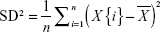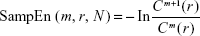Back to Journals » Clinical Interventions in Aging » Volume 12
Correlation between movement complexity during static standing and balance function in institutionalized older adults
Authors Yamagata M, Ikezoe T, Kamiya M, Masaki M, Ichihashi N
Received 14 January 2017
Accepted for publication 11 February 2017
Published 8 March 2017 Volume 2017:12 Pages 499—503
DOI https://doi.org/10.2147/CIA.S132425
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Richard Walker
Momoko Yamagata,1 Tome Ikezoe,1 Midori Kamiya,1 Mitsuhiro Masaki,2,3 Noriaki Ichihashi1
1Human Health Sciences, Graduate School of Medicine, Kyoto University, Kyoto, 2Department of Physical Therapy, 3Institute for Human Movement and Medical Sciences, Niigata University of Health and Welfare, Niigata, Japan
Purpose: Sample entropy (SampEn) is an analysis to evaluate movement complexity of the center of pressure (COP). A lower value of SampEn indicates lower complexity of COP variability, that is, rigidity, and lower degrees of freedom. Previous studies reported the association of increased SampEn with improved standing balance ability in young subjects. However, no studies have examined these relationships among older adults. Thus, we aimed to investigate the relationship between SampEn and standing balance ability in older adults.
Subjects and methods: The subjects were 33 institutionalized older adults (aged 82.2±6.5 years). COP during static standing was measured. The standard deviation (SD) values of COP and SampEn in the sagittal and frontal planes were calculated using time series data. One-leg standing test (OLST), functional reach (FR) test, and lateral reach (LR) test were also measured to evaluate standing balance ability.
Results: OLST, FR, and LR were 6.5±8.3 s, 19.8±5.9 cm, and 18.2±6.4 cm, respectively. Pearson correlation analysis revealed that SampEn in the sagittal plane significantly correlated with OLST (r=-0.35) and FR (r=-0.36). However, SampEn in the frontal plane and SD of COP in both sagittal and frontal planes had no relationship with any of the clinical balance tests.
Conclusion: Lower SampEn implies rigidity for postural control. In the present study, it was found that lower SampEn in the sagittal plane was related to a higher balance function, which suggests that older adults utilized body rigidity to maintain postural stability as a compensative strategy.
Keywords: older adults, posture, balance, standing, complexity, entropy
Introduction
The center of pressure (COP) displacement is commonly used as an index for postural stability. The magnitude of COP variability during static standing, which reflects balance function to maintain the center of mass within the base of support,1 is influenced by aging or environment.2,3 Generally, a larger COP variability indicates increased postural instability. However, some previous studies found no correlation between the magnitude of COP variability and balance ability.4 Hughes et al4 showed that although the magnitude of COP variability such as the area of ellipse and path length of COP has been associated with sensorimotor deficits, it had no relationship with standing balance measurements such as the functional reach (FR) test in older adults. Based on previous investigations, it has been pointed out that the magnitude of COP variability may not reflect balance function during static standing.5,6 On the other hand, some authors focus on the complexity of COP variability in postural control.7
The movement complexity in the system of postural control, which is different from random noise, is determined by the behavioral system factors, such as integration of the sensory signals and muscle activities, and physiological system factors, such as the control of blood pressure and heart rate.8,9 The change in the complexity of the postural control system is associated with the change in the ability to adjust to perturbations or change in environments.10 Thus, the complexity is crucial to evaluate adaptive responses or strategy for postural control.
In the framework of analysis for the complexity, entropy is defined as a quantity of the predictability of complex dynamics system. Recently, sample entropy (SampEn) is being increasingly used to measure COP variability in time series during quiet standing. SampEn is an effective method for evaluating postural control because it has statistically little bias and produces consistent results even with small data points.11 A smaller value for SampEn is associated with smaller complexity or larger regularity of physical movement.12,13 As complexity is crucial to having flexible adaptations to one’s surroundings, this lower complexity of physical movement translates to lower flexibility and higher rigidity for postural control.12,13 Conversely, a larger SampEn, which is increased complexity, is interpreted as increased self-organization and effective strategy in postural control.12,13 A previous study demonstrated that SampEn increases after standing balance training for postural control in healthy young adults,14 which suggests that the complexity of COP variability may be associated with their standing balance ability. However, there are no studies investigating the relationship between the standing balance ability and SampEn during quiet standing in older adults. Generally, standing balance ability is evaluated by one-leg standing test (OLST), FR, or lateral reach (LR) in the clinical field, which are useful tests because of the high reliability and easiness to access.5
Another study that examined the muscle activity around the ankle joint when reaching forward in standing position indicated that older adults showed greater muscle coactivation compared to healthy young adults.15 This indicates that older adults with poor balance ability often utilize rigid postural strategy for maintaining postural stability in the sagittal plane. However, the postural strategy in the frontal plane among older adults with poor balance ability is unclear.
