Back to Journals » Clinical Ophthalmology » Volume 12
Comparison of central corneal thickness measurements in corneal edema using ultrasound pachymetry, Visante anterior-segment optical coherence tomography, Cirrus optical coherence tomography, and Pentacam Scheimpflug camera tomography
Authors Wongchaisuwat N, Metheetrairut A, Chonpimai P, Nujoi W, Prabhasawat P
Received 24 April 2018
Accepted for publication 17 July 2018
Published 25 September 2018 Volume 2018:12 Pages 1865—1873
DOI https://doi.org/10.2147/OPTH.S172159
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Scott Fraser
Nida Wongchaisuwat,1,2 Ankana Metheetrairat,1 Pratuangsri Chonpimai,1 Waree Nujoi,1 Pinnita Prabhasawat1
1Department of Ophthalmology, Faculty of Medicine, Siriraj Hospital, Mahidol University, Bangkok, Thailand; 2Department of Ophthalmology, Golden Jubilee Medical Center, Mahidol University, Nakhon Pathom, Thailand
Purpose: To compare the central corneal thickness (CCT) measurements in subjects with corneal edema using ultrasound pachymetry, Visante anterior-segment optical coherence tomography (OCT), Cirrus OCT, and Pentacam Scheimpflug camera tomography.
Methods: This prospective cross-sectional study included 46 eyes of 33 patients with corneal edema and a CCT exceeding 550 μm evaluated by ultrasound pachymetry, Visante OCT, Cirrus OCT, and Pentacam. Two observers measured each eye twice. Intraobserver and interobserver reproducibility were determined and agreement among the devices calculated.
Results: CCT was measured in 40 eyes of 29 patients. Regardless of the CCT, the measurements obtained using Visante OCT, Cirrus CCT, and ultrasound pachymetry were well correlated. Interobserver and intraobserver reproducibility were high among the three devices. Pentacam overestimated the results compared with the other devices, and ultrasound pachymetry was unmeasurable in six (13%) eyes with very thick and opaque corneas. In eyes with mild corneal edema (CCT 551–650 μm), measurements from the four devices were comparable.
Conclusion: All devices reliably measured the CCT <650 μm. In eyes with edema exceeding 650 μm, CCT measurements from the Visante OCT, Cirrus OCT, and ultrasound pachymetry devices showed good reproducibility and were well correlated, while the Pentacam overestimated the values compared to the other devices. Pentacam and ultrasound pachymetry should not be used in eyes with extreme corneal edema and opacity.
Keywords: central corneal thickness, ultrasonography, optical coherence tomography, Scheimpflug camera tomography
Introduction
Central corneal thickness (CCT) measurements are useful for measuring intraocular pressure (IOP), evaluating corneal endothelial function, and assessing patients undergoing keratorefractive surgery preoperatively and postoperatively.1 Ultrasound pachymetry is the gold standard for measuring CCT.2 Intraobserver and interobserver repeatability values are highly reproducible in normal corneas. However, the values are limited when using techniques in which a probe is in contact with the cornea and in decentration, both of which require individual user skill to place the probe perpendicularly to the central corneal apex. The former techniques also increase the risk of transmission of infections or development of corneal epithelial defects.3,4 Inaccuracies might result from changes in sound speed associated with different levels of corneal hydration.
The Ocular Response Analyzer (Reichert Technologies, Depew, NY, USA) can also be used for ultrasound pachymetry. High correlations have been reported among measurements obtained using this device, standard ultrasound pachymetry, and optical coherence tomography (OCT).5 Other devices, including rotating Scheimpflug camera tomography (Pentacam; Oculus Optikgeräte, Wetzlar, Germany), specular microscopy, optical low-coherence reflectometry, and OCT, have been developed using a light-based system, the advantages of which are the absence of contact with the cornea, short measurement times, and objective determination of the central corneal apex.6 Ultrasound pachymetry, optical low-coherence reflectometry, and the Pentacam have exhibited good repeatability and agreement when measuring CCTs in normal corneas.7,8 CCTs measured by Fourier-domain (FD) OCT, the Pentacam, and ultrasound pachymetry are also correlated with slightly thinner CCTs visualized by FD-OCT compared to other devices.9
After laser in situ keratomileusis performed to treat myopia in patients with thin CCTs, the Pentacam and Visante OCT (Carl Zeiss Meditec, Jena, Germany) underestimated the CCT results compared with standard ultrasound measurements. No significant differences were seen between ultrasound and Orbscan measurements (Bausch & Lomb, Rochester, NY, USA).10 When the Pentacam and ultrasound pachymetry were used to measure CCTs in patients with keratoconus, the Pentacam was more accurate and CCTs thinner compared to ultrasound pachymetry.11
Few studies have compared CCT measurements obtained using different devices in pathological corneal conditions, such as pseudophakic bullous keratopathy, corneal guttata, or Fuchs’s corneal endothelial dystrophy. After keratoplasty in patients with clear corneas, mean CCT measurements using the Pentacam were higher than measurements obtained using ultrasound pachymetry, and they also had higher intraobserver and interobserver variability.12 One study13 reported the repeatability and comparability of the CCT measurements among the Pentacam, Heidelberg anterior-segment OCT (Spectralis; Heidelberg Engineering, Heidelberg, Germany), and ultrasound pachymetry in postoperative edematous corneas. However, intraobserver variability remained high.
