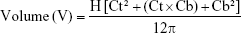Back to Journals » Clinical Interventions in Aging » Volume 13
Comparison of the effectiveness of complex decongestive therapy and compression bandaging as a method of treatment of lymphedema in the elderly
Authors Zasadzka E , Trzmiel T , Kleczewska M, Pawlaczyk M
Received 9 December 2017
Accepted for publication 24 February 2018
Published 14 May 2018 Volume 2018:13 Pages 929—934
DOI https://doi.org/10.2147/CIA.S159380
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Richard Walker
Ewa Zasadzka,1 Tomasz Trzmiel,1 Maria Kleczewska,2 Mariola Pawlaczyk1
1Department of Geriatric Medicine and Gerontology, Karol Marcinkowski University of Medical Sciences, Poznan, Poland; 2Day Rehabilitation Center, Hospicjum Palium, Poznań, Poland
Background: Lymphedema is a chronic condition which significantly lowers the quality of patient life, particularly among elderly populations, whose mobility and physical function are often reduced.
Objectives: The aim of the study was to compare the effectiveness of multi-layer compression bandaging (MCB) and complex decongestive therapy (CDT), and to show that MCB is a cheaper, more accessible and less labor intensive method of treating lymphedema in elderly patients.
Patients and methods: The study included 103 patients (85 women and 18 men) aged ≥60 years, with unilateral lower limb lymphedema. The subjects were divided into two groups: 50 treated with CDT and 53 with MCB. Pre- and post-treatment BMI, and average and maximum circumference of the edematous extremities were analyzed.
Results: Reduction in swelling in both groups was achieved after 15 interventions. Both therapies demonstrated similar efficacy in reducing limb volume and circumference, but MCB showed greater efficacy in reducing the maximum circumference.
Conclusion: Compression bandaging is a vital component of CDT. Maximum lymphedema reduction during therapy and maintaining its effect cannot be achieved without it. It also demonstrates its effectiveness as an independent method, which can reduce therapy cost and accessibility.
Keywords: lymphedema, elderly, therapy, compression bandaging
Introduction
Lymphedema is a distressing medical problem which significantly lowers the quality of patient life. Primary and secondary forms of lymphedema may be distinguished. Primary lymphedema occurs rarely (in approximately 1 in 100,000 people), and is the consequence of inherited genetic predisposition and abnormality in the construction of the lymphatic system.1,2 Secondary lymphedema occurs more frequently. In the developing countries, its etiology is associated with parasitic infections which affect as many as 300 million people globally,3 making them the most common cause of lymphedema worldwide. In the developed countries, where parasitic infections are rare, lymphedema is one of the major complications of cancer treatment. According to various studies, lymphedema following cancer treatment appears in up to 50% of the affected cases.1,3,4
Lymphedema causes functional impairment, increases body weight (leading to the asymmetric overload of the motor system), promotes the development of skin and subcutaneous tissue infections, and negatively affects patient psyche and self-perception, thus reducing the quality of patient life.5,6 This is particularly important for elderly populations, whose mobility and physical functions are often already reduced due to progressive degenerative changes.7 Complex decongestive therapy (CDT) remains the gold standard in the therapy of lymphedema.5 The first stage is performed by therapists in specialized centers and includes manual lymphatic drainage, compression therapy, exercise, and skin care. Manual lymphatic drainage (MLD) is performed in an upward direction to encourage the lymphatic flow. It mainly involves the use of pressures and movements directed toward the nearest groups of lymph nodes, proximal to the drainage area. It is important to start the drainage from the central part and next move to more distal parts of the body. Compression therapy increases tissue pressure by inducing external compressive pressure using bandaging or wearing compression garments. Multi-layer compression bandaging (MCB) uses bandages of varying degree of compression, additional materials, and support materials, eg, cotton-wool bandages.8–11 The second phase, consisting of self-administered lymphatic massage, daily use of compression garments, and self-directed continuation of the exercises, should be implemented only after the completion of the first phase.9,10 CDT is a costly treatment which may only be carried out in specialized centers. It is often associated with burdensome commute and prolonged wait for admission to the center, which in the case of elderly people may further limit the accessibility of this treatment. The estimated cost of therapy for one patient may exceed 2,500 EUR annually.12 Thus, the search for other methods and comparison of their effectiveness are both necessary.
The aim of the study was to investigate the effectiveness of MCB as a cheaper, more accessible and less labor-intensive method of treating lymphedema as compared to CDT in elderly populations.