In the current study, SampEn was used as an index to assess the movement complexity as a strategy for postural control during quiet standing. The purpose of this study was to investigate the correlation between the complexity and magnitude of COP variability and standing balance ability in older adults living in care facilities. We hypothesized that 1) the complexity of COP variability is more closely related to the standing balance ability rather than the magnitude of COP variability among older adults and 2) older adults with poor balance ability apply lower complexity, that is, rigid strategy for postural control to increase stability during standing.
Subjects and methods
Subjects
A total of 33 healthy older adults (25 females and 8 males; aged 82.2±6.5 years) living in two nursing homes in Kyoto, Japan, participated in this study (Table 1). The subjects were able to ambulate independently or with an assistive device and did not have an unstable physical condition. The subjects with physical dysfunctions that may influence outcome measures, such as neurological impairment or severe musculoskeletal impairment, were excluded. The study was approved by the ethical review board of Graduate School of Medicine, Kyoto University, Kyoto, Japan. All participants provided a written informed consent prior to this study.
  | Table 1 Characteristics of participants (N=33) |
Measurements of postural sway and SampEn
The COP variability during quiet standing was assessed using the foot pressure plate (Win-Pod; Finggal Link Co., Tokyo, Japan) in eyes-open condition. Subjects stood on the plate with their feet shoulder-width apart and their arms resting comfortably at their side. They were instructed to keep their balance to sway naturally without any intentional motion. The COP trajectories in the frontal plane (X-direction) and sagittal plane (Y-direction) were recorded during 35 s at a sampling rate of 50 Hz, and the data of first 5 s were discarded.
The standard deviation (SD) values for COP trajectories were calculated in the frontal and sagittal planes (X-SD and Y-SD, respectively). The X-SD and Y-SD values were used to represent the magnitude of COP variability, which was defined as an index for postural sway.2 The SD was calculated using the following formula:
|
where n is the total number of data and 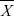 is the mean of the values.
is the mean of the values.
The SampEn for COP complexity was calculated in the frontal and sagittal planes (X-SampEn and Y-SampEn, respectively) using MATLAB (MathWorks). SampEn, which is used to analyze complex stochastic systems, including deterministic and random processes, was used to represent regularity and complexity of COP time series.13 SampEn is determined as the negative of the natural logarithm of conditional probability, which is a sequence of data points, having repeated itself within a tolerance r for a window length m, which will also repeat itself for m+1 points, without allowing self-matches.16 SampEn was calculated for each time series as follows:
|
where m is the length of a repeated vector, r is the similarity criterion, and N is the dataset length of the time series used to calculate SampEn. Here, SampEn was quantified using m=3 and r=0.3.17 Lower SampEn values imply more regular and less complex COP time series. Conversely, higher SampEn values imply more complex and random time series.
Clinical tests of standing balance ability
OLST, FR, and LR were measured as clinical tests of standing balance ability. OLST was used to assess the static balance function during standing on a narrow base of support.18 Subjects were instructed to keep the balance on their dominant foot with eyes open and their arms resting comfortably at both sides. The time before the supporting foot was shifted or the raised foot touched the ground was measured using a stopwatch. The measurement was conducted twice for a maximum of 60 s, and the maximum value was recorded.
FR and LR tests were used to evaluate dynamic balance function in the sagittal and frontal planes, respectively.4,19 FR and LR tests measured the distance that subjects could reach forward or to the right side beyond arm’s length while maintaining a fixed base. Two trials were performed, and the greater value was recorded.
Statistical analysis
Pearson correlation coefficient was used to investigate the relationship of postural sway (X-SD and Y-SD) and SampEn (X-SampEn and Y-SampEn) with balance ability (OLST, FR, and LR). SPSS 18 (PASW Statistics; SPSS Inc., Chicago, IL, USA) software was used for the data analysis. The statistical significance was set at 0.05.
Results
SD values in the frontal and sagittal planes were 5.5±3.6 cm and 7.4±3.7 cm, respectively. SampEn values in the frontal and sagittal planes were 1.0±0.3 and 0.8±0.2, respectively. The results of clinical balance tests were 6.5±8.3 s for OLST, 19.8±5.9 cm for FR, and 18.2±6.4 cm for LR (Table 2).
The Pearson correlation coefficient analysis revealed that SampEn in the sagittal plane was significantly and negatively associated with OLST (r=−0.35, P<0.05) and FR (r=−0.36, P<0.05), but not with LR (Table 3). There was no significant relationship of SampEn in the frontal plane or SD in either plane to OLST, FR, or LR (Table 3).
