Back to Journals » ClinicoEconomics and Outcomes Research » Volume 8
Cost-effectiveness of edoxaban versus rivaroxaban for stroke prevention in patients with nonvalvular atrial fibrillation (NVAF) in the US
Authors Miller J, Ye X, Lenhart GM, M. Farr A ![]() , Tran O, Kwong WJ, Magnuson E, Weintraub W
, Tran O, Kwong WJ, Magnuson E, Weintraub W
Received 4 November 2015
Accepted for publication 16 January 2016
Published 20 May 2016 Volume 2016:8 Pages 215—226
DOI https://doi.org/10.2147/CEOR.S98888
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Professor Giorgio Colombo
Jeffrey D Miller,1 Xin Ye,2 Gregory M Lenhart,1 Amanda M Farr,1 Oth V Tran,1 W Jackie Kwong,2 Elizabeth A Magnuson,3 William S Weintraub4
1Truven Health Analytics Inc, Cambridge, MA, 2Daiichi Sankyo Inc, Parsippany, NJ, 3St Luke Mid-America Heart Institute, Kansas City, MO, 4Center for Heart and Vascular Health, Christiana Care Health System, Newark, DE, USA
Background: Understanding the value of new anticoagulation therapies compared with existing therapies is of paramount importance in today’s cost-conscious and efficiency-driven health care environment. Edoxaban and rivaroxaban for stroke prevention in nonvalvular atrial fibrillation (NVAF) patients with CHADS2 scores ≥2 have been evaluated in pivotal trials versus warfarin. The relative value of edoxaban versus rivaroxaban would be of interest to health care stakeholders and patients who prefer a once-daily treatment option for long-term stroke prevention in NVAF.
Objective: To evaluate the relative cost-effectiveness of two once-daily regimens of novel oral anticoagulation therapy – edoxaban (60 mg/30 mg dose-reduced) versus rivaroxaban (20 mg/15 mg dose-reduced) – for stroke prevention in NVAF patients from a US health-plan perspective.
Materials and methods: A Markov model simulated lifetime risk and treatment of stroke, systemic embolism, major bleeding, clinically relevant nonmajor bleeding, myocardial infarction, and death in NVAF patients treated with edoxaban or rivaroxaban. Efficacy and safety data were derived from a network meta-analysis that utilized data from patients enrolled in ENGAGE AF-TIMI 48 and ROCKET-AF. Health care cost and utility data were obtained from published sources. Incremental cost-effectiveness ratios of $150,000 per quality-adjusted life year (QALY) gained were used as thresholds for “highly cost-effective”, “cost-effective”, and “not cost-effective” treatment options, respectively, as per American Heart Association/American College of Cardiology guidelines.
Results: Edoxaban was dominant relative to rivaroxaban, such that it was associated with lower total health care costs and better effectiveness in terms of QALYs in the base-case analysis. Results were supported by probabilistic sensitivity analyses that showed edoxaban as either dominant or a highly cost-effective alternative (incremental cost-effectiveness ratio <$50,000) to rivaroxaban in 88.4% of 10,000 simulations.
Conclusion: Results of this study showed that the once-daily edoxaban (60 mg/30 mg dose-reduced) regimen is a cost-saving or highly cost-effective treatment relative to rivaroxaban (20 mg/15 mg dose-reduced) for stroke prevention in NVAF patients with CHADS2 ≥2.
Keywords: edoxaban, rivaroxaban, cost-effectiveness, nonvalvular atrial fibrillation, oral anticoagulation, stroke, NOAC, SPAF, economic model, economic analysis
Introduction
Atrial fibrillation (AF) affects approximately 2.3 million people in the US, and is associated with a fivefold increase in stroke compared to patients without AF.1–3 Oral anticoagulation with the vitamin K antagonist (VKA) warfarin has been the standard of care for stroke prevention in people with nonvalvular AF (NVAF).4–6 However, VKAs have numerous limitations, including a variety of food and drug interactions, slow onset of action, high discontinuation rates, narrow therapeutic range, and variable patient dose response, which require routine laboratory monitoring.6–8 In recent years, several non-VKA novel oral anticoagulants (NOACs; dabigatran, rivaroxaban, apixaban, and edoxaban) have been approved in the US as alternatives to warfarin for stroke prevention in NVAF patients. These newer agents do not require routine monitoring, have faster onset of action, bear fewer food and drug interactions compared to VKA therapy, and offer the promise to provide practical and more convenient oral anticoagulation (OAC) treatment.6–8
As new OAC therapies come to market, the need to understand their “value” is of paramount importance in today’s cost-conscious health care environment. Recently, the American College of Cardiology/American Heart Association Task Force on Performance Measures and Task Force on Practice Guidelines proposed the inclusion of cost-effectiveness/value assessment and recommendations in practice guidelines and performance measures.9 The cost-effectiveness of warfarin therapy compared to aspirin/no therapy for stroke-prevention NVAF anticoagulation has been well established.10–12 Consensus has emerged from the large and ever-growing body of economic literature about the comparative costs and cost-effectiveness of anticoagulation therapies for stroke prevention in NVAF patients that NOACs are cost-effective alternatives to warfarin for stroke prevention in NVAF.13–19 While data on the particular value of one NOAC versus another NOAC are scant, this is a subject of growing interest to health care decision makers.
