Back to Archived Journals » Comparative Effectiveness Research » Volume 4
Current issues in comparative effectiveness research for hepatitis C
Authors Yoo M, Cheng Y, Rhien T, Biskupiak J
Received 20 November 2013
Accepted for publication 29 January 2014
Published 26 June 2014 Volume 2014:4 Pages 13—27
DOI https://doi.org/10.2147/CER.S40939
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Minkyoung Yoo, Yan Cheng, Taylor J Rhien, Joseph Biskupiak
Pharmacotherapy Outcomes Research Center, University of Utah College of Pharmacy, Salt Lake City, UT, USA
Background: With increasing treatment options for hepatitis C, evidence of comparative effectiveness of these treatment options is required to improve treatment outcomes. The purpose of this study was to evaluate the most recent comparative effectiveness research and suggest future directions for hepatitis C research.
Methods: We identified and evaluated the literature on comparative effectiveness research and conducted a literature search for additional studies since the most current review. A review of ongoing clinical trials in hepatitis C was performed to assess how forthcoming research is addressing the research gaps and limitations.
Results: Since a comprehensive comparative effectiveness research review by Chou et al new studies have been published, which were mostly consistent with the consensus in the literature. A few of them added to comparative effectiveness research knowledge by addressing issues of the likelihood of sustained virologic response in an older cohort, the effect of genomics and individualizing treatment duration, or the effect of delayed treatment. Research gaps and limitations of the existing comparative effectiveness research and future study needs were well identified in the second study from Chou et al. Some of the gaps and limitations were filled by additional research over the past year, though many of them still remained unanswered.
Conclusion: To have complete information on the effectiveness of alternative treatments for hepatitis C virus, further research is needed on results in the general population, the effectiveness of treatment methods such as noninvasive treatment and individualized treatment, and the long-term effects of triple therapies. Additionally, evidence from a real-world setting is lacking. Methodologically thorough and independently funded retrospective research will help to generalize the effectiveness of current therapies for hepatitis C virus.
Keywords: PEG-interferon, ribavirin, telaprevir, boceprevir
Background
Hepatitis C virus (HCV) infection is among the most frequent of liver diseases. Infection is estimated in 4.4 million individuals in the US and Canada and 184 million worldwide.1 It is the most common cause of cirrhosis and primary liver cancer in the world,2 and results in over 15,000 US deaths per year.3 Within HCV, multiple genotypes exist. Genotype 1 is the most common in the US, comprising 74% of cases, and is also the most difficult to treat.4
The goal of HCV therapy is to achieve sustained virologic response (SVR), defined as the inability to detect HCV ribonucleic acid (RNA) 6 months after treatment. Patients who achieve SVR show decreased rates of mortality, liver cancer, and need for transplant.5 To attain SVR, treatment employs the use of antiviral combinations, and in the early 2000s dual therapy with polyethylene glycol (PEG)ylated-interferon (PEG-IFN) and ribavirin (RBV) was standardized as the treatment of choice. This regimen proved more successful in genotypes 2 and 3 (nearly 80% SVR) compared to genotype 1, where SVR was reached in only about 40% of patients.6
The past 2 decades have brought increased knowledge of the HCV, including the production of a viral culture in 2005.7 This knowledge is being used to develop direct-acting antivirals which specifically target HCV replication factors, such as NS3/4A serine protease. These drugs have the potential to shorten treatment, decrease adverse effects, and improve SVR.7 As of January 2013, over 30 new agents were being tested, and two HCV NS3/4A protease inhibitors, telaprevir and boceprevir, were approved by the US Food and Drug Administration (FDA) in 2011.8 Guidelines for genotype 1 treatment have changed from 48 weeks of dual PEG-IFN + RBV therapy to 24–48 weeks of triple PEG-IFN + RBV + boceprevir/telaprevir therapy,9 and genotype 1 SVR rates have increased from approximately 40% to 70%.10,11 Treatment options progressed further in November and December 2013 with the approval of simeprevir and sofosbuvir. While this will significantly affect HCV treatment, these approvals and their supporting evidence were published after our literature review took place. Recommended treatment duration is 24 weeks for genotypes 2 and 3 and 48 weeks for genotype 1.
Despite these advances, HCV management has room for improvement. IFN-based therapy is associated with significant adverse effects, including fatigue, pain, influenza-like symptoms, neuropsychiatric effects, and others.12 Consequently, many patients are unable or unwilling to proceed with treatment. Among those that do, SVR is not always achieved.
With increasing treatment options, ever-improving SVR rates, and yet significant remaining adverse effects, evidence of comparative effectiveness of these treatment options is required to achieve better outcomes. Patients and health care providers can then use this information to inform therapy decisions. This review identified and evaluated recently published and ongoing comparative effectiveness research (CER) pertaining to HCV. The review also identified current limitations in HCV outcomes research and assessed the identified studies against these limitations.
Methods
There has been one comprehensive review on the comparative effectiveness research for HCV by Chou et al published in 2013.13 Our study identified the most recent publication on the effectiveness of HCV therapies beyond this study, and also provided a careful review of the study.13 Additionally, the search for ongoing clinical trials for HCV treatment was performed.
