Back to Journals » Clinical and Experimental Gastroenterology » Volume 13
Effect of Ginger Powder Supplementation in Patients with Non-Alcoholic Fatty Liver Disease: A Randomized Clinical Trial
Authors Rafie R , Hosseini SA, Hajiani E, Saki Malehi A , Mard SA
Received 15 October 2019
Accepted for publication 30 December 2019
Published 23 January 2020 Volume 2020:13 Pages 35—45
DOI https://doi.org/10.2147/CEG.S234698
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Everson L.A. Artifon
Roya Rafie,1 Seyed Ahmad Hosseini,1 Eskandar Hajiani,2 Amal Saki Malehi,3 Seyed Ali Mard4
1Nutrition and Metabolic Diseases Research Center, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran; 2Research Center for Infectious Diseases of the Digestive System, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran; 3Health Research Institute, Thalassemia and Hemoglobinopathy Research Center, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran; 4Alimentary Tract Research Center, Imam Khomeini Hospital Clinical Research Development Unit, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran
Correspondence: Seyed Ahmad Hosseini
Nutrition and Metabolic Diseases Research Center, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran
Tel +98-6133367543
Fax +98-6133720299
Email [email protected]
Background: Non-alcoholic fatty liver disease (NAFLD) is one of the most common chronic liver disorders. The main causes of NAFLD are associated with insulin resistance, severe lipid metabolism disorders, oxidative stress and inflammation. Previous studies have reported that ginger has positive metabolic results.
Aim: The aim of this study was to determine the effect of ginger powder supplement on lipid profiles, insulin resistance, liver enzymes, inflammatory cytokines and antioxidant status in patients with NAFLD.
Methods: In this randomized clinical trial, 46 people with NAFLD were parted into two groups and subjected to the ginger or placebo capsules (3 capsules daily, each containing 500 mg of ginger or wheat flour) over 12 weeks. All patients received a diet with balanced energy and physical activity during the intervention period. Liver ultrasonography, anthropometric indices and biochemical parameters were measured before and after intervention.
Results: No significant difference was found between the two groups in the baseline variables at the beginning of the study. At the end of the study, serum levels of alanine aminotransferase (ALT), total cholesterol, low-density lipoprotein (LDL-C), fasting blood glucose, and insulin resistance index (HOMA), C-reactive protein (hs-CRP), and fetuin-A in the group receiving a ginger supplement significantly decreased compared to placebo. However, there was no significant difference between the two groups in body weight, fasting insulin, HDL-C, triglyceride, adiponectin, alpha-tumor necrosis factor (TNF-α), total antioxidant capacity (TAC), gamma-glutamyl transferase (GGT), aspartate aminotransferase (AST), fatty liver index (FLI), fatty liver grade and blood pressure.
Conclusion: The ginger supplement may be used as a complementary therapy along with existing therapies to reduce insulin resistance, liver enzymes and inflammation in patients with non-alcoholic fatty liver.
Keywords: ginger, liver enzymes, non-alcoholic fatty liver disease, lipid profiles, insulin resistance
Background
Non-alcoholic fatty liver disease (NAFLD) was identified by Ludwig and colleagues in people who did not have any alcohol history for the first time in 1980. The accumulation of more than 5% triglyceride in the hepatic parenchyma without notable alcohol consumption is considered to be NAFLD.1 NAFLD includes a wide range of liver disorders from hepatocellular steatosis to more severe non-alcoholic steatohepatitis (NASH), which may progress to hepatic fibrosis and cirrhosis.2 About 20–30% of adults in developed countries have an increased fat accumulation in the liver.3 In Iran, the prevalence of this disease is estimated to be 21.5%.4
The cause of NAFLD is not fully understood. The accepted hypothesis to explain the pathogenicity of the NAFLD is the two-hit hypothesis, which was first expressed by James and Day. This hypothesis states that insulin resistance, as the first hit, leads to stimulation of the synthesis of liver fatty acids and accumulation of fat in the liver and steatosis, and the liver is more susceptible to the second hit, which is oxidative stress from different sources.5 There is currently no definitive treatment for NAFLD, so there is an urgent need to find new therapeutic approaches to replace or help with existing fatty liver treatments. Previous studies have indicated that diets containing antioxidants and anti-inflammatory agents in some medicinal plants may be beneficial in the treating of NAFLD.6–8 Ginger (Zingiber Officinale Roscoe) is widely used worldwide as a spice. In the past, this plant has been used to treat neurological diseases, diabetes, rheumatism, gingivitis, toothache, asthma, constipation, maldigestion and nausea and vomiting. Food and Drug Administration has approved ginger as a food supplement. Ginger has various pharmacological effects, such as immune regulator, tumor formation inhibiting, anti-inflammatory, anti-apoptotic and antiemetic effects. Most of these medicinal effects of ginger are attributed to the presence of various compounds such as gingerol and shogaols. More than 40 antioxidant compounds have been identified in ginger.9