Patients and methods
The study included 103 patients (85 women and 18 men; aged ≥60 years) with unilateral, lower limb lymphedema, treated at the Poznań Rehabilitation Centre in 2016. The subjects were recruited among patients reporting to the center for the treatment of lymphedema. Primary lymphedema was diagnosed in 5 and secondary lymphedema in 98 patients (73 after radical lymph node dissection and radiotherapy due to cancer and 25 with damage of the lymph vessels caused by trauma or inflammation). Unilateral lower limb lymphedema, untreated for the last 12 months, and age ≥60 constituted the inclusion criteria. The exclusion criteria were as follows: active neoplastic disease, chronic inflammation, and arteriosclerosis confirmed on ultrasound. Written informed consent was obtained from all participants. The patients were randomly divided into two groups: the CDT group – 50 subjects treated with the CDT method, and the MCB group – 53 subjects treated with the MCB method. The same bandaging scheme was used for both groups (Table 1). Compression was maintained for 21 to 23 hours a day. Emollients were applied prior to bandaging to avoid skin damage which may result from wearing a compression bandage. The therapy included 15 interventions performed by qualified therapists 5 days a week (Monday to Friday), for the duration of 3 weeks.
Prior to the start of therapy, medical records were reviewed, edema was evaluated, and BMI was calculated for all patients. The circumference of each limb was measured immediately before treatment and after 3 weeks (following 15 treatments), with the same tape measure (accurate to 1 mm), and in the same exact places, in accordance with the standard procedure.13 The measurement of the lower limb circumference started with the marking (in the standing position) of a point located 2 cm proximal to the center of the medial malleolus. Then, the measurement points were marked every 4 cm in the proximal direction to the point 2 cm below the gluteal line. After marking all points, the limbs were measured in the lying position, so that the upper edge of the tape measure was in line with the measuring point. All measurements were performed by certified lymphedema therapists.
Based on the measurements of the points and circumferences, the volume of the extremities affected by lymphedema was calculated using the formula for the volume of a truncated cone. The volume of individual segments was calculated using the following formula:
|
where H is the distance between the individual measuring points, ie, the height of the segment, Ct is the circumference at the top of the segment, and Cb is the circumference at the bottom of the segment.14 The volume of the entire measured limb was calculated by adding the volumes of all the segments.14–16 Circumferences were given in cm, and volumes in cm3. BMI, average and maximum limb circumference, and limb volume were analyzed before and after (last day) the treatment.
This study was approved by the Ethics Committee of Poznan University of Medical Sciences (No. 487/16).
Statistical analysis
Statistical analysis was performed using Statistica 12. The Shapiro-Wilk test was used to check normality of the distribution. The selected data were presented by mean and standard deviation of the evaluated parameters. The Mann–Whitney test for independent variables was used to compare two groups of quantitative variables. The Wilcoxon parallel test was used to compare two dependent variables. A p-value of <0.05 was considered statistically significant.
Results
The study included 103 patients, aged 60–80 years (mean: 68.4±6.4), 85 (82.5%) women (mean 69.0±6.5 years, range 60.0–80.0) and 18 (17.5%) men (mean 65.4±4.9 years, range 60.0–73.0). The women were significantly older than the men (p=0.033). Mean age was 68±6.7 years in the CDT group (50 subjects: 46 women and 7 men), and 68.8±6.0 years in the MCB group (53 subjects: 42 women and 11 men). The difference was not statistically significant (p=0.382).
Significant differences in lower limb dimensions before and after treatment were found in both groups (Table 2). In the CDT group, the maximum circumference before and after therapy ranged from 28.0 to 75.0 cm (mean 51.4±12.9 cm, median 50.7 cm) and from 26.5 to 69.5 cm (mean 48.9±11.6 cm, median 48.8 cm), respectively (p<0.001). Mean circumference before and after therapy was 38.2±8.6 cm and 35.0±6.1 cm, respectively (p<0.001). Mean volume before and after therapy was 7,419.4±3,721.9 cm3 and 6,402.5±3,106.5 cm3, respectively (p<0.001). In the MCB group, the maximum circumference before and after treatment ranged from 34.0 to 83.3 cm (mean 47.2±13.2 cm, median 41.9 cm), and from 29.3 to 79.7 cm (mean 43.7±12.7 cm, median 39.6 cm), respectively (p<0.001). Mean circumference before and after therapy was 35.8±6.7 cm and 32.9±6.6 cm, respectively (p<0.001). Mean volume before and after therapy was 5,858.0±3,988.1 cm3, and 5,041.9±3,718.0 cm3, respectively (p<0.001). Both therapies resulted in notable improvement of the patient condition, ie, decreased lymphedema after 15 days of treatment. No significant differences in BMI among women and men in the study group were observed.