The search strategy for this review was consistent with Chou et al,13 and details are summarized in Table S1. Studies for HCV treatments were identified in National Center for Biotechnology Information (NCBI) Ovid Medline, The Cochrane Library, Scopus (includes EMBASE records from 1996 to present), PsycINFO, and clinical trial registries during the period of September 2012 to August 2013. These inclusion/exclusion criteria were used: 1) English-language articles and conference abstracts were included; 2) studies with pregnant women, patients who received a transplant, HIV-infected patients, patients undergoing hemodialysis, and previously treated patients were excluded; and 3) studies utilizing regimens with antiviral drugs not approved by the FDA for HCV infection were excluded.1
The flow chart of the literature search is shown in Figure 1. We initially identified 249, ten, 619, 22, and two studies from NCBI Medline, The Cochrane Library, Scopus, PsycINFO, and clinical trial registries, respectively. After a manual review, 118 duplicates and 650 studies irrelevant to our objectives were excluded, and 134 studies remained for further screening. Applying the inclusion and exclusion criteria, a total of 19 studies (four randomized control trials [RCTs],14–17 ten meta-analysis studies,18–27 three nonrandomized intervention studies,28–30 one cross-sectional study,31 and one cohort study32) were included for the updated studies for this review (Table 1). Additionally, eight RCTs were also included in this review that were published during the study period of Chou et al13 and met the inclusion/exclusion criteria, but were not reviewed by them.
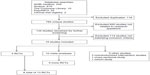  | Figure 1 Flow chart for updated literature search. |
Another study by Chou et al discussed research gaps and limitations and future research needs.33 This review adds to the work of Chou et al13 by updating the list of clinical trials that may address HCV research limitations. Ongoing comparative studies regarding HCV were searched by one reviewer at ClinicalTrials.gov through August 2013. The search strategy mirrored that of Chou et al33 and began with all trials listed under the topic “hepatitis C”. After limiting the results to Phase III studies in adults ≥18 years, 244 studies remained. Thirty-eight studies were excluded due to termination, withdrawal, or unknown status; full-text summaries of the remaining 206 studies were reviewed. Study participants were assessed for being treatment naïve, chronic HCV infected, and without coinfections such as human immunodeficiency virus (HIV) or hepatitis B virus (HBV). Studies with completion dates prior to the literature review in Chou et al13 (August 2012) were also excluded. Beyond the 46 studies addressed in Chou et al,13 22 additional ongoing studies were identified (Figure 2).
  | Figure 2 Flow chart for updated ongoing clinical trial search. |
Every investigator was actively involved in all phases of the study. Two investigators conducted the literature search for the published literatures and ongoing trials, and the remaining investigators were involved in the discussion of the uncertain cases.
Literature on CER for hepatitis C infection
A comprehensive comparative effectiveness research review for hepatitis C was conducted in a recent publication by Chou et al13 funded by the Agency for Healthcare Research and Quality (AHRQ). It systematically compared effectiveness and safety of antiviral treatments for HCV infection in treatment-naïve adults by answering four key questions: Q1) comparative effectiveness of antiviral treatments on health outcomes; Q2) SVR by patient characteristics; Q3) comparative harms of antiviral treatments by patient characteristics; and Q4) improvements in SVR on reducing adverse health outcomes from HCV infection.
Studies published from 1947 to August 2012 from several literature databases, including Ovid Medline, The Cochrane Library, Embase, Scopus, PsycINFO, and clinical trial registries were included in this study. For key questions Q1 through Q3, it included RCTs of dual therapies (PEG-IFN alfa-2b plus RBV, PEG-IFN alfa-2a plus RBV), triple therapies (PEG-IFN, RBV, and either telaprevir or boceprevir), and different doses or duration of HCV therapies. For key question Q4, it included cohort studies comparing an SVR with no SVR after treatment on clinical outcomes (including mortality, cirrhosis, hepatic decompensation, hepatocellular carcinoma, and need for transplantation) and harms (including withdrawals due to adverse events, serious adverse events, neutropenia, anemia, psychological adverse events, influenza-like symptoms, and rash).
The study13 identified 90 studies for the systematic review (Figure 3). No study evaluated the comparative effectiveness of antiviral treatments on long-term clinical outcomes (ie, mortality, complications of chronic HCV infection, or quality of life). For dual-therapy regimens, it found that: 1) dual therapy with PEG-IFN alfa-2b plus RBV was slightly less likely to achieve SVR compared to dual therapy with PEG-IFN alfa-2a plus RBV 2) in patients with genotype 2 or 3 infection, dual therapy for 12 to 16 weeks was less likely to achieve SVR than dual therapy for 24 weeks, while there was no difference between 24 weeks and longer duration of therapy; 3) standard doses of PEG-IFN alfa-2b were more effective than lower doses, while no differential effects of RBV dosing were seen in the published RCTs; 4) lower response rates were observed in older patients, minorities (blacks), patients with high viral load, patients with liver-related complications, or genotype 1-infected patients; and 5) the tolerability did not significantly vary among different dual therapy treatments, while PEG-IFN alfa-2b had a slightly improved safety profile due to its lower risk of serious adverse events.
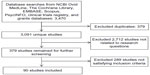  | Figure 3 Flow chart for literature search in the review by Chou et al.13 |
For triple-therapy regimens, it found that: 1) triple-therapy regimens were associated with an increased likelihood of achieving an SVR than dual therapy; 2) triple-therapy regimens had similar effects on achieving an SVR as dual therapy in patient groups with different demographic characteristics; 3) triple therapy was associated with increased risk of adverse events, such as hematological adverse events for boceprevir and anemia and rash for telaprevir; and 4) flu-like symptoms were the most commonly reported adverse events of all antiviral regimens. As for SVR after antiviral therapy and clinical outcomes, they reported that 19 published studies indicated that patients with an SVR after the antiviral therapy had a lower risk of all-cause mortality, liver-related mortality, and other hepatic complications than patients with no SVR; however, more than half of the studies were conducted in Asian countries.