Edoxaban, a factor-Xa inhibitor, was approved in 2015 by the US Food and Drug Administration as a once-daily NOAC for stroke prevention in NVAF.20 Data from the Phase III pivotal trial ENGAGE AF-TIMI 48 have shown that a once-daily edoxaban 60 mg regimen was noninferior in the risk of stroke/systemic embolism (SE) when compared to warfarin, with significantly lower risk of major bleeding (Daiichi Sankyo, data on file, 2013).21 Among patients with creatinine clearance ≤95 mL/min, edoxaban reduced the risk of stroke/SE by 32%, with lower risk of major bleeding by 16% relative to warfarin.20 Although no head-to-head randomized controlled studies have evaluated the efficacy and safety of edoxaban in direct comparison with other NOACs, several network meta-analyses have been presented or published that indirectly compared the efficacy and safety of NOACs (including edoxaban).22–28
The objective of this study was to evaluate the relative cost-effectiveness of two once-daily NOAC-therapy options – edoxaban (60 mg/30 mg dose-reduced) versus rivaroxaban (20 mg/15 mg dose-reduced) – for stroke prevention in NVAF patients from a US health-plan perspective. We were interested in comparing edoxaban to rivaroxaban for multiple reasons. First, rivaroxaban is the most commonly prescribed NOAC in the US and dominates the NVAF stroke-prevention market.29 Second, the pivotal trials of edoxaban – ENGAGE AF-TIMI 48 (Daiichi Sankyo, data on file, 2013)21 and rivaroxaban ROCKET-AF30 – were more similar in study design than the trials for other NOACs, as both studies utilized once-daily treatment and limited enrollment to patients with CHADS2 scores ≥2. Finally, we believed the relative value of edoxaban versus rivaroxaban would be of interest to health care stakeholders and patients who prefer a once-daily treatment option for chronic stroke prevention in NVAF.
Materials and methods
Model design
A health-state transition (semi-Markov) model was developed using Microsoft Excel 2013. The Markov modeling approach has been used for other published economic evaluations of NOAC therapy in recent years, as reviewed and summarized in recent publications.13,14,16,17 The economic model compared two hypothetical NVAF patient cohorts initiating treatment with either once-daily edoxaban (60 mg/30 mg dose-reduced) regimen or once-daily rivaroxaban (20 mg/15 mg dose-reduced) for stroke prevention on a chronic basis. The patient cohorts simulated in the model consisted of CHADS2 score ≥2 patients (consistent with the study populations in ENGAGE AF-TIMI 48 (Daiichi Sankyo, data on file, 2013)21 and ROCKET-AF30) with a starting age of 72 years (the median ages of patients in ENGAGE AF-TIMI 48 and ROCKET-AF were 72 years and 73 years, respectively). These patients transitioned between discrete health states in the model to replicate the natural course of changes in health status on a monthly basis over a remaining lifetime horizon (up to maximum age of 100 years).
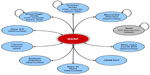
A schematic presenting the model health states and transition pathways is shown in Figure 1. All patients begin in the NVAF health state. During any given model cycle, patients can either remain in their current health state, experience a clinical event, or die. Consistent with other published models evaluating OAC therapy, the following clinical events were included in the model: ischemic stroke (first and recurrent), transient ischemic attack (TIA), SE, hemorrhagic stroke (first and recurrent), other intracranial hemorrhage (ie, subdural and epidural hematoma), major gastrointestinal bleed, other major nongastrointestinal extracranial bleed, clinically relevant nonmajor extracranial bleed, myocardial infarction (MI), event-related death, and death from other causes. Also consistent with other models,31,32 both ischemic stroke and hemorrhagic stroke were modeled as closed health states, such that after experiencing the first stroke patients would transition to a post-stroke state where they could experience only stroke recurrence or die. Nonfatal stroke events were stratified by severity: mild stroke (modified Rankin score 0–2), moderate stroke (modified Rankin score 3–4), and severe stroke (modified Rankin score 5). Patients experiencing an MI entered a post-MI health state where they were allowed to experience other clinical events, including subsequent MI. The post-stroke and post-MI states were included to account for residual deficits as a result of these events. All other clinical events were modeled as transient health states, such that patients who survived an event would return to the NVAF “well” state without any residual deficit. All clinical events have the potential of being fatal, except TIA and clinically relevant nonmajor bleeds. Institutional Review Board (IRB) approval was not required for this economic model using simulated cohorts of patients.
Clinical event risks and health state-transition probabilities
Warfarin served as the reference treatment in the Markov model, and its transition probabilities were based on the warfarin event rates stratified by CHADS2 score (as per the format of the data from the overall population in ENGAGE AF-TIMI 48 (Daiichi Sankyo, data on file, 2013)21 and reflecting differing underlying risk of thrombotic events) and consolidated into a single value weighted by CHADS2-score distribution (Table 1). The rationale for this was supported by the data from the pivotal trials of edoxaban (Daiichi Sankyo, data on file, 2013)21 and rivaroxaban,30 which showed that their relative efficacy versus warfarin did not differ by CHADS2 score, so there was no need to stratify the thrombotic event risks by CHADS2 score (Table 1). Consistent with the approach taken by other published cost-effectiveness analyses, the risk of ischemic stroke, TIA, MI, and bleeding events (including hemorrhagic stroke) in our model increased with age, using factors of 1.40,33–36 1.73,37 1.30,31,32,34,38,39 and 1.97,31,32,34,36,40 respectively, for each decade increase in age. The distribution of stroke severity was based on data from ENGAGE AF-TIMI 48 (Daiichi Sankyo, data on file, 2013).21
  | Table 1 Patient characteristics, clinical event rates, and stroke-severity distribution |
Event rates for patients receiving edoxaban or rivaroxaban were calculated based on risk ratios relative to warfarin obtained from a network meta-analysis of data from Phase III pivotal trials where rivaroxaban and edoxaban were compared to warfarin (Table 1).22 Patients either continue OAC therapy or discontinue OAC therapy temporarily or permanently at any time. Patients who discontinue edoxaban and rivaroxaban were assumed in the model to receive aspirin therapy. Event rates for second-line aspirin therapy were obtained from a published NOAC cost-effectiveness modeling study that used data from a meta-analysis of clinical trials (Table 1).41,42 The risk of recurrent ischemic or hemorrhagic stroke was independent of treatment, and was set to 2.7% for both the edoxaban- and rivaroxaban-treatment arms to be consistent with other recently published OAC-therapy economic evaluations.31,32,43
Mortality
Case-fatality rates for modeled clinical events are presented in Table 2. Cardiovascular mortality outside clinical events explicitly represented in the model was separately accounted for using a cardiovascular mortality risk-ratio estimate of 0.87 for both edoxaban versus warfarin and for rivaroxaban versus warfarin, as estimated from the network meta-analysis.22 In addition to event-related mortality, allowance was made in the model for patients to die from other causes. This underlying mortality risk unrelated to clinical events was based on age-specific US life tables.44 A multiplier of 1.34 was applied to the general mortality rate to account for the increased mortality risk for NVAF patients relative to the general population.31,32 In addition, mortality risk for patients who survived clinical events was further increased according to the mortality-risk multipliers presented in Table 2.
