Back to Journals » Clinical Interventions in Aging » Volume 12
Comparative study of two different respiratory training protocols in elderly patients with chronic obstructive pulmonary disease
Authors Mehani SHM ![]()
Received 6 July 2017
Accepted for publication 29 August 2017
Published 12 October 2017 Volume 2017:12 Pages 1705—1715
DOI https://doi.org/10.2147/CIA.S145688
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Richard Walker
Sherin Hassan Mohammed Mehani1,2
1Physical Therapy Department for Internal Medicine, 2Education and Student Affairs, Faculty of Physical Therapy, Beni-Suef University, Beni Suef, Egypt
Aim: The aim of the present study was to compare threshold inspiratory muscle training (IMT) and expiratory muscle training (EMT) in elderly male patients with moderate degree of COPD.
Materials and methods: Forty male patients with moderate degree of COPD were recruited for this study. They were randomly divided into two groups: the IMT group who received inspiratory training with an intensity ranging from 15% to 60% of their maximal inspiratory pressure, and the EMT group who received expiratory training with an equal intensity which was adjusted according to the maximal expiratory pressure. Both groups received training three times per week for 2 months, in addition to their prescribed medications.
Results: Both IMT and EMT groups showed a significant improvement in forced vital capacity, forced expiratory volume in the first second, forced expiratory volume in the first second% from the predicted values, and forced vital capacity% from the predicted value, with no difference between the groups. Both types of training resulted in a significant improvement in blood gases (SaO2%, PaO2, PaCO2, and HCO3), with the inspiratory muscle group showing the best results. Both groups showed a significant improvement in the 6-min walking distance: an increase of about 25% in the inspiratory muscle group and about 2.5% in the expiratory muscle group.
Conclusion: Both IMT and EMT must be implemented in pulmonary rehabilitation programs in order to achieve improvements in pulmonary function test, respiratory muscle strength, blood oxygenation, and 6-min walking distance.
Keywords: respiratory muscle training, respiratory muscle strength, arterial blood gases, walking, chronic obstructive pulmonary disease
Introduction
- COPD is a chronic progressive lung syndrome with a high incidence worldwide. Because it is incurable, patients must undergo treatment for the rest of their lives. It is a disabling disease causing the patients to restrict their activities, and becomes a social burden with high financial cost due to repeated hospital admissions. One of the most important causes of this disability is respiratory muscle weakness.
- Inspiratory muscle training (IMT) has been extensively used to improve inspiratory muscle strength and hence mitigate the sense of dyspnea. On the other hand, expiratory muscle training (EMT) has been used in some limited studies with contradictory results, and therefore the aim of the present study was to compare IMT and EMT in patients with COPD.
- This is the first study to compare the effects of IMT and EMT, with 40 patients and a sample size power of 0.87. All the patients had a baseline weakness in the inspiratory muscles (as represented by a reduction in the maximal inspiratory pressure [MIP]) and weakness in the expiratory muscle strength (as represented by a reduction in the maximal expiratory pressure [MEP]), and after the training program, they showed a greater percentage of improvement in their respiratory muscle strength, arterial blood gases, and 6-min walk test (6MWT).
- Comparing the absolute and relative differences in both groups showed that both IMT and EMT are effective in improving respiratory muscle strength, arterial blood gases, and functional capacity represented by an improvement in the 6MWT. However, the IMT group showed greater improvements.
Despite advances in the medical management of patients with COPD, these patients remain symptomatic and suffer from exceptional dyspnea and limited daily living activities.1 COPD is a partially reversible disease and is characterized by progressive airflow limitation. Cigarette smoking is a primary predisposing risk factor for COPD, which affects the lungs and also has a systemic effect.2,3 COPD is expected to be the seventh leading cause of disability-adjusted life years and the fifth leading cause of death by 2030.4
One of the most prominent symptoms in COPD is exercise intolerance, which is extremely variable even in patients with the same degree of airflow limitation.5
In patients with COPD, the diaphragm does not show a reduction in the oxidative capacity, since hypoxia serves as an endurance training effect with a predominance of aerobic metabolism. Therefore, including diaphragm strength training and training of the inspiratory muscles rather than endurance training in pulmonary rehabilitation programs for these patients is of great importance.6–8
Ultrastructural studies have shown a reduction in sarcomere length and increased mitochondria concentration in the diaphragm. On the other hand, expiratory muscles show a reduction in force and endurance.9 Expiratory muscle weakness may be attributed to generalized myopathy in COPD and it is usually accompanied by ineffective coughing.10
COPD patients, mainly hypercapnic patients (with CO2 retention), have an inappropriate length–tension of the respiratory muscles, overriding the chemical input for dyspnea. As a result, more expiratory muscles are activated when breathlessness occurs during exertion.11
The 6MWT is a sustained submaximal popular test for measuring functional capacity in COPD patients, due to its simplicity, familiarity, and reproducibility.12,13 Respiratory muscle strength can be indirectly measured at the mouth level as an indicator for the pressure generated by the inspiratory and expiratory muscles.2
Although it has been shown that training the inspiratory muscles can produce modifications in fiber type toward type IIa and also more strength, no additional benefits were demonstrated with regard to exercise capacity.14 IMT is currently approved as an important component of pulmonary rehabilitation, with a well-accepted evidence base. Strengthening of inspiratory or expiratory muscles could be achieved through resistive or pressure threshold equipment. Pressure threshold systems are more reliable as the amount of load can be accurately determined and graduated.15
It has been concluded that patients with inspiratory muscle weakness, lower PaO2, or higher PaCO2 could gain more benefits from IMT with regard to functional capacity and inspiratory muscle strength.16 On the other hand, there are contradictory results concerning the effects of different breathing exercises on the level of oxygen saturation.17 Only scarce data are available as regards the effect of EMT on respiratory muscle strength and exercise tolerance,18,19 and therefore the present study could be used as a trial of evidence-based practice in this contradictory zone for managing COPD patients.