Because insulin resistance, severe disorders of lipid metabolism, oxidative stress and inflammation play a role in the pathogenesis of NAFLD; therefore, each potential therapeutic agent should target one or some of these pathological events. Due to the fundamental role of insulin resistance and as a result of hyperinsulinemia in fat accumulation in the liver, an increase in insulin sensitivity is considered as a treatment approach against NAFLD. A previous study reported that the insulin sensitivity to adipocytes could be improved using ginger.10 On the other hand ginger with effect on the liver reduces biosynthesis of cholesterol and possibly increases the transformation of cholesterol to bile acids.11
As the conversion of simple steatosis to more advanced forms of NAFLD is also affected by oxidative stress and inflammation.12 Therefore, reducing inflammatory and oxidative stress factors in NAFLD patients may play an important role in slowing the rate of NAFLD progression and reducing the risk of cardiovascular disorders due to this disease. Studies have indicated that ginger can inhibit arachidonic acid metabolism with the suppression of cyclooxygenase and lipooxygenase enzymes, thereby acting as an anti-inflammatory agent.13 Ginger has potential antioxidant activities due to its polyphenolic compounds as gingerol and curcumin. Including these antioxidant activities of radical trapping and inhibition lipid peroxidation. Ginger also prevents liver toxicity by increasing its antioxidant activity.14 A number of studies have reported that adiponectin and TNF-α play a key role in the hepatic steatosis progression to NASH. Adiponectin protects against NAFLD by increasing insulin sensitivity and decreasing fat hepatic accumulation, while TNF- α stimulates hepatic steatosis. Thus, reduction the expression of TNF-α can be an important mechanism to improve liver damage. Previous studies have shown that ginger bioactive compounds can downregulate TNF-α. Also, ginger significantly inhibited the reduction of adiponectin expression by TNF-α.15
Fetuin–A (a2-Heremans–Schmid glycoprotein), is a serum protein that is mainly synthesized in the liver. A number of studies have shown that serum level of fetuin-A is considerably enhanced in people with NAFLD. Thus, the reduction of serum levels of fetuin-A can be associated with improvement in NAFLD.16
The use of ginger in the treatment of cardiovascular diseases has long been posed. The diuretics and lowering blood pressure effect of ginger have been shown in a number of studies.17 Ghayur et al showed that the intake of the raw extract of ginger reduces the arterial blood pressure by blocking the voltage-dependent calcium channels in rats.18
The present study was designed to investigate the effects of ginger on the lipid and sugar profiles, inflammatory conditions, oxidative stress and reduction of the cardiovascular complications of NAFLD.
Methods
Materials
Ginger powder supplement used in this study is a ready-made product. The contents of each ginger capsule were 500 mg of ginger rhizome powder. Placebo containing wheat flour was prepared in the same form and color as a ginger supplement in Pharmacy Faculty Lab of Ahvaz Jundishapur University of Medical Sciences.
Study Population
100 women and men aged 20–70 years old, whose fatty liver was diagnosed by elevated levels of alanine aminotransferase enzyme and ultrasonography, were enrolled in the study from among the patients referring to the gastroenterology clinic of Imam Khomeini Hospital in Ahvaz.
Inclusion Criteria
Having a willingness to participate, aged 20 to 70 years, diagnosis of NAFLD based on high levels of liver enzymes (more than 30 U/L in men and more than19 U/L in women), ultrasound and 24.9˂BMI˂35.19
Exclusion Criteria
1) Patients who are reluctant to continue the study; 2) patients who used alcohol more than 20 g per day; 3) patients taking drugs with a liver toxicity such as calcium channel blockers, methotrexate, nonsteroidal anti-inflammatory drugs, NSAIDs, estrogens, progesterones, immunosuppressed, diuretics and corticosteroids; 4) patients with liver disorder other than NAFLD (like hereditary hemochromatosis, Wilson’s disease, cirrhosis, hepatitis C, hepatitis B and autoimmune hepatitis), diabetes and other metabolic diseases; 5) history of diseases such as Cushing’s syndrome, hypothyroidism, heart failure, renal failure and renal stones, weight loss medications; 6) patients receiving any supplements in the last 6 months, 7) Patients who have had a history of gastric bypass surgery, or severe weight loss during the last 6 months; 8) patients receiving hormone therapy, and 9) patient who do not use more than 10% of the capsules given in each follow-up.
Ethical Approval
The clinical trial was conducted in accordance with the Declaration of Helsinki. The Medical Ethics Committee of Ahvaz Jundishapur University of Medical Sciences has approved the implementation of this research. Moreover, it has been recorded in the Iranian registry of clinical trial (No.IRCT2016042827652N1) and available through www.irct.ir. All participants signed a written informed consent form.
Study Design
This study is a double-blind randomized clinical trial. Of the 100 patients enrolled in this project, 50 were selected based on inclusion criteria. Then, the patients were randomly assigned (1:1 ratio) (block randomized method) to two groups receiving ginger supplements and placebo. Before the beginning of the study, the cans containing the capsules were coded by a person other than the researcher, in the form of B and A, to ensure that the researchers did not know the type of capsules received by each group (given the double-blindness of the study).
