Back to Journals » Therapeutics and Clinical Risk Management » Volume 18
Cumulative Doses Predict the Risk of Furosemide-Induced Electrolyte Abnormalities in Critically Ill Neonates
Authors Sridharan K , Al Madhoob A, Al Jufairi M
Received 9 April 2022
Accepted for publication 2 June 2022
Published 9 June 2022 Volume 2022:18 Pages 633—641
DOI https://doi.org/10.2147/TCRM.S369737
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Garry Walsh
Kannan Sridharan,1 Abdulraoof Al Madhoob,2 Muna Al Jufairi2,3
1Department of Pharmacology & Therapeutics, College of Medicine & Medical Sciences, Arabian Gulf University, Manama, Kingdom of Bahrain; 2Neonatal Intensive Care Unit, Salmaniya Medical Complex, Manama, Kingdom of Bahrain; 3Department of Pediatrics, College of Medicine & Medical Sciences, Arabian Gulf University, Manama, Kingdom of Bahrain
Correspondence: Kannan Sridharan, Department of Pharmacology & Therapeutics, College of Medicine & Medical Sciences, Arabian Gulf University, Manama, Kingdom of Bahrain, Email [email protected]
Background: Furosemide has limited indications in the term neonates. Its use in preterm neonates is off-label. Considering the dearth of data, we carried out a retrospective study evaluating the furosemide use and its effects on the electrolyte abnormalities in critically ill neonates.
Methods: Critically ill neonates receiving at least one dose of furosemide during their stay in the NICU were recruited. The following details were obtained: demographic characteristics and furosemide details (dose, frequency, route, and duration). Urine output, body weight, serum creatinine, electrolytes (sodium, potassium, calcium, bicarbonate, chloride, and magnesium) during furosemide therapy were extracted.
Results: Ninety neonates were recruited. Elevated serum creatinine was observed in 21.1% of the patients, and electrolyte disturbances were observed in 52.2%. Those with electrolyte disturbances had significantly greater cumulative doses compared to those without [5.5 (1– 34) vs 3.9 (0.9– 30.2) mg/kg; p = 0.01]. Cumulative doses adjusted to body-weight were significantly lower in very preterm and extremely preterm neonates compared to late preterm category. A significant area-under-the-curve was observed for the cumulative dose (0.66; 95% CI: 0.54– 0.78; p = 0.01) in predicting the risk of electrolyte abnormalities with a cut-off value of 4 mg/kg. Eight neonates received more than 10 mg/kg cumulative dose of furosemide of which one died. No significant differences were observed between the proportion of neonates with electrolyte disturbances with cumulative furosemide dose above 10 mg/kg (p = 0.3) and with mortality (p = 0.3).
Conclusion: We observed that our critically ill neonates received relatively higher cumulative doses of furosemide and electrolyte disturbances were observed in nearly half of the population. A cumulative dose of 4 mg/kg increases the risk of electrolyte abnormalities, particularly in preterm neonates. More diligence in the dose titration coupled with deprescribing and intense monitoring of all the potential adverse effects in this vulnerable population is needed.
Keywords: furosemide, preterm neonates, off-label drug use
Background
Furosemide, a loop-diuretic, has been approved for use in term neonates for treating edema due to congestive heart failure, renal failure, and liver failure.1,2 However, it is often used in preterm neonates as an off-label drug for treating chronic lung disease, bronchopulmonary dysplasia, and transient tachypnea of newborns.3 Off-label use of drugs is rampant in neonates to an extent of 96.4% that is associated with several ethical and legal challenges particularly in the assessment of risk–benefit ratio.4 The well-characterized adverse effects of furosemide in adults and older children include electrolyte abnormalities (such as hyponatremia, hypokalemia, hypochloremia, hypocalcemia, and hypomagnesemia), sensorineural hearing defects, and nephrocalcinosis.5 The first few days of neonatal life are characterized by rapid development and maturation of nephrons. Hence, furosemide, whose target organ of action is the kidney, can be characterized by varied therapeutic and adverse effects in this population. Additionally, the spectrum of indications for which furosemide is being used in neonates significantly differs from that of other populations. There is a dearth of conclusive evidence related to the extent and severity of adverse events of furosemide in the neonatal population. A recent study by Williams et al6 reveals that 44.6% of extremely preterm neonates received diuretics for at least seven days and highlighted the absence of high-quality clinical studies evaluating the use of diuretic class in this population. Furthermore, furosemide dosing regimens vary widely between the neonatal intensive care units (NICUs) worldwide.7 In our NICU, the dose of furosemide ranges between 0.5 and 1 mg/kg/dose and depending on the clinical condition, dosing regimen is titrated. Hence, we carried out the present study to evaluate the furosemide use and its effects on the various clinical and laboratory parameters in our critically ill neonates.
