Back to Journals » Risk Management and Healthcare Policy » Volume 14
Dental Practitioners’ Knowledge, Attitude and Practices for Mouthwash Use Amidst the COVID-19 Pandemic
Authors Imran E, Khurshid Z , Adanir N , Ashi H, Almarzouki N, Baeshen HA
Received 19 October 2020
Accepted for publication 27 January 2021
Published 15 February 2021 Volume 2021:14 Pages 605—618
DOI https://doi.org/10.2147/RMHP.S287547
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Marco Carotenuto
Eisha Imran,1 Zohaib Khurshid,2 Necdet Adanir,3 Heba Ashi,4 Nawaf Almarzouki,5 Hosam Ali Baeshen6
1Department of Dental Materials, Islamic International Dental College, Riphah International University, Islamabad, 44000, Pakistan; 2Department of Prosthodontics and Dental Implantology, College of Dentistry, King Faisal University, Al Ahsa, 31982, Saudi Arabia; 3Department of Restorative Dentistry, College of Dentistry, King Faisal University, Al Ahsa, 31982, Saudi Arabia; 4Department of Dental Public Health, College of Dentistry, King Abdulaziz University, Jeddah, Saudi Arabia; 5Department of Ophthalmology, College of Medicine, King Abdulaziz University, Jeddah, Saudi Arabia; 6Department of Orthodontics, College of Dentistry, King Abdulaziz University, Jeddah, Saudi Arabia
Correspondence: Zohaib Khurshid
Department of Prosthodontics and Dental Implantology, College of Dentistry, King Faisal University, Al Ahsa, 31982, Saudi Arabia
Tel +966-558-420410
Email [email protected]
Introduction: COVID-19 created a peculiar situation worldwide, thus altering the fundamental dynamics of clinical dentistry. This KAP survey was carried out to evaluate the knowledge, attitude and practices among dental practitioners regarding the use of mouthwashes and emphasize on pre-procedural utilization of mouthwashes.
Methodology: A descriptive, cross-sectional study was conducted during 1st to 15th July 2020, among working dental practitioners across the globe. A questionnaire was formed on Kwiksurveys.com, it comprised of demographic details, it further investigated the level of knowledge and new precautionary measures adopted. A total number of 707 dental practitioners from eighteen different countries responded.
Results: Data were analyzed using SPSS version 23. A “fisher exact test” was applied to assess the difference between the mouthwashes prescribed by various countries. Knowledge section revealed a requisite understanding regarding the disease and its transmission. Only 38.9% of the participants knew that Povidone-iodine (PVP-I) mouthwashes are more efficient in reducing coronaviruses in contrast to mouthwashes made of chlorhexidine (CHX). Whereas 33.9% knew that 0.23% of PVP-I had substantial virucidal activity against SARS-COV, MERS-CoV, influenza virus and rotavirus while 31.1% recognized that oral rinses of Cetylpyridinium chloride (CPC) remained successful in the oral cavity for up to 180– 300 minutes.
Conclusion: There is an immense need to raise awareness among practitioners, regarding the viricidal activity of commercially available mouthwashes as demonstrated by numerous in-vitro studies and urge health workers to carry out more clinical trials and to get a translational step towards clinical practice.
Keywords: mouthwash, povidone-iodine, PVP-I, SARS-CoV-2, COVID-19, dental procedures
Introduction
Coronaviruses are originated from Coronaviridae family of viruses and were declared as a causative agent for COVID-19 on 8th January 2020 by the Chinese Center for Disease Control (CDC), afflicting humans along with other mammals. In December 2019, the epidemic emerged from Wuhan, city of China and since then it has become a global health challenge. Prodrome stage of the disease includes mild respiratory disease while the symptoms may aggravate to severe acute respiratory distress syndrome (SARS-CoV-2) and organ failure.1 Following the pattern of antecedent respiratory outbreaks in humans which include SARS-Co V and MERS-CoV (Middle East Respiratory Syndrome), SARS-CoV-2 is zoonotic in origin while the Chinese horseshoe bats are believed to be the principal cause.2 Manifested as a pandemic by the World Health Organization (WHO) on 11th March 2020, COVID-19 affected multitudinous individuals across the globe thus altering fundamental dynamics of clinical dentistry.3 Epidemiologic and genetic studies documented that sole animal to human transmission was the starting point of this outbreak, while continuous human to human interaction is now liable for its spread mainly through respiratory droplets, contact transfer, or faecal-oral route.4 Symptomatic patients are predominantly responsible for transmission5 while asymptomatic patients are also known to be potential carriers of the disease thus augmenting the spread of the virus, and making the control even more challenging.6 Studies signified a similar CT scan pattern in asymptomatic COVID-19 patients when compared with symptomatic virus carriers; therefore an incubation period of 14 days is recommended for quarantine of people exposed to patients or virus carriers.7 Researchers have observed that people of all ages are susceptible to COVID-19 while the healthcare staff is at the greatest risk because of their close contact with patients. As yet no specific COVID-19 treatment has been proposed, consequently supportive medications are prescribed.2