Intervention
Patients in the intervention group (n = 25) daily received 3 capsules of 500 mg ginger, and patients in the placebo group (n = 25) daily took 3 capsules of 500 mg of placebo that were apparently similar to the ginger supplement. They were advised to take one capsule an hour after breakfast and one hour after lunch and another capsule for an hour after dinner for 12 weeks. It should be noted that the dosage of ginger used and the duration of the intervention have been determined according to previous studies.20 To calculate the compliance rate of patients for the supplementation, all patients received supplementation for 4 weeks and they were asked to deliver the cans of capsules at each visit, and then they were given supplements for the next four weeks. Patients were also followed up on a weekly basis by telephone in order to be aware of possible side effects and to ensure the use of supplements.
Patients in both groups were advised to receive a diet with energy balanced, according to guidelines published by the North American Association and all patients were asked to exercise at least three times a week for 30 mins a day.21
Clinical and Paraclinical Assessments
Anthropometric measurement of height, weight, waist circumference and hip circumference of the patients was performed at the beginning and end of the intervention according to the WHO definition. Body mass index (BMI) was calculated by division of weight by squared height, and also waist to hip ratio (WHR) was calculated by dividing waist to hip circumference.22 Blood (10cc) was taken from all participants at the beginning of the study and at the end of the 12th week after 10–12 hrs of fasting. Samples taken were centrifuged and their serum was separated. For further tests, the serum was kept at −80° C.
Serum triglyceride level was measured by enzymatic calorimetric and total cholesterol, high- density lipoprotein and low-density lipoprotein of plasma were measured by the photometric enzymatic method. The serum levels of liver enzymes, including GGT, ALT, and AST were measured by colorimetry (Parsazmun Co., Tehran, Iran). Fasting blood sugar was measured using the glucose oxidase method (Pars Azmoon Co, Tehran, Iran). Enzyme-linked immunosorbent assay (ELISA) was used to measure fasting serum insulin level (Insulin-R monobind, Lake Forest, USA), high-sensitivity c-reactive protein (hs-CRP) (LDN Labor Diagnostics Nord GmbH & Co. KG, Nordhorn, Germany), Total antioxidant capacity of the serum (ZellBio GmbH, Germany), Adiponectin (Orgenium Co, Vantaa, Finland), TNF-α (IBL Co., Ltd. Hamburg, Germany) and fetuin-A (Hangzhou Eastbiopharm Co., Ltd., Hangzhou, China) were measured by available enzyme-linked immunosorbent assay (ELISA) and based on the kit’s instructions. HOMA-IR was evaluated via the formula mentioned below.
(1)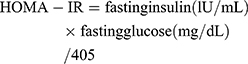
The fatty liver index was also calculated using FLI calculator software that computed FLI based on BMI, waist circumference, serum TG and GGT.
In order to control the effect of confounding the intake of diet and physical activity of the individual, a 3-day diet record (one day off and two days non-off) was taken from patients at the before and after intervention. Energy, macronutrients, and some of the dietary micronutrients received by people were measured using the Software modified Nutritionist IV software (version 3.5.2, First Data-Bank; Hearst Corp., San Bruno, CA). The physical activity level of patients was evaluated via the IPAQ (short form of the international physical activity questionnaire) at the beginning and end of the study.
A scoring system was employed to determine a semi-quantitative assessment of the severity of fat accumulation in the liver. The grade of liver fat infiltration was scored from I to III According to the liver radiance, liver echo tissue, liver-kidney contrast ratio, and bladder veins.
Dietary Intake Assessments
In order to control the effect of confounding the intake of diet and physical activity of the individual, a 3-day diet record (one day off and two days non-off) was taken from patients at the before and after intervention. Energy, macronutrients, and some of the dietary micronutrients received by people were measured using the Software modified Nutritionist IV software (version 3.5.2, First Data-Bank; Hearst Corp., San Bruno, CA). The physical activity level of each patient was evaluated using the short form of the international physical activity questionnaire (IPAQ) at the beginning and end of the study.
Statistical Analysis
The Shapiro–Wilk test was used to determine the data subordination of normal distribution. Intragroup comparison of quantitative variables with non-normal distribution, quantitative variables with normal distribution and categorical variables was analyzed by Wilcoxon paired rank test, paired-sample t-test, and Mc Nemar test, respectively. In order to intergroup comparison of the quantitative variables with non-normal distribution, quantitative variables with normal distribution and categorical variables, we used Mann–Whitney U-test, independent samples t-test, Mann–Whitney U-test, and chi-square test, respectively. Statistical analyzes were done using the SPSS version 16. P-value less than 0.05 was considered significant statistically.
Result
Patient Inclusion, Compliance and Safety
Out of 100 patients, 50 people who had inclusion criteria were entered into the study. According to the flow chart in Figure 1, 23 patients in the control group and 23 patients in the intervention group completed the stud. Compliance with supplements was 94% for the control group and 96% for the intervention group. None of the patients showed an allergic reaction or side effect of supplementation.
 |
Figure 1 Flow chart of the study. |
Baseline Characteristics
Baseline characteristics of the study participants are shown in Table 1. There was no statistically significant difference between the two groups in terms of the age, smoking, gender, height, and BMI at the beginning of the study (p> 0.05 for all variables).
