Back to Journals » Therapeutics and Clinical Risk Management » Volume 16
Comparison of Percutaneous Endoscopic Lumbar Discectomy with Minimally Invasive Transforaminal Lumbar Interbody Fusion as a Revision Surgery for Recurrent Lumbar Disc Herniation after Percutaneous Endoscopic Lumbar Discectomy
Received 25 September 2020
Accepted for publication 9 November 2020
Published 8 December 2020 Volume 2020:16 Pages 1185—1193
DOI https://doi.org/10.2147/TCRM.S283652
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Deyun Wang
Anqi Wang, Zhengrong Yu
Department of Orthopedics, Peking University First Hospital, Peking, People’s Republic of China
Correspondence: Zhengrong Yu
Department of Orthopedics, Peking University First Hospital, Xishiku Street No. 8, Xicheng District, Peking 100034, People’s Republic of China
Tel +86-10-8357-2655
Email [email protected]
Objective: The purpose of this study was to compare the outcomes between percutaneous endoscopic lumbar discectomy (PELD) and minimally invasive transforaminal lumbar interbody fusion (MIS-TLIF) for the revision surgery for recurrent lumbar disc herniation (rLDH) after PELD surgery.
Patients and Methods: A total of 46 patients with rLDH were retrospectively assessed in this study. All the patients had received a PELD in Peking University First Hospital between January 2015 and June 2019, before they underwent a revision surgery by either PELD (n=24) or MIS-TLIF (n=22). The preoperative data, perioperative conditions, complications, recurrence condition, and clinical outcomes of the patients were compared between the two groups.
Results: Compared to the MIS-TLIF group, the PELD group had significantly shorter operative time, less intraoperative hemorrhage, and shorter postoperative hospitalization, but higher recurrence rate (P< 0.05). Complication rates were comparable between the two groups. Both groups had satisfactory clinical outcomes at a 12-month follow-up after the revision surgery. The PELD group also showed significantly lower visual analog scale (VAS) scores of back pain and Oswestry disability index (ODI) in one month after the revision surgery, whereas the difference was not detectable at six- and 12-month follow-ups.
Conclusion: Both PELD and MIS-TLIF are effective as a revision surgery for rLDH after primary PELD. PELD is superior to MIS-TLIF in terms of operative time amount of intraoperative hemorrhage and postoperative hospitalization. However, its higher postoperative recurrence rate must be considered and patients should be well informed, when making a decision between the two surgical approaches.
Keywords: recurrent lumbar disc herniation, revision surgery, percutaneous endoscopic lumbar discectomy, minimally invasive transforaminal lumbar interbody fusion
Introduction
Microdiscectomy technique has been considered the “gold standard” procedure for lumbar disc herniation (LDH, a degenerative disease with a reported prevalence of 1–3%),1 when conservative treatment cannot relieve its clinical symptoms.2,3 With the development of high-resolution endoscopes and corresponding instruments in recent years, percutaneous endoscopic lumbar discectomy (PELD), a nonfusion surgery, has become an alternative for the treatment of LDH.4
PELD is associated with the improvement of back pain and radicular or cauda equina syndromes caused by LDH, and therefore higher quality of life of patients.5–7 However, the occurrence of recurrent LDH (rLDH) after PELD has raised the concerns of many researchers,13–15 despite of its advantages of minimal invasiveness, shorter hospital stays, faster rehabilitation, and earlier return to work.7–12 rLDH is considered the same level disc herniation after primary discectomy with a more than one month pain-free interval.16 Yin et al17 demonstrated that PELD was correlated with a certain rate of recurrence (3.6%), which usually occurred within six months postoperatively. Leven et al18 reported that 9.3% of patients underwent reoperation because of rLDH in eight years after primary lumbar discectomy.
Minimally invasive transforaminal lumbar interbody fusion (MIS-TLIF), first reported by Foley in 2003,19 is a lumbar fusion surgery correlated with less damage to the back muscles and bony structures.20–25 Both MIS-TLIF and PELD have been well accepted as revision surgery for rLDH.26–29 In this study, we aim to compare the outcomes of these two approaches for patients with rLDH after primary PELD, to explore some helpful insights on making decisions between the two procedures for rLDH.