A well-documented fact is that the severity of disease caused by the virus depends on its ability to penetrate the cells. Furin, a regulator of numerous proteins is produced by salivary glands and has a property to cleave viral molecules, thus is known to be involved in multiple infectious diseases including coronavirus. The presence of “Furin gene expression” in salivary glands results in rapid activation and consequentially rapid spread of virus through salivary droplets.8 Secondly, the angiotensin-converting enzyme (ACE2) receptors function as a cellular door and primary receptor for the entrance of the virus. Literature signifies that mucosal cells in the epithelium of the oral cavity, especially tongue has a considerable expression for ACE2 receptors thus, resulting in excessive viral load in the oral cavity. The oral cavity fosters a wide array of bacterial and viral strains, it poses a serious threat for cross-infection especially viral in aetiology thus, significantly declining dental practice amidst COVID-19 era.9 Studies imply that respiratory droplets are a major source of cross-infection in a dental environment, usage of the handpiece or ultrasonic devices generate and release secretions known as aerosols and splatters, constituting saliva and blood along with bacteria and viruses (0.2–2.0µm in diameter), which aerosolize into the surrounding environment, these aerosols can travel up-to a distance of 3 feet (1m) and remain suspended in the air for 10 minutes,10,11 thus contaminating dental apparatus causing problems for other patients.12 Secondly, these contaminated aerosols might be inhaled by nearby people, causing respiratory complications among dental staff.12 One of the foremost challenges encountered by a dental practitioner in COVID-19 aeon is reducing cross-infection in a dental setting but still many catastrophes occur predominantly due to professional negligence.3
Oral cavity acts as a pool for SARS-CoV-2 virus. Infected respiratory particles play a primary role in the transmission of SARS-CoV infection thus, effective strategies to disrupt the lipid envelop of these viruses need to be established. Therefore, an approach for pre-procedural decontamination of mouth should be implemented in a dental setting.13 Oral rinses may offer promising results and a prophylactic role as they can exterminate the lipid envelope of the virus. Various in-vitro investigations have demonstrated the viricidal effect of different mouthwashes on enveloped viruses which include ethanol, hydrogen peroxide, chlorhexidine (CHX) and Cetylpyridinium-chloride. Fortified on the guidelines provided by CDC, CHX oral rinses gained widespread recognition to diminish the spread of COVID-19 virus via aerosols, however, literature mentions its poor antiviral properties in contrast to its antibacterial property.14,15 While 0.23% formulation of povidone-iodine (PVP-I) exceptionally inactivated SARS-CoV strains within 15 seconds of exposure,16 However till to date, a gap in literature and clinical trials exists that could determine the antiviral activity of these agents on SARS-CoV-2.
PVP-I dissociates into free iodine, which is capable to destroy the cell membrane of the virus and alter its metabolic pathway thus causing irreversible damage. These virucidal molecules inhibit neuraminidase and hemagglutinin which prevents attachment of the virus receptors with cells thus blocking the release of infectious cells17 hence, making PVP-I an auspicious anti-viral agent.18 Latterly, two protocols were published for utilization of PVP-I amidst COVID-19, the protocol proposed by United-kingdom was bisected and categorized instructions for three groups of individuals; Conscious COVID patients; 9mL of 0.5% formulation, constituting PVP-I was prescribed via the oral route, while 0.3mL solution in the same concentration was directed via nostrils. The second category comprised instructions for front-line medical staff, they were suggested to use these formulations after every 2 to 3 hours. While the third class included unconscious patients, 2mL oral application of PVP-I formulation was advised.19,20 Pittsburgh in their protocol counselled COVID-19 patients, people at risk and front-line medical staff to use 0.4% and 0.5% of PVP-I solution via nasal route and oral rinse, respectively.21 Taking PVP-I toxicity into consideration, Frank et al after a literature search indicated that a concentration of 2.5% via the oral route is safe and can routinely be used as a decontaminating agent. However, its use is contraindicated in patients with thyroid problems, patients undergoing radiotherapy and pregnant women. A minute amount of PVP-I is strictly advised for unconscious patients as there are chances for aspiration pneumonia.16
Other valuable ingredients in mouthwashes include Citrox, cyclodextrin and hydrogen peroxide. Citrox’s oxidizing ability exposes and damages the viral lipid envelop, therefore significantly reducing the viral load14,18 while Cyclodextrin has a potential to attract and subsequently inactivate the outer shell of virus thus destroying it completely.14 Hydrogen peroxide disrupts the lipid membrane of virus by its oxygen-free radicles.18,22
Decontaminating role and antiviral properties of various agents in mouthwashes has gained considerable attention during the pandemic. Recently a randomized controlled trial has been planned at Aga Khan University (Pakistan) to determine and compare efficacy of PVP-I, hydrogen-peroxide, hypertonic saline and neem extract in reducing viral load in COVID-19 patients.23 While another clinical trial in France is evaluating antiviral properties of the active ingredients in mouthwashes such as cyclodextrin and Citrox.24 Fortified on the remarkable effects of PI and CPC mouthwashes to reduce SARS CoV-2 levels in saliva of COVID-19 patients in a randomized clinical trial in Singapore by Seneviratne et al, which is the only clinical trial in our knowledge;25 the main emphasis of our survey is on the use of PVP-I mouthwashes as its available in the market under the commercial name Betadine.