 |
Table 1 Baseline Characteristics in Two Groups |
Anthropometric Variables and NAFLD Severity
Changes in all variables between the two intervention and control groups and in each group are shown in Table 2. Weight, hip circumference, waist circumference and BMI were significantly decreased in both groups (p< 0.001), but this difference was not significant between the two groups. There was no significant difference between the waist circumference (WHR) in the intragroup and inter-group comparisons. There was no significant difference between the two groups in the grade of fatty liver at the end of the intervention period.
 |
Table 2 Intra- and Inter-Group Comparisons of the Changes from Baseline to the End of the Intervention for Fatty Liver Status, Anthropometric and Nutritional Variables in Both Groups |
Physical Activity and Nutritional Variables
There was no significant difference in the improvement of physical activity between the two groups (p = 0.959). Although the physical activity increased significantly in both groups, the estimated energy intake in both groups significantly decreased, but in comparison to the changes, there was no significant difference between the two groups in the estimated energy received and the percentage of carbohydrate, protein, and fat in the diet.
Biochemical Parameters
The variations of all biochemical variables in both groups at the beginning and end of the study are briefly illustrated in Tables 3 and 4.
 |
Table 3 Intra- and Inter-Group Comparisons of the Changes from Baseline to the End of the Intervention for Biochemical Parameters |
 |
Table 4 Intra- and Inter-Group Comparisons of the Changes from Baseline to the End of the Intervention for Biochemical Parameters |
Liver Enzymes
Serum GGT and GGT ALT levels decreased meaningfully in both groups after intervention (p <0.05). However, no significant differences were found in serum levels GGT in between group comparisons (p = 0.06). There was no significant change in serum AST level in any group (Table 3).
Insulin Resistance Indices
After the intervention, FBS and HOMA-IR and FBS were significantly lower in the group receiving ginger than the placebo group (p = 0.047 and 0.029, respectively). The level of fasting insulin in the ginger group significantly decreased compared to the beginning of the study (p = 0.017). However, there was no statistically significant change in the comparison between the groups (p = 0.559) (Table 3).
Serum Level of Lipid
After intervention, total serum total cholesterol and LDL-C levels in the ginger group decreased meaningfully compared to placebo (p = 0.026 and p = 0.032, respectively), while HDL-C and TG had no significant changes in any of the groups. No significant difference was observed in terms of improvement of the ratio of LDL to HDL between the two groups (P = 0.267). But, this ratio was meaningfully decreased in the ginger group compared to the beginning of the study (P = 0.033) (Table 3).
Antioxidant and Inflammatory Markers
Concentration of hs-CRP and fetuin-A in the ginger group decreased significantly compared to the placebo group (P = 0.006 and P = 0.008, respectively). Conversely, changes in TNF-α, TAC and adiponectin level were not significant in any of the groups (Table 4).
Fatty Liver Index (FLI)
The FLI reduced meaningfully in both groups, although there was no difference between the two groups significantly (Table 4).
Discussion
Human and animal studies have reported several pharmacological effects including weight lowering, anti-inflammatory, antioxidant, lipid lowering and improving insulin sensitivity and glucose tolerance for ginger.23 The gingerol and shogaol are considered as bioactive compounds in fresh and dried ginger, respectively, and are responsible for a large number of pharmacological properties of ginger.9 Overall, the results of this study showed that daily consuming 1500 mg of ginger for 12 weeks meaningfully reduced serum levels of total cholesterol, LDL-C, FBS, ALT, fetuin-A, hs-CRP and HOMA-IR.
Recently, Ginger’s beneficial effects on weight control have been attracted attention. The precise mechanisms of the ginger effects on weight loss are not fully understood. However, several possible mechanisms, including suppression of the pancreatic lipase enzyme and decreasing intestinal absorption of dietary fat, increasing lipolysis of white adipose tissue by increasing the enzyme activation of hormone-sensitive lipase, suppression of lipogenesis and lipid accumulation by decreasing the induction of the fatty acid synthase and acetyl CoA carboxylase, increasing thermogenesis and controlling appetite have been proposed in studies.23
In this study, although weight loss in both groups was significant compared to the beginning of the study, supplementation of ginger compared with placebo was not associated with more weight loss (Table 2). Reduction of receiving energy and improvement of physical activity observed in end evaluations of the study compared to the baseline may explain the weight loss caused in both groups. There are few human studies examining the effect of ginger on anthropometric indices and the results of these studies are inconsistent. Atashak et al had studied the effects of ginger supplementation (1 g/day) for 10 weeks on obese men (n=8). The results of their study showed no significant differences in body composition and anthropometric indices between the ginger and placebo groups,24 which was consistent with our study results. Contrary to our study, in Ebrahimzadeh et al’s study, ginger supplementation (2 g/day) for 12 weeks, reduced the BMI and anthropometric indices compared with placebo in 80 obese women.25 Most animal studies support the weight-reducing effects of ginger in the obese animal models, while based on the results of the available human studies available, or no changes in anthropometric properties have been made or variations are very slight. Of course, it should be taken into account that in most animal studies, ginger extract or its active ingredients have been used instead of ginger powder, which can justify the difference observed in the results of animal and human studies.23
Due to the fundamental role of insulin resistance and as a result of hyperinsulinemia in fat accumulation in the liver, an increase in insulin sensitivity is considered as a treatment approach against NAFLD.10