Patients and Methods
Subjects and Grouping
Between January 2015 and June 2019, a total of 1536 patients received PELD in Peking University First Hospital. Fifty of them, who developed rLDH after the PELD and then received another PELD or MIS-TLIF, were analyzed retrospectively with approval from the Human Subjects Institutional Review Board at Peking University First Hospital. Patient outcomes were collected independently from the participants with written informed consent, and the data were analyzed anonymously.
The inclusion criteria were as follows: patients who (1) had at least an one-month pain-free interval after the primary PELD; (2) showed recurrent pain symptoms, and a herniated disc fragment on the same level as that in the previous PELD, confirmed by MRI; and (3) conservative therapy failed to relieve the recurrent pain. In addition, to avoid scar formation from real rLDH, only patients showing the following were enrolled: (1) definite neurological symptom; (2) space-occupying lesions in the lumbar spinal canal that were confirmed by MRI; (3) a herniation of the nucleus pulposus was observed intraoperatively.
Patients were excluded if the following criteria are met: (1) the recurrent pain symptoms and MRI confirmation of residual disc fragment occurred within a month post PELD (which is defined as a surgical failure); and (2) the herniated disc fragment did not appear at the same level as that in the previous PELD. Besides, the patients with vertebral instability or spondylolisthesis, who preferred MIS-TLIF, were also excluded.14
Based on the above criteria, 46 of the 50 patients were finally included into the study. Among them, 24 received PELD, and 22 had MIS-TLIF. All the patients had signed a detailed written informed consent before the surgical procedures. The final choice between the two approaches were made by the patients, after they were well informed about the surgical procedures, experience of their surgeon, complications and recurrence, as well as the total cost of both treatments.
Surgical Technique and Postoperative Management
Both PELD and MIS-TLIF were performed by the same experienced surgeon for the enrolled patients.
PELD
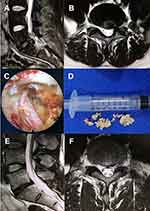
PELD was performed as described by Hoogland et al30 as well as Mayer and Brock.4 The procedure was performed under local anesthesia with patients in the prone position on a radiolucent table. After infiltration of the entry point (8–12 cm from the midline) with local anesthetics, an 18-gauge spinal needle was introduced under fluoroscopic guidance. The final target point of the spinal needle was the medial pedicular line on the anteroposterior image and the posterior vertebral line on the lateral image. The subsequent steps were as follows: (1) a guide wire was inserted through the spinal needle; (2) the spinal needle was removed; (3) a small skin incision was created at the entry point; (4) a tapered cannulated obturator was inserted along the guide wire; (5) after contacting the annulus, the obturator was inserted into the disc with hammering, and the timing of foraminoplasty was selected based on the type of prolapse of the disc; and (6) a bevel-ended, oval-shaped working cannula was inserted into the disc along the obturator. The obturator was then removed, and an endoscope was inserted through the cannula. Finally, the herniated disc was removed using small forceps. A targeted fragmentectomy was performed in all cases. After the herniated fragment was completely removed, the endoscope was also withdrawn, and a sterile dressing was applied with a one-point suture (Figure 1).