Treatment discontinuation
All patients were assumed to discontinue NOAC therapy temporarily for the model cycle (1 month) after an acute clinical event occurred. In addition, patients could continue OAC therapy, or discontinue OAC therapy temporarily or permanently in future model cycles subsequent to clinical events. Assumptions about permanent discontinuation of OAC therapy following clinical events mirrored those used in other published economic evaluations.31,32 All patients discontinue NOAC therapy permanently following hemorrhagic stroke or MI, which is consistent with assumptions used in other models.31,32 Fifty-six percent of patients discontinue NOAC therapy permanently and 44% temporarily stop therapy for 1 month after intracranial hemorrhage (other ICH).31,32 Following a major extracranial hemorrhage, 25% of patients will discontinue NOAC therapy permanently, and 75% will stop therapy temporarily for 1 month.31,32 Patients who discontinue NOAC therapy permanently were assumed to receive aspirin.31,32 In addition, the model assumed an annual permanent treatment discontinuation rate of 5% unrelated to clinical events to account for medication nonadherence.
Utilities and disutilities
NVAF patients entered the model with a baseline utility of 0.836, consistent with baseline utility for patients enrolled in ENGAGE-AF TIMI 48 assessed by the EQ-5D™, a standardized instrument for use as a measure of health outcome.45 Disutility associated with all clinical events was applied for the full 1-month cycle in which an acute clinical event occurred; otherwise, disutility associated with chronic health states was applied across the full duration of time in which a patient remained in a particular chronic health state (Table 3).
Costs
The wholesale acquisition cost of edoxaban (US$9.24 per day for both 60 mg and 30 mg) and rivaroxaban ($10.49 per day for both 20 mg and 15 mg) were used in the analysis.46 Health care expenditures associated with each health state in the model were derived from published literature, diagnosis-related group data from the Healthcare Cost and Utilization Project,47 and from Centers for Medicare & Medicaid Services (CMS) fee schedules, adjusted where necessary to 2014 levels using the “Medical care” component of the US Consumer Price Index (Table 4). Future costs and life years (LYs) and quality-adjusted LYs (QALYs) were discounted by 3% annually after the first year, as recommended by pharmacoeconomic guidelines published by the Academy of Managed Care Pharmacy.48
Analyses
The cost-effectiveness of edoxaban relative to rivaroxaban was assessed using an incremental cost-effectiveness ratio (ICER), which is calculated as the incremental cost per QALY gained. ICERs of <$50,000, $50,000–$150,000, and >$150,000 per QALY gained were proposed as thresholds for “high value/highly cost-effective”, “intermediate value/cost-effective”, and “low value/not cost-effective” treatment options, respectively, as per the American Heart Association/American College of Cardiology statement on cost/value methodology in clinical practice guidelines and performance measures.9
Probabilistic sensitivity analysis, which allows all model parameters to be varied simultaneously, was conducted to test the robustness of model parameter values and their impact on the ICERs. Second-order Monte Carlo simulation with 10,000 iterations where the value of each model parameter was randomly sampled from the probability distribution (either normal, γ-, lognormal, or triangular distribution, as uniquely determined for each type of model parameter) of base-case value was performed. Results were depicted on a cost-effectiveness plane and transformed into a cost-effectiveness acceptability curve. In addition, a series of one-way sensitivity analyses was performed to determine the independent impact of key model parameters on model results. The mean starting age of the cohort was varied from 60 years to 80 years. The acquisition cost of edoxaban was varied ±13.5% to match the acquisition cost of rivaroxaban on the upper end of the bound, while all clinical event costs were varied ±10% and all utility decrements varied ±25%, as is typically done in modeling sensitivity analyses. An additional one-way sensitivity analysis was performed where clinical events were varied from the lower and upper bounds of their 95% confidence intervals.
Results
Base-case analysis
In the base-case analysis, while the estimated lifetime mean number of thrombotic events per patient was similar between treatment groups, the mean number of hemorrhagic events was lower for edoxaban relative to rivaroxaban (Table 5). These findings resulted in a numerically higher mean number of QALYs for edoxaban (7.299 versus 7.238), and lower mean total health care cost for edoxaban than rivaroxaban ($45,358 versus $49,472 per patient) (Table 5). Therefore, edoxaban was economically dominant over rivaroxaban in the base-case analysis. Across both therapies, about 24% of total health care cost was attributable to acute events, 31% was attributable to time spent in chronic health states, and the remaining 45% of total costs came from the OAC drugs themselves.