The results of the present study indicated that the ginger intake compared to the placebo resulted in a significant decrease in FBS and HOMA-IR, which is consistent with the result of previous studies. Similar to our article, Rahimlou et al indicated that consumption of 2000 mg of ginger over12 weeks, meaningfully reduced insulin and fasting blood sugar.26 Similarly, as Mozaffari et al reported supplementation of ginger powder (3g/day) in patients with type 2 diabetes for 8 weeks caused a meaningful decrease in fasting blood sugar and hemoglobin A1C and a meaningful increase in the insulin sensitivity index compared to the beginning of the study.27 Findings from similar animal studies also confirm these results.28,29
The effect of ginger on insulin sensitivity is probably due to the effect of its bioactive compounds on PPARγ or adiponectin upregulation. PPARα and PPARγ can affect the accumulation of liver triglycerides and thus pathogenesis of NAFLD. It has been reported that PPARγ can improve insulin sensitivity and decrease the flow of free fatty acids to the liver. 6-Shogaol has been reported in ginger as a significant agonist of PPARγ in adipose tissue.30 Another mechanism of ginger effect is the inhibition of liver phosphorylase to prevent degradation of glycogen stored in liver cells. By inhibiting glucose 6-phosphatase activity, ginger also reduces the degradation of glucose 6-phosphate into glucose and, consequently, reducing blood glucose. Ginger seems to reduce the absorption of glucose in the body through inhibition of enzymes of glucose metabolism pathway, such as α-glucosidase and amylase in the intestine.31 There are many antioxidant compounds, including gingerols, shogaol, paradols and zingerone in ginger. These antioxidant compounds probably increase the expression of GLUT4 proteins, insulin receptors, and improve the function of β-pancreatic cells, thereby improving glucose tolerance.32
Another finding of our study was the antilipidemic effects of ginger. The results of the present study indicated that daily consumption of ginger compared to placebo significantly reduced LDL-C and total cholesterol levels. But Serum levels of HDL-C and TG did not change meaningfully. The results of previous clinical trials investigating the effect of ginger on serum lipid levels are contradictory. In the study of Rahimlou et al, it was shown that an intervention with 2000 mg of ginger for 12 weeks reduced serum triglyceride, but HDL-C, LDL-C and total cholesterol levels between two groups did not have a significant difference.26 Alizadeh et al reported that intervention with 3g/ginger for 45 days in hyperlipidemic patients significantly decreased serum triglyceride and serum cholesterol compared to the placebo.33 Mozaffari et al observed that daily intake of three grams of ginger for eight weeks in diabetic patient leads to a reduction in serum LDL-C level, but does not affect the levels of total cholesterol, TG and HDL-C.24 The finding of the present study on the effect of ginger on serum cholesterol concentration is consistent with the results of many animal studies.28,34
The reduction of cholesterol by ginger can be explained as follows: Ginger can induce cholesterol conversion to bile acids by increasing activity the cholesterol-7 -α-hydroxylase enzyme and ultimately lead to, decline serum cholesterol level.33 Also, it has been reported that ginger compounds suppress the synthesis of cholesterol in the mice liver cells.35 The findings of this study regarding the lack of effect of Ginger Supplement on serum HDL-C levels are consistent with the results of the studies conducted by Mozaffari,27 Alizadeh,33 and Bordia.34 The reason for not changing HDL-C levels in this study and other studies are probably that HDL-C changes are less affected by dietary components.
The conversion of simple steatosis to more advanced forms of NAFLD is also affected by oxidative stress and inflammation.12 A number of previous studies have shown that the serum levels of oxidative stress markers and inflammatory cytokines in NAFLD patients are significantly higher.36,37 Therefore, reducing inflammatory and oxidative stress factors in NAFLD patients may play an important role in slowing the rate of NAFLD progression and reducing the risk of cardiovascular disorders due to this disease.
The results of this study showed that supplementation with ginger (1500mg/day) for 12 weeks decreased the serum levels of hs-CRP and fetuin-A significantly, but TNF-α, TAC and adiponectin levels were not meaningfully changed. The hs-CRP is mostly produced in the liver and its serum level is linked to the rate of liver inflammation. In a study by Arablou et al daily consumption of 1600 mg of ginger over 12 weeks significantly decreased serum level hs-CRP in diabetic patients.38 Furthermore, in the study conducted by Atashak et al supplementation with ginger powder (1g/day) for 10 weeks reduced the mean hs-CRP concentration in obese men, which is similar to our study results.39
It looks like that the effect of ginger on reducing inflammation is due to the effect of some active ingredients (gingerol and zerombon) in the inhibition of NFκB and TNF-α. Ginger decreases the activity of NFκB by inhibition of TNFα gene and thus production of acute phase positive protein such as CRP is inhibited.40
Fetuin-A is a protein abundant in the serum, mainly produced in the liver. Recently, studies have reported that fetuin-A may influence the development of metabolic syndrome through two mechanisms. It has been shown that fetuin-A inhibits the autophosphorylation of insulin receptors. Improving insulin sensitivity in mice with low serum levels of fetuin-A suggests that fetuin-A may play a role in increasing insulin resistance.41 Fetuin-A inhibits the expression of adiponectin and increases the expression of inflammatory cytokines, as well as a strong association was demonstrated in liver biopsies between fetuin-A gene expression and major enzymes involved in glucose and lipid metabolism.42
In the study of Reinehr and Roth was observed that serum levels of fetuin-A were significantly increased in obese children with non-alcoholic fatty liver disease.43 A study by Yilmaz et al showed that the serum level of the fetuin-A enhanced in adult patients with NAFLD diagnosed with liver biopsy.44 This study examined the effect of ginger on the serum level of fetuin-A in patients with NAFLD for the first time.