MIS-TLIF
MIS-TLIF was performed as described by Foley et al.19 All the participants received general anesthesia before MIS-TLIF. C-arm machine and Quadrant System were prepared before the operation. The patients were placed in the prone position on a radiolucent table. Under C-arm fluoroscopy, the targeted level was confirmed with a self-made locator. Based on the spatial relationship, the intervertebral spaces and the pedicle positions were marked on the body surface. An incision was planned by connecting a line between the outer portions of both ends pedicles (approximately 3 cm off midline). Then a 2–3 cm skin incision was made on the more symptomatic side or more severe side suggested by imaging. The paravertebral muscles were split and retracted laterally to the outer edge of the facet joint, and the zygapophysis was confirmed. Expansion tube was then inserted and Quadrant System was placed. X-ray examination was repeated to confirm the target segments and the placement of Quadrant System. We conducted the decompression by cutting the inferior portion of the lamina, hypertrophied superior and inferior articular processes, and ligamenta flava. Then we enlarged the intervertebral space and appropriate bullet-type interbody cage (Medtronic Sofamor Danek, Memphis, TN, USA) filled with autologous bone was packed into the center of intervertebral space. Following these, ipsilateral percutaneous pedicle screws (Medtronic Sofamor Danek) were inserted through the same skin incision, and contralateral percutaneous pedicle screw was also placed through a mirror incision under fluoroscopic guidance, also titanium rods (Medtronic Sofamor Danek) were inserted and tightened on both sides. Finally, hemostasis and proper irrigation of the incision was performed and negative pressure drainage was placed followed by closure in layers (Figure 2).
Off-bed activities were allowed with a waist support on the second day after the surgery in both groups. In MIS-TLIF group, drainage tube was removed when 24-h drainage fluid was less than 30 mL.23 The patients could return to nonmanual work or school two weeks postoperatively, and then back to full activity three months after the surgery. We encourage sufficient bed-rest and off-bed activities with a waist support within three months after surgery.
Clinical Assessment
The preoperative data, including demographic information (age, gender, body mass index [BMI], marital status, alcohol use history, and smoking history) and clinical conditions (herniation level, paramedian/central herniation, migrated/nonmigrated herniation, and Modic change); and perioperative data, including operation time, intraoperative hemorrhage, postoperative hospitalization, complications, and recurrence condition of the patients were collected and evaluated.
Clinical outcomes were assessed by the improvement of back and leg pain, level of disability, and rate of clinical satisfaction. The intensity of back and leg pain were evaluated using visual analog scores (VAS) preoperatively, and then at one, six, and 12 months postoperatively. Disability was assessed using the Oswestry disability index (ODI) version 2.0 at one, six, and 12 months after the procedures. Clinical satisfaction was assessed by an independent surgeon at 12 months postoperatively by using the MacNab criteria.31 Excellent outcome was defined as no pain and no limitation of daily life activities; good outcome as occasional pain or paresthesia, but no need for medication, and no limitation of daily life activities; fair outcome as pain is somewhat improved but needs medication, and some limitation on daily activities; poor outcome as no improvement or worsening, additional operation is needed due to incomplete decompression, or development of instability. The excellent and good were rated as clinically satisfactory outcomes.
Statistical Analysis
Data were analyzed by Statistical Package for Social Sciences (Version 12, SPSS, Chicago, IL, USA). The statistic was demonstrated as mean ±SD. Independent Student’s t test was used to compare the difference of continuous variables between the two groups. Chi-squared test was used to compare the difference of dichotomous variables between the two groups. These two tests were used to compare the baseline data and observation parameters between the two groups. P<0.05 was regarded as statistically significant.
Results
Preoperative Data of the Patients
All the enrolled patients received one-segment PELD or MIS-TLIF, and then were followed up for at least 12 months. The preoperative demographic and clinical characteristics were not significantly different between the two groups (Table 1).
 |
Table 1 Preoperative Data of PELD and MIS-TLIF Groups |
Perioperative Outcomes, Complications, and Recurrent Condition
Comparing to MIS-TLIF group, PELD group was associated with significantly shorter operative time, less intraoperative hemorrhage, and shorter postoperative hospitalization (Table 2). Complications occurred in two patients (8.33%) in PELD group and one patient (4.55%) in MIS-TLIF group (Table 2). One patient in MIS-TLIF group experienced epidural hematoma and was later cured by evacuation of the hematoma. One case in PELD group complained of headache during the operation, and then improved with sufficient bed rest in a day after the surgery. Another patient in PELD group complained of dysesthesia on the posterior thigh, which improved in a week with neurotrophic drugs.