Updated literature and findings (September 2012–August 2013)
A total of 29 publications were identified by the search criteria described earlier and carefully reviewed; they are summarized in this section. The types of studies reviewed include RCTs, nonrandomized intervention studies, observational studies, and meta-analyses studies.
RCTs
Dual therapy
An open-label Phase III noninferiority trial among German adults with acute HCV discovered that symptomatic patients treated with delayed treatment with PEG-IFN alfa-2b plus RBV (for 24 weeks) starting at 12 weeks had a slightly but not statistically significantly lower SVR (67% versus [vs] 54%, P=0.071) compared to those treated with immediate PEG-IFN alfa-2b treatment for 24 weeks.14 However, symptomatic patients with delayed treatment were more likely to have discontinuation than symptomatic and asymptomatic patients assigned immediate treatment (42% vs 25%, P=0.037).14 Another randomized, open-label trial was conducted among Canadian adult drug users with chronic HCV to compare immediate treatment (treating 24 weeks for HCV genotype 1 and 48 weeks for genotypes 2 or 3) to delayed treatment (starting treatment after 24 weeks of observation for HCV genotype 1 and 48 weeks of observation for genotypes 2 or 3) with PEG-IFN alfa-2a plus RBV and followed for 96 months.15 Similarly, patients who received the delayed treatment had a slightly lower SVR than the patients who received the immediate treatment (39% vs 65%, P=0.06). However, drop rates between immediate and delayed treatment were not significantly different (23% vs 33%, P=0.389). The effects of PEG-IFN alfa-2a plus RBV and PEG-IFN alfa-2b plus RBV were compared among Egyptian patients with chronic HCV genotype 4.17 In this trial, the SVR for the patients treated with PEG-IFN alfa-2a were higher than that for the patients treated with PEG-IFN alfa-2b (59.6% vs 53.9%, P<0.05) at similar discontinuation rates (rates not reported); however, significantly higher rates of aspartate aminotransferase elevation and lower thyroid-stimulating hormone abnormalities were observed in patients with PEG-IFN alfa-2a (72.5% vs 69.2%, P=0.03; 10.3% vs 12.5%, P=0.04).
Triple versus dual therapy
A study utilizing triple therapy explored the risk of developing anemia in an international randomized trial in which patients were assigned to one of three arms: placebo plus PEG-IFN-α-2b and RBV for 44 weeks; boceprevir plus PEG-IFN-α-2b and RBV using response-guided therapy; and boceprevir plus PEG-IFN-α-2b and RBV for 44 weeks.16 The study found that patients in the boceprevir arms had much higher rates of anemia than those on dual therapy (50% vs 31%, P-value not reported); nevertheless, the SVR rate was higher in patients with anemia than those without anemia (72% vs 58%, P-value not reported).
Studies other than RCTs
Dual therapy
Hu et al28 evaluated the efficacy and safety of weekly PEG-IFN plus RBV among middle-aged adults (50–64 years) compared to the elderly adults (≥65 years) with chronic HCV infection in Taiwan. They found that SVR rate was significantly lower in the elderly than in the middle aged (40.7% vs 61.5%, P=0.005) with follow-up for 24 weeks after the completion of treatment.28 Interestingly, the significant difference only occurred in nongenotype 1 HCV patients. Compared to the middle-aged group, the elderly had a significantly higher rate of anorexia, dyspnea, skin rash, and hematological adverse events, including thrombocytopenia and anemia. Due to the intolerance to adverse events, withdrawal rates in the elderly were higher compared to the middle-aged (13.2% vs 7.7%, P-value not reported). A cross-sectional study compared the safety of HCV treatment with PEG-IFN alfa-2a vs PEG-IFN alfa-2b among 340 individuals at the Reference Center for Special Immunobiologicals of the Hospital Mario Covas, Brazil.31 Compared to PEG-IFN alfa-2a, PEG-IFN alfa-2b was more likely to be associated with fever (69.6% vs 45.5%, P=0.001) and weight loss (80.1% vs 62.1%, P=0.001). Another retrospective multicenter trial conducted in Korea discovered that PEG-IFN alfa-2a and PEG-IFN alfa-2b had similar effects on SVR and adverse events rate, even after controlling for age, HCV viral load, and hepatic fibrosis.29
A meta-analysis study, Flori et al,18 investigated eleven randomized and 15 nonrandomized trials to evaluate the effectiveness of PEG-IFN alfa-2a vs PEG-IFN alfa-2b.18 This study suggested that PEG-IFN alfa-2a plus RBV was more effective than PEG-IFN alfa-2b plus RBV, especially in genotype 1 or 4 HCV patients. Another meta-analysis study by Minami et al19 found that PEG-IFN alfa-2a was associated with a higher risk of severe adverse events than PEG-IFN alfa-2b (7.45% vs 6.74%, P-value not reported).19 Additionally, patients with higher doses of PEG-IFN treatment had a higher risk of severe adverse events than those with lower doses (11.94% vs 6.99% for PEG-IFN alfa-2a; 7.10% vs 5.05% for PEG-IFN alfa-2b, P-value not reported). Patients also had a higher rate of severe adverse events with the extended duration (>48 weeks) than with the standard duration (48 weeks) (15.5% vs 6.67%, P-value not reported). However, another meta-analysis based on seven randomized and nonrandomized clinical trials did not find a statistically significant difference in rapid virological response and SVR between PEG-IFN alfa-2a and PEG-IFN alfa-2b, although it found a positive relationship between PEG-IFN alfa-2a and early complete virological response and sustained virological response.20 Another meta-analysis compared the treatment duration of PEG-IFN plus RBV and found that extended PEG-IFN plus RBV for 72 weeks was more effective than standard PEG-IFN plus RBV for 48 weeks among patients with HCV genotype 1, while no difference in adverse events was observed for the two treatment strategies.21 Finally, a pooled analysis based on five RCTs assessed the safety of standard-dose (180 μg/week) vs high-dose (360 μg/week) PEG-IFN alfa-2a plus RBV in HCV genotype 1 or 4 patients.22 Although it did not find a statistically significant difference in frequencies of severe adverse events (3.2% vs 4.2%, P>0.05) or discontinuation rates (2.8% vs 2.9%, P>0.05), it showed that patients receiving high doses were more likely to experience weight loss compared to those receiving the standard dose (7.7% vs 3.3%, P<0.05).