Finally, the findings of our study showed that the intake of ginger compared with placebo resulted in a significant reduction in serum concentration of ALT, although there was no significant change in AST and steatosis level in any of the groups. Both groups showed significant improvement in serum ALT and GGT levels due to receiving nutritional recommendations and increasing physical activity, but only ALT changes in the intervention group were meaningfully more than the placebo group. These findings were consistent with previous research findings. Rahimlou et al reported supplementation with 2000 mg of ginger for 12 weeks significantly decreased serum levels of ALT and GGT compared to the placebo.25 In a study conducted by Motawi et al at the National Research Center in Cairo in 2011, the ginger effect on liver fibrosis was surveyed in Wistar rats. The results of this study showed that treatment with ginger extract with a significant decrease in aspartate aminotransferase and alanine aminotransferase has a protective effect against hepatic fibrosis.45
The Fatty Liver (FLI) index helps to diagnose fatty liver based on BMI, triglyceride, GGT, and waist circumference for referral for ultrasonography. In the research, FLI is used to identify people at high risk of liver damage to participate in the interventional and observational studies. The FLI ranges from 1 to 100. FIL <30 means that the presence of fatty liver is unlikely, FLI≥60 means that there is a fatty liver, and FLI <60 <30 means that a conclusion is impossible.2 In our study, FLI was significantly reduced in both ginger and placebo groups, but the changes between the two groups were not significant.
In this study, ultrasonography method was used to detect the degree of steatosis. This method cannot determine changes as precisely as exact techniques such as fibroscan. Also, the small sample size is another weakness of the present study.
Conclusions
In total, the results of the present study showed that daily receiving 1500 mg of ginger for 12 weeks could increase the efficacy of lifestyle modification (increased physical activity and Nutritional recommendations) in improving patients with NAFLD. As regards that receiving ginger supplement in this study did not have any side effects, it can be understood that supplementation with ginger in NAFLD patients can increase the success rate of treatment.
However, due to a few clinical trials in this field, more human research with different amount of ginger supplement and longer duration of the intervention is recommended. It is also suggested that since, according to the view of traditional medicine, ginger is hot and dry, it is recommended that studies be conducted to evaluate the effect of ginger in patients with NAFLD, taking into account their temperament.
Data Sharing Statement
The data sets used and/or analyzed during this study are available from the corresponding author on reasonable request and permission for use was received by the ethics committee of Ahvaz Jundishapur University of Medical Sciences.
Acknowledgments
The reported results were a part of the MSc thesis of Roya Rafie (NRC-9501) that is registered in the Nutrition and Metabolic Diseases Research. This study was supported by the Vice Chancellor for research affairs of Ahvaz Jundishapur University of Medical Sciences.
Author Contributions
All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors declare that they have no competing interests in this work.
References
1. Vuppalanchi R, Chalasani N. Nonalcoholic fatty liver disease and nonalcoholic steatohepatitis: selected practical issues in their evaluation and management. Hepatology. 2009;49(1):306–317. doi:10.1002/hep.22603
2. Marchesini G, Bugianesi E, Forlani G, et al. Nonalcoholic fatty liver, steatohepatitis, and the metabolic syndrome. Hepatology. 2003;37(4):917–923. doi:10.1053/jhep.2003.50161
3. Chalasani N, Younossi Z, Lavine JE, et al. The diagnosis and management of non-alcoholic fatty liver disease: practice guideline by the American Association for the Study of Liver Diseases, American College of Gastroenterology, and the American Gastroenterological Association. Hepatology. 2012;55(6):2005–2023. doi:10.1002/hep.25762
4. Lankarani KB, Ghaffarpasand F, Mahmoodi M, et al. Non alcoholic fatty liver disease in southern Iran: a population based study. Hepat Mon. 2013;13:5.