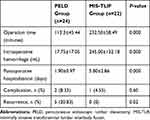 |
Table 2 Perioperative Outcomes, Complications, and Recurrence |
The patients in PELD group showed significantly higher recurrence rate (20.83%) than those in MIS-TLIF group (0%, P=0.02) within 12 months postoperation (Table 2). For the patients suffering recurrence, the symptoms were managed with conservative treatment, including bed rest and drug therapy (neurotrophic and nonsteroidal anti-inflammatory drugs). Five patients from PELD group had re-recurrence after the operation, three of them reported that their pain was remitted until the last follow-up, the other two received reoperation by MIS-TLIF, and their pain had been largely relieved until the last follow-up.
Clinical Outcomes
VAS scores for back and leg pain, and ODI were significantly improved in both groups at one, six, and 12 months after operation. There were no differences in the average VAS scores for back and leg pain, as well as ODI, between the two groups before, and at six and 12 months after the operation (P>0.05). However, VAS scores for back pain and ODI were found significantly lower in PELD group than that in MIS-TLIF group at one-month follow-up (P<0.05, Table 3).
 |
Table 3 Clinical Outcomes of PELD and MIS-TLIF Groups |
According to the MacNab criteria, satisfactory (excellent or good) results were revealed in 21 of the patients (87.5%) from PELD group; and 20 (90.9%) from MIS-TLIF group. There was no significant difference in the rate of satisfaction between these two groups (P=0.92, Table 3).
Discussion
With the development of endoscopic surgical devices, the indications of spine endoscopy have been extended,32,33 and PELD has been more popular over the last decade for LDH. However, PELD is a nonfusion surgery, which means rLDH may occur afterwards. Hence, many researchers have focused on the causes of rLDH after PELD. Age, BMI, Modic change, disc height, spinal canal occupancy, postoperative instability or hypermobility, smoking are the factors correlated to rLDH post PELD.13–15,17,34,35 The surgeon can make decisions on surgical strategies, evaluate prognosis, and predict postoperative responses based on the factors. However, except for the patients with LDH combined with vertebral instability or spondylolisthesis, which are more suitable for fusion surgery, most of the time, the patient would be the one who makes the final decision.
With the advantages of fewer injuries to the back muscles and bony structures, reduced intraoperative hemorrhage, shorter hospitalization, and faster recovery comparing to conventional TLIF and open discectomy, PELD and MIS-TLIF have been considered good alternatives.8,10,11,20–25,36 In this study, we compared the outcomes of PELD and MIS-TLIF for patients with PELD recurrence. We found that the PELD group showed shorter operative time, less intraoperative hemorrhage, and shorter postoperative hospitalization than the MIS-TLIF group. These findings are consistent with the previous studies.37,38 Shorter operative time in the PELD group might have resulted from a bigger visual scope and simpler operating steps than MIS-TLIF. Less intraoperative hemorrhage and shorter postoperative hospitalization in PELD might be benefit from less muscle and bone damages, and shorter operative time, which also contributes to faster recovery compared with MIS-TLIF. These outcomes might influence the duration of disability and mental health of the patients, as well as their obedience and trust to their doctors.