Methods
Ethics and Study Design
This was a retrospective study carried out in the neonatal intensive care unit (NICU) of the largest tertiary care hospital in the Kingdom of Bahrain, between September 2020 and August 2021 after obtaining approval from the Institutional Ethics Committees (Arabian Gulf University: E030-PI-9/21; and Salmaniya Medical Complex: 138011121). Informed consent was waived as the study was retrospective in design. The study team took all the efforts for maintaining the patient confidentiality. We adhered to the latest Declaration of Helsinki guidelines.
Study Procedure
Critically ill neonates who received at least one dose of furosemide during their stay in the NICU were the study participants. The following details were obtained: demographic characteristics (gestational age, sex, length, birth weight, APGAR scores, duration of stay) and furosemide details (dose, frequency, route, and duration). Cumulative furosemide doses were estimated for the entire stay of neonates in the critical care unit. Urine output, body weight, serum creatinine, electrolytes (sodium, potassium, calcium, bicarbonate, chloride, and magnesium) during furosemide were extracted.
Statistical Analysis
Descriptive statistics were used for representing the demographic characteristics of the study participants. Pearson’s correlation test was used for evaluating the association between numerical variables. Mann–Whitney U-test was used for evaluating the differences in the cumulative doses between those with electrolyte disturbances. Receiver-operating characteristics (ROC) curve was generated for evaluating the cumulative furosemide doses in predicting the risk of electrolyte abnormalities in the overall population, in preterm, and term neonates. The Youden’s index was used for determining the cut-off values. Neonates were classified based on their gestational age as follows: extremely preterm (<28 weeks); very preterm (28 to <32 weeks); late preterm (32 to <37 weeks); and term (≥37 weeks of gestation).8 Birth weights were classified as follows: ≥2.5 kg – normal; 1.5 to <2.5 kg – low; 1 to <1.5 kg – very low; and <1 kg – extremely low. The reference ranges adhered in the present study were as follows: serum sodium – 133–146 mmol/L; potassium – 3.5–5.5 mmol/L; chloride – 100–117 mmol/L; calcium – 1.5–2.9 mmol/L; magnesium – 0.62–1 mmol/L; creatinine – 26.5–88.4 mmol/L; and bicarbonate – 16–24 mmol/L. Percent changes in serum electrolytes were estimated from the observed maximum changes in the electrolyte levels during the neonates’ stay in NICU compared to baseline. Cumulative dose adjusted for body-weight was calculated by dividing the cumulative doses upon body weights of the neonates. A p-value of ≤0.05 was considered statistically significant. SPSS (IBM Corp. Released 2020. IBM SPSS Statistics for Windows, Version 27.0. Armonk, NY: IBM Corp.) was used for statistical analysis.
Results
Demographic Characteristics
Ninety neonates were enrolled, and a summary of their demographic characteristics is mentioned in Table 1. The following were the diagnoses amongst the study participants: sepsis (n = 35), respiratory distress syndrome (n = 33), patent ductus arteriosus (PDA) (n = 28), intraventricular hemorrhage (n = 15), metabolic disorder (n = 11), seizure disorder (n = 7), congenital diaphragmatic hernia (n = 4), pneumonia (n = 4), birth asphyxia (n = 3), persistent pulmonary hypertension (n = 2), retinopathy of prematurity (n = 2), and one each with apnea of prematurity, tachypnea of newborn, and pneumothorax.