The prospect of this study is to assess knowledge, attitude, and practice (KAP survey) regarding the use of mouthwashes amidst COVID-19 pandemic among dental practitioners and emphasize on pre-procedural utilization of mouthwashes.
Materials and Methods
A cross-sectional, descriptive analysis was carried out during 1st to 15th July 2020 among working dental practitioners across the globe. Anonymity was maintained, the purpose of the study was explained to them in detail and the questionnaire was filled with their consent. The Ethical approval for this survey was obtained from the Institutional Review Committee of Islamic International Dental College (Ref. no. IIDC/IRC/2020/07/004). The questionnaire was formed after scrutinizing literature, pertinent to COVID-19, SARS, mouthwashes, and international guidelines and was in English language. The layout for the survey was designed on Kwiksurveys.com and the link was circulated on social media accounts like LinkedIn, WhatsApp, Facebook, Twitter, and E-mails to reach the target population which included general practitioners and consultants. The questionnaire comprised of demographic details in the first section which included sex, health sector and designation. The second section included knowledge part comprising eight questions, responded by the participants with either a yes or no answer. The third section ascertained attitude and practice, associated with five questions, four were designed on a 5-point Likert scale, while the fifth was an open-ended question.
A sample size of 747 was estimated using statistics of knowledge regarding the recommendation of 0.23% to 7% Povidone-Iodine (PVP I) as a pre-procedural rinse to reduce viral load ie, 22.6% absolute precision as 3% and 95% confidence level.26 For the validation of questionnaire, a pilot study of 15 samples was conducted. Cronbach’s alpha value was estimated by taking questions related to knowledge, attitude and practice together. Overall Cronbach’s alpha value was estimated as 70.3%. Data were analyzed using SPSS version 23. Frequency and percentage were estimated for all categorical variables. Fisher exact test was applied to assess the statistical difference between various oral rinses, prescribed by different countries. p≤0.05 was taken as statistically significant.
Results
After inflating the sample size by 10% for non-respondent, a total of 822 dental practitioners were approached which included undergraduate/post-graduate residents, general practitioners and consultants, wherein only 707 participants responded (86%) and 570 completed the survey (80.6%). Most of the participants were from India (19.8%), followed by Pakistan (18.6%), USA (17.4%), Saudi Arab (16.3%), United Kingdom (13.3%), Canada (7.5%) and Australia (5.6%) respectively. About 1.4% participants were from other countries such as Germany, Hungary, Nigeria, China, and South Africa. (Figure 1)
 |
Figure 1 Country-wise distribution of respondents. |
Of the 570 participants, 329 (57.7%) were females and 241 (42.3%) were males. About 205 participants were working at the public institute (36%) and 365 participants were working at a private institute (64%). One hundred and ninety-one participants were specialist/consultant (33.5%) and 379 were general dental practitioners (66.5%).
Approximately 88.2% of participants knew that SARS-CoV-2 transmission was mainly through respiratory droplets and aerosols during a dental procedure, 78.9% knew that aerosols produced during dental treatment could reach alveoli in the lungs that caused respiratory problems, while 72.5% knew that aerosols remained in the air for at least 10 minutes after the dental procedure and have the capacity to spread to a distance of at least 2 feet from the dental chair. Only 32.6% of participants recognized that mouthwashes give positive outcomes against coronavirus transmission because they can kill the virus’ lipid membrane, thereby being successful against all mutated COVID-19 strains, and 53.3% participants were considerate that pre-procedural usage of mouthwashes would efficiently minimize the bacterial and viral load in dental aerosols. Only 38.9% of the participants knew that Povidone-iodine (PVP-I) mouthwashes are more efficient in reducing coronaviruses than mouthwashes made of chlorhexidine (CHX). Of the 570 participants, 33.9% knew that 0.23% of PVP-I had substantial virucidal activity against SARS-COV, MERS-CoV, influenza virus and rotavirus while 31.1% recognized that oral rinses of Cetylpyridinium chloride (CPC) remained successful in the oral cavity for up to 180–300 minutes (Table 1).
 |
Table 1 Assessment of Knowledge |
Out of 570 participants, 71% strongly agreed that dental practitioners are exposed to the highest risk of getting infected by coronavirus not only due to their close contact with infected patients but also due to inhalation of aerosols (Figure 2).