With regard to corneal edema, many factors, including the degree of corneal edema, corneal opacity, and corneal surface irregularity, might affect measurements obtaining using each instrument. Accurate CCT measurements in patients with corneal edema are necessary and affect the treatment plan, eg, the follow-up of progression of stromal and endothelial corneal dystrophy, corneal decompensation, postoperative cataract or corneal surgery, corneal graft survival, or the obtaining of precise CCT values, when planning lamellar keratoplasty surgery. For these reasons, it is interesting to know if different tools provide equivalent results. The current study compared CCT measurements in eyes with corneal edema using ultrasound pachymetry, Visante anterior-segment OCT, Cirrus OCT (Carl Zeiss Meditec), and Pentacam Scheimpflug camera tomography, and evaluated intraobserver and interobserver reproducibility values and agreement among the four devices.
Methods
Study design
The current prospective cross-sectional study was performed to evaluate the reproducibility, correlation, and agreement of CCT measurements in patients with corneal edema. The study was conducted in accordance with the principles of the Declaration of Helsinki. The Committee for the Protection of Human Participants in Research at the Faculty of Medicine, Siriraj Hospital, Mahidol University, Bangkok, Thailand (Siriraj Institutional Review Board, certificate of approval Si271/2013, protocol 165/2556 [EC4]) approved the study protocol. The trial was registered with the Thai Clinical Trials Registry (primary site) ID TCTR20161005001.
Patients
After providing written informed consent, patients with corneal edema who met the inclusion criteria were enrolled in the study at the Department of Ophthalmology, Faculty of Medicine, Siriraj Hospital from 2013 to 2015. Outpatients of the Ophthalmology Department were included if they had corneal edema from corneal diseases and a previous CCT measurement >550 μm. Patients were excluded who were younger than 18 years, could not cooperate during examinations, refused to undergo CCT measurements, were normal subjects with high CCT, or had at least one CCT measurement obtained from the four devices that was below 550 μm. Normal CCTs vary among different populations. Lekskul et al reported that the average CCT was 535.2±29.9 μm in 467 Thai patients aged 12–60 years. This current study comprised patients with CCTs >550 μm, which was estimated to be approximately the average CCT in the population plus 1 SD.14
Study procedures and outcome measures
CCTs were measured in 46 eyes of 33 patients with corneal edema >550 μm. Patient age, sex, visual acuity, IOP, and CCT were recorded and anterior-segment photography performed. CCT was measured using four methods. To avoid the effects of ultrasound probe impression and variations in tear film, the three noncontact devices, ie, Visante OCT, Cirrus OCT, and Pentacam, were used first. The time difference between the use of the noncontact devices and ultrasound pachymetry was 5 minutes to allow the anesthetic drop to take effect.
Visante OCT uses low-coherence interferometry with a 1,310 nm-wavelength light source and a scanning speed of 512 A-scans in 250 ms, which results in higher speed without loss of the signal:noise ratio. All participants were instructed to fixate on the target, the center of the measurement was aligned with the corneal apex, and the pachymetry scan was performed. The machine automatically displays CCT results from the corneal center.
The Cirrus high-definition OCT model 4,000 (Carl Zeiss Meditec) also uses low-coherence interferometry with an 840 nm wavelength five parallel-line scan (4,096 A-scans/line) to view high-resolution corneal images. The space between the lines was 0.25 mm, and the scanning beam was 3 mm long and 2 mm deep. All participants were instructed to fixate on the target, the center of the measurement was aligned with the corneal apex, and the pachymetry scan was performed.
