eyes with vitreous VEGF levels under 1,000 pg/mL did not undergo IVB injection (non-IVB group). The two groups were compared with respect to vitreous VEGF concentration and clinical data including postoperative complications. Vitreous VEGF level was significantly higher in the IVB group (median, 1,761 pg/mL) than in the non-IVB group (median, 412 pg/mL) (P=0.01) (Table 4). The prevalence of hypertension (64% vs 26%, P=0.02) was significantly higher in the IVB group. However, the two groups did not differ in intraoperative findings including previous PRP (55% vs 65%, P=0.56), neovascularization of the iris (9% vs 10%, P=0.95), traction retinal detachment (36% vs 39%, P=0.76), FVP (36% vs 55%, P=0.29) and neovascularization of the disc (72% vs 94%, P=0.12). These findings suggest that previous PRP did not affect vitreous VEGF concentration and that there were no differences in clinical severity between the two groups. Regarding postoperative complications, which were the primary outcome of this study, none of the 11 eyes in the IVB group developed early VH (<4 weeks) while 2 of 31 eyes in the non-IVB group developed early VH, although there was no significant difference between two groups (0% vs 6%, P=0.99). The two groups also did not differ in the incidence of late postoperative VH (>4 weeks) (18% vs 10%, P=0.66). No postoperative NVG was observed in both groups during the follow-up periods (0% vs 0%). For visual outcome, BCVA improvement by 3 lines or more in 10 patients in the IVB group and 27 patients in the non-IVB group, with no difference between two groups (91% vs 87%, P=0.91).
 |
Table 4 IVB and clinical data with or without IVB in PDR |
Discussion
Vitrectomy is an effective surgical treatment for VH in PDR patients.6 The number of PDR patients is expected to increase,1–6 and the demand of vitrectomy for DR cases with VH will also increase. However, postoperative VH, one of the complications after vitrectomy, occurs in some patients, and may delay recovery of visual activity. Ahn et al9 reported that the incidence of early postoperative VH was reduced to 22% with prophylactic IVB injection given before vitrectomy. Other clinical studies reported reduction in incidence of early postoperative VH to 11–12% with prophylactic IVB injection given at the end of vitrectomy.9,10 These reports indicate the effectiveness of VEGF antagonists for the prevention of postoperative VH. Especially in patients with severe PDR and high VEGF level, even after adequate vitrectomy, elevated VEGF concentration as a result of inflammation after surgery may induce early postoperative VH and NVG. Prophylactic IVB injection may reduce postoperative complications through inhibiting the increase of VEGF after surgery. The demand of IVB injection will increase accompanying the increase in number of patients with DR. However, the use of VEGF antagonists has to be rationalized, because of medical financial stress17,18 and complications of IVB injection such as cerebral infarction and myocardial infarction.19 Anti-VEGF agents such as pegaptanib, bevacizumab, and ranibizumab are costly; a single dose costs >1,000 dollars in USA and >150,000 yens in Japan. Therefore, increasing injection of VEGF antagonists will become a major liability on medical finance.17,18 Moreover, several recent reports indicate that VEGF antagonists may augment TGF-β expression and increase the numbers of patients with retinal detachment and endophthalmitis after IVB injection.20–28 In addition, cerebral infarction and myocardial infarction are well known to be major complications of IVB injection.19 In the present study, no complications of IVB injection were observed (Table 2) and the incidence of early post-vitrectomy VH was reduced to 5% with prophylactic IVB injection given to patients with high vitreous VEGF levels. This result shows that the beneficial effect of selective prophylactic IVB injection to prevent early VH after vitrectomy is not inferior compared with giving IVB injection to all patients before vitrectomy9 or at the end of vitrectomy.9,10 In other words, the present result indicates that selective IVB injection may reduce the number of IVB injections and avoid the risk of IVB injection-related complications.
In the clinical setting, however, patients with high vitreous VEGF concentrations who do not develop postoperative VH, and, on the contrary, patients with low vitreous VEGF concentrations who develop postoperative VH are encountered. Some reports attribute these phenomena to be associated with the type of VEGF and individual differences in VEGF concentration.29–32 This information suggests that the VEGF types and individual differences in VEGF concentration have to be investigated to select better candidates for anti-VEGF injection. In this study, the average VEGF concentration in patients with early VH was 940 pg/mL (884–996). Therefore, the criterion of IVB injection should be set at vitreous VEGF concentration lower than 1,000 pg/mL. Furthermore, IVB injections were performed around 11 days after vitrectomy, because it took approximately 7 days to measure the vitreous VEGF concentration in this study. There was a lag of a median of 11 days between vitrectomy and IVB due to the time taken to assay vitreous VEGF concentrations. Any changes that might have occurred during this period could not be accounted for. Further investigations on the timing of vitreous VEGF measurement and IVB injection for the prevention of postoperative VH are needed.