Triple therapy versus dual therapy
A meta-analysis study, Sitole et al,23 of Phase II and Phase III placebo-controlled studies, suggested that chronic HCV patients treated with telaprevir or boceprevir + PEG-IFN + RBV were each about three times more likely to achieve SRV than the control group (odds ratio =3.31 or 3.55; P<0.0001).23 Additionally, telaprevir patients were more likely to incur rash, pruritus, and anemia, while boceprevir patients were more likely to incur anemia and dysgeusia.23 Another meta-analysis study found that both boceprevir and telaprevir could achieve better SVR (relative risk [RR] =1.91, P<0.05 for boceprevir; RR =1.69, P<0.05 for telaprevir), reduce relapse rate (RR =0.24, P>0.05; RR =0.30, P<0.05), and decrease the discontinuation rate (RR =0.65, P<0.05; RR =0.81, P>0.05) than placebo.24 This study also found that patients treated with boceprevir were more likely to have anemia (RR =1.63) while those treated with telaprevir were more likely to have rash (RR =1.49, P<0.05) and pruritus (RR =1.41, P<0.05). A meta-analysis by Qin et al25 discovered that the addition of telaprevir to PEG-IFN plus RBV increased the risk of severe adverse events (RR =1.56, P=0.0007), treatment discontinuation (RR =2.10, P<0.0001), nausea (RR =1.39, P<0.0001), diarrhea (RR =1.32, P=0.004), pruritus (RR =1.56; P=0.0006), rash (RR =1.60, P<0.0001), and anemia (RR =1.55, P=0.007).25 The study by Gu et al26 showed that telaprevir with PEG-IFN plus RBV significantly lowered the SVR without the higher incidence of severe adverse events or treatment discontinuation among genotype 1 HCV patients.26
Triple therapy
Furusyo et al30 suggested that telaprevir plus PEG-IFN and RBV had similar effects on HCV RNA and SVR in both genotype 1b HCV Japanese patients aged >60 years and aged ≤60 years. Additionally, in both age groups, patients with IL28B TT allele had a significantly better response than patients with IL28 TG/GG allele according to their SVR rates.30
SVR and mortality
An international, multicenter, long-term follow-up study in Europe and Canada investigated the association between SVR and mortality in patients with chronic HCV or advanced hepatic fibrosis.32 The study found that SVR was associated with a reduced risk of all-cause mortality (hazard ratio =0.26; P<0.001) and a reduced risk of liver-related mortality or transplantation (hazard ratio =0.06; P<0.001). This study also found that patients with SVR were less likely to have hepatocellular carcinoma (5.1% vs 21.8%) and liver failure (2.1% vs 29.9%, P<0.001) than patients without SVR. A meta-analysis on observational studies by Morgan et al27 further supported the finding that SVR had an effect on reducing hepatocellular carcinoma, with a pooled RR of 0.24.27
Quality assessment for included studies
We evaluated the quality of the RCTs using the Cochrane Collaboration’s tool, which is commonly used to evaluate RCTs in many systematic reviews and meta-analysis studies.34 It consists of five components: random sequence generation, concealment of allocation, blinding, attrition, and funding source. The quality assessment results for RCTs are reported in Table 2. Among 13 RCTs, seven RCTs provided a description about how the allocation sequence was generated randomly, with an illustration of the adequacy of the procedures.14,15,34–38 All but one RCT discussed the concealment of allocation.35 Eight RCTs were open-label trials;14–17,36–39 only one RCT was blinded adequately,35 and four RCTs were unclear about the blinding.40–43 Ten RCTs justified their data sets in terms of discontinuation or drop-off rates for each arm,14–16,35,37,38,40–43 while three RCTs provided inadequate attrition data.17,36,39 As for funding sources, five RCTs were supported fully or partially by pharmaceutical companies,15,16,35,37,39 two RCTs were supported by governments or other nonprofit organizations,14,42 and six RCTs did not report their funding source.17,36,38,40,41,43
  | Table 2 Quality assessment for RCTs included in the updated publication |
As for non-RCTs, we adopted the Grading of Recommendations Assessment, Development and Evaluation (GRADE) approach to access study quality.44 GRADE considers three factors: sample size, effect size, and bias to adjust the magnitude of treatment effects. The quality assessment results for non-RCT studies were reported in Table 3. Most of the studies in our review were rated as moderate quality except for a cross-sectional study that compared PEG-IFN alfa-2a and PEG-IFN alfa-2b in terms of safety profiles.31
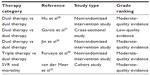  | Table 3 Quality assessment for non-RCT studies included in the updated publication |
Study needs in CER for hepatitis C
Research gaps and limitations of the existing CER literature and future study needs to address them were identified in the study by Chou et al;33 these are listed in Table 4. The limitations were organized according to the PICOT (Population-related gaps, Intervention-related gaps, Comparator-related gaps, Outcome-related gaps, and Timing-related gaps) framework, then sent by survey to stakeholders who prioritized their importance and gave additional feedback.33 Stakeholders included clinicians, researchers, policy makers, payors, and consumer advocates. The resulting list included 12 limitations ranked in order of importance from highest to lowest. Issues 1–7 were classified as top-tier issues, and the remainder as second-tier issues (Table 4).