The Pentacam Scheimpflug camera with a charge-coupled-device camera uses a 475 nm wavelength to create Scheimpflug images of the entire anterior segment and perform multiple slit-scan analyses. All participants were instructed to fixate on the target. The device determined automatically when the image was in focus, and the corneal curvature and anterior and posterior elevation values were obtained. CCT was measured as the difference between anterior and posterior elevations in the central cornea.
Ultrasound pachymetry (AL-4000; Tomey, Nuremberg, Germany) uses sound velocity, a frequency of 20 MHz, and a tip diameter of 1.5 mm. One drop of 0.5% tetracaine hydrochloride was instilled on the cornea 5 minutes before measurement, and patients were instructed to look straight when the examiner placed the probe perpendicularly on the corneal center. The mean of three measurements was recorded.
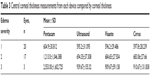
The examiner determined that the measurement was successful when the corneal apex alignment was positioned correctly. Good signal strength was found, and CCT values were obtained automatically with every device. CCT values obtained with each device were derived from the apical corneal center. The two examiners performed the four methods of CCT measurement in the same order, with a time difference between examiners of 5 minutes. A meta-analysis showed that CCT values measured 5 minutes after instillation of the anesthetic drops did not differ significantly.15 Data were collected in the case record, and the two examiners repeated the CCT measurements in each participant using the four devices within 1 hour of the first measurement. To evaluate intraobserver and interobserver reproducibility of the CCT measurements, the two examiners measured the CCTs in 17 subjects with normal CT in a small pilot study. High intraobserver and interobserver reproducibility values were found in measurements of all devices (Table 1).
In the current study, CCTs were graded into three groups by Visante OCT, with grade 1 indicating CCTs 551–650 μm, grade 2 CCTs 651–850 μm, and grade 3 CCTs >850 μm.
Statistical analysis
All data were analyzed using MedCalc statistical software (version 18.2.1; MedCalc Software, Ostend, Belgium). All CCT results are shown in scatterplots (Figures 1–3). Bland–Altman plots were used to detect agreement among the devices between pairs of Pentacam, Visante, Cirrus, and ultrasound pachymetry data. Mean differences, SD, P-values, 95% limits of agreement (LOA), intraclass correlation coefficients (ICCs), and correlations were derived from these comparisons. Repeated measure analysis of variance with Bonferroni post hoc comparison, LOA, and ICCs were used to calculate interobserver and intraobserver reproducibility values (Table 2).
Results
A total of 46 eyes of 33 patients (18 men, 15 women, mean age 66 years, range 41–86 years) met the inclusion criteria (CCT >550 μm measured by Visante OCT). Visual acuity ranged from 6/6 to light perception. Corneal edema was diagnosed in association with corneal decompensation (n=25, 54%), Fuchs’s endothelial corneal dystrophy (n=18, 39%), and other stromal corneal dystrophies (n=3, 7%). CCTs were measured in all 46 eyes, whereas ultrasound pachymetry failed to measure CCTs in six (13%) of the 46 eyes. CCTs were graded based on Visante OCT images on a scale of 1–3 as described previously. To compare the results and determine statistical significance, we used 40 eyes (29 patients) that were measured using all four devices and eliminated the six eyes that could not be measured by ultrasound pachymetry (Table 3).
  | Table 3 Central corneal thickness measurements from each device compared by corneal thickness |
We identified a linear correlation between CCT measurements of corneal edema among the four devices (Figure 1). In eyes with CT <650 μm, good correlations of CCT measurements were seen among the four devices (ICC between Pentacam and Visante 0.82, between Pentacam and Cirrus 0.83, between Pentacam and ultrasound 0.75, between ultrasound and Visante 0.86, between Visante and Cirrus 0.97, and between ultrasound and Cirrus 0.87).
With regard to overall CT, Visante OCT, Cirrus OCT, and ultrasound pachymetry measurements were well correlated, while the Pentacam overestimated the CCT values compared to the measurements obtained from the other devices (ICC between Pentacam and Visante 0.09, between Pentacam and Cirrus 0.09, between Pentacam and ultrasound 0.09, between ultrasound and Visante 0.95, between Visante and Cirrus 0.98, and between ultrasound and Cirrus 0.97). The highest correlation in any degree of CCT was between Visante and Cirrus OCT corneal measurements (Table 4).