Sensitivity analyses
In one-way sensitivity analyses, when the acquisition cost of edoxaban was increased by 13.5% to the same level as rivaroxaban, at $10.49 per day, edoxaban remained dominant over rivaroxaban, with cost savings of $1,382 per patient (mean per patient cost of $48,090 edoxaban versus $49,472 rivaroxaban) and higher QALYs (7.299 versus 7.238) (Table 6). The cost-effectiveness of edoxaban relative to rivaroxaban was not sensitive to age. Edoxaban remained the economically dominant therapy versus rivaroxaban when cost parameters were varied ±10% from their base values and utilities were varied ±25% from their base values. Although not reported in Table 6, edoxaban also remained the economically dominant therapy versus rivaroxaban when risks of clinical events were varied from the lower and upper bounds of their 95% confidence intervals.
In the Monte Carlo probabilistic sensitivity analysis with 10,000 model iterations, in which all model parameters were randomly sampled from their distributions, edoxaban yielded an ICER <$50,000 per QALY gained in 88.4% of the 10,000 simulations (Figure 2).
  | Figure 2 Results of the probabilistic sensitivity analysis. |
Discussion
This study assessed the cost-effectiveness of edoxaban versus rivaroxaban as treatment for stroke prevention among patients with NVAF from a US health-plan perspective. Our analysis found that edoxaban was associated with greater quality-adjusted life expectancy at lower total health care costs than rivaroxaban. This finding of economic dominance was robust under a series of one-way sensitivity analysis where event-treatment cost and health state-utility estimates were varied. The cost-effectiveness of edoxaban relative to rivaroxaban for stroke prevention was further supported by probabilistic sensitivity analysis where all model parameters were randomly sampled from their distribution, and ICER estimates fell below $50,000 per QALY gained in 88.4% of model simulations. These results were primarily driven by the lower number of nonintracranial major and clinically relevant nonmajor bleeding events in the edoxaban cohort, resulting in lower bleeding-related health care cost and bleeding-related quality-of-life impairment and mortality.
As new OAC therapies come to market, the need to understand their value compared with existing therapies is of paramount importance in today’s cost-conscious and efficiency-driven health care environment. There have been numerous published cost-effectiveness analyses comparing edoxaban and rivaroxaban with warfarin.34,45,49–59 These studies have consistently showed that edoxaban and rivaroxaban are cost-effective relative to warfarin. However, few studies have set out to assess the particular value or cost-effectiveness of one NOAC versus another NOAC, and where such analyses have been conducted (albeit using an indirect comparison approach), there has been substantial inconsistency and contradiction between their findings.13,17
Edwards et al60 found rivaroxaban to be economically dominant over dabigatran in the UK setting, but Kansal et al58 found dabigatran to dominate rivaroxaban in the Canadian setting. In the Canadian model by Wells et al,61 rivaroxaban was dominated by apixaban, yet apixaban is either a dominant strategy or a dominated strategy compared with dabigatran, depending on the dose of dabigatran evaluated. In the UK model by Lip et al,31 apixaban was cost-effective relative to both rivaroxaban and dabigatran, with ICERs well below the commonly assumed threshold of £20,000 per QALY gained. Similarly, Lanitis et al62 reported that in the French setting, apixaban dominates both rivaroxaban and dabigatran, with rivaroxaban either dominating or having favorable ICERs compared with dabigatran. By contrast, the UK model by Zheng et al63 showed dabigatran to be economically dominant over both rivaroxaban and apixaban, while Coyle et al64 found both apixaban and dabigatran dominating rivaroxaban in the Canadian setting. None of these studies were from the perspective of US health care payers. In consideration of the fact that rivaroxaban is the most commonly prescribed NOAC in the US, the relative value of edoxaban relative to rivaroxaban is an important consideration for clinicians and payers. To the best of our knowledge, this is the first study formally to evaluate the cost-effectiveness of edoxaban relative to rivaroxaban.
In the absence of head-to-head comparative trials, cost-effectiveness analysis comparing one NOAC to another requires reliance on clinical data derived from network meta-analysis that indirectly compares one treatment versus another. Several researchers have conducted network meta-analyses of pivotal trial data to assess the relative efficacy and safety of edoxaban versus other NOACs. While these studies differ in methodology and the inclusion of data from patients with CHADS2 scores of 1, they consistently showed rivaroxaban 20 mg was associated with a higher risk of major bleeding and the composite of major and clinically relevant major bleeding, than edoxaban 60 mg. To be consistent with the study population in ENGAGE AF-TIMI 48 and ROCKET-AF (both studies enrolled patients with CHADS2 scores ≥2), we purposefully chose to use data from a network meta-analysis of data from patients with CHADS2 scores ≥2, which showed that among patients with NVAF and CHADS2 scores ≥2, edoxaban (60 mg/30 mg dose-reduced) had similar efficacy to rivaroxaban for the risk of stroke and SE (relative risk 0.90, 95% confidence interval 0.70–1.16 for composite of stroke/SE) and substantially lower risk of major bleeding compared with rivaroxaban (relative risk 0.76, 95% confidence interval 0.52–1.10).22 In other indirect comparisons analyses, Skjøth et al25 and Fu et al26 drew similar conclusions that edoxaban has similar efficacy to rivaroxaban for the risk of stroke and SE, but substantially lower risk of major bleeding.