Our data showed that both PELD and MIS-TLIF improved the back and leg pain VAS scores, and reduced ODI in 12 months, comparing to preoperative baselines. Most of the patients from both groups reported clinical satisfaction measured by the MacNab criteria, indicating that the two procedures are equally effective for rLDH as a revision surgery. These outcomes are similar to those reported previously.37–39 However, back pain VAS scores and ODI in the PELD group decreased quicker than those in MIS-TLIF group in one month postoperation, indicating that PELD could remit back and leg pain, and improve quality of life in the short-term. This was highlighted by reduced neural tissue retraction, also decreased trauma to paravertebral muscles in discectomy surgery compared with interbody fusion surgery.37,38
Complications are nightmares for spine surgeons, especially for re-operation. In our study, one case in the MIS-TLIF group underwent unilateral approach for bilateral decompression and experienced postoperative epidural hematoma. Than et al40 reported that unilateral approach for bilateral decompression with MIS-TLIF increased the risks of dural laceration and cerebrospinal fluid leak. Similarly, postoperative epidural hematoma might attribute to limited intraoperative view, tissue or scar adhesion around the spinal dural sac, insufficient hemostasis, and postoperative obstructed drainage. In addition, in PELD group, one patient complained of headache and one reported dysesthesia on the posterior thigh. The working sheath might compress the nerve root during the procedure,41 leading to radicular symptoms after the surgery. Sairyo et al reported that if the endoscopic maneuver duration was too long, the intracranial pressure might increase,42 resulting in headaches. Because of distorted anatomy, altered landmarks, and scar formation, revision spine surgery is more challenging than a primary surgery, and is associated with a relatively higher complication rate, especially nerve root injury, dural laceration, and surgical site infection.43,44 In addition, the steep learning curves of either MIS-TLIF or PELD need to be considered,45–47 expert knowledge of spine anatomy and experience accumulation are required to avoid these complications.
Recurrences are difficult to accept after a re-operation. In our study, five patients from the PELD group suffered from rLDH after the revision surgery. The rate of recurrence was significantly higher than that in MIS-TLIF group (no cases). As mentioned above, some common risk factors, for instance elderly age, obesity, Modic change can be predictive for rLDH after PELD surgery. Those five patients who experienced recurrence were either over 60 years old or had a BMI >25, indicating a high risk for recurrent herniation after PELD. The risk factors for recurrence after a primary surgery might also predict a recurrence after a revision surgery. In addition, after a primary PELD, the artificial incision in the annulus fibrosus could alter the interlaminar shear stress, causing the residual nucleus pulposus being more prone to prolapse.48 Hence, for patients with high risk factors for rLDH, surgeons should completely inform them about the fusion surgery option, and risks of recurrent herniation after either primary or revision nonfusion surgery.
The present study has some limitations. First, it is a retrospective study with a small sample size, and relatively short follow-up period. A multicenter and prospective study with a larger sample size is needed to confirm the long-term clinical and radiographic outcomes. In addition, some more observation parameters, such as adjacent degeneration and stability of lumbar spine, need to be compared in patients who have had different surgeries. A larger sample size is also required to increase the accuracy of the occurrence rates of complications and recurrence. Second, surgical choices in this study is limited. Micro-endoscopic discectomy (MED), open transforaminal lumbar interbody fusion (O-TLIF), posterior lumbar intervertebral fusion (PLIF), etc can also be used for rLDH as a revision surgery. These procedures also need to be discussed in future studies. Third, all the operations were done by the same surgeon, there may be a bias therefore, resulting from his specific learning curves.
Conclusions
This study compared the outcomes of PELD and MIS-TLIF as a revision surgery of for rLDH after PELD. PELD shows advantages in nongeneral anesthesia, shorter operative time, less intraoperative hemorrhage, and shorter postoperative hospitalization over MIS-TLIF. However, it is also associated with a higher recurrence rate. PELD could also remit back pain and improve the quality of life more quickly than MIS-TLIF surgery, but both methods provide patients with satisfactory outcomes. When evaluating a patient for a revision surgery for PELD recurrence, the advantages and disadvantages of the two procedures must be carefully balanced, and the patients should be completely informed.
Ethics
This study complied with the Declaration of Helsinki.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Frymoyer JW. Lumbar disk disease: epidemiology. Instr Course Lect. 1992;41:217–223.
2. Yeung AT, Tsou PM. Posterolateral endoscopic excision for lumbar disc herniation: surgical technique, outcome, and complications in 307 consecutive cases. Spine. 2002;27(7):722–731. doi:10.1097/00007632-200204010-00009
3. Hamawandi SA, Sulaiman II, Al-Humairi AK. Open fenestration discectomy versus microscopic fenestration discectomy for lumbar disc herniation: a randomized controlled trial. BMC Musculoskelet Disord. 2020;21(1):384. doi:10.1186/s12891-020-03396-x
4. Mayer HM, Brock M. Percutaneous endoscopic lumbar discectomy (PELD). Neurosurg Rev. 1993;16(2):115–120.
5. Chen C, Fan P, Huang L, Zhen H, Liu L, Wang Y. Percutaneous Endoscopic Lumbar Discectomy as an Emergent Surgery for Cauda Equina Syndrome Caused by Lumbar Disc Herniation. Pain Physician. 2020;23(3):E259–E264.