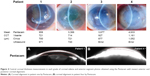
Bland–Altman analysis showed significant (P<0.05 for all comparisons) differences in CCT measurements between the Pentacam and the other devices. Measurements between the Visante, Cirrus, and ultrasound showed good agreement with some variations. The highest agreement was seen in CCT measurements between the Visante and Cirrus OCT. The mean difference between the Visante and Cirrus OCT devices was 0.2 μm, with the 95% LOA between −38.8 and −38.4 (Figure 2, Table 4). Ultrasound pachymetry failed to measure the CCT in six eyes, and four of the six eyes had extreme corneal edema and severe corneal opacities (Figure 4).
A difference in CCT exceeding 2 mm between the Pentacam and other devices was observed in nine cases with CCTs >650 μm. All cases with a large difference in CCTs had corneal opacity at the corneal apex (Figure 4). All devices showed good interobserver and intraobserver reproducibility of CCT measurements. The Visante OCT, Cirrus OCT, and ultrasound pachymetry yielded high interobserver and intraobserver reproducibility, while Pentacam had lower reproducibility values with high variations (Figure 3, Table 2).
Discussion
The current study compared CCT measurements obtained from edematous corneas using Visante OCT, Cirrus OCT, Pentacam, and ultrasound pachymetry. Overall, the results showed that corneal edema in eyes with CCTs <650 μm had CCT values obtained from the four devices that were correlated. In eyes with corneal edema with CCTs >650 μm, Visante OCT, Cirrus OCT, and ultrasound pachymetry were also well correlated, but the Pentacam measurements were higher compared with the others. Visante OCT, Cirrus OCT, and ultrasound pachymetry provided good reproducibility with every degree of corneal edema.
The current study found that ultrasound pachymetry had limited ability to determine CCTs in six eyes, because it measures the time interval between the echoes of sound wave reflection from the anterior and posterior corneal surfaces. When the sound wave scan passes through thick and opaque corneal tissue, the sound propagation speed changes, and the error occurs because the posterior corneal surface is not detected.6 Ultrasound pachymetry might not be suitable for use in patients with severe corneal edema with opacity.
In eyes with CCT >650 μm, the Pentacam was more likely to overestimate the measurement and provide inaccurate results compared to the other devices (Table 3). Nine cases with CCT >650 μm showed large differences the CCTs of more than 2 mm between the Pentacam and the other devices. The limitation of the device is evident when a thick opaque cornea is detected (Figure 4). The Pentacam calculates the pachymetry map based on the difference between the anterior and posterior curvatures. The map had false alignment of both the anterior and posterior corneal surfaces because the light wave cannot pass easily through opaque media and the resultant light dispersion affects the measurements. Misalignment of the corneal surfaces caused extreme variations in CCT values. The current study also showed that the Pentacam had the lowest interobserver and intraobserver reproducibility values compared with the other three devices (Figure 3).
Wu et al reported higher CCT measurements with the Pentacam compared to ultrasound pachymetry in normal healthy corneas, but the Pentacam tended to underestimate the CCT in keratoconic eyes.16 In contrast, Sadoughi et al reported that the Pentacam overestimated CCTs in normal eyes and a more significant difference was found with CCTs <500 μm.17 Yap et al also reported that the highest CCTs were obtained using the Pentacam compared with ultrasound and OCT in normal eyes.18 Kuerten et al found that postoperative edematous corneas had overestimated CCTs when measured by the Pentacam compared to anterior-segment OCT and ultrasound pachymetry.13 The current results supported those of Kuerten et al, and also found that the Pentacam overestimated the cases with extreme corneal edema and opacity. Use of the Pentacam should be avoided in these patients.
OCT is considered a promising new noncontact instrument for evaluating pachymetry.19 Visante and Cirrus OCT use low-coherence interferometry scanning with high penetration, low scatter, and low absorption. These devices have high-speed scans and provide high-definition images. Consequently, they were substantially more precise and accurate than the other two technologies. OCT provides high-resolution cross-sectional corneal images in vivo and more detail, such as localization and quantification of corneal scars and edema.
CCT measurements obtained using Visante and Cirrus OCT provided good reproducibility compared to measurements obtained using other devices. Visante and Cirrus OCT show very high agreement and good correlation of CCT measurements in every degree of corneal edema. The mean difference between the devices was only 0.2 μm (Table 3). Small variations were observed between the measurements. However, despite the high agreement, they might not be used interchangeably in clinical practice.