Limitations
The economic model we developed for this study has several potential limitations. First, many of the model’s data inputs were derived from a network meta-analysis of clinical trial data. Indirect treatment comparisons through network meta-analysis of clinical trials has its own inherent limitations. Although we used data from an analysis of patients with CHADS2 scores ≥2 as an attempt to minimize biases, the comparison did not control for all differences in patient baseline characteristics, such as warfarin time in therapeutic range. The relative efficacy of an NOAC to warfarin depends on the quality of warfarin control. The amount of time warfarin patients spent in the target therapeutic range was higher in ENGAGE AF-TIMI 48 (median 68.4%) than ROCKET-AF (median 58%). This variation would be expected to favor the rivaroxaban-versus-warfarin comparison rather than the edoxaban-versus-warfarin comparison, thus leading to a conservative estimate for the cost-effectiveness of edoxaban.
Another potential study limitation comes from the fact that participants in clinical trials are likely to have received closer management and had better adherence to therapy than patients who would be treated in a nontrial, real-world population. Therefore, the efficacy, safety, and tolerability observed from clinical trials may be better than what would occur in actual clinical practice.
We also note that many data parameters required for an economic model that would include dabigatran and apixaban, the other two NOACs marketed in the US, were not available in the published literature for the subgroup of patients with CHADS2 scores ≥2, as the pivotal trials of these agents included patients with CHADS2 scores ≥1. Other researchers who have attempted indirectly to compare apixaban and dabigatran to rivaroxaban have also acknowledged the challenges for adequately controlling the heterogeneity in patient baseline characteristics and study design.13,31,65–72 For similar reasons, we were unable to pursue analysis for apixaban and dabigatran. The comparative effectiveness of edoxaban relative to all marketed NOACs should be addressed in future observational studies in the real-world setting. Finally our model and analyses were based on costs and resource-use data specific for the US, and thus results are not necessarily generalizable to other countries, due to differences in health care practices and financing.
Conclusion
Our cost-effectiveness analysis over a remaining lifetime horizon provides a comprehensive assessment of the health care resources, mortality risk, and quality-of-life impairment incurred in NVAF patients receiving lifetime once-daily OAC therapy for stroke prevention. Our base-case analysis findings, along with the results of sensitivity analyses, suggest that edoxaban is an economically attractive alternative to rivaroxaban for stroke prevention in NVAF patients. In an era of evidence-based medicine, comparative effectiveness and economics research can inform health care stakeholders in their resource-allocation decisions. Future research is warranted to evaluate further the economic implications of edoxaban therapy in the real-world setting.
Disclosure
Funding for this study was provided by Daiichi Sankyo Inc. (Parsippany, NJ, USA), manufacturer of edoxaban. At the time the study was conducted, WJK and XY were employees of Daiichi Sankyo Inc.; EAM and WSW served as consultants to Daiichi Sankyo Inc. and received honoraria for their services. WSW also discloses funding in part by an Institutional Development Award (IDeA) from the National Institute of General Medical Sciences of the National Institutes of Health under grant number U54-GM104941 (PI: Binder-Macleod). JDM, GML, AMF, and OVT were employed by Truven Health Analytics Inc., which received contracted research funding from Daiichi Sankyo Inc. for conducting the study. An earlier version of this study was presented as a poster (I22) at the Academy of Managed Care Pharmacy (AMCP) Nexus 2015, Orlando, FL, October 26–29, 2015, with the abstract published in Circulation: Cardiovascular Quality and Outcomes (2015;8:A104). The authors report no other conflicts of interest in this work.
References
Go AS, Hylek EM, Phillips KA, et al. Prevalence of diagnosed atrial fibrillation in adults: national implications for rhythm management and stroke prevention: the AnTicoagulation and Risk Factors in Atrial Fibrillation (ATRIA) study. JAMA. 2001;285(18):2370–2375. | |
Gersh BJ, Tsang TS, Seward JB. The changing epidemiology and natural history of nonvalvular atrial fibrillation: clinical implications. Trans Am Clin Climatol Assoc. 2004;115:149–159; discussion 159–160. | |
Kannel WB, Benjamin EJ. Current perceptions of the epidemiology of atrial fibrillation. Cardiol Clin. 2009;27(1):13–24, vii. | |
You JJ, Singer DE, Howard PA, et al. Antithrombotic therapy for atrial fibrillation: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 Suppl):e531S–e575S. | |
Furie KL, Goldstein LB, Albers GW, et al. Oral antithrombotic agents for the prevention of stroke in nonvalvular atrial fibrillation: a science advisory for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2012;43(12):3442–3453. Errata in: Stroke. 2013;44(3):e20; Stroke. 2012;43(12):e181. | |