6. Li X, Dou Q, Hu S, et al. Treatment of cauda equina syndrome caused by lumbar disc herniation with percutaneous endoscopic lumbar discectomy. Acta Neurol Belg. 2016;116(2):185–190.
7. Peng CW, Yeo W, Tan SB. Percutaneous endoscopic lumbar discectomy: clinical and quality of life outcomes with a minimum 2 year follow-up. J Orthop Surg Res. 2009;4:20.
8. Ruan W, Feng F, Liu Z, Xie J, Cai L, Ping A. Comparison of percutaneous endoscopic lumbar discectomy versus open lumbar microdiscectomy for lumbar disc herniation: A meta-analysis. Int J Surg. 2016;31:86–92.
9. Cao J, Huang W, Wu T, Jia J, Cheng X. Percutaneous endoscopic lumbar discectomy for lumbar disc herniation as day surgery - short-term clinical results of 235 consecutive cases. Medicine. 2019;98(49):e18064.
10. Kim M, Lee S, Kim H-S, Park S, Shim S-Y, Lim D-J. A Comparison of Percutaneous Endoscopic Lumbar Discectomy and Open Lumbar Microdiscectomy for Lumbar Disc Herniation in the Korean: A Meta-Analysis. Biomed Res Int. 2018;2018:9073460. doi:10.1155/2018/9073460
11. Qin R, Liu B, Hao J, et al. Percutaneous Endoscopic Lumbar Discectomy Versus Posterior Open Lumbar Microdiscectomy for the Treatment of Symptomatic Lumbar Disc Herniation: A Systemic Review and Meta-Analysis. World Neurosurg. 2018;120:352–362. doi:10.1016/j.wneu.2018.08.236
12. Ahn -S-S, Kim S-H, Kim D-W, Lee B-H. Comparison of Outcomes of Percutaneous Endoscopic Lumbar Discectomy and Open Lumbar Microdiscectomy for Young Adults: A Retrospective Matched Cohort Study. World Neurosurg. 2016;86:250–258. doi:10.1016/j.wneu.2015.09.047
13. Kim HS, You JD, Ju CI. Predictive Scoring and Risk Factors of Early Recurrence after Percutaneous Endoscopic Lumbar Discectomy. Biomed Res Int. 2019;2019:6492675. doi:10.1155/2019/6492675
14. Wang Y, Ning C, Xu F, et al. Recurrent lumbar disc herniation recurrence after percutaneous endoscopic lumbar discectomy: A case report. Medicine. 2018;97(34):e11909. doi:10.1097/MD.0000000000011909
15. Kim JM, Lee SH, Ahn Y, Yoon DH, Lee CD, Lim ST. Recurrence after successful percutaneous endoscopic lumbar discectomy. Minim Invasive Neurosurg. 2007;50(2):82–85. doi:10.1055/s-2007-982504
16. Yaman ME, Kazanci A, Yaman ND, Bas F, Ayberk G. Factors that influence recurrent lumbar disc herniation. Hong Kong Med J. 2017;23(3):258–263.
17. Yin S, Du H, Yang W, Duan C, Feng C, Tao H. Prevalence of Recurrent Herniation Following Percutaneous Endoscopic Lumbar Discectomy: A Meta-Analysis. Pain Physician. 2018;21(4):337–350.
