Back to Journals » Clinical, Cosmetic and Investigational Dermatology » Volume 15
Disseminated Kaposi Sarcoma
Received 31 May 2022
Accepted for publication 9 August 2022
Published 20 August 2022 Volume 2022:15 Pages 1711—1714
DOI https://doi.org/10.2147/CCID.S376060
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Jeffrey Weinberg
Jin-Yuan Ma,1 Jia-Wei Liu2
1The Second School of Clinical Medicine, Zhujiang Hospital, Southern Medical University, Guangzhou, 510282, People’s Republic of China; 2Department of Dermatology, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, National Clinical Research Center for Dermatologic and Immunologic Diseases, Beijing, 100730, People’s Republic of China
Correspondence: Jia-Wei Liu, Department of Dermatology, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, National Clinical Research Center for Dermatologic and Immunologic Diseases, Beijing, 100730, People’s Republic of China, Tel +86-10-69151543, Fax +86-10-69151502, Email [email protected]
Abstract: AIDS-associated Kaposi sarcoma (KS) is the most common HIV-associated neoplasm. Disseminated Kaposi sarcoma became rare with the application of antiretroviral therapy. Oral AIDS-associated KS has prognostic relevance, indicating higher mortality than those with cutaneous lesions only. In this study, we reported a 40-year-old man presented with ulcerated violaceous plaques on his hard palate. Similar lesion can be observed on his left groin and anus, as well as on esophagus and gastric fundus under upper gastrointestinal endoscopy. Histological examination accorded with KS. After five cycles of doxorubicin, his oral, skin and esophagus lesions regressed considerably.
Keywords: oral Kaposi sarcoma, AIDS-associated Kaposi sarcoma, AIDS, disseminated Kaposi sarcoma, chemotherapy
Introduction
Kaposi sarcoma (KS) is a multicentric vascular neoplasm of endothelial origin with low-grade malignancy. The disease predominantly involves mucocutaneous regions but can also affect viscera, bone, and lymph nodes. Histologically, the disease is characterized by neoangiogenesis, spindle-shaped cells and a chronic inflammatory infiltrate. KS is classified into four clinical variants: classic, endemic (African), iatrogenic (transplant-associated), and epidemic (AIDS-associated). AIDS-associated KS is the most prevalent and is more aggressive than the other three types.1 AIDS-associated KS is also the most common HIV-associated neoplasm, occurring in >50% of patients with late-stage HIV infection2 and accounting for 90% of cancers in patients with AIDS.3
Case Report
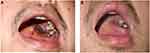
A 40-year-old man who have sex with man (MSM) presented with a purple ulcerated plaque 2cm × 2cm in diameter on the left side of his hard palate which started a month before presentation. He had been diagnosed with human immunodeficiency virus (HIV) infection on the basis of serum antibodies 3 years before presentation with a marked reduction in the T4:T8 lymphocyte ratio. His CD4 count was 250 cells per microlitre at the time of diagnosis. He was under treatment with antiretroviral therapy (ART-Stavudine/Lamivudine/Efavirenz) for five months. He denied any history of trauma or injury in his mouth. On physical examination, slightly tender lymphadenopathies were found on both sides of the neck and groins. Another non-ulcerated nodule 1cm × 1cm in diameter was present on the hard palate. A small patch of violet plaque was visible behind the ulcerated plaque (Figure 1A). Several purple papules and nodules were visible on his left groin and anus. Upper gastrointestinal endoscopy revealed multiple violaceous plaques on the esophagus and gastric fundus (Figure 2). Lab test revealed his CD4 count was 70 (normal 500–1400) cells per microlitre, and his viral load in plasma was 44,913 copies per milliliter.
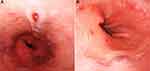 |
Figure 2 (A) Violaceous ulcerative plaques on esophagus and (B) violaceous plaques on gastric fundus under upper gastrointestinal endoscopy. |
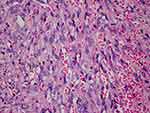
Biopsies of the ulcerated palatal lesion and left groin lesion were performed. Histopathology revealed proliferating spindle cells with atypia, and irregular vascular slits filled with blood cells (Figure 3). Immunohistochemistry is positive for HHV-8. The diagnosis of AIDS-associated disseminated Kaposi sarcoma was confirmed. Chemotherapy with doxorubicin 40mg on a 3-weekly schedule was started, performing 5 cycles. The patient’s lesions steadily improved from the first cycle of doxorubicin. After five cycles of doxorubicin, his oral, skin, and esophagus lesions had regressed considerably (Figure 1B).
Discussion
First reported in 1981, AIDS-associated KS was considered aggressive and frequently fatal. The disease can be disseminated, affecting multi-system including skin, lung, lymphatic tissue, bone, oral cavity, and gastrointestinal tract. While nowadays, the incidence of disseminated Kaposi sarcoma has become rare with the wide application of ART.