January CT, Wann LS, Alpert JS, et al. 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2014;64(21):e1–e76. | |
Saraf K, Morris P, Garg P, Sheridan P, Storey R. Non-vitamin K antagonist oral anticoagulants (NOACs): clinical evidence and therapeutic considerations. Postgrad Med J. 2014;90(1067):520–528. | |
Katsnelson M, Sacco RL, Moscucci M. Progress for stroke prevention with atrial fibrillation: emergence of alternative oral anticoagulants. Circulation. 2012;125(12):1577–1583. | |
Anderson JL, Heidenreich PA, Barnett PG, et al. ACC/AHA statement on cost/value methodology in clinical practice guidelines and performance measures: a report of the American College of Cardiology/American Heart Association Task Force on Performance Measures and Task Force on Practice Guidelines. Circulation. 2014;129(22):2329–2345. | |
Gage BF, Cardinalli AB, Albers GW, Owens DK. Cost-effectiveness of warfarin and aspirin for prophylaxis of stroke in patients with nonvalvular atrial fibrillation. JAMA. 1995;274(23):1839–1845. | |
Sorensen SV, Dewilde S, Singer DE, Goldhaber SZ, Monz BU, Plumb JM. Cost-effectiveness of warfarin: trial versus “real-world” stroke prevention in atrial fibrillation. Am Heart J. 2009;157(6):1064–1073. | |
Jowett S, Bryan S, Mant J, et al. Cost effectiveness of warfarin versus aspirin in patients older than 75 years with atrial fibrillation. Stroke. 2011;42(6):1717–1721. | |
Limone BL, Baker WL, Kluger J, Coleman CI. Novel anticoagulants for stroke prevention in atrial fibrillation: a systematic review of cost-effectiveness models. PLoS One. 2013;8(4):e62183. | |
Limone BL, Baker WL, Mearns ES, White CM, Kluger J, Coleman CI. Common flaws exist in published cost-effectiveness models of pharmacologic stroke prevention in atrial fibrillation. J Clin Epidemiol. 2014;67(10):1093–1102. | |
von Schéele B, Fernandez M, Hogue SL, Kwong WJ. Review of economics and cost-effectiveness analyses of anticoagulant therapy for stroke prevention in atrial fibrillation in the US. Ann Pharmacother. 2013;47(5):671–685. | |
Kasmeridis C, Apostolakis S, Ehlers L, Rasmussen LH, Boriani G, Lip GY. Cost effectiveness of treatments for stroke prevention in atrial fibrillation: focus on the novel oral anticoagulants. Pharmacoeconomics. 2013;31(11):971–980. | |
Kansal AR, Zheng Y, Pokora T, Sorensen SV. Cost-effectiveness of new oral anticoagulants in the prevention of stroke in patients with atrial fibrillation. Best Pract Res Clin Haematol. 2013;26(2):225–237. | |
Mani H, Lindhoff-Last E. New oral anticoagulants in patients with nonvalvular atrial fibrillation: a review of pharmacokinetics, safety, efficacy, quality of life, and cost effectiveness. Drug Des Devel Ther. 2014;8:789–798. | |
Ferreira J, Mirco A. Systematic review of cost-effectiveness analyses of novel oral anticoagulants for stroke prevention in atrial fibrillation. Rev Port Cardiol. 2015;34(3):179–191. | |
Daiichi Sankyo. Savaysa (edoxaban) [package insert]. Available from: http://dsi.com/prescribing-information-portlet/getPIContent?productName=Savaysa&inline=true. Accessed January 2, 2016. | |
Giugliano RP, Ruff CT, Braunwald E, et al. Edoxaban versus warfarin in patients with atrial fibrillation. N Engl J Med. 2013;369(22):2093–2104. | |
Fernandez MM, Wang J, Ye X, et al. Systematic review and network meta-analysis of the relative efficacy and safety of edoxaban versus other nonvitamin K antagonist oral anticoagulants among patients with nonvalvular atrial fibrillation and CHADS2 score ≥2. SAGE Open Med. 2015;3:1–11. | |
Messori A, Fadda V, Maratea D, Trippoli S, Marinai C. Testing the therapeutic equivalence of novel oral anticoagulants for thromboprophylaxis in orthopedic surgery and for prevention of stroke in atrial fibrillation. Int J Clin Pharmacol Ther. 2015;53(3):211–219. | |
Verdecchia P, Angeli F, Bartolini C, et al. Safety and efficacy of non-vitamin K oral anticoagulants in non-valvular atrial fibrillation: a Bayesian meta-analysis approach. Expert Opin Drug Saf. 2015;14(1):7–20. | |
Skjøth F, Larsen TB, Rasmussen LH, Lip GY. Efficacy and safety of edoxaban in comparison with dabigatran, rivaroxaban and apixaban for stroke prevention in atrial fibrillation: an indirect comparison analysis. Thromb Haemost. 2014;111(5):981–988. | |
Fu W, Guo H, Guo J, et al. Relative efficacy and safety of direct oral anticoagulants in patients with atrial fibrillation by network meta-analysis. J Cardiovasc Med (Hagerstown). 2014;15(12):873–879. | |
Assiri A, Al-Majzoub O, Kanaan AO, Donovan JL, Silva M. Mixed treatment comparison meta-analysis of aspirin, warfarin, and new anticoagulants for stroke prevention in patients with nonvalvular atrial fibrillation. Clin Ther. 2013;35(7):967–984.e2. | |
Biondi-Zoccai G, Malavasi V, D’Ascenzo F, et al. Comparative effectiveness of novel oral anticoagulants for atrial fibrillation: evidence from pair-wise and warfarin-controlled network meta-analyses. HSR Proc Intensive Care Cardiovasc Anesth. 2013;5(1):40–54. | |
Drugs.com. US pharmaceutical sales – Q4 2013. 2014. Available from: http://www.drugs.com/stats/top100/sales. Accessed January 2, 2016. | |
Patel MR, Mahaffey KW, Garg J, et al. Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med. 2011;365(10):883–891. | |