Materials and methods
Patients
Forty male patients with moderate degree of COPD according to the criteria of the American Thoracic Society participated in the present study. They were recruited from the outpatient clinic of Imbaba National Institute of Allergy and Chest Diseases, Giza. The degree of expiratory airflow limitation was determined based on spirometry testing with forced expiratory volume in the first second (FEV1) ≤50%–70% of the predicted value. The ages of the participants ranged between 50 and 60 years. None of the patients had experienced a respiratory infection in the previous 2 months and none of them were enrolled in a program of pulmonary rehabilitation.
All the participants were smokers, with an index of <400 (mild to moderate degree). Exclusion criteria included the following: severe airflow obstruction (FEV1 <50% of the predicted value), chest infection in the previous 2 months, chronic respiratory failure requiring supplemental O2, CO2 retention, use of drugs such as corticosteroids or anabolic steroids within the previous 3 months which could affect exercise capacity, cardiac disease, neurological or musculoskeletal disorders, pulmonary hypertension, and mental disorders that could affect cooperation.
A randomization process was performed using closed envelopes. The investigator prepared 50 closed envelopes, each envelope containing a card labeled either group A or group B. Each patient was asked to draw a closed envelope to determine whether he was allocated to group A or group B. The patients were randomly allocated to either the IMT group (n=25) or the EMT group (n=25). The randomization (active dynamic adaptive randomization; minimization) was performed by a colleague who was independent and blind with respect to the study, using closed envelopes with the group description randomly placed within them. All patients were under medical control and were medically stable with no acute exacerbation in the previous 2 months. For all patients, several practice tests were performed for evaluation purposes, to correct possible training and learning effects. All the data were collected by the same examiner who was blinded to the patients and their training protocol. The study was conducted in accordance with the Declaration of Helsinki and approved by the ethical committee of the National Institute of Allergy and Chest Diseases, and informed consent was obtained from all patients. The study was registered in the Pan African Clinical Trials Registry with the number PACTR201703002095224.
Instrumentation
Assessment instrument
Spirometry
A micro respiratory pressure meter (ZAN, Berlin, Germany) was used to determine the degree of expiratory airflow obstruction, in order to fulfill the inclusion criteria, that is, to measure forced vital capacity (FVC), FEV1, FEV1/FVC, FEV1% deviation from the predicted value, and FVC% deviation from the predicted value, to measure the MIP and MEP for evaluation before and after the pulmonary rehabilitation program and for adjusting the intensity of the resisted breathing exercises for both groups. MIP and MEP can provide an indirect measurement for respiratory muscle strength.
A gas analyzer was used to obtain the arterial blood gas analysis (SaO2%, PaO2, PaCO2, HCO3, and pH). The measurement was obtained at rest, while breathing room air.
Training instrument
Inspiratory/expiratory muscle trainer
The trainers were used for applying resisted breathing exercises for both the inspiratory muscles (Group A) and the expiratory muscles (Group B).
Equipment used in the present study for IMT or EMT was manufactured by Respironics Inc., with the same principle of action, measurement range (5–41 cm H2O), resolution (2 cm H2O), length (5.1 cm), diameter (1.6 cm), mouthpiece and nose clip made of polypropylene, and adjustable pressure (expiratory trainer, 1 cm H2O and inspiratory trainer, 2 cm H2O).
The threshold loading device allowed variable loading at a detectable intensity by providing near flow-independent resistance to inspiration or expiration using a spring-loaded poppet valve. The spring characteristics are linear such that a given change in spring length results in the same change in valve-opening pressure at each spring length. The valve opens only when the inspiratory pressure in the inspiratory muscle trainer or expiratory pressure in expiratory muscle trainer generated by the patient exceeds the spring tension. The expiration in the inspiratory muscle trainer or inspiration in the expiratory muscle trainer was unimpeded and occurred via a flap valve.