Chou et al then looked at ongoing clinical trials to assess how forthcoming research was addressing these issues.33 Using ClinicalTrials.gov, they identified and reviewed 46 ongoing studies that might address top-tier issues through August 2012. Two addressed issue 2 (broader spectrum of patients), four addressed issue 3 (new IFN-free drug regimens), and three addressed issue 6 (long-term outcomes). This review updated the list of clinical trials following the same search strategy of Chou et al.33 In addition to the 46 studies identified in Chou’s article, 22 new studies were found after exclusions. These studies were then evaluated to determine whether they addressed the identified limitations that were initially classified in Chou et al33 (Table 4).
Several trends regarding the progress of HCV treatment are apparent in these results. Most notably, some limitations are being addressed more heavily than others. This imbalance is best illustrated by issue 3 (new IFN-free drugs). While many limitations were addressed in few or no studies, issue 3 was addressed in ten of the 22 new studies. Orally delivered, IFN-free drug regimens are becoming much more common, and it is expected that such treatment will become standard. IFN-free treatment should also decrease adverse event rates and increase compliance. Longer-term studies (issue 6) are also receiving some attention, with three more studies looking at relapse and safety outcomes beyond 1 year post-treatment. Population-related gaps were addressed in three studies: two recruiting geriatric patients (issue 10) and one recruiting active drug users (issue 2). Lastly, one study was independently funded (issue 11) and looked at long-term harms of telaprevir/boceprevir (issue 12). These ongoing trials indicate that some progress is being made in filling in current research gaps. However, five of the identified limitations were not addressed in any of the published papers or ongoing trials identified in this review. Issues such as real-world effects, poor adherence, and clinically relevant outcomes are not being seen in ongoing trials. Rather, the majority of studies continue to be industry-sponsored RCTs using highly selective inclusion criteria and short-term SVR as outcomes. Although RCTs are known to be the best available study method of treatment outcomes, a retrospective study may better answer the long-term outcomes of HCV treatment options.
Discussion
Alternative antiviral therapies for HCV continue to be improved, so careful evaluation of their effectiveness and safety is required to assist patients, clinicians, and policy makers to make informed decisions. There has been only one comparative effectiveness research study of HCV treatment by Chou et al.13 Although no more comparative effectiveness research has been conducted since then, 19 new studies have been published, and they reinforce most of the conclusions made by Chou et al. Concerning double therapy, three additional articles reported an advantage of PEG-IFN alfa-2a over PEG-IFN alfa-2b.17,18,20 The duration of dual therapy continued to be a common research question, and another three articles reinforced the thought that the longer duration of dual therapy was associated with higher SVR in HCV genotypes 2 and 3.19,21,35 In genotype 1, a clear advantage was still seen in triple therapy over dual therapy. However, five articles reported that this advantage did come at the cost of a higher rate of adverse effects, such as anemia with boceprevir and rash with telaprevir.16,23–25 Two articles, including one meta-analysis study in our search, examined the association of achieving SVR and health outcomes, and they agreed that patients who attain SVR are less likely to experience liver carcinoma, liver failure, and liver-related death.32 All of these findings are in harmony with the conclusions made by Chou et al. Additionally, consistent with Chou et al, we were unable to find any study measuring long-term clinical outcomes associated with treatment.
A small number of new articles since Chou et al13 also add to CER knowledge by addressing issues that were not seen in the AHRQ article. First, some studies in our search looked at the likelihood of SVR in specific subgroups. Two of these looked at response rates in elderly and nonelderly patients. One found lower rates of SVR in the elderly undergoing dual therapy.28 The other found no difference in SVR between elderly and nonelderly undergoing triple therapy.30 One study included genetic data, looking at the effect of the IL28B gene on SVR.30 Another study researched the effect of individualizing treatment duration based on early virological response.38 Lastly, two studies addressed the effect of delayed PEG-IFN treatment, but neither one was able to show a statistically significant difference.14,15 While most articles in our search addressed the same issues as the AHRQ article, these few articles show that some research is being done to fill in gaps.
Some of the gaps and limitations of current CER literature for HCV were filled by additional research over the past year, yet many of them still remained unanswered. According to the ongoing research identified by Chou et al,33 effectiveness of alternative therapies for a broader range of patients, long-term clinical outcomes of current treatment options, and evidence on new IFN-free drug regimens may further be answered with the completion of ongoing clinical trials. We identified 22 new additional clinical trials since August 2012. Although one of our updated ongoing clinical trials may fill the gap in the literature by analyzing the long-term effect and harm of telaprevir and boceprevir in an independently funded setting, most of the updated ongoing clinical trials focus more on the same issues as clinical trials listed in Chou et al.33 One limitation of this review is the lack of data concerning the recently approved simeprevir and sofosbuvir. While clinical trials of these drugs were included in our identified list of ongoing studies, the results and subsequent approvals occurred after our literature search was completed.