  | Table 2 Comparison of the lower limbs dimensions before and after the therapy |
Table 3 shows the BMI parameters of the lymphedema measurements in both groups. The analysis of the efficacy in reducing the volume and medium circumference of the affected limbs revealed similar results for both treatments. However, the analysis of the reduction of maximum circumferences showed significantly better results in the CBG group.
Discussion
MCB is always performed with lymphatic drainage, as one of the components of CDT, in every rehabilitation center in Poland which treats lymphedema. However, the CDT method, recognized as the gold standard for the treatment of lymphedema,5,14 is often criticized as being time-consuming and costly.17 The high cost of therapy is often accompanied by a prolonged wait for treatment. Chronic lymphedema significantly deteriorates the quality of patient life.5,6,18 This is a particularly distressing experience for elderly people, who may already suffer from reduced mobility or other limitations. Walking impairment makes it difficult for the elderly to leave home, which may result in weakened social and family connections, as well as decreased self-esteem. This, in turn, has a negative impact on their psychosocial functioning, and may either lead to depression or worsen the existing problems.19 Thus, an alternative treatment, one that will allow the patient to start therapy at home while waiting for a place at a CDT center, seems necessary. This study was designed to test the effectiveness of MCB as an alternative approach to treat lymphedema, specifically designed for the elderly.
Lymphedema was reduced in both groups, CDT and MCB. What is more, the level of improvement in patients from either of the groups did not differ significantly, which may indicate a similar efficacy for both methods. The literature offers very few papers which compare these two methods in outpatient settings. Johansson et al, in 1999,20 in their study in 38 females with upper limb lymphedema, previously treated for breast cancer, compared the effects of low stretch MCB alone (MCB group, 18 subjects, mean age: 64 years) and combined with manual lymph drainage (MCB + MLD; 20 subjects, mean age: 58 years). These authors showed that bandaging is an effective method of reducing lymphedema, especially if it is applied daily for 2 weeks, after which time its effectiveness significantly decreases. Tidhar et al,21 examined 30 patients (27 women and 3 men) who underwent self-bandage training. In their study, the patients who used the learned bandage techniques and protocols achieved significant lymphedema reductions, which also confirmed the effectiveness of bandaging as an anti-edema treatment. The results of the 2013 study by Dayes et al,22 in a group of 56 patients (mean age: 60 years) with CDT and a parallel group of 39 subjects (mean age: 59 years) with only elastic compression garments are consistent with our findings. In their study, reduction in lymphedema was observed in both groups although, in terms of percentage, the difference between the groups was not statistically significant. Their results might indicate that MCB is the most important component of CDT. Similar efficacy was observed for both therapies in their study and our results. Several other authors23–27 have confirmed the efficacy of bandaging as a method of reducing lymphedema, and identify it as the most effective and basic component of anti-edema treatment.
Although patients often consider MLD to be the most important component of CDT, and generally CDT is not performed without it, in a prospective cohort study by Vignes et al,24 in 2011, involving 682 women (mean age: 62 years), a relationship between MLD and increased risk for therapy failure was demonstrated. In the same study, combination of MCB with MLD also reduced the chance of limb volume retention as compared to patients who only received the MCB intervention. As can be seen, it is difficult to assess the significance of individual CDT components used separately without raising doubt. Further studies on the effectiveness of the isolated methods constituting CDT are necessary.13,28 After all, compression is the common component of most CDT programs.29
In our study, CDT consisted of MCB, a 60-min MLD, and a 45-min training routine. While the training routine of a group of patients may be supervised by one professional, MLD requires separate sessions with individual patients. The estimated saving on time would be 1 hour. Owing to that, and with the assumption that CB takes approximately 30 minutes, it would be possible to perform CB in 16 patients or in 14 patients and supervise their training during 1 working day of a single therapist. In case of the abovementioned CDT scheme, it would be possible for a single therapist to attend to 4–5 patients only during 1 working day. According to the calculations of Gutknecht et al,12 the total annual cost of CDT may significantly exceed 2,500 EUR (mean: 2,509.73 EUR – MLD, 243.70 EUR – rehabilitation, 29.50 EUR – bandaging, without general practitioner and physician fees) for one patient. At the same time, the annual cost of MCB has been estimated at 29.50 EUR. Thus, MCB instead of CDT might significantly lower the costs of treating lymphedema. Further research to compare these two methods is necessary, with particular emphasis on the long-term efficacy of both treatments. Should the results of our study be confirmed, it might be advisable to modify the management standards for lymphedema patients. Physical activity and adequate skin care, combined with MCB, may prove to be less burdensome for the health care system than CDT, while maintaining the same treatment efficacy.