 |
Table 1 Demographic Characteristics of the Study Participants (N = 90) |
Dosage Regimen of Furosemide
The median (range) of the furosemide dose used in the study participants was 1 (0.2–5) mg. Furosemide was administered once daily (n = 54), twice daily (n = 92), thrice daily (n = 5), and four times a day (n = 2). The median (range) duration of furosemide therapy was 2 (1–12) days. Two neonates received oral furosemide, while the remaining received intravenously. Indications for receiving furosemide were as follows: respiratory distress syndrome (n = 33), patent ductus arteriosus (n = 28), chronic lung disorder (n = 28), and transient tachypnea in newborn (n = 1). Median (range) cumulative dose of furosemide amongst the study participants was 5.5 (0.6–119) mg. A weak positive correlation (r: 0.4; 95% CI: 0.3–0.6; p < 0.001) was observed between the gestational age and cumulative furosemide doses (Figure 1). The cumulative furosemide doses were significantly lower in extremely preterm, and very preterm neonates compared to late preterm and term neonates (Figure 2). Similarly, cumulative doses were significantly (p = 0.04) lower amongst those that died compared to alive [2.7 (1.4–9.8) mg vs 6 (0.6–119) mg]. The median (range) cumulative dose adjusted to body-weight was 4.7 (0.9–34.5) mg. Late preterm neonates received significantly higher cumulative dose adjusted to body-weight compared to other gestational age categories (Figure 3). Eight neonates received more than 10 mg/kg cumulative dose of furosemide of which one died.
 |
Figure 1 Correlation between gestational age and cumulative furosemide doses. The observations are represented by blue circles. The diagonal line represents the line of best fit. |
The median (range) of the daily dose of furosemide was 1 (0.2–4.7) mg/kg body weight. A significant correlation was observed between the gestational age and daily dose per kilogram of furosemide (r: 0.4; 95% CI: 0.2–0.5; p < 0.0001). Lower gestational age categories of neonates received lesser daily per kilogram body weight doses of furosemide compared to older categories (Figure 4). None of the neonates received furosemide doses of more than 6 mg/kg body weight.
Clinical and Laboratory Parameters
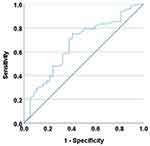
The baseline clinical and laboratory parameters for the study participants are listed in Table 2. Percent changes in the clinical and laboratory parameters following furosemide are depicted in Figure 5. Elevated serum creatinine was observed in 19/90 (21.1%), hyponatremia in 18 (20%), hypernatremia in 4 (4.4%), hypokalemia in 9 (10%), hyperkalemia in 11 (12.2%), hypochloremia in 36 (40%), hyperchloremia in 1 (1.1%), low bicarbonate in 3 (3.3%), hypercalcemia in 1 (1.1%), hypermagnesemia in 4 (4.4%), and hypomagnesemia in 3 (3.3%). Forty-seven (52.2%) had one or more electrolyte abnormalities as stated above. Those with electrolyte disturbances had significantly greater cumulative doses compared to those without [5.5 (1–34) vs 3.9 (0.9–30.2) mg/kg; p = 0.01]. ROC curve (Figure 6) revealed a significant area under the curve for cumulative dose (0.66; 95% CI: 0.54–0.78; p = 0.01). Youden’s index revealed a cut-off cumulative dose of 4 mg/kg in predicting the risk of electrolyte abnormalities. Sub-group analyses also revealed significant area under the curve for preterm neonates (0.65; 95% CI: 0.5–0.8; p = 0.03) but not for term (p = 0.3). In preterm neonates, a cut-off cumulative dose of 4.2 mg/kg was observed to predict the risk of electrolyte abnormalities. No significant differences were observed in the proportion of electrolyte disturbances between gestational age categories (term: 9, 69.2%; late preterm: 9, 52.7%; very preterm: 13, 50%; and extremely preterm: 16, 59.3%) (p = 0.7). No significant differences were observed between the proportion of neonates with electrolyte disturbances with cumulative furosemide dose above 10 mg/kg (p = 0.3), duration of furosemide (p = 0.3) and with mortality (p = 0.3). However, six neonates out of eight with cumulative doses >10 mg/kg had electrolyte abnormalities.
 |
Table 2 Baseline Clinical and Laboratory Parameters of the Study Participants (N = 90) |
 |
Figure 5 Percent changes in the clinical and laboratory parameters from baseline. The individual observations are represented by the colored circles. |
No documentation on the hearing assessment/abdominal ultrasonogram during the neonates’ stay in NICU could be retrieved. However, all the neonates were advised to follow-up with an otorhinolaryngologist following discharge from the hospital.