Discussion
The current study is the first research to investigate the relationship between SampEn, which is defined as the index of complexity of COP variability, and the standing balance ability in older adults living in care facilities who have lower balance function. Our results indicated that SampEn in the sagittal plane was related to the standing balance ability, although there were no relationships between the magnitudes of COP variability (the SD values for COP) and standing balance ability. In addition, the results of clinical balance tests were 19.8±5.9 cm for FR and 6.5±8.3 s for OLST in this study. The mean values among community-dwelling older adults were 34.13 cm20 for FR and 14.2 s18 for OLST; thus, we assumed that older adults in this study had relatively poor balance ability. These results support our hypothesis that standing balance ability is related to the complexity of COP variability rather than the magnitude of COP variability and that rigid strategy for postural control may lead to good balance ability among older adults with poor balance.
In this study, older adults with a lower value of SampEn in the sagittal plane had high values for clinical balance tests such as OLST and FR. On the other hand, lower SampEn is known to represent more regular pattern for COP variability, that is, the rigid strategy, and lower adjustment to perturbations.21,22 Previous studies showed that older adults utilized greater muscle coactivation or joint rigidity as their postural strategy during difficult conditions to maintain balance, such as standing on a narrow base of support.23,24 Furthermore, greater muscle coactivation during forward-reaching task has also been shown in older adults compared with healthy young adults.15 This finding suggests that rigid strategy for maintaining balance may be an important compensative strategy for older adults, especially for those who have lower postural control.24,25 Our results support these reports and suggest that older adults who utilize rigid strategy as compensative strategy for maintaining posture may have greater balance ability.
This study showed that standing balance ability was associated with SampEn in the sagittal plane, but not with SampEn in the frontal plane. Movements in the sagittal plane have larger degrees of freedom, such as in the hip, knee, and ankle joint, than those in the frontal plane. Therefore, it was also shown that the effects or the adaptive response for difficult environment on postural control were greater in the sagittal plane and that the postural stability in the frontal plane was relatively maintained even under perturbations.26,27 In our measurements of SampEn, the base of support during standing in the frontal plane was larger than that in the sagittal plane, since subjects stood with their feet shoulder-width apart. Therefore, it is expected that maintaining stability in the frontal plane was relatively easier even for older adults, which may lead to no correlation between SampEn in the frontal plane and standing balance ability.
In our study, there was no correlation between SD values for COP variability and standing balance ability. Previous studies showed that the magnitude of COP variability had less correlation with the clinical balance tests such as FR4 or OLST,28 which is in agreement with the results of the present study. The magnitude of COP variability, which is commonly used as an index of postural control, does not include their time structure in the analytical process. Considering this fact, the magnitude of COP variability might be insufficient to reflect the postural control strategy.7 Our results suggest that standing balance ability in older adults with poor balance may be more closely related to the complexity rather than the magnitude of the COP variability.
The limitation of this study is that subjects living in care facilities have lower balance ability compared to community-dwelling older adults. It is possible not to observe the same result for all older adults owing to differences in the postural balance ability. Future studies are required to clarify the associations between movement complexity and balance ability among older adults, including community-dwelling healthier elderly people with higher balance function as control group for comparison.
Conclusion
Our results showed that lower SampEn in the sagittal plane was related to the higher balance function, which suggests that older adults with poor balance might be used to adopt the rigid strategy to maintain balance. Moreover, the standing balance ability for older adults may be more closely associated with the complexity rather than the magnitude of COP variability.
Acknowledgment
We thank special nursing home “Sawarabi-en” and “Kenko-en” for recruiting the older adults.
Author contributions
MY carried out the designing of the study and wrote the initial draft of the manuscript. TI participated in the design of the study and assisted in the preparation of the manuscript. MK and MM contributed to analysis and interpretation of data. NI participated in the design and coordination of the study and helped to draft the manuscript. All authors contributed toward data analysis, drafting and revising the paper and agree to be accountable for all aspects of the work. All authors read and approved the final manuscript.
Disclosure
The authors report no conflicts of interest in this work.