Finally, although further studies are required to verify the present result, selective IVB injection may be an effective approach to prevent the development of early post-vitrectomy VH in high-risk patients with high vitreous VEGF concentrations. For patients undergoing vitrectomy for PDR, selective IVB injection for high-risk patients may have advantages over IVB injection for all patients with respect to medical finance by reducing the number of anti-VEGF injections as well as safety by avoiding the risk of IVB injection-related complications.
Data availability
The clinical data used to support the findings of this study are restricted by the Review Ethics Committee of the Tokyo Medical University in order to protect patient privacy. Data are available from Kinya Tsubota, who is the corresponding author of this manuscript, for researchers who meet the criteria for access to confidential data.
Acknowledgments
An earlier version of this study has been presented as an abstract meeting in 2013 ARVO Annual Meeting Abstract (https://iovs.arvojournals.org/article.aspx?articleid=2146636). This work was not supported by any funding source.
Author contributions
YW conceived and designed the experiments; YU, YW, and JS performed operations and collected the sample data; KT contributed to the writing of the manuscript and tables; YU reviewed the manuscript. All authors contributed to data analysis, drafting and revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Fong DS, Aiello LP, Ferris FL
2. International Diabetes Federation. IDF Diabetes Atlas,
3. Cheung N, Mitchell P, Wong TY. Diabetic retinopathy. Lancet. 2010;376(9735):124–136. doi:10.1016/S0140-6736(09)62124-3
4. Xie XW, Xu L, Wang YX, Jonas JB. Prevalence and associated factors of diabetic retinopathy. The Beijing Eye Study 2006. Graefes Arch Clin Exp Ophthalmol. 2008;246(11):1519–1526. doi:10.1007/s00417-008-0884-6
5. Shaw JE, Sicree RA, Zimmet PZ. Global estimates of the prevalence of diabetes for 2010 and 2030. Diabetes Res Clin Pract. 2010;87(1):4–14. doi:10.1016/j.diabres.2009.10.007
6. Lee R, Wong TY, Sabanayagam C. Epidemiology of diabetic retinopathy, diabetic macular edema and related vision loss. Eye Vis (Lond). 2015;30(2):17. doi:10.1186/s40662-015-0026-2
7. Yorston D, Wickham L, Benson S, Bunce C, Sheard R, Charteris D. Predictive clinical features and outcomes of vitrectomy for proliferative diabetic retinopathy. Br J Ophthalmol. 2008;92(3):365–368. doi:10.1136/bjo.2007.124495
8. Yang CM, Yeh PT, Yang CH. Intravitreal long-acting gas in the prevention of early postoperative vitreous hemorrhage in diabetic vitrectomy. Ophthalmology. 2007;114(4):710–715. doi:10.1016/j.ophtha.2006.10.027
9. Ahn J, Woo SJ, Chung H, Park KH. The effect of adjunctive intravitreal bevacizumab for preventing postvitrectomy hemorrhage in proliferative diabetic retinopathy. Ophthalmology. 2011;118(11):2218–2226. doi:10.1016/j.ophtha.2011.03.036
10. Park DH, Shin JP, Kim SY. Intravitreal injection of bevacizumab and triamcinolone acetonide at the end of vitrectomy for diabetic vitreous hemorrhage: a comparative study. Graefes Arch Clin Exp Ophthalmol. 2010;248(5):641–650. doi:10.1007/s00417-009-1275-3
11. Wakabayashi Y, Usui Y, Okunuki Y, et al. Intraocular VEGF level as a risk factor for postoperative complications after vitrectomy for proliferative diabetic retinopathy. Invest Ophthalmol Vis Sci. 2012;53(10):6403–6410. doi:10.1167/iovs.12-10367
12. Adamis AP, Miller JW, Bernal MT, et al. Increased vascular endothelial growth factor levels in the vitreous of eyes with proliferative diabetic retinopathy. Am J Ophthalmol. 1994;118(4):445–450.