5. Day C, James O. Steatohepatitis: a tale of two” hits”. Gastroenterology. 1998;114:842–845. doi:10.1016/S0016-5085(98)70599-2
6. Shahmohammadi HA, Hosseini SA, Hajiani E, Malehi AS, Alipour M. Effects of green coffee bean extract supplementation on patients with non-alcoholic fatty liver disease: a randomized clinical trial. Hepat Mon. 2017;17(4):e12299. doi:10.5812/hepatmon.45609
7. Abbasnezhad A, Choghakhori R, Kashkooli S, Alipour M, Asbaghi O, Mohammadi R. Effect of L-carnitine on liver enzymes and biochemical factors in hepatic encephalopathy: a systematic review and meta-analysis. J Gastroenterol Hepatol. 2019 Des;34(12):2062–2070. doi:10.1111/jgh.14765
8. Mahdavinia M, Alizadeh S, Raesi Vanani A, et al. Effects of quercetin on bisphenol A-induced mitochondrial toxicity in rat liver. Iran J Basic Med Sci. 2019;22(5):499–505. doi:10.22038/ijbms.2019.32486.7952
9. Ali BH, Blunden G, Tanira MO, Nemmar A. Some phytochemical, pharmacological and toxicological properties of ginger (Zingiber officinale Roscoe): a review of recent research. Food Chem Toxicol. 2008;46(2):409–420. doi:10.1016/j.fct.2007.09.085
10. Sahebkar A. Potential efficacy of ginger as a natural supplement for nonalcoholic fatty liver disease. World J Gastroenterol. 2011;17(2):271. doi:10.3748/wjg.v17.i2.271
11. Verma S, Singh M, Jain P, Bordia A. Protective Effect of Ginger, Zingiber Officinale Rosc on Experimental Atherosclerosis in Rabbits. Indian J Exp Biol. 2004;42(7):736738.
12. Jou J, Choi SS, Diehl AM, editors. Mechanisms of disease progression in nonalcoholic fatty liver disease. Semin Liver Dis. 2008;28(4):370–379. doi:10.1055/s-0028-1091981
13. Mustafa T, Srivastava K, Jensen K. Drug development report. 9. Pharmacology of ginger, zingiber-officinale. J Drug Dev. 1993;6(1):25–39.
14. Stoilova I, Krastanov A, Stoyanova A, Denev P, Gargova S. Antioxidant activity of a ginger extract (Zingiber officinale). Food Chem. 2007;102(3):764–770. doi:10.1016/j.foodchem.2006.06.023
15. Polyzos S, Kountouras J, Zavos C. The multi-hit process and the antagonistic roles of tumor necrosis factor-alpha and adiponectin in non alcoholic fatty liver disease. Hippokratia. 2009;13(2):127.
16 Haukeland JW, Dahl TB, Yndestad A, et al. Fetuin A in nonalcoholic fatty liver disease: in vivo and in vitro studies. Eur J Endocrinol. 2012;166(3):503–510. doi:10.1530/EJE-11-0864
17. Gilani A, Ghayur M. Ginger: From Myths to Reality. Ethnotherapies in the Cycle of Life. BOD-Books on Demand/Ethnomed Institut Für Ethnomedizin eV: Munich; 2005:307–315.
18. Ghayur MN, Gilani AH. Ginger lowers blood pressure through blockade of voltage-dependent calcium channels. J Cardiovasc Pharmacol. 2005;45(1):74–80. doi:10.1097/00005344-200501000-00013
19. Prati D, Taioli E, Zanella A, et al. Updated definitions of healthy ranges for serum alanine aminotransferase levels. Ann Intern Med. 2002;137(1):1–10. doi:10.7326/0003-4819-137-1-200207020-00006
20. Khandouzi N, Shidfar F, Rajab A, Rahideh T, Hosseini P, Taheri MM. The effects of ginger on fasting blood sugar, hemoglobin a1c, apolipoprotein B, apolipoprotein aI and malondialdehyde in type 2 diabetic patients. Iran j Pharm Res. 2015;14(1):131.
21. Initiative NOE, Heart N, Lung, Institute B, Obesity NAAftSo, Identification EPot, et al. The Practical Guide: Identification, Evaluation, and Treatment of Overweight and Obesity in Adults. The Institute; 2000.
22. Ashtary-Larky D, Daneghian S, Alipour M, et al. Waist circumference to height ratio: better correlation with fat mass than other anthropometric indices during dietary weight loss in different rates. Int J Endocrinol Metab. 2018;16(4):e55023. doi:10.5812/ijem
23. Ebrahimzadeh Attari V, Malek Mahdavi A, Javadivala Z, Mahluji S, Zununi Vahed S, Ostadrahimi A. A systematic review of the anti-obesity and weight lowering effect of ginger (Zingiber officinale Roscoe) and its mechanisms of action. Phytother Res. 2017;32:577–585.
24. Atashak S, Peeri M, Azarbayjani MA, Stannard SR, Haghighi MM. Obesity-related cardiovascular risk factors after long-term resistance training and ginger supplementation. J Sports Sci Med. 2011;10(4):685.