Clinically, KS presented with multiple purple-blue or reddish-brown plaques and nodules. The histopathological features of KS are identical in all subtypes and comprise proliferating spindle-shaped tumor cells, lymphoplasmacytic inflammatory infiltration, incomplete vascular slit formation, and extravascular hemorrhage. These microscopic features may not be evident in the early macular stage but may become more prominent with clinical progression into nodules and plaques.4 Immunohistochemistry is positive for CD31, CD34, D2-40, LYVE-1, VEGFR-3, Prox-1, and LNA-1.5,6
Human herpesvirus-8 (HHV-8) is an essential factor in the etiology of all variants of KS. In rare cases, HHV-8 infection in HIV patients can result in lethal complications such as Kaposi Sarcoma Inflammatory Cytokine Syndrome (KICS). KICS clinically presents with lymphadenopathy, pancytopenia, HIV and KSHV viremia, and signs of systemic inflammatory syndrome (SIRS), requiring a ventilator and vasopressor support.7
Oral KS is prevalent in all stages of HIV infection. It occurs in up to 71% of patients with AIDS-associated KS and is the initial presentation of KS in 22% of patients with HIV infection.3 The high incidence of oral AIDS-associated KS may result from a higher viral copy number in saliva than in other bodily secretions. Additionally, the oropharyngeal epithelial cells are preferential susceptibility to HHV-8 infection, where the virus can be harbored, further replicated, and shed.4,8
Clinically, oral KS presents as violaceous patches, nodules, or plaques with pain and hemorrhage after trivial trauma. It can be unifocal or multifocal and may affect the palate, gingiva, and dorsum of the tongue; the hard and soft palates are most commonly involved.9 Oral KS can also cause the erosion of alveolar bone, resulting in tooth mobility.5 Oral AIDS-associated KS has prognostic relevance. Among patients who are naive to antiviral treatment, those with oral AIDS-associated KS have far higher mortality than those with cutaneous lesions only, especially in patients with exophytic lesions and facial lymphedema.4,5
Oral KS must be clinically distinguished from hemangioma, bacillary angiomatosis, pyogenic granuloma, lymphangioma, angiosarcoma, melanoma, and Kaposi-like hemangioendothelioma. It may also mimic an odontogenic abscess, peripheral giant cell granuloma, or gingival enlargement caused by medications or hematologic malignancies.8,9
Treatment for KS aims to alleviate symptoms, restore function, and improve cosmetic appearance. Reconstitution of immune function with highly active antiretroviral therapy is critical for the management of AIDS-associated KS. Asymptomatic cutaneous lesions can be monitored during antiretroviral therapy, symptomatic, limited cutaneous lesions should be treated with minimally invasive therapy, while systemic chemotherapy should be initiated in those with advanced cutaneous lesions or oral, visceral, and nodal involvement.10 Treatments range from local interventions such as radiotherapy, intralesional chemotherapy, intralesional sclerosing agents, surgical excision, laser ablation, cryosurgery, topical alitretinoin gel, and topical imiquimod to systemic chemotherapy based on liposomal anthracyclines and taxanes.8 Other therapeutic options including bevacizumab, thalidomide, anti-HHV-8 agents, metalloproteinase inhibitors, and cytokine signaling pathway inhibitors are available but require further testing.9,10
Conclusion
Here, we report a typical case of disseminated Kaposi’s sarcoma in a 40-year-old Chinese man with AIDS. Diagnosis of Kaposi sarcoma was confirmed by histopathological examination and a positive immunohistochemistry result for HHV-8. As the patient was already under treatment with highly active antiretroviral therapy, chemotherapy was initiated and the patient responded well. Oral Kaposi sarcoma indicates a poor prognosis in AIDS patients and disseminated Kaposi’s sarcoma should be identified in time for timely intervention.
Consent Statement
A formal written consent was obtained for publication the case details and associated images from the patient. No institutional approval is required to publish the case details.
Funding
The article had no funding source.
Disclosure
All authors stated no potential conflicts of interest, including specific financial interests and relationships and affiliations relevant to the subject of the manuscript.
References
1. Antman K, Chang Y. Kaposi’s sarcoma. N Engl J Med. 2000;342(14):1027–1038. doi:10.1056/NEJM200004063421407
2. La Ferla L, Pinzone MR, Nunnari G, et al. Kaposi’ s sarcoma in HIV-positive patients: the state of art in the HAART-era. Eur Rev Med Pharmacol Sci. 2013;17(17):2354–2365.
3. Lager I, Altini M, Coleman H, Ali H. Oral Kaposi’s sarcoma: a clinicopathologic study from South Africa. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2003;96(6):701–710. doi:10.1016/S1079-2104(03)00370-6
4. Bunn BK, Carvalho Mde V, Louw M, Vargas PA, van Heerden WF. Microscopic diversity in oral Kaposi sarcoma. Oral Surg Oral Med Oral Pathol Oral Radiol. 2013;115(2):241–248. doi:10.1016/j.oooo.2012.11.009
5. Pantanowitz L, Khammissa R, Lemmer J, Feller L. Oral HIV-associated Kaposi sarcoma. J Oral Pathol Med. 2013;42(3):201–207. doi:10.1111/j.1600-0714.2012.01180.x
6. Pantanowitz L, Otis CN, Dezube BJ. Immunohistochemistry in Kaposi’s sarcoma. Clin Exp Dermatol. 2010;35(1):68–72. doi:10.1111/j.1365-2230.2009.03707.x
7. Karass M, Grossniklaus E, Seoud T, Jain S, Goldstein DA. Kaposi sarcoma inflammatory cytokine syndrome (KICS): a rare but potentially treatable condition. Oncologist. 2017;22(5):623–625. doi:10.1634/theoncologist.2016-0237
8. Fatahzadeh M. Kaposi sarcoma: review and medical management update. Oral Surg Oral Med Oral Pathol Oral Radiol. 2012;113(1):2–16. doi:10.1016/j.tripleo.2011.05.011
9. Fatahzadeh M, Schwartz RA. Oral Kaposi’s sarcoma: a review and update. Int J Dermatol. 2013;52(6):666–672. doi:10.1111/j.1365-4632.2012.05758.x
10. Reid E, Suneja G, Ambinder RF, et al. AIDS-related Kaposi sarcoma, version 2.2019, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw. 2019;17(2):171–189. doi:10.6004/jnccn.2019.0008
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.