Conclusion
A fair amount of research has been conducted to evaluate efficacy and effectiveness of antiviral therapies and provide evidence. To have complete information on the effectiveness of alternative treatments for HCV, however, further research on their effectiveness in the general population, data on treatment methods such as noninvasive treatment and individualized treatment, and the long-term effect of triple therapies is needed. Additionally, evidence in a real-world setting is lacking. Methodologically thorough and independently funded retrospective research will help to generalize the effectiveness of current therapies for HCV.
Disclosure
The authors report no conflicts of interest in this work.
References
Mohd Hanafiah K, Groeger J, Flaxman AD, Wiersma ST. Global epidemiology of hepatitis C virus infection: new estimates of age-specific antibody to HCV seroprevalence. Hepatology. 2013;57(4):1333–1342. | |
Perz JF, Armstrong GL, Farrington LA, Hutin YJ, Bell BP. The contributions of hepatitis B virus and hepatitis C virus infections to cirrhosis and primary liver cancer worldwide. J Hepatol. 2006;45(4):529–538. | |
Ly KN, Xing J, Klevens RM, Jiles RB, Ward JW, Holmberg SD. The increasing burden of mortality from viral hepatitis in the United States between 1999 and 2007. Ann Intern Med. 2012;156(4):271–278. | |
Alter MJ, Kruszon-Moran D, Nainan OV, et al. The prevalence of hepatitis C virus infection in the United States, 1988 through 1994. N Engl J Med. 1999;341(8):556–562. | |
Veldt BJ, Heathcote EJ, Wedemeyer H, et al. Sustained virologic response and clinical outcomes in patients with chronic hepatitis C and advanced fibrosis. Ann Intern Med. 2007;147(10):677–684. | |
McHutchison JG, Lawitz EJ, Shiffman ML, et al. Peginterferon alfa-2b or alfa-2a with ribavirin for treatment of hepatitis C infection. N Engl J Med. 2009;361(6):580–593. | |
Jazwinski AB, Muir AJ. Direct-acting antiviral medications for chronic hepatitis C virus infection. Gastroenterol Hepatol (NY). 2011;7(3):154–162. | |
Stedman CA. Current prospects for interferon-free treatment of hepatitis C in 2012. J Gastroenterol Hepatol. 2013;28(1):38–45. | |
Ghany MG, Nelson DR, Strader DB, Thomas DL, Seeff LB; American Association for Study of Liver Diseases. An update on treatment of genotype 1 chronic hepatitis C virus infection: 2011 practice guideline by the American Association for the Study of Liver Diseases. Hepatology. 2011;54(4):1433–1444. | |
Jacobson IM, McHutchison JG, Dusheiko G, et al; ADVANCE Study Team. Telaprevir for previously untreated chronic hepatitis C virus infection. N Engl J Med. 2011;364(25):2405–2416. | |
Poordad F, McCone J, Bacon BR, et al; SPRINT-2 Investigators. Boceprevir for untreated chronic HCV genotype 1 infection. N Engl J Med. 2011;364(13):1195–1206. | |
Fried MW. Side effects of therapy of hepatitis C and their management. Hepatology. 2002;36(5 Suppl 1):S237–S244. | |
Chou R, Hartung D, Rahman B, Wasson N, Cottrell EB, Fu R. Comparative effectiveness of antiviral treatment for hepatitis C virus infection in adults: a systematic review. Ann Intern Med. 2013;158(2):114–123. | |
Deterding K, Grüner N, Buggisch P, et al; Hep-Net Acute HCV-III Study Group. Delayed versus immediate treatment for patients with acute hepatitis C: a randomised controlled non-inferiority trial. Lancet Infect Dis. 2013;13(6):497–506. | |
Hilsden RJ, Macphail G, Grebely J, Conway B, Lee SS. Directly observed pegylated interferon plus self-administered ribavirin for the treatment of hepatitis C virus infection in people actively using drugs: a randomized controlled trial. Clin Infect Dis. 2013;57 Suppl 2:S90–S96. | |
Sulkowski MS, Poordad F, Manns MP, et al; SPRINT-2 Trial Investigators. Anemia during treatment with peginterferon Alfa-2b/ribavirin and boceprevir: Analysis from the serine protease inhibitor therapy 2 (SPRINT-2) trial. Hepatology. 2013;57(3):974–984. | |
El Raziky M, Fathalah WF, El-Akel WA, et al. The effect of peginterferon Alpha-2a vs peginterferon Alpha-2b in treatment of naive chronic HCV genotype-4 patients: A Single Centre Egyptian Study. Hepat Mon. 2013;13(5):e10069. | |
Flori N, Funakoshi N, Duny Y, et al. Pegylated interferon-α2a and ribavirin versus pegylated interferon-α2b and ribavirin in chronic hepatitis C: a meta-analysis. Drugs. 2013;73(3):263–277. | |
Minami T, Kishikawa T, Sato M, Tateishi R, Yoshida H, Koike K. Meta-analysis: mortality and serious adverse events of peginterferon plus ribavirin therapy for chronic hepatitis C. J Gastroenterol. 2013;48(2):254–268. | |