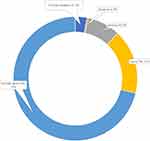 |
Figure 2 Dental practitioners are at the highest risk of getting coronavirus (attitude assessment). |
About 12% strongly agreed, while 22% agreed that coronavirus can be effectively controlled by pre-procedural use of mouthwashes in a dental setting whereas 13% strongly disagreed with the statement and 34% responded neutral (Figure 3).
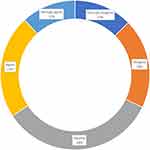 |
Figure 3 Coronavirus can successfully be controlled by pre-procedural use of mouth-washes (attitude assessment). |
About 12.3% of participants strongly agreed that they educate their patient about COVID-19 disease and its transmission, whereas 14.6% strongly agreed that they use mouthwashes prior to dental procedures amidst COVID pandemic (Figure 4).
 |
Figure 4 Practices of dental practitioners (practice assessment). |
Most of the dental practitioners recommended chlorhexidine (67%), followed by Povidone-Iodine (12%) normally prior to any dental procedures (Figure 5).
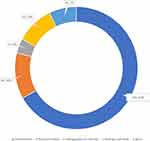 |
Figure 5 Normally recommended mouthwash prior to dental procedure (practice assessment). |
In Saudi Arab, about 69.9% recommend chlorhexidine while 10.8% recommend povidone-iodine prior to dental procedures. Whereas in Pakistan, 67% of dental practitioners normally recommend chlorhexidine and 12.3% recommend povidone-iodine prior to dental procedures. In United Kingdom, about 69.7% recommend chlorhexidine while only 15.2% recommend povidone-iodine and 8.1% recommended hydrogen peroxide prior to dental procedures. A statistically significant difference in proportions of recommended mouthwash was observed across different countries (p≤0.05) (Table 2).
 |
Table 2 Country-Wise Comparison of Normally Recommended Mouthwash Prior to Dental Procedures (* Significant at p-value ≤0.05) |
General dental practitioners had higher awareness regarding pre-procedural use of mouthwashes (p=0.031) and 0.23% PVP-I efficacy against virus (p=0.001) than specialist/consultants. About 77.1% of the general dental practitioners agreed that they are exposed to highest risk of getting infected by coronavirus not only due to their close contact with infected patients but also due to inhalation of aerosols and 65% of the general dental practitioners strongly agreed with the same statement. Almost 57% of the general dental practitioners strongly agreed that they educate their parents about COVID-19 disease and its transmission whereas 43% of the consultants strongly agreed that they educate their parents about COVID-19 disease and its transmission (p=0.001) see the Table 3.
 |
Table 3 Comparison Between the Level of Qualification of the Dental Healthcare Workers and Their Knowledge Attitude and Practices (*Significant at p-value ≤0.05) |
Discussion
Presently COVID 19 is a debatable subject among healthcare workers and the public. The prospect of the analysis was to evaluate awareness level regarding mouthwashes among dental practitioners since the future is going to witness significant crises and collapse of the healthcare system if proper precautionary measures are not adopted. A flourishing body of research has been developed recently addressing infection control protocols but the findings of our analysis were documented on an unprecedented scale since the significance of mouthwashes remained a neglected area for aerosol reduction during COVID crises. The outcomes are valuable to organize “infection control” training programs accordingly. Secondly, it would also welcome researchers for further clinical trials.
A requisite understanding of the disease and its transmission is essential to depreciate the spread of infection. A creditable response was achieved as 83.3% of participants correctly identified that respiratory droplets and aerosols generated during a procedure are the preeminent parameters accountable for the transmission of SARS-CoV-2, these findings contradict the findings of Quadri et al, in his survey less than 50% of healthcare workers in Saudi-Arab correctly identified that COVID-19 is an airborne disease.11 In our survey 71.2% clinicians believed with certitude that the aerosols linger for 10 minutes, following a dental procedure. 71.2% of practitioners were acquainted with the fact that respiratory problems initiate once the causative factors (aerosols) reach the lungs. The findings of our study were in accordance with a previous KAP analysis by Ahmed et al, who documented 97% awareness level among dental practitioners regarding the mode of SARS-CoV-2 transmission.34 To construct a paradigm for infection control protocols, such information holds immense importance in a dental setting over the course of a pandemic. Protocols emphasizing on aerosol generation and reduction should be implemented.