References
Winter DA, Patla AE, Ishac M, Gage WH. Motor mechanisms of balance during quiet standing. J Electromyogr Kinesiol. 2003;13(1):49–56. | ||
Amiridis IG, Hatzitaki V, Arabatzi F. Age-induced modifications of static postural control in humans. Neurosci Lett. 2003;350(3):137–140. | ||
Redfern MS, Moore PL, Yarsky CM. The influence of flooring on standing balance among older persons. Hum Factors. 1997;39(3):445–455. | ||
Hughes MA, Duncan PW, Rose DK, Chandler JM, Studenski SA. The relationship of postural sway to sensorimotor function, functional performance, and disability in the elderly. Arch Phys Med Rehabil. 1996;77(6):567–572. | ||
Mancini M, Horak FB. The relevance of clinical balance assessment tools to differentiate balance deficits. Eur J Phys Rehabil Med. 2010;46(2):239–248. | ||
Horak FB, Henry SM, Shumway-Cook A. Postural perturbations: new insights for treatment of balance disorders. Phys Ther. 1997;77(5):517–533. | ||
Cavanaugh JT, Mercer VS, Stergiou N. Approximate entropy detects the effect of a secondary cognitive task on postural control in healthy young adults: a methodological report. J Neuroeng Rehabil. 2007;4(42):1–7. | ||
Kaplan DT, Furman MI, Pincus SM, Ryan SM, Lipsitz LA, Goldberger AL. Aging and the complexity of cardiovascular dynamics. Biophys J. 1991;59(4):945–949. | ||
Lipsitz LA. Dynamics of stability: the physiologic basis of functional health and frailty. J Gerontol A Biol Sci Med Sci. 2002;57(3):B115–B125. | ||
Seigle B, Ramdani S, Bernard PL. Dynamical structure of center of pressure fluctuations in elderly people. Gait Posture. 2009;30(2):223–226. | ||
Richman JS, Moorman JR. Physiological time-series analysis using approximate entropy and sample entropy. Am J Physiol Heart Circ Physiol. 2000;278(6):H2039–H2049. | ||
Georgoulis AD, Moraiti C, Ristanis S, Stergiou N. A novel approach to measure variability in the anterior cruciate ligament deficient knee during walking: the use of the approximate entropy in orthopaedics. J Clin Monit Comput. 2006;20(1):11–18. | ||
Donker SF, Roerdink M, Greven AJ, Beek PJ. Regularity of center-of-pressure trajectories depends on the amount of attention invested in postural control. Exp Brain Res. 2007;181(1):1–11. | ||
Lakhani B, Mansfield A. Visual feedback of the centre of gravity to optimize standing balance. Gait Posture. 2015;41(2):499–503. | ||
Nagai K, Yamada M, Uemura K, Yamada Y, Ichihashi N, Tsuboyama T. Differences in muscle coactivation during postural control between healthy older and young adults. Arch Gerontol Geriatr. 2011;53(3):338–343. | ||
Pincus SM, Gladstone IM, Ehrenkranz RA. A regularity statistic for medical data analysis. J Clin Monit. 1991;7(4):335–345. | ||
Ramdani S, Seigle B, Lagarde J, Bouchara F, Louis P. On the use of sample entropy to analyze human postural sway data. Med Eng Phys. 2009;31(8):1023–1031. | ||
Bohannon RW, Larkin PA, Cook AC, Gear J, Singer J. Decrease in timed balance test scores with aging. Phys Ther. 1984;64(7):1067–1070. | ||
Thapa PB, Gideon P, Fought RL, Kormicki M, Ray WA. Comparison of clinical and biomechanical measures of balance and mobility in elderly nursing home residents. J Am Geriatr Soc. 1994;42(5):493–500. | ||
Isles RC, Choy NLL, Steer M, Nitz JC. Normal values of balance tests in women aged 20–80. J Am Geriatr Soc. 2004;52(8):1367–1372. | ||
Gerontol J, Sci AB, Sci M, et al. Frailty and the degradation of complex balance dynamics during a dual-task protocol. J Gerontol A Biol Sci Med Sci. 2009;64(12):1304–1311. | ||
Schmit JM, Regis DI, Riley MA. Dynamic patterns of postural sway in ballet dancers and track athletes. Exp Brain Res. 2005;163(3):370–378. | ||
Hsu WL, Lin KH, Yang RS, Cheng CH. Use of motor abundance in old adults in the regulation of a narrow-based stance. Eur J Appl Physiol. 2014;114(2):261–271. | ||
Benjuya N, Melzer I, Kaplanski J. Aging-induced shifts from a reliance on sensory input to muscle cocontraction during balanced standing. J Gerontol A Biol Sci Med Sci. 2004;59(2):166–171. | ||
Hay L, Bard C, Fleury M, Teasdale N. Availability of visual and proprioceptive afferent messages and postural control in elderly adults. Exp Brain Res. 1996;108(1):129–139. | ||
Connor SMO, Kuo AD. Direction-dependent control of balance during walking and standing. J Neurophysiol. 2009;102(3):1411–1419. | ||
Iqbal K. Mechanisms and models of postural stability and control. Conf Proc IEEE Eng Med Biol Soc. 2011;2011:7837–7840. | ||
Gil AWO, Oliveira MR, Coelho VA, Carvalho CE, Teixeira DC, da Silva RA. Relationship between force platform and two functional tests for measuring balance in the elderly. Rev Bras Fisioter. 2011;15(6):429–435. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.