13. Aiello LP, Avery RL, Arrigg PG, et al. Vascular endothelial growth factor in ocular fluid of patients with diabetic retinopathy and other retinal disorders. N Engl J Med. 1994;331(22):1480–1487. doi:10.1056/NEJM199407073310103
14. Nakao S, Arima M, Ishikawa K, et al. Intravitreal Anti-VEGF therapy blocks inflammatory cell infiltration and re-entry into the circulation in retinal angiogenesis. Inves Ophthalmol Vis Sci. 2012;53(7):4323–4328. doi:10.1167/iovs.11-9119
15. Funatsu H, Yamashita H, Sakata K, et al. Vitreous levels of vascular endothelial growth factor and intercellular adhesion molecule 1 are related to diabetic macular edema. Ophthalmology. 2005;112(5):806–816. doi:10.1016/j.ophtha.2004.11.045
16. Yoshida S, Yoshida A, Ishibashi T, Elner SG, Elner VM. Role of MCP-1 and MIP-1 alpha in retinal neovascularization during postischemic inflammation in a mouse model of retinal neovascularization. J Leukoc Biol. 2003;73(1):137–144. doi:10.1189/jlb.0302117
17. Ip MS, Domalpally A, Hopkins JJ, Wong P, Ehrlich JS. Long-term effects of ranibizumab on diabetic retinopathy severity and progression. Arch Ophthalmol. 2012;130(9):1145–1152. doi:10.1001/archophthalmol.2012.1043
18. Saeed MU, Gkaragkani E, Ali K. Emerging roles for antiangiogenesis factors in management of ocular disease. Clin Ophthalmol. 2013;6:533–543. doi:10.2147/OPTH.S31016
19. Schlenker MB, Thiruchelvam D, Redelmeier DA. Intravitreal anti-vascular endothelial growth factor treatment and the risk of thromboembolism. Am J Ophthalmol. 2015;160(3):569–580. doi:10.1016/j.ajo.2015.06.011
20. Dinc E, Yildirim O, Ayaz L, Ozcan T, Yilmaz SN. Effects of intravitreal injection of bevacizumab on nitric oxide levels. Eye. 2015;29(3):436–442. doi:10.1038/eye.2014.297
21. Oshima Y, Shima C, Wakabayashi T, et al. Microincision vitrectomy surgery and intravitreal bevacizumab as a surgical adjunct to treat diabetic traction retinal detachment. Ophthalmology. 2009;116(5):927–938. doi:10.1016/j.ophtha.2008.11.005
22. Forooghian F, Kertes PJ, Eng KT, Agrón E, Chew EY. Alterations in the intraocular cytokine milieu after intravitreal bevacizumab. Invest Ophthalmol Vis Sci. 2010;51(5):2388–2392. doi:10.1167/iovs.09-4065
23. Shah CP, Hsu J, Garg SJ, Fischer DH, Kaiser R. Retinal pigment epithelial tear after intravitreal bevacizumab injection. Am J Ophthalmol. 2006;142(6):1070–1072. doi:10.1016/j.ajo.2006.07.037
24. Guber J, Praveen A, Saeed MU. Higher incidence of retinal pigment epithelium tears after ranibizumab in neovascular age-related macular degeneration with increasing pigment epithelium detachment height. Br J Ophthalmol. 2013;97(11):1486–1487. doi:10.1136/bjophthalmol-2013-303978
25. Chang LK, Flaxel CJ, Lauer AK, Sarraf D. RPE tears after pegaptanib treatment in age-related macular degeneration. Retina. 2007;27(7):857–863. doi:10.1097/IAE.0b013e3180342c42
26. Doguizi S, Ozdek S. Pigment epithelial tears associated with anti-VEGF therapy: incidence, long-term visual outcome, and relationship with pigment epithelial detachment in age-related macular degeneration. Retina. 2014;34(6):1156–1162. doi:10.1097/IAE.0000000000000056
27. Oshima Y, Apte RS, Nakao S, Yoshida S, Ishibashi T. Full thickness macular hole case after intravitreal aflibercept treatment. BMC Ophthalmol. 2015;15:30. doi:10.1186/s12886-015-0021-3
28. Sato T, Emi K, Ikeda T, et al. Severe intraocular inflammation after intravitreal injection of bevacizumab. Ophthalmology. 2010;117(3):512–516. doi:10.1016/j.ophtha.2009.07.041
29. Perrin RM, Konopatskaya O, Qiu Y, Harper S, Bates DO, Churchill AJ. Diabetic retinopathy is associated with a switch in splicing from anti- to pro-angiogenic isoforms of vascular endothelial growth factor. Diabetologia. 2005;48(11):2422–2427. doi:10.1007/s00125-005-1951-8
30. Konopatskaya O, Churchill AJ, Harper SJ, Bates DO, Gardiner TA. VEGF165b, an endogenous C-terminal splice variant of VEGF, inhibits retinal neovascularization in mice. Mol Vis. 2006;26(12):626–632.
31. Barańska P, Jerczyńska H, Pawłowska Z. Vascular endothelial growth factor structure and functions. Postepy Biochem. 2005;51(1):12–21.
32. Cooper M, Vranes D, Youssef S, et al. Increased renal expression of vascular endothelial growth factor (VEGF) and its receptor VEGFR-2 in experimental diabetes. Diabetes. 1999;48(11):2229–2239. doi:10.2337/diabetes.48.11.2229
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