The level of awareness regarding the potential decontaminating role of mouthwashes (by disrupting the membrane of the virus) is alarming as only 31.5% participants endorsed with the statement while 52.3% respondents acknowledged the viricidal activity of mouthwashes. A global survey by Tariq et al reported that 27.1% dental practitioners accord with the fact that mouthwashes could reduce the risk of COVID-19 infection,35 however, majority of healthcare workers reported for using mouthwashes prior to dental services in a study by Mouhat et al.36 The key challenge to control COVID-19 infection is to reduce the number of virus particles in the oral cavity since infected respiratory particles play a primary role in the transmission of SARS-CoV-2 infection, effective strategies to disrupt the lipid envelop of these viruses need to be established. Oral rinses exhibit a prophylactic role in decontamination of mouth and subsequently destroying the membrane.37 Established on previous literature, a strong interrelationship has been developed between the diminished risk of respiratory infections, viral in aetiology and good oral hygiene. Systemic reviews concluded a significant improvement in ICU patients suffering from ventilator linked pneumonia when their oral health was improved. Oral cavity and lungs harbour numerous pathogenic bacterial species which are acknowledged for their role in carrying homeostasis and conserving tissues. There is constant immigration of these microbes between the lungs and oral cavity. An infection drastically disturbs the equilibrium, allowing overgrowth of these microbes hence injuring the lungs as inhalation of aerosols and splatters pollutes the epithelium of lower airways. Pneumonia and sepsis may develop when respiratory tract aspirates, biofilm bacteria from the oral cavity. Complications of systemic diseases such as autoimmune or cardiovascular diseases aggravate when the bacteria and viral load in the mouth are high thus emphasizing on the need to reduce viral load in the mouth.38
Subsequently, another confounding finding of our study for evaluating the level of awareness about the different mouthwashes revealed that only 38.3% respondents accord with the fact that PVP-I has supercilious antiviral properties in contrast to CHX mouthwashes while only 31.1% dental practitioners were cognizant of the viricidal activity of PVP-I against antecedent viral pandemics, for instance, SARS-CoV, MERS-CoV, influenza and rotavirus. Whereas, for CPC oral rinses, only 27.2% of participants were aware of its effectiveness. Numerous experiments on oral rinses were conducted and it was concluded that pre-procedural utilization of rinses consisting of chlorohexidine and cetyl pyridinium chloride substantially reduced bacterial count in aerosols thus reduced any potential risks of cross-infection. Clinicians have a wide assortment of mouthwashes with different active ingredients such as chlorhexidine, povidone-iodine, hydrogen peroxide and cetyl-pyridinium chloride, each owing divergent viricidal and anti-bacterial activity, however, there is a dearth of knowledge in the literature regarding the awareness for the use of mouthwashes.37 In a recent literature search by Mosavi et al, it was concluded that CHX oral rinses are the finest antibacterial agents used world-wide.39 However, in terms of antiviral properties, PVP-I can progress in being a potential gold standard in future especially to eradicate COVID-19 virus.40,41 On April 2020, the American Dental Association (ADA) and the Centers for Disease Control and Prevention (CDC) proposed pre-procedural utilization of 1.5% hydrogen peroxide and 0.2% povidone-iodine mouthwashes. Inactivation of SARS-CoV within 15 seconds of contact with 0.23% PVP-1 solutions are noteworthy in-vitro findings available in the literature.3
Harrel and Molinari documented three protective coatings to avoid the spread of these airborne virus particles since neither any sole chemical agent nor any specific procedure is available. Therefore, the quest for protective barriers has been intensified that could eliminate the novel coronavirus. The three protective coatings incorporate the use of personal protective equipment (PPE) followed by oral mouthwashes and the use of high-speed suction. PPE along with high-speed suction gained notable recognition while the use of oral rinses was regrettably neglected.3 Therefore, the framework of the second part of the questionnaire was designed to determine the attitude and practices for the use of mouthwashes amidst COVID-19 pandemic. Majority of practitioners used (CHX) mouthwashes prior to dental procedures. These results were different with the findings of Ahmed et al,34 and Lodhi et al,42 who reported that a majority of dental practitioners are ignoring the use of oral mouthwashes in their dental practice. However, the use of (CHX) mouthwashes in-spite of their low virucidal activity, necessitates prompt attention by public health authorities to emphasize clinicians and the general population for the benefits of pre-procedural use of PVP-I, CPC or hydrogen peroxide mouthwashes during the course of this pandemic.