In clinical practice, Visante OCT was more beneficial with its high-resolution anterior-segment evaluations, while Cirrus OCT was superior for visualizing the various tissue types of both the anterior and posterior segments, such as the anterior chamber angle, retinal nerve-fiber layer, and ganglion cells, and for macular analysis. Even though they are both OCT devices, Visante OCT provides automatic machine centration, while Cirrus OCT needs manual centration that can cause more errors in corneal apex detection.
de Sanctis et al and Kuerten et al reported the CCTs of mildly edematous corneas after surgery.12,13 The current study was conducted in eyes with varying degrees of corneal edema. CCTs >650 μm were considered for the first time in this study, and the results provide information that will benefit patients in clinical practice. The limitations of the current study included the small sample and the analysis of both eyes. These limitations might have introduced bias in the calculations and had significant effects on the interpretation of the results and clinical application. Another limitation was that the study included a few corneal diseases. Patients with severe corneal edema usually also have severe corneal irregularities and opacities, due to the disease course. Other confounding factors might also have added bias to interpretation of the results.
Khurana et al studied corneal opacity, and reported that CCT measurements were equivalent in ultrasound pachymetry and OCT, whereas the Orbscan II slit-scanning tomography significantly underestimated CCT.20 Further studies should evaluate the impact of each factor separately, including corneal opacity and irregularities. The main purpose of this study was to compare CCT measurements in real-world practice, including corneal edema in many pathologies. We also wanted to observe the relationships among devices.
Many new pachymetry devices have been introduced recently, such as the Casia OCT (Tomey), a swept-source OCT device with a scan speed of 30,000 A-scans/second designed specifically for anterior-segment imaging. However, the principles of pachymetric measurement using low-coherence interferometry used in Casia and Visante OCT are the same. CCTs measured using the Visante and Casia devices in eyes with pseudoexfoliation syndrome are comparable.21
Conclusion
Based on the current results, Visante OCT, Cirrus OCT, ultrasound pachymetry, and Pentacam Scheimpflug camera tomography reliably measured CCTs <650 μm. In eyes with moderate–severe corneal edema, CCTs obtained by Visante, Cirrus OCT, and ultrasound pachymetry devices showed good reproducibility and were well correlated, while the Pentacam overestimated the values compared to the other devices. In eyes with severe corneal edema and opacity, ultrasound pachymetry and Pentacam might not be suitable options for measuring CCT.
Acknowledgments
The authors gratefully acknowledge Anupong Veeraburinon from the Research Division, Faculty of Medicine, Siriraj Hospital, Mahidol University, for research assistance, Mathuwan Srikong from the Medical Education Technology Center, Faculty of Medicine, Siriraj Hospital, Mahidol University, for providing the figures, and Assistant Professor Chulaluk Komoltri from Siriraj Medical Research Center for statistical analysis. The abstract of this paper was presented at the ninth Annual Congress on Controversies in Ophthalmology Europe (COPHy) as a poster presentation with interim findings. The poster’s abstract was published in “Poster abstracts” in an abstract book of the ninth COPHy. The authors alone are responsible for the content and writing of the paper.
Disclosure
The authors report no conflicts of interest in this work.