18. Leven D, Passias PG, Errico TJ, et al. Risk Factors for Reoperation in Patients Treated Surgically for Intervertebral Disc Herniation: A Subanalysis of Eight-Year SPORT Data. J Bone Joint Surg Am. 2015;97(16):1316–1325. doi:10.2106/JBJS.N.01287
19. Foley KT, Holly LT, Schwender JD. Minimally invasive lumbar fusion. Spine. 2003;28(15 Suppl):S26–35. doi:10.1097/01.BRS.0000076895.52418.5E
20. Sheng S-R, Geng Y-B, Zhou K-L, Wu A-M, Wang X-Y, Ni W-F. Minimally invasive surgery for degenerative spondylolisthesis: transforaminal or oblique lumbar interbody fusion. J Comp Eff Res. 2020;9(1):45–51. doi:10.2217/cer-2019-0055
21. Chen Y-C, Zhang L, Li E-N, et al. An updated meta-analysis of clinical outcomes comparing minimally invasive with open transforaminal lumbar interbody fusion in patients with degenerative lumbar diseases. Medicine. 2019;98(43):e17420. doi:10.1097/MD.0000000000017420
22. Lin G-X, Park C-K, Hur J-W, Kim J-S. Time Course Observation of Outcomes between Minimally Invasive Transforaminal Lumbar Interbody Fusion and Posterior Lumbar Interbody Fusion. Neurologia medico-chirurgica. 2019;59(6):222–230. doi:10.2176/nmc.oa.2018-0194
23. Yang Y, Liu Z-Y, Zhang L-M, et al. Microendoscopy-Assisted Minimally Invasive Versus Open Transforaminal Lumbar Interbody Fusion for Lumbar Degenerative Diseases: 5-Year Outcomes. World Neurosurg. 2018;116:e602–e610. doi:10.1016/j.wneu.2018.05.049
24. Karikari IO, Isaacs RE. Minimally invasive transforaminal lumbar interbody fusion: a review of techniques and outcomes. Spine. 2010;35(Supplement):S294–S301. doi:10.1097/BRS.0b013e3182022ddc
25. Parker SL, Adogwa O, Witham TF, Aaronson OS, Cheng J, McGirt MJ. Post-operative infection after minimally invasive versus open transforaminal lumbar interbody fusion (TLIF): literature review and cost analysis. Minim Invasive Neurosurg. 2011;54(1):33–37. doi:10.1055/s-0030-1269904
26. Ahn Y, Lee S-H, Park W-M, Lee H-Y, Shin S-W, Kang H-Y. Percutaneous endoscopic lumbar discectomy for recurrent disc herniation: surgical technique, outcome, and prognostic factors of 43 consecutive cases. Spine. 2004;29(16):E326–E332. doi:10.1097/01.BRS.0000134591.32462.98
27. Lee DY, Shim CS, Ahn Y, Choi Y-G, Kim HJ, Lee S-H. Comparison of percutaneous endoscopic lumbar discectomy and open lumbar microdiscectomy for recurrent disc herniation. Journal of Korean Neurosurgical Society. 2009;46(6):515–521. doi:10.3340/jkns.2009.46.6.515
28. Li X, Hu Z, Cui J, et al. Percutaneous endoscopic lumbar discectomy for recurrent lumbar disc herniation. Int J Surg. 2016;27:8–16. doi:10.1016/j.ijsu.2016.01.034
29. Yao Y, Zhang H, Wu J, et al. Minimally invasive transforaminal lumbar interbody fusion versus percutaneous endoscopic lumbar discectomy: Revision surgery for recurrent herniation after microendoscopic discectomy. World Neurosurg. 2017;99:89–95. doi:10.1016/j.wneu.2016.11.120
30. Hoogland T, Schubert M, Miklitz B, Ramirez A. Transforaminal posterolateral endoscopic discectomy with or without the combination of a low-dose chymopapain: a prospective randomized study in 280 consecutive cases. Spine. 2006;31(24):E890–E897. doi:10.1097/01.brs.0000245955.22358.3a
31. Macnab I. Negative disc exploration. An analysis of the causes of nerve-root involvement in sixty-eight patients. J Bone Joint Surg Am. 2016;86(5):891–903. doi:10.2106/00004623-197153050-00004
32. Patel MS, Braybrooke J, Newey M, Sell P. A comparative study of the outcomes of primary and revision lumbar discectomy surgery. Bone Joint J. 2013;95-B(1):90–94.