Lip GY, Kongnakorn T, Phatak H, et al. Cost-effectiveness of apixaban versus other new oral anticoagulants for stroke prevention in atrial fibrillation. Clin Ther. 2014;36(2):192–210.e20. | |
Dorian P, Kongnakorn T, Phatak H, et al. Cost-effectiveness of apixaban vs. current standard of care for stroke prevention in patients with atrial fibrillation. Eur Heart J. 2014;35(28):1897–1906. | |
Pisters R, Lane DA, Marin F, Camm AJ, Lip GY. Stroke and thromboembolism in atrial fibrillation. Circ J. 2012;76(10):2289–2304. | |
Wang Y, Xie F, Kong MC, Lee LH, Ng HJ, Ko Y. Cost-effectiveness of dabigatran and rivaroxaban compared with warfarin for stroke prevention in patients with atrial fibrillation. Cardiovasc Drugs Ther. 2014;28(6):575–585. | |
[No authors listed]. Risk factors for stroke and efficacy of antithrombotic therapy in atrial fibrillation: analysis of pooled data from five randomized controlled trials. Arch Intern Med. 1994;154(13):1449–1457. Erratum in: Arch Intern Med. 1994;154(19):2254. | |
National Institute for Health and Clinical Excellence (NICE). Apixaban (Eliquis®) for the prevention of stroke and systemic embolism in people with non-valvular atrial fibrillation. Single technology appraisal (STA) submitted by Bristol-Myers Squibb and Pfizer. 17 August 2012. Available from: http://www.nice.org.uk/guidance/ta275/documents/stroke-and-systemic-embolism-prevention-nonvalvular-atrial-fibrillation-apixaban-bristolmyers-squibb-and-pfizer2. Accessed January 2, 2016. | |
Bos MJ, van Rijn MJ, Witteman JC, Hofman A, Koudstaal PJ, Breteler MM. Incidence and prognosis of transient neurological attacks. JAMA. 2007;298(24):2877–2885. | |
Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Executive summary of the third report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). JAMA. 2001;285(19):2486–2497. | |
Freeman JV, Zhu RP, Owens DK, et al. Cost-effectiveness of dabigatran compared with warfarin for stroke prevention in atrial fibrillation. Ann Intern Med. 2011;154(1):1–11. | |
Ariesen MJ, Claus SP, Rinkel GJ, Algra A. Risk factors for intracerebral hemorrhage in the general population: a systematic review. Stroke. 2003;34(8):2060–2065. | |
Sorensen SV, Kansal AR, Connolly S, et al. Cost-effectiveness of dabigatran etexilate for the prevention of stroke and systemic embolism in atrial fibrillation: a Canadian payer perspective. Thromb Haemost. 2011;105(5):908–919. | |
Hart RG, Benavente O, McBride R, Pearce LA. Antithrombotic therapy to prevent stroke in patients with atrial fibrillation: a meta-analysis. Ann Intern Med. 1999;131(7):492–501. | |
Easton JD, Lopes RD, Bahit MC, et al. Apixaban compared with warfarin in patients with atrial fibrillation and previous stroke or transient ischaemic attack: a subgroup analysis of the ARISTOTLE trial. Lancet Neurol. 2012;11(6):503–511. Erratum in: Lancet Neurol. 2012; 11(12):1021. | |
Heron M, Hoyert DL, Murphy SL, Xu J, Kochanek KD, Tejada-Vera B. Deaths: final data for 2006. Natl Vital Stat Rep. 2009;57(14):1–134. | |
Magnuson EA, Vilain K, Wang K, et al. Cost-effectiveness of edoxaban vs warfarin in patients with atrial fibrillation based on results of the ENGAGE AF-TIMI 48 trial. Am Heart J. 2015;170(6):1140–1150. | |
First Databank. FDB MedKnowledge: drug pricing. Available from: http://www.fdbhealth.com/fdb-medknowledge-drug-pricing. Accessed January 22, 2016. | |
Agency for Healthcare Research and Quality. Healthcare Cost and Utilization Project (HCUP). 2013. Available from: http://www.ahrq.gov/research/data/hcup/index.html. Accessed January 2, 2016. | |
Academy of Managed Care Pharmacy (AMCP). The AMCP Format for Formulary Submissions: Version 3.1. Alexandria (VA): AMCP; 2012. Available from: http://amcp.org/practice-resources/amcp-format-formulary-submisions.pdf. Accessed January 2, 2016. | |
Krejczy M, Harenberg J, Wehling M, Obermann K, Lip GY. Cost-effectiveness of anticoagulation in patients with nonvalvular atrial fibrillation with edoxaban compared to warfarin in Germany. Biomed Res Int. 2015;2015:876923. | |
Rognoni C, Marchetti M, Quaglini S, Liberato NL. Edoxaban versus warfarin for stroke prevention in non-valvular atrial fibrillation: a cost-effectiveness analysis. J Thromb Thrombolysis. 2015;39(2):149–154. | |
Mearns E, Doleh Y, Coleman CI. Cost-effectiveness of high-dose edoxaban compared to adjusted-dose warfarin for prevention of stroke and systemic embolism in non-valvular atrial fibrillation. Poster presented at: European Society for Cardiology Congress 2014; August 30–September 3, 2014; Barcelona, Spain. | |
Mensch A, Stock S, Stollenwerk B, Müller D. Cost effectiveness of rivaroxaban for stroke prevention in German patients with atrial fibrillation. Pharmacoeconomics. 2015;33(3):271–283. | |
Morais J, Aguiar C, McLeod E, Chatzitheofilou I, Fonseca Santos I, Pereira S. Cost-effectiveness of rivaroxaban for stroke prevention in atrial fibrillation in the Portuguese setting. Rev Port Cardiol. 2014;33(9):535–544. | |
Krejczy M, Harenberg J, Marx S, Obermann K, Frölich L, Wehling M. Comparison of cost-effectiveness of anticoagulation with dabigatran, rivaroxaban and apixaban in patients with non-valvular atrial fibrillation across countries. J Thromb Thrombolysis. 2014;37(4):507–523. | |
Rognoni C, Marchetti M, Quaglini S, Liberato NL. Apixaban, dabigatran, and rivaroxaban versus warfarin for stroke prevention in non-valvular atrial fibrillation: a cost-effectiveness analysis. Clin Drug Investig. 2014;34(1):9–17. | |