MCB may lead to a significant improvement in patient condition or may improve lymphedema control in the affected patients. However, Protz et al,30 in 2014, and Heyer et al,31 in 2017, revealed worrisome lack of skill and knowledge about compression therapy among the medical staff in their 891 and 1,476 respective respondents. The study participants were not only unaware of the products available on the market, but also had outdated or incorrect information on the methodology of the treatment, or had no information or knowledge of it at all. A patient, especially an elderly one, should receive the most reliable and up-to-date information and explanation of the procedures for any type of treatment. In our study, the therapy was performed by qualified lymphedema therapists. However, as the literature shows, compression therapy can successfully be performed by the patients themselves, at their homes.21 Correct instructions and assistance of the medical staff before selecting the compression products remain important elements which boost the efficacy of self-therapy.
In case of the elderly, self-bandage may be hindered by age-related impaired mobility and the bandages may additionally hamper lower limb mobility. However, lower extremity lymphedema also limits patient mobility so the bandage-related inconvenience should not be greater than the lymphedema itself.
It is necessary to educate both patients and medical staff. The latter, in particular, ought to be the source of reliable information for the affected patients. Proper education will lower the high cost of patient care,12 and will also reduce the prolonged wait for specialist treatment. Auto-therapy in the form of bandaging may help elderly patients to reduce the swelling of the limbs. At the same time, through active participation in the treatment, they will increase the sense of control over their own health and body, as well as the sense of security and self-reliance.21 This, in turn, will have a positive effect on the quality of their lives.19
Conclusion
MCB is a vital component of CDT, without which the maximum reduction in lymphedema cannot be achieved and the effect of the therapy cannot be maintained. It also demonstrates its effectiveness as an independent method, which can reduce the cost of therapy and increase the availability of lymphedema therapy to older patients.
Disclosure
The authors report no conflicts of interest in this work.
References
Klernäs P, Johnsson A, Horstmann V, Johansson K. Health-related quality of life in patients with lymphoedema – a cross-sectional study. Scand J Caring Sci. Epub 2017 Sep 11. | ||
Greene AK. Epidemiology and morbidity of lymphedema. In: Greene AK, Slavin SA, Brorson H, editors. Lymphedema: Presentation, Diagnosis and Treatment. Cham: Springer International Publishing; 2015:33–44. | ||
Dayan JH, Ly CL, Kataru RP, Mehrara BJ. Lymphedema: pathogenesis and novel therapies. Annu Rev Med. 2018;69:263–279. | ||
Nguyen TT, Hoskin TL, Habermann EB, Cheville AL, Boughey JC. Breast cancer-related lymphedema risk is related to multidisciplinary treatment and not surgery alone: results from a large cohort study. Ann Surg Oncol. 2017;24(10):2972–2980. | ||
Bakar Y, Tuğral A. Lower extremity lymphedema management after gynecologic cancer surgery: a review of current management strategies. Ann Vasc Surg. 2017;44:442–450. | ||
Randheer S, Kadambari D, Srinivasan K, Bhuvaneswari V, Bhanumathy M, Salaja R. Comprehensive decongestive therapy in postmastectomy lymphedema: an Indian perspective. Indian J Cancer. 2011;48(4):397–402. | ||
Shafrin J, Sullivan J, Goldman DP, Gill TM. The association between observed mobility and quality of life in the near elderly. PLoS One. 2017;12(8):e0182920. | ||
Whitaker J, Williams A, Pope D, et al. Clinical audit of a lymphoedema bandaging system: a foam roll and cohesive short stretch bandages. J Wound Care. 2015;24(3):83–84. | ||
International Society of Lymphology. The diagnosis and treatment of peripheral lymphedema: 2013 Consensus Document of the International Society of Lymphology. Lymphology. 2013;46(1):1–11. | ||