33. Kim HS, Paudel B, Jang JS, Lee K, Oh SH, Jang IT. Percutaneous Endoscopic Lumbar Discectomy for All Types of Lumbar Disc Herniations (LDH) Including Severely Difficult and Extremely Difficult LDH Cases. Pain Physician. 2018;21(4):E401–E408.
34. Xu J, Li Y, Wang B, et al. Percutaneous Endoscopic Lumbar Discectomy for Lumbar Disc Herniation with Modic Changes via a Transforaminal Approach: A Retrospective Study. Pain Physician. 2019;22(6):E601–E608.
35. Yao Y, Liu H, Zhang H, et al. Risk Factors for Recurrent Herniation After Percutaneous Endoscopic Lumbar Discectomy. World Neurosurg. 2017;100:1–6.
36. Choi KC, Kim JS, Park CK. Percutaneous Endoscopic Lumbar Discectomy as an Alternative to Open Lumbar Microdiscectomy for Large Lumbar Disc Herniation. Pain Physician. 2016;19(2):E291–E300.
37. Liu C, Zhou Y. Comparison Between Percutaneous Endoscopic Lumbar Discectomy and Minimally Invasive Transforaminal Lumbar Interbody Fusion for Lumbar Disc Herniation with Biradicular Symptoms. World Neurosurg. 2018;120:e72–e79.
38. Liu C, Zhou Y. Percutaneous endoscopic lumbar discectomy and minimally invasive transforaminal lumbar interbody fusion for massive lumbar disc herniation. Clin Neurol Neurosurg. 2019;176:19–24.
39. Yao Y, Zhang H, Wu J, et al. Comparison of Three Minimally Invasive Spine Surgery Methods for Revision Surgery for Recurrent Herniation After Percutaneous Endoscopic Lumbar Discectomy. World Neurosurg. 2017;100(641–647):e641.
40. Than KD, Mummaneni PV. Unilateral approach for bilateral decompression with MIS TLIF. World Neurosurg. 2014;82(5):646–647.
41. Choi I, Ahn JO, So WS, Lee SJ, Choi IJ, Kim H. Exiting root injury in transforaminal endoscopic discectomy: preoperative image considerations for safety. Eur Spine J. 2013;22(11):2481–2487.
42. Sairyo K, Matsuura T, Higashino K, et al. Surgery related complications in percutaneous endoscopic lumbar discectomy under local anesthesia. J Med Invest. 2014;61(3–4):264–269.
43. Bari TJ, Karstensen S, Sorensen MD, Gehrchen M, Street J, Dahl B. Revision surgery and mortality following complex spine surgery: 2-year follow-up in a prospective cohort of 679 patients using the Spine AdVerse Event Severity (SAVES) system. Spine Deform. 2020.
44. Hu X, Lieberman IH. Revision spine surgery in patients without clinical signs of infection: how often are there occult infections in removed hardware? Eur Spine J. 2018;27(10):2491–2495.
45. Lau D, Lee JG, Han SJ, Lu DC, Chou D. Complications and perioperative factors associated with learning the technique of minimally invasive transforaminal lumbar interbody fusion (TLIF). J Clin Neurosci. 2011;18(5):624–627.
46. Lee DY, Lee SH. Learning curve for percutaneous endoscopic lumbar discectomy. Neurol Med Chir (Tokyo). 2008;48(9):383–388.
47. Wang H, Huang B, Li C, et al. Learning curve for percutaneous endoscopic lumbar discectomy depending on the surgeon’s training level of minimally invasive spine surgery. Clin Neurol Neurosurg. 2013;115(10):1987–1991.
48. Hadjipavlou AG, Tzermiadianos MN, Bogduk N, Zindrick MR. The pathophysiology of disc degeneration: a critical review. J Bone Joint Surg Br. 2008;90(10):1261–1270.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.