In the event of a pandemic, ascertaining strategies to prevent or reduce infection serves well in public interest in contrast to identifying the disease and evaluating its response.43 Individuals can better defend themselves and would seek immediate medical care in case of emergencies if their level of awareness is raised regarding the disease.44 An appalling practitioner’s attitude was revealed in our study since only 14.9% strongly agreed about educating their patient about COVID-19 disease and its transmission. An investigation on the MERS outbreak unveiled that lack of knowledge was one of the aggravating factors for the disease spread.45 An overwhelming response to the utilization of chlorohexidine was exhibited. Presently there are no clinical trials on the effectiveness of PVP-I against SARS-CoV-2 virus, this might be a possible explanation of this response.
Data collection for this KAP analysis was done over a short period as the authors intend to raise awareness among practitioners and hope for immediate attention by researchers so that future clinical studies could be conducted that would serve well in the interest of the community. Secondly, data could not be generalized since few countries (Europe) responded while others gave limited responses due to language barriers and limitation of resources resulting in small sample size. Since most responses collected were from Pakistan and Saudi Arabia, the findings of our study cannot be globalized. For future research, clinical trials and descriptive studies emphasizing on the anti-viral properties of different active ingredients in the mouthwashes such as Citrox®, Cyclodextrin and essential oils are highly recommended since they demonstrate a reduction in salivary viral load.14
Conclusions
An intricate and perplexing situation has been created world-wide and various effective treatment strategies are proposed to reduce viral load while a perceptible ignorant attitude of clinicians is observed for the pre-procedural utilization of mouth-rinses. Mouthwashes especially PVP-I is a potential anti-viral agent which can significantly reduce viral load in saliva and subsequently in aerosols thus limiting the spread COVID-19 infection. However, there is a dearth of literature, consequentially lack of knowledge among dental practitioners regarding the significance of using oral mouthwashes. Therefore, there is an immense need for further clinical trials with an aspiration to achieve a translational step towards clinical practice.
Acknowledgments
We acknowledged the College of Dentistry of King Faisal University and King Abdul Aziz University, Saudi Arabia.
Author Contributions
All authors mutually contributed to the conception, study design, execution, acquisition of data, analysis and interpretation. All authors contributed to the drafting or writing of the article, or substantially revised or critically reviewed the article. All authors have agreed on the journal to which the article will be submitted and have reviewed and agreed on all versions of the article before submission, during revision, the final version accepted for publication, and any significant changes introduced at the proofing stage. All authors agreed to take responsibility and be accountable for the contents of the article.
Funding
There is no funding to report.
Disclosure
The authors declare no conflicts of interest for this work.
References
1. Mubeen SM, Kamal S, Kamal S, Balkhi F. Knowledge and awareness regarding spread and prevention of COVID-19 among the young adults of Karachi. J Pak Med Assoc. 2020;70(5):S169–S74. doi:10.5455/JPMA.40
2. Meng L, Hua F, Bian Z. Coronavirus disease 2019 (COVID-19): emerging and future challenges for dental and oral medicine. J Dent Res. 2020;99(5):481–487. doi:10.1177/0022034520914246
3. Bidra AS, Pelletier JS, Westover JB, et al. Rapid in‐vitro inactivation of severe acute respiratory syndrome Coronavirus 2 (SARS‐CoV‐2) using Povidone‐Iodine oral antiseptic rinse. J Prosthodontics. 2020.
4. Del Rio C, Malani PN. 2019 novel coronavirus—important information for clinicians. JAMA. 2020;323(11):1039–1040. doi:10.1001/jama.2020.1490
5. Chan JF-W, Yuan S, Kok K-H, et al. A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: a study of a family cluster. Lancet. 2020;395(10223):514–523. doi:10.1016/S0140-6736(20)30154-9
6. Rothe C, Schunk M, Sothmann P, et al. Transmission of 2019-nCoV infection from an asymptomatic contact in Germany. New England J Med. 2020;382(10):970–971. doi:10.1056/NEJMc2001468
7. Backer JA, Klinkenberg D, Wallinga J. Incubation period of 2019 novel coronavirus (2019-nCoV) infections among travellers from Wuhan, China, 20–28 January 2020. Eurosurveillance. 2020;25(5):2000062. doi:10.2807/1560-7917.ES.2020.25.5.2000062
8. Zupin L, Pascolo L, Crovella S. Is FURIN gene expression in salivary glands related to SARS-CoV-2 infectivity through saliva? J Clin Pathol. 2020. doi:10.1136/jclinpath-2020-206788
9. Herrera D, Serrano J, Roldán S, Sanz M. Is the oral cavity relevant in SARS-CoV-2 pandemic? Clin Oral Investig. 2020;1–6.