Discussion
We evaluated the use and effects of furosemide in 90 critically ill neonates. A positive correlation was observed between furosemide doses and gestational age categories. Elevated serum creatinine was observed in 21.1% of patients, and electrolyte disturbances were observed in 52.2%. Cumulative furosemide doses were significantly lower in the lower gestational age categories as well as those that died. Cumulative doses adjusted to body-weight are significantly lower in the very and extremely preterm neonates compared to late preterm. A cumulative dose above 4 mg/kg increases the risk of electrolyte abnormalities, particularly in preterm neonates. None of the neonates were administered furosemide more than 6 mg/kg body weight. Eight neonates received more than 10 mg/kg cumulative dose of furosemide of which one died.
We did not observe any neonates receiving furosemide doses above 6 mg/kg per day. However, a recent study from Italian intensive care units revealed that nearly 32.1% administered furosemide at more than the recommended doses.7 The United States Food and Drug Administration (USFDA) recommends initiating furosemide at 1 mg/kg dose, and titration can be done with 1 mg/kg until optimum clinical response, to a maximum of 6 mg/kg daily.9 We observed neonates in the very and extremely preterm neonates receiving a daily dose of furosemide more than 1 mg/kg. The USFDA label indicates that such neonates are at increased risk of adverse events such as ototoxicity following furosemide. However, a recent systematic evaluation of 32 observational studies revealed a low risk of sensorineural hearing loss in neonates <31 weeks following furosemide.10 However, high-quality randomized clinical trials are needed for attaining any firm conclusion. Until then, periodic monitoring of hearing and ultrasonogram of the abdomen are recommended for all neonates, particularly in preterm for early detection of hearing abnormalities and nephrocalcinosis. A recent report from North American NICUs revealed an average daily dose was 1 mg/kg (0.88, 1.1) with a median cumulative dose of 2 mg/kg (1.0, 4.5) and a shorter duration of 2 days (1, 4).11 However, our doses were relatively higher compared to this report, and differences in the population, and severity of illnesses could explain this difference. This also calls for improving the neonatologists’ awareness of the importance of rational use and potential de-prescription of furosemide. We observed that cumulative doses above 4 mg/kg increase the risk of electrolyte abnormalities. A recent study revealed that 85% of preterm neonates receiving 8 mg/kg and 64% of above 4 mg/kg cumulative doses of diuretics had an increased risk of metabolic bone disease.12 A cumulative dose above 10 mg/kg was observed to increase the risk of sensorineural defects.13 We had eight neonates receiving more than 10 mg/kg cumulative doses, and six of them had electrolyte abnormalities. Cumulative doses adjusted for body-weight were significantly higher in later preterm compared to very preterm and extremely preterm neonates in the present study. Interestingly, a recent study observed that neonates born between 36 and 41 weeks had a greater incidence of adverse events with furosemide compared to other diuretics.14 In a recent survey amongst the Italian neonatologists, nearly half of the centers (28 out of the total 57) were observed to administer furosemide at higher doses than those recommended by neonatal guidelines.7 There is an urgent need to standardize the furosemide doses according to gestational age categories. Also, we could not obtain the details of clinical impact of the observed electrolyte abnormalities as they were not captured electronically. However, during furosemide therapy, there was no documentation on either hearing assessment or abdominal ultrasound amongst the study participants. There is a lack of consensus in the literature on the criteria for carrying out evaluations for hearing defects and nephrocalcinosis, which is an urgent need of the hour. Additionally, we could not evaluate the relationship between the timing of occurrences of electrolyte abnormalities with cumulative doses as serum electrolytes are not monitored on stipulated intervals. Hypochloremia was observed more commonly than other electrolyte abnormalities in the present study. Hypochloremia has often been ignored despite its association with nephrocalcinosis, hypochloremic alkalosis, and poor growth in neonatal life.15
We observed that nearly 28 neonates in the presents study with PDA received furosemide. Furosemide administration in neonates with PDA has been debated and reports indicate delayed closure of the ductus.9 However, our previous experience revealed no such significant interaction.16 Perhaps, improvement in the monitoring techniques and advancement in the medical therapies for PDA have mitigated this drug–disease interaction.
Conclusion
We observed that our critically ill neonates received relatively higher cumulative doses of furosemide and electrolyte disturbances were observed in nearly half of the population. A cumulative dose of 4 mg/kg increases the risk of electrolyte abnormalities, particularly in preterm neonates. More diligence in the dose titration coupled with deprescribing and intense monitoring of all the potential adverse effects in this vulnerable population is needed.