Coppola N, Pisaturo M, Tonziello G, Sagnelli C, Sagnelli E, Angelillo IF. Efficacy of Pegylated interferon α-2a and α-2b in patients with genotype 1 chronic hepatitis C: a meta-analysis. BMC Infect Dis. 2012;12:357. | |
Katz LH, Goldvaser H, Gafter-Gvili A, Tur-Kaspa R. Extended peginterferon plus ribavirin treatment for 72 weeks versus standard peginterferon plus ribavirin treatment for 48 weeks in chronic hepatitis C genotype 1 infected slow-responder adult patients. Cochrane Database Syst Rev. 2012;9:CD008516. | |
Marcellin P, Roberts SK, Reddy KR, et al. Safety profile of standard- vs high-dose peginterferon alfa-2a plus standard-dose ribavirin in HCV genotype 1/4 patients: pooled analysis from 5 randomized studies. Expert Opin Drug Saf. 2012;11(6):901–909. | |
Sitole M, Silva M, Spooner L, Comee MK, Malloy M. Telaprevir versus boceprevir in chronic hepatitis C: a meta-analysis of data from phase II and III trials. Clin Ther. 2013;35(2):190–197. | |
Cooper CL, Druyts E, Thorlund K, et al. Boceprevir and telaprevir for the treatment of chronic hepatitis C genotype 1 infection: an indirect comparison meta-analysis. Ther Clin Risk Manag. 2012;8:105–130. | |
Qin H, Li H, Zhou X, et al. Safety of telaprevir for chronic hepatitis C virus infection: a meta-analysis of randomized controlled trials. Clin Drug Investig. 2012;32(10):665–672. | |
Gu L, Chen Q, Xie J, et al. Telaprevir for genotype 1 chronic hepatitis C: a systematic review and meta-analysis. Pharmazie. 2012;67(12):963–972. | |
Morgan RL, Baack B, Smith BD, Yartel A, Pitasi M, Falck-Ytter Y. Eradication of hepatitis C virus infection and the development of hepatocellular carcinoma: a meta-analysis of observational studies. Ann Intern Med. 2013;158(5 Pt 1):329–337. | |
Hu CC, Lin CL, Kuo YL, et al. Efficacy and safety of ribavirin plus pegylated interferon alfa in geriatric patients with chronic hepatitis C. Aliment Pharmacol Ther. 2013;37(1):81–90. | |
Jin YJ, Lee JW, Lee JI, et al. Multicenter comparison of PEG-IFN α2a or α2b plus ribavirin for treatment-naïve HCV patient in Korean population. BMC Gastroenterol. 2013;13:74. | |
Furusyo N, Ogawa E, Nakamuta M, et al; Kyushu University Liver Disease Study (KULDS) Group. Telaprevir can be successfully and safely used to treat older patients with genotype 1b chronic hepatitis C. J Hepatol. 2013;59(2):205–212. | |
Garcia TJ, Lara PH, Morimoto TP, Higasiaraguti M, Perejão AM, Ayub MA. Side effects of the hepatitis C treatment at the ABC application center. Rev Assoc Med Bras. 2012;58(5):543–549. | |
van der Meer AJ, Veldt BJ, Feld JJ, et al. Association between sustained virological response and all-cause mortality among patients with chronic hepatitis C and advanced hepatic fibrosis. JAMA. 2012;308(24):2584–2593. | |
Chou R, Bougatsos C, Blazina I, Dana T, Cantor A. Treatment of Hepatitis C virus infection in adults: future research needs: identification of future research needs from comparative effectiveness review no 76 [webpage on the Internet]. Available from: http://www.effectivehealthcare.ahrq.gov/ehc/products/502/1470/hepatitis-C-treatment-future-130415.pdf. Accessed February 4, 2014. | |
Higgins JP, Altman DG, Gøtzsche PC, et al. Cochrane Bias Methods Group; Cochrane Statistical Methods Group. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928. | |
Kamal SM, El Kamary SS, Shardell MD, et al. Pegylated interferon alpha-2b plus ribavirin in patients with genotype 4 chronic hepatitis C: The role of rapid and early virologic response. Hepatology. 2007;46(6):1732–1740. | |
Glue P, Rouzier-Panis R, Raffanel C, et al. A dose-ranging study of pegylated interferon alfa-2b and ribavirin in chronic hepatitis C. The Hepatitis C Intervention Therapy Group. Hepatology. 2000;32(3):647–653. | |
Lee SD, Yu ML, Cheng PN, et al. Comparison of a 6-month course peginterferon alpha-2b plus ribavirin and interferon alpha-2b plus ribavirin in treating Chinese patients with chronic hepatitis C in Taiwan. J Viral Hepat. 2005;12(3):283–291. | |
Lee SS, Sherman M, Ramji A, et al. Randomised clinical trial: the efficacy of treatment, guided by a shorter duration of response, using peginterferon alfa-2a plus ribavirin for hepatitis C virus other than genotypes 2 or 3. Aliment Pharmacol Ther. 2012;35(1):37–47. | |
Sjogren MH, Sjogren R, Lyons MF, et al. Antiviral response of HCV genotype 1 to consensus interferon and ribavirin versus pegylated interferon and ribavirin. Dig Dis Sci. 2007;52(6):1540–1547. | |
Bosques-Padilla F, Trejo-Estrada R, Campollo-Rivas O, et al. Peginterferon alfa-2a plus ribavirin for treating chronic hepatitis C virus infection: analysis of Mexican patients included in a multicenter international clinical trial. Ann Hepatol. 2003;2(3):135–139. | |