10. Organization WH. Rolling updates on coronavirus disease (COVID-19); 2020. Available from: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/events-as-they-happen.
11. Quadri MF, Jafer MA, Alqahtani AS, et al. Novel corona virus disease (COVID-19) awareness among the dental interns, dental auxiliaries and dental specialists in Saudi Arabia: a nationwide study. J Infect Public Health. 2020;13:856–864. doi:10.1016/j.jiph.2020.05.010
12. Harrel SK, Molinari J. Aerosols and splatter in dentistry: a brief review of the literature and infection control implications. J Am Dental Association. 2004;135(4):429–437. doi:10.14219/jada.archive.2004.0207
13. Khurshid Z, Asiri FYI, Al Wadaani H. Human saliva: non-invasive fluid for detecting novel coronavirus (2019-nCoV). Int J Environ Res Public Health. 2020;17(7):2225. doi:10.3390/ijerph17072225
14. Carrouel F, Conte MP, Fisher J, et al. COVID-19: a recommendation to examine the effect of mouthrinses with β-cyclodextrin combined with citrox in preventing infection and progression. J Clin Med. 2020;9(4):1126. doi:10.3390/jcm9041126
15. Peng X, Xu X, Li Y, et al. Transmission routes of 2019-nCoV and controls in dental practice. Int J Oral Sci. 2020;12(1):1–6. doi:10.1038/s41368-020-0075-9
16. Frank S, Capriotti J, Brown SM, Tessema B. Povidone-Iodine use in sinonasal and oral cavities: a review of safety in the COVID-19 Era. Ear Nose Throat J. 2020;0145561320932318.
17. Sriwilaijaroen N, Wilairat P, Hiramatsu H, et al. Mechanisms of the action of povidone-iodine against human and avian influenza A viruses: its effects on hemagglutination and sialidase activities. Virol J. 2009;6(1):124. doi:10.1186/1743-422X-6-124
18. Carrouel F, Gonçalves L, Conte M. et al. <? Covid19?> Antiviral activity of reagents in mouth rinses against SARS-CoV-2. J Dent Res;2020. 0022034520967933. doi: 10.1177/0022034520967933
19. Kirk-Bayley J, Challacombe S, Sunkaraneni S, Combes J. The use of povidone iodine nasal spray and mouthwash during the current COVID-19 Pandemic may protect healthcare workers and reduce cross infection. SSRN Electronic J. 2020. doi:10.2139/ssrn.3563092
20. Imran E, Khurshid Z, Al Qadhi AAM, Al-Quraini AAA, Tariq K. Preprocedural use of povidone-iodine mouthwash during dental procedures in the COVID-19 Pandemic. Eur J Dent. 2020.
21. Mady LJ, Kubik MW, Baddour K, Snyderman CH, Rowan NR. Consideration of povidone-iodine as a public health intervention for COVID-19: utilization as “Personal Protective Equipment” for frontline providers exposed in high-risk head and neck and skull base oncology care. Oral Oncol. 2020;105:104724. doi:10.1016/j.oraloncology.2020.104724
22. O’Donnell VB, Thomas D, Stanton R, et al. Potential role of oral rinses targeting the viral lipid envelope in SARS-CoV-2 infection. Function. 2020;1(1):zqaa002. doi:10.1093/function/zqaa002
23. Khan FR, Kazmi SMR, Iqbal NT, et al. A quadruple blind, randomised controlled trial of gargling agents in reducing intraoral viral load among hospitalised COVID-19 patients: a structured summary of a study protocol for a randomised controlled trial. Trials. 2020;21(1):1–4. doi:10.1186/s13063-020-04634-2
24. Carrouel F, Viennot S, Valette M, et al. Salivary and nasal detection of the SARS-CoV-2 Virus after antiviral mouthrinses (BBCovid): a structured summary of a study protocol for a randomised controlled trial. Trials. 2020;21(1):1–3. doi:10.1186/s13063-020-04846-6
25. Seneviratne CJ, Balan P, Ko KKK, et al. Efficacy of commercial mouth-rinses on SARS-CoV-2 viral load in saliva: randomized control trial in Singapore. Infection. 2020;1–7.
26. Sarfaraz S, Shabbir J, Mudasser MA, et al. Knowledge and attitude of dental practitioners related to disinfection during the COVID-19 Pandemic. Paper presented at: Healthcare, 2020.
27. Ather A, Patel B, Ruparel NB, Diogenes A, Hargreaves KM. Coronavirus disease 19 (COVID-19): implications for clinical dental care. J Endod. 2020.