25. Attari VE, Ostadrahimi A, Jafarabadi MA, Mehralizadeh S, Mahluji S. Changes of serum adipocytokines and body weight following Zingiber officinale supplementation in obese women: a RCT. Eur J Nutr. 2016;55(6):2129–2136. doi:10.1007/s00394-015-1027-6
26. Rahimlou M, Yari Z, Hekmatdoost A, Alavian SM, Keshavarz SA. Ginger supplementation in nonalcoholic fatty liver disease: a randomized, double-blind, placebo-controlled pilot study. Hepat Mon. 2016;16:1. doi:10.5812/hepatmon
27. Mozaffari-Khosravi H, Talaei B, Jalali B-A, Najarzadeh A, Mozayan MR. The effect of ginger powder supplementation on insulin resistance and glycemic indices in patients with type 2 diabetes: a randomized, double-blind, placebo-controlled trial. Complement Ther Med. 2014;22(1):9–16. doi:10.1016/j.ctim.2013.12.017
28. Nammi S, Sreemantula S, Roufogalis BD. Protective effects of ethanolic extract of Zingiber officinale rhizome on the development of metabolic syndrome in high-fat diet-fed rats. Basic Clin Pharmacol Toxicol. 2009;104(5):366–373. doi:10.1111/pto.2009.104.issue-5
29. Shanmugam KR, Mallikarjuna K, Kesireddy N, Reddy KS. Neuroprotective effect of ginger on anti-oxidant enzymes in streptozotocin-induced diabetic rats. Food Chem Toxicol. 2011;49(4):893–897. doi:10.1016/j.fct.2010.12.013
30. Isa Y, Miyakawa Y, Yanagisawa M, et al. 6-Shogaol and 6-gingerol, the pungent of ginger, inhibit TNF-α mediated downregulation of adiponectin expression via different mechanisms in 3T3-L1 adipocytes. Biochem Biophys Res Commun. 2008;373(3):429–434. doi:10.1016/j.bbrc.2008.06.046
31. Priya Rani M, Padmakumari K, Sankarikutty B, Lijo Cherian O, Nisha V, Raghu K. Inhibitory potential of ginger extracts against enzymes linked to type 2 diabetes, inflammation and induced oxidative stress. Int J Food Sci Nutr. 2011;62(2):106–110. doi:10.3109/09637486.2010.515565
32. Li Y, Tran VH, Duke CC, Roufogalis BD. Preventive and protective properties of Zingiber officinale (ginger) in diabetes mellitus, diabetic complications, and associated lipid and other metabolic disorders: a brief review. Evid Based Complement Alternat Med. 2012;2012:516870.
33. Alizadeh-Navaei R, Roozbeh F, Saravi M, Pouramir M, Jalali F, Moghadamnia AA. Investigation of the effect of ginger on the lipid levels. A Double Blind Controlled Clinical Trial. Saudi Med J. 2008;29(9):1280–1284.
34. ElRokh E-SM, Yassin NA, El-Shenawy SM, Ibrahim BM. Antihypercholesterolaemic effect of ginger rhizome (Zingiber officinale) in rats. Inflammopharmacology. 2010;18(6):309–315. doi:10.1007/s10787-010-0053-5
35. Bordia A, Verma S, Srivastava K. Effect of ginger (Zingiber officinale Rosc.) and fenugreek (Trigonella foenumgraecum L.) on blood lipids, blood sugar and platelet aggregation in patients with coronary artery disease. Prostaglandins Leukotrienes Essent Fatty Acids. 1997;56(5):379–384. doi:10.1016/S0952-3278(97)90587-1
36. Haukeland JW, Damås JK, Konopski Z, et al. Systemic inflammation in nonalcoholic fatty liver disease is characterized by elevated levels of CCL2. J Hepatol. 2006;44(6):1167–1174. doi:10.1016/j.jhep.2006.02.011
37. Belfort R, Harrison SA, Brown K, et al. A placebo-controlled trial of pioglitazone in subjects with nonalcoholic steatohepatitis. N Engl J Med. 2006;355(22):2297–2307. doi:10.1056/NEJMoa060326
38. Arablou T, Aryaeian N, Valizadeh M, Sharifi F, Hosseini A, Djalali M. The effect of ginger consumption on glycemic status, lipid profile and some inflammatory markers in patients with type 2 diabetes mellitus. Int J Food Sci Nutr. 2014;65(4):515–520. doi:10.3109/09637486.2014.880671
39. Atashak S, Azarbayjani M, Piri M, Jafari A. Effects of combination of long-term ginger consumption and resistance training on lipid peroxidation and insulin resistance in obese men. J Med Plants. 2012;2(42):179–188.
40. Kim SO, Chun K-S, Kundu JK, Surh Y-J. Inhibitory effects of [6]-gingerol on PMA-induced COX-2 expression and activation of NF-κB and p38 MAPK in mouse skin. Biofactors. 2004;21(1–4):27–31. doi:10.1002/biof.v21:1/4
41. Mathews ST, Singh GP, Ranalletta M, et al. Improved insulin sensitivity and resistance to weight gain in mice null for the Ahsg gene. Diabetes. 2002;51(8):2450–2458. doi:10.2337/diabetes.51.8.2450
42. Hennige AM, Staiger H, Wicke C, et al. Fetuin-A induces cytokine expression and suppresses adiponectin production. PLoS One. 2008;3(3):e1765. doi:10.1371/journal.pone.0001765
43. Reinehr T, Roth CL. Fetuin-A and its relation to metabolic syndrome and fatty liver disease in obese children before and after weight loss. J Clin Endocrinol Metab. 2008;93(11):4479–4485. doi:10.1210/jc.2008-1505
44. Yilmaz Y, Yonal O, Kurt R, et al. Serum fetuin A/α 2HS-glycoprotein levels in patients with non-alcoholic fatty liver disease: relation with liver fibrosis. Ann Clin Biochem. 2010;47(6):549–553. doi:10.1258/acb.2010.010169
45. Motawi TK, Hamed MA, Shabana MH, Hashem RM, Aboul Naser AF. Zingiber officinale acts as a nutraceutical agent against liver fibrosis. Nutr Metab. 2011;8:40.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