Kleintjens J, Li X, Simoens S, et al. Cost-effectiveness of rivaroxaban versus warfarin for stroke prevention in atrial fibrillation in the Belgian healthcare setting. Pharmacoeconomics. 2013;31(10):909–918. | |
Harrington AR, Armstrong EP, Nolan PE Jr, Malone DC. Cost-effectiveness of apixaban, dabigatran, rivaroxaban, and warfarin for stroke prevention in atrial fibrillation. Stroke. 2013;44(6):1676–1681. | |
Kansal AR, Sharma M, Bradley-Kennedy C, et al. Dabigatran versus rivaroxaban for the prevention of stroke and systemic embolism in atrial fibrillation in Canada: comparative efficacy and cost-effectiveness. Thromb Haemost. 2012;108(4):672–682. | |
Lee S, Anglade MW, Pham D, Pisacane R, Kluger J, Coleman CI. Cost-effectiveness of rivaroxaban compared to warfarin for stroke prevention in atrial fibrillation. Am J Cardiol. 2012;110(6):845–851. | |
Edwards SJ, Hamilton V, Nherera L, Trevor N, Barton S. Rivaroxaban for the Prevention of Stroke and Systemic Embolism in People with Atrial Fibrillation: A Single Technology Appraisal. London: BMJ-TAG; 2011. | |
Wells G, Coyle D, Cameron C, et al. Therapeutic Review: Safety, Effectiveness, and Cost-Effectiveness of New Oral Anticoagulants Compared with Warfarin in Preventing Stroke and Other Cardiovascular Events in Patients with Atrial Fibrillation. Ottawa: Canadian Collaborative for Drug Safety, Effectiveness and Network Meta-Analysis; 2012. Available at http://cadth.ca/media/pdf/NOAC_Therapeutic_Review_final_report.pdf. Accessed January 2, 2016. | |
Lanitis T, Cotté FE, Gaudin AF, Kachaner I, Kongnakorn T, Durand-Zaleski I. Stroke prevention in patients with atrial fibrillation in France: comparative cost-effectiveness of new oral anticoagulants (apixaban, dabigatran, and rivaroxaban), warfarin, and aspirin. J Med Econ. 2014;17(8):587–598. | |
Zheng Y, Sorensen SV, Gonschior AK, et al. Comparison of the cost-effectiveness of new oral anticoagulants for the prevention of stroke and systemic embolism in atrial fibrillation in a UK setting. Clin Ther. 2014;36(12):2015–2028.e2. | |
Coyle D, Coyle K, Cameron C, et al. Cost-effectiveness of new oral anticoagulants compared with warfarin in preventing stroke and other cardiovascular events in patients with atrial fibrillation. Value Health. 2013;16(4):498–506. | |
Cope S, Clemens A, Hammès F, Noack H, Jansen JP. Critical appraisal of network meta-analyses evaluating the efficacy and safety of new oral anticoagulants in atrial fibrillation stroke prevention trials. Value Health. 2015;18(2):234–249. | |
Lip GY, Larsen TB, Skjøth F, Rasmussen LH. Indirect comparisons of new oral anticoagulant drugs for efficacy and safety when used for stroke prevention in atrial fibrillation. J Am Coll Cardiol. 2012;60(8):738–746. | |
Cameron C, Coyle D, Richter T, et al. Systematic review and network meta-analysis comparing antithrombotic agents for the prevention of stroke and major bleeding in patients with atrial fibrillation. BMJ Open. 2014;4(6):e004301. | |
Pink J, Pirmohamed M, Hughes DA. Comparative effectiveness of dabigatran, rivaroxaban, apixaban, and warfarin in the management of patients with nonvalvular atrial fibrillation. Clin Pharmacol Ther. 2013;94(2):269–276. | |
Baker WL, Phung OJ. Systematic review and adjusted indirect comparison meta-analysis of oral anticoagulants in atrial fibrillation. Circ Cardiovasc Qual Outcomes. 2012;5(5):711–719. | |
Schneeweiss S, Gagne JJ, Patrick AR, Choudhry NK, Avorn J. Comparative efficacy and safety of new oral anticoagulants in patients with atrial fibrillation. Circ Cardiovasc Qual Outcomes. 2012;5(4):480–486. | |
Rasmussen LH, Larsen TB, Graungaard T, Skjøth F, Lip GY. Primary and secondary prevention with new oral anticoagulant drugs for stroke prevention in atrial fibrillation: indirect comparison analysis. BMJ. 2012;345:e7097. | |
Mantha S, Ansell J. An indirect comparison of dabigatran, rivaroxaban and apixaban for atrial fibrillation. Thromb Haemost. 2012;108(3):476–484. | |
Canestaro WJ, Patrick AR, Avorn J, et al. Cost-effectiveness of oral anticoagulants for treatment of atrial fibrillation. Circ Cardiovasc Qual Outcomes. 2013;6(6):724–731. | |
Sullivan PW, Lawrence WF, Ghushchyan V. A national catalog of preference-based scores for chronic conditions in the United States. Med Care. 2005;43(7):736–749. | |
Sullivan PW, Slejko JF, Sculpher MJ, Ghushchyan V. Catalogue of EQ-5D scores for the United Kingdom. Med Decis Making. 2011;31(6):800–804. | |
Eckman MH, Rosand J, Greenberg SM, Gage BF. Cost-effectiveness of using pharmacogenetic information in warfarin dosing for patients with nonvalvular atrial fibrillation. Ann Intern Med. 2009;150(2):73–83. | |
Vilain KR, Magnuson EA, Li H, et al. Costs and cost-effectiveness of carotid stenting versus endarterectomy for patients at standard surgical risk: results from the Carotid Revascularization Endarterectomy Versus Stenting Trial (CREST). Stroke. 2012;43(9):2408–2416. | |
You JH. Novel oral anticoagulants versus warfarin therapy at various levels of anticoagulation control in atrial fibrillation – a cost-effectiveness analysis. J Gen Intern Med. 2014;29(3):438–446. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.