References
Lackner B, Schmidinger G, Pieh S, Funovics MA, Skorpik C. Repeatability and reproducibility of central corneal thickness measurement with Pentacam, Orbscan, and ultrasound. Optom Vis Sci. 2005;82(10):892–899. | ||
Grewal DS, Brar GS, Grewal SP. Assessment of central corneal thickness in normal, keratoconus, and post-laser in situ keratomileusis eyes using Scheimpflug imaging, spectral domain optical coherence tomography, and ultrasound pachymetry. J Cataract Refract Surg. 2010;36(6):954–964. | ||
Shafiq IHS. Comparison between central corneal thickness measurements obtained with orbscan II topographer and ultrasonic pachymeter. Pak J Ophthalmol. 2007;23:87–92. | ||
Bayhan HA, Aslan Bayhan S, Can I. Comparison of central corneal thickness measurements with three new optical devices and a standard ultrasonic pachymeter. Int J Ophthalmol. 2014;7(2):302–308. | ||
Chang S-W, Su P-F, Lo AY, Huang J-Y. Central corneal thickness measurement by Fourier domain optical coherence tomography, ocular response analyzer and ultrasound pachymetry. Taiwan J Ophthalmol. 2014;4(4):163–169. | ||
Bao F, Wang Q, Cheng S, et al. Comparison and evaluation of central corneal thickness using 2 new noncontact specular microscopes and conventional pachymetry devices. Cornea. 2014;33(6):576–581. | ||
Tai LY, Khaw KW, Ng CM, Subrayan V, Cm N, Visvaraja AL. Central corneal thickness measurements with different imaging devices and ultrasound pachymetry. Cornea. 2013;32(6):766–771. | ||
Barkana Y, Gerber Y, Elbaz U, et al. Central corneal thickness measurement with the Pentacam Scheimpflug system, optical low-coherence reflectometry pachymeter, and ultrasound pachymetry. J Cataract Refract Surg. 2005;31(9):1729–1735. | ||
Ishibazawa A, Igarashi S, Hanada K, et al. Central corneal thickness measurements with Fourier-domain optical coherence tomography versus ultrasonic pachymetry and rotating Scheimpflug camera. Cornea. 2011;30(6):615–619. | ||
Ho T, Cheng AC, Rao SK, Lau S, Leung CK, Lam DS. Central corneal thickness measurements using Orbscan II, Visante, ultrasound, and Pentacam pachymetry after laser in situ keratomileusis for myopia. J Cataract Refract Surg. 2007;33(7):1177–1182. | ||
de Sanctis U, Missolungi A, Mutani B, Richiardi L, Grignolo FM. Reproducibility and repeatability of central corneal thickness measurement in keratoconus using the rotating Scheimpflug camera and ultrasound pachymetry. Am J Ophthalmol. 2007;144(5):712–718. | ||
de Sanctis U, Missolungi A, Mutani B, Grignolo FM. Graft central thickness measurement by rotating Scheimpflug camera and ultrasound pachymetry after penetrating keratoplasty. Ophthalmology. 2007;114(8):1461–1468. | ||
Kuerten D, Plange N, Koch EC, Koutsonas A, Walter P, Fuest M. Central corneal thickness determination in corneal edema using ultrasound pachymetry, a Scheimpflug camera, and anterior segment OCT. Graefes Arch Clin Exp Ophthalmol. 2015;253(7):1105–1109. | ||
Lekskul M, Aimpun P, Nawanopparatskul B, et al. The correlations between Central Corneal Thickness and age, gender, intraocular pressure and refractive error of aged 12–60 years old in rural Thai community. J Med Assoc Thai. 2005;88(Suppl 3):175–179. | ||
Perez-Bermejo M, Cervino A, Calvo-Maroto AM, Moscardo M, Murillo-Llorente M, Sanchis-Gimeno JA. Corneal thickness response after anesthetic eye drops: our own results and meta-analysis. Biomed Res Int. 2018;2018:9–9. | ||
Wu W, Wang Y, Xu L. Meta-analysis of Pentacam vs. ultrasound pachymetry in central corneal thickness measurement in normal, post-LASIK or PRK, and keratoconic or keratoconus-suspect eyes. Graefes Arch Clin Exp Ophthalmol. 2014;252(1):91–99. | ||
Sadoughi MM, Einollahi B, Einollahi N, Rezaei J, Roshandel D, Feizi S. Measurement of central corneal thickness using ultrasound pachymetry and Orbscan II in normal eyes. J Ophthalmic Vis Res. 2015;10(1):4–9. | ||
Yap TE, Archer TJ, Gobbe M, Reinstein DZ. Comparison of central corneal thickness between fourier-domain OCT, very high-frequency digital ultrasound, and Scheimpflug imaging systems. J Refract Surg. 2016;32(2):110–116. | ||
Correa-Pérez ME, Olmo N, López-Miguel A, Fernández I, Coco-Martín MB, Maldonado MJ. Dependability of posterior-segment spectral domain optical coherence tomography for measuring central corneal thickness. Cornea. 2014;33(11):1219–1224. | ||
Khurana RN, Li Y, Tang M, Lai MM, Huang D. High-speed optical coherence tomography of corneal opacities. Ophthalmology. 2007;114(7):1278–1285. | ||
Krysik K, Dobrowolski D, Polanowska K, Lyssek-Boron A, Wylegala EA. Measurements of corneal thickness in eyes with pseudoexfoliation syndrome: comparative study of different image processing protocols. J Healthc Eng. 2017;2017:6. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.