Data Sharing Statement
The data is available with the corresponding author and will be shared upon a reasonable request.
Ethics
The study was carried out after obtaining approval from the Institutional Ethics Committee with the approval number: E030-PI-9/21.
Funding
There is no funding to report.
Disclosure
The authors report no conflicts of interest in relation to this work.
References
1. Pacifici GM. Clinical pharmacology of furosemide in neonates: a review. Pharmaceuticals. 2013;6(9):1094–1129. doi:10.3390/ph6091094
2. Bestic ML, Reed MD. Common diuretics used in the preterm and term infant: What’s changed? Neoreviews. 2012;13(7):e410–e419. doi:10.1542/neo.13-7-e410
3. Blaisdell CJ, Troendle J, Zajicek A. Acute responses to diuretic therapy in extremely low gestational age newborns: results from the PROP cohort study. J Pediatr. 2018;197:42–47. doi:10.1016/j.jpeds.2018.01.066
4. Costa HTML, Costa TX, Martins RR, Oliveira AG. Use of off-label and unlicensed medicines in neonatal intensive care. PLoS One. 2018;13(9):e0204427. doi:10.1371/journal.pone.0204427
5. Khan TM, Patel R, Siddiqui AH. Furosemide. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2021. Available from https://www.ncbi.nlm.nih.gov/books/NBK499921/.
6. Williams EE, Gunawardana S, Donaldson NK, Dassios T, Greenough A. Postnatal diuretics, weight gain and home oxygen requirement in extremely preterm infants. J Perinat Med. 2021;50(1):100–107. doi:10.1515/jpm-2021-0256
7. Manfredini VA, Cerini C, Clavenna A, et al.; on the behalf of study Group of Neonatal Pharmacotherapy of the Italian Society of Neonatology. Furosemide use in Italian neonatal intensive care units: a national survey. Ital J Pediatr. 2020;46(1):86. doi:10.1186/s13052-020-00851-2
8. Quinn JA, Munoz FM, Gonik B, et al.; Brighton Collaboration Preterm Birth Working Group. Preterm birth: case definition & guidelines for data collection, analysis, and presentation of immunisation safety data. Vaccine. 2016;34(49):6047–6056. doi:10.1016/j.vaccine.2016.03.045
9. Furosemide. Package insert. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2016/018902s027lbl.pdf.
10. Jackson W, Taylor G, Selewski D, Smith PB, Tolleson-Rinehart S, Laughon MM. Association between furosemide in premature infants and sensorineural hearing loss and nephrocalcinosis: a systematic review. Matern Health Neonatol Perinatol. 2018;4(1):23. doi:10.1186/s40748-018-0092-2
11. Thompson EJ, Benjamin DK, Greenberg RG, et al. Pharmacoepidemiology of Furosemide in the Neonatal Intensive Care Unit. Neonatology. 2020;117(6):780–784. doi:10.1159/000510657
12. Orth LE, O’Mara KL. Impact of early versus late diuretic exposure on metabolic bone disease and growth in premature neonates. J Pediatr Pharmacol Ther. 2018;23(1):26–33.
13. Ketkeaw K, Thaithumyanon P, Punnahitananda S. Nephrocalcinosis in very low birth weight infants: a single center experience. J Med Assoc Thai. 2004;87(Suppl 2):S72–7.
14. Dartois LL, Levek C, Grover TR, Murphy ME, Ross EL. Diuretic use and subsequent electrolyte supplementation in a level IV Neonatal Intensive Care Unit. J Pediatr Pharmacol Ther. 2020;25(2):124–130.
15. Kalikkot Thekkeveedu R, Ramarao S, Dankhara N, Alur P. Hypochloremia secondary to diuretics in preterm infants: should clinicians pay close attention? Glob Pediatr Health. 2021;8:2333794X21991014. doi:10.1177/2333794X21991014
16. Sridharan K, Al Madhoob A, Al Jufairi M, Al Ansari E, Al Marzooq R. Intravenous frusemide does not interact pharmacodynamically with Acetaminophen in critically ill preterm neonates with patent ductus arteriosus. Eur Rev Med Pharmacol Sci. 2021;25(3):1612–1615. doi:10.26355/eurrev_202102_24871
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.