Vojáčková N, Fialová J, Hercogová J. Management of lymphedema. Dermatol Ther. 2012;25(4):352–357. | ||
Szuba A, Rockson SG. Lymphedema: classification, diagnosis and therapy. Vasc Med. 1998;3(2):145–156. | ||
Gutknecht M, Herberger K, Klose K, et al. Cost-of-illness of patients with lymphedema. J Eur Acad Dermatol Venereol. 2017;31(11):1930–1935. | ||
Moffatt C, Doherty D, Morgan P, editors. Lymphoedema Framework. Best Practice for the Management of Lymphoedema. International Consensus. London: MEP Ltd; 2006. | ||
Franks PJ, Moffatt CJ, Murray S, Reddick M, Tilley A, Schreiber A. Evaluation of the performance of a new compression system in patients with lymphoedema. Int Wound J. 2013;10(2):203–209. | ||
Brown J. A clinically useful method for evaluating lymphedema. Clin J Oncol Nurs. 2004;8(1):35–38. | ||
Mori T, Lustman A, Katz-Leurer M. Self-measurement of upper extremity volume in women post-breast cancer: reliability and validity study. Physiother Theory Pract. 2015;31(4):283–287. | ||
Lasinski BB, McKillip Thrift K, Squire D, et al. A systematic review of the evidence for complete decongestive therapy in the treatment of lymphedema from 2004 to 2011. PM R. 2012;4(8):580–601. | ||
Melam GR, Buragadda S, Alhusaini AA, Arora N. Effect of complete decongestive therapy and home program on health-related quality of life in post mastectomy lymphedema patients. BMC Women’s Health. 2016;16:23. | ||
Morgan PA, Franks PJ, Moffatt CJ. Health-related quality of life with lymphoedema: a review of the literature. Int Wound J. 2005;2(1):47–62. | ||
Johansson K, Albertsson M, Ingvar C, Ekdahl C. Effects of compression bandaging with or without manual lymph drainage treatment in patients with postoperative arm lymphedema. Lymphology. 1999;32(3):103–110. | ||
Tidhar D, Hodgson P, Shay C, Towers A. A lymphedema self-management programme: report on 30 cases. Physiother Can. 2014;66(4):404–412. | ||
Dayes IS, Whelan TJ, Julian JA, et al. Randomized trial of decongestive lymphatic therapy for the treatment of lymphedema in women with breast cancer. J Clin Oncol. 2013;31(30):3758–3763. | ||
McNeely ML, Magee DJ, Lees AW, Bagnall KM, Haykowsky M, Hanson J. The addition of manual lymph drainage to compression therapy for breast cancer related lymphedema: a randomized controlled trial. Breast Cancer Res Treat. 2004;86(2):95–106. | ||
Vignes S, Porcher R, Arrault M, Dupuy A. Factors influencing breast cancer-related lymphedema volume after intensive decongestive physiotherapy. Support Care Cancer. 2011;19(7):935–940. | ||
Moffatt CJ, Franks PJ, Hardy D, Lewis M, Parker V, Feldman JL. A preliminary randomized controlled study to determine the application frequency of a new lymphoedema bandaging system. Br J Dermatol. 2012;166(3):624–632. | ||
Moseley AL, Carati CJ, Piller NB. A systematic review of common conservative therapies for arm lymphoedema secondary to breast cancer treatment. Ann Oncol. 2007;18(4):639–646. | ||
Gradalski T, Ochalek K, Kurpiewska J. Complex decongestive lymphatic therapy with or without Vodder II manual lymph drainage in more severe chronic postmastectomy upper limb lymphedema: a randomized noninferiority prospective study. J Pain Symptom Manage. 2015;50(6):750–757. | ||
Shao Y, Zhong D-S. Manual lymphatic drainage for breast cancer-related lymphoedema. Eur J Cancer Care (Engl). 2017;26(5). Epub 2016 May 11. | ||
King M, Deveaux A, White H, Rayson D. Compression garments versus compression bandaging in decongestive lymphatic therapy for breast cancer-related lymphedema: a randomized controlled trial. Support Care Cancer. 2012;20(5):1031–1036. | ||
Protz K, Heyer K, Dörler M, Stücker M, Hampel-Kalthoff C, Augustin M. Compression therapy: scientific background and practical applications. J Dtsch Dermatol Ges. 2014;12(9):794–801. | ||
Heyer K, Protz K, Augustin M. Compression therapy – cross-sectional observational survey about knowledge and practical treatment of specialised and non-specialised nurses and therapists. Int Wound J. 2017;14(6):1148–1153. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.