28. Saini R. Efficacy of preprocedural mouth rinse containing chlorine dioxide in reduction of viable bacterial count in dental aerosols during ultrasonic scaling: a double-blind, placebo-controlled clinical trial. Dent Hypotheses. 2015;6(2):65. doi:10.4103/2155-8213.158479
29. Anderson EL, Turnham P, Griffin JR, Clarke CC. Consideration of the aerosol transmission for COVID‐19 and public health. Risk Analysis. 2020;40:902–907. doi:10.1111/risa.13500
30. Mohan M, Jagannathan N. The efficacy of pre-procedural mouth rinse on bacterial count in dental aerosol following oral prophylaxis. Dental Med Problems. 2016;53(1):78–82. doi:10.17219/dmp/60694
31. Farzan A, Firoozi P. Common Mouthwashes for Pre-procedural Rinsing in Dental Practice: Wich One is Appropriate for Eliminating Coronaviruses? A Mini Literature Review. Journal of “Regeneration, Reconstruction & Restoration“ (Triple R). 2020;5:e2. doi:10.22037/rrr.v5i1.29543
32. Roberts W, Addy M. Comparison of the in vivo and in vitro antibacterial properties of antiseptic mouthrinses containing chlorhexidine, alexidine, cetyl pyridinium chloride and hexetidine: relevance to mode of action. J Clin Periodontol. 1981;8(4):295–310. doi:10.1111/j.1600-051X.1981.tb02040.x
33. Elworthy A, Greenman J, Doherty F, Newcombe R, Addy M. The substantivity of a number of oral hygiene products determined by the duration of effects on salivary bacteria. J Periodontol. 1996;67(6):572–576. doi:10.1902/jop.1996.67.6.572
34. Ahmed MA, Jouhar R, Ahmed N, et al. Fear and practice modifications among dentists to combat Novel Coronavirus Disease (COVID-19) outbreak. Int J Environ Res Public Health. 2020;17(8):2821. doi:10.3390/ijerph17082821
35. Hamid H, Tariq R, Mashood S, et al. Common misconceptions regarding COVID-19 among health care professionals: an online global cross-sectional survey. J Oral Res. 2020;2020. doi: 10.34133/2020/9426453
36. Stangvaltaite-Mouhat L, Uhlen -M-M, Skudutyte-Rysstad R, et al. Dental health services response to COVID-19 in Norway. Int J Environ Res Public Health. 2020;17(16):5843. doi:10.3390/ijerph17165843
37. Kelly N, Íomhair AN, McKenna G. Can oral rinses play a role in preventing transmission of Covid 19 infection? Evid Based Dent. 2020;21(2):42–43. doi:10.1038/s41432-020-0099-1
38. Sampson V, Kamona N, Sampson A. Could there be a link between oral hygiene and the severity of SARS-CoV-2 infections? Br Dent J. 2020;228(12):971–975. doi:10.1038/s41415-020-1747-8
39. Haydari M, Bardakci AG, Koldsland OC, et al. Comparing the effect of 0.06% -, 0.12% and 0.2% Chlorhexidine on plaque, bleeding and side effects in an experimental gingivitis model: a parallel group, double masked randomized clinical trial. BMC Oral Health. 2017;17(1):118. doi:10.1186/s12903-017-0400-7
40. Kariwa H, Fujii N, Takashima I. Inactivation of SARS coronavirus by means of povidone-iodine, physical conditions and chemical reagents. Dermatology. 2006;212(Suppl. 1):119–123. doi:10.1159/000089211
41. Khan MM, Parab SR, Paranjape M. Repurposing 0.5% povidone iodine solution in otorhinolaryngology practice in Covid 19 pandemic. Am J Otolaryngol. 2020;41:102618. doi:10.1016/j.amjoto.2020.102618
42. Lodhi SK, Ehsan S, Rafique A, Tahir S, Khan T. Knowledge and practices of dentists regarding personal protective equipment during COVID-19 Pandemic: a cross-sectional study among pakistani dentists. Biomedica. 2020;36(COVID19-S2):275–280.
43. Heymann DL, Dar OA. Prevention is better than cure for emerging infectious diseases. BMJ. 2014;348:g1499. doi:10.1136/bmj.g1499
44. Lu S, Tian B, Kang X, et al. Public awareness of tuberculosis in China: a national survey of 69 253 subjects. Int j Tuberculosis Lung Disease. 2009;13(12):1493–1499.
45. Willman M, Kobasa D, Kindrachuk J. A comparative analysis of factors influencing two outbreaks of Middle Eastern respiratory syndrome (MERS) in Saudi Arabia and South Korea. Viruses. 2019;11(12):1119. doi:10.3390/v11121119
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