Bruno S, Camma C, Di Marco V, et al. Peginterferon alfa-2b plus ribavirin for naive patients with genotype 1 chronic hepatitis C: a randomized controlled trial. J Hepatol. 2004;41(3):474–481. | |
Fried MW, Shiffman ML, Reddy KR, et al. Peginterferon alfa-2a plus ribavirin for chronic hepatitis C virus infection. N Engl J Med. 2002;347(13):975–982. | |
Mangia A, Minerva N, Bacca D, et al. Individualized treatment duration for hepatitis C genotype 1 patients: A randomized controlled trial. Hepatology. 2008;47(1):43–50. | |
Guyatt GH, Oxman AD, Vist GE, et al; GRADE Working Group. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ. 2008;336(7650):924–926. | |
Miyase S, Haraoka K, Ouchida Y, Morishita Y, Fujiyama S. Randomized trial of peginterferon α-2a plus ribavirin versus peginterferon α-2b plus ribavirin for chronic hepatitis C in Japanese patients. J Gastroenterol. 2012;47(9):1014–1021. | |
Gilead Sciences. Safety and Efficacy of Sofosbuvir/Ledipasvir Fixed-Dose Combination (FDC) With and Without Ribavirin for the Treatment of HCV. Available from: http://clinicaltrials.gov/show/NCT01701401. NLM identifier: NCT01701401. Accessed February 27, 2014. | |
AbbVie (prior sponsor, Abbott). A Study to Evaluate Chronic Hepatitis C Infection. Available from: http://clinicaltrials.gov/show/NCT01716585. NLM identifier: NCT01716585. Accessed February 27, 2014. | |
Bristol-Myers Squibb. A Phase 3, Comparative Study of Asunaprevir and Daclatasvir Combination Therapy Versus Telaprevir Therapy in Japanese HCV Subjects. Available from: http://clinicaltrials.gov/show/NCT01718145. NLM identifier: NCT01718145. Accessed February 27, 2014. | |
Boehringer Ingelheim. Phase 3 Study of BI 207127 in Combination With Faldaprevir and Ribavirin for Treatment of Patients With Hepatitis C Infection, Including Patients Who Are Not Eligible to Receive Peginterferon: HCVerso2. Available from: http://clinicaltrials.gov/show/NCT01728324. NLM identifier: NCT01728324. Accessed February 27, 2014. | |
Boehringer Ingelheim. IFN-free Combination Therapy in HCV-infected Patients Treatment-naive:HCVerso1. Available from: http://clinicaltrials.gov/show/NCT01732796. NLM identifier: NCT01732796. Accessed February 27, 2014. | |
AbbVie (prior sponsor, Abbott). A Study to Evaluate Chronic Hepatitis C Infection in Adults With Genotype 1b Infection (PEARL-III). Available from: http://clinicaltrials.gov/show/NCT01767116. NLM identifier: NCT01767116. Accessed February 27, 2014. | |
AbbVie. A Study to Evaluate Chronic Hepatitis C Infection in Adults With Genotype 1a Infection (PEARL-IV). Available from: http://clinicaltrials.gov/show/NCT01833533. NLM identifier: NCT01833533. Accessed February 28, 2014. | |
Gilead Sciences. Safety and Efficacy of Sofosbuvir/Ledipasvir Fixed-Dose Combination ± Ribavirin for the Treatment of HCV (ION-3). Available from: http://clinicaltrials.gov/ct2/show/NCT01851330. NLM identifier: NCT01851330. Accessed February 28, 2014. | |
AbbVie. A Study to Evaluate the Efficacy and Safety of Three Experimental Drugs Compared With Telaprevir (a Licensed Product) in People With Hepatitis C Virus Infection Who Have Not Had Treatment Before (MALACHITE 1). Available from: http://clinicaltrials.gov/ct2/show/NCT01854697. NLM identifier: NCT01854697. Accessed February 28, 2014. | |
Boehringer Ingelheim. Open Label Trial to Compare BI 207127 to Telaprevir in HCV Patients. Available from: http://clinicaltrials.gov/show/NCT01858961. NLM identifier: NCT01858961. Accessed February 28, 2014. | |
Bristol-Myers Squibb. Safety and Efficacy Study of Peginterferon Lambda-1a vs. Peginterferon Alfa-2a, Plus Ribavirin in Subjects With Genotype 1 Hepatitis C (BASIS). Available from: http://clinicaltrials.gov/show/NCT01754974. NLM identifier: NCT01754974. Accessed February 28, 2014. | |
Hoffmann-La Roche. An Observational Study on Dual And Triple Therapies Based on Peginterferon Alfa (eg, Pegasys) in Patients With Chronic Hepatitis C. Available from: http://clinicaltrials.gov/show/NCT01447446. NLM identifier: NCT01447446. Accessed February 28, 2014. | |
Hoffmann-La Roche. An Observational Study of Dual and Triple Therapies Based on Pegasys (Peginterferon Alfa-2a) in Patients With Chronic Hepatitis C. Available from: http://clinicaltrials.gov/show/NCT01604291. NLM identifier: NCT01604291. Accessed February 28, 2014. | |
Southern Illinois University. Comparison of Two Triple Regimens for Treatment and Retreatment of Chronic Hepatitis C Infection. Available from: http://clinicaltrials.gov/show/NCT01771653. NLM identifier: NCT01771653. Accessed February 28, 2014. |
Supplementary materials
  | Table S1 Search strategies |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