Procedures
Assessment procedure
Spirometry evaluation
FVC, FEV1, FEV1/ FVC, FVC%, and FEV1% were measured three times and the largest value was recorded to determine the degree of airflow obstruction before and after the study. To ensure reproducibility of the test, the two largest values of FVC or FEV1 should not differ by more than 5%. Testing was done in a sitting position.
Respiratory muscle strength
MIP and MEP were measured using a mouthpiece with a small air leak to prevent closure of the glottis and to reduce the pressure developed by the cheeks and buccal muscles. MIP was measured when the patient maximally inhaled from residual volume and MEP was measured when the patient maximally exhaled from total lung capacity. The mouthpiece was connected to a pneumotach with an electronically adjusted shutter, which could be adjusted to resist the inspiration and measure the MIP, and also resist expiration and measure the MEP, both expressed in cm H2O. The leaning forward position was avoided, as this can result in overestimation of the values. To ensure reproducibility, testing was repeated three times with a variability of about 5%, and the largest value which could be maintained for 1 s was recorded as the MIP or MEP. Both MIPs and MEPs were measured for both groups before and after the training program.
6-Min walk test
The test was performed in a corridor 30 m long, with a standardized verbal encouragement to walk as fast as possible. Patients were allowed to stop if they could not go on and to resume walking subsequently. Pulse oximetry was used during the test, in order to stop the test if the SpO2 dropped to 4% below the resting value or became <90%. The test was repeated twice with 15 min rest between the two trials, and the mean value was recorded to correct for the training effect.
Blood gas analysis
The blood sample was obtained from the radial artery during rest in room air as described by Rupple.20 The patient’s arm was placed palm-up on a flat surface, with the wrist dorsiflexed at 45°, then the blood sample was delivered for analysis using an acid–base analyzer (Siemens 284). The sample was analyzed as soon as possible.
Training procedure
The IMT group received training in the form of six sets and each set consisted of five deep breaths against the trainer which contained a stainless-steel, spring-loaded, one-way valve offering resistance to the inspiratory muscles. The resistance of the valve was adjusted to 15% of the MIP at the beginning of the training program and was increased by 5%–10% each week up to 60% of the MIP at the end of the 2 months.21 This group also received sham or placebo EMT with the expiratory muscle trainer using a fixed load of about 7 cm H2O. The MIP was measured weekly in order to periodically adjust the training intensity.
The EMT group received resisted breathing exercises for the expiratory muscles with a threshold expiratory muscle trainer using the same intensity, which ranged from 15% of MEP up to 60% at the end of the 2 months. The group also received sham or placebo IMT using the inspiratory muscle trainer with a fixed low load of 7 cm H2O. The MEP was also measured weekly in order to periodically adjust the training intensity.
Each group started training at 15% of their initial maximal pressure (MIP for the IMT group and MEP for the EMT group). Intermediate measurements of the maximal pressure were performed every week. These maximal pressure values were used to calculate the training loads to be implemented in the following week. In other words, the training load in each week was increased continuously over time by adjusting at least 50% of the maximal pressure value recorded in the previous week. Rates of perceived exertion according to the Borg scale were also used to support decisions on training load increments.
Training for both groups took place under the supervision of the physiotherapist, and the valve-opening pressure was continuously monitored to ensure the achievement of the target pressure. Ratings of exertion were collected according to the modified Borg scale of perceived exertion during the training sessions for both groups. The rating was used and adjusted to show a score of ~3 (moderate) to reflect the perception of the patients regarding the difficulty of the physical exertion.
Training was conducted three times per week instead of four or more times per week to improve the retention of the patients in the present study, as most of the patients suffered from problems with transportation (most of them lived in rural areas) and also had low incomes (poverty). In most studies which use training six times per week, at least three of the training sessions per week were conducted unsupervised at home. In the present study, there was no funding to give each patient a device for use at home.
Data analysis
Subject characteristics were compared for both groups using the t-test. Mixed multivariate analysis of variance (MANOVA) analysis was conducted to investigate the effect of training on FEV1, FVC, FEV1/FVC, FEV1%, FVC%, MIP, MEP, pH, SaO2%, PaO2, PaCO2, HCO3, and 6MWT. Main effects of treatment, main effects of time, and interaction effects of treatment and time were investigated. The Bonferroni test was conducted as a post hoc test to follow the significant interactions or main effects, mean values of the dependent variables for both groups, and pre- and post-training results in each group. The Pearson correlation coefficient was used to determine the correlation between functional performance and other parameters. All tests were two tailed and the level of significance for all statistical tests was set at p<0.05. All statistical analyses were conducted using SPSS (statistical package for social sciences, version 19). A sample size calculation was performed at the beginning of the study using G*POWER statistical software (version 3.1.9.2; Franz Faul, Universität Kiel, Kiel, Germany) which revealed that the appropriate sample size for this study was N=40, giving an observed power of 0.87. Calculations were made using α=0.05, β=0.2, and an effect size of 0.6 to control for type I errors.
Results
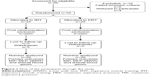
Flow chart for study procedure
All the patients were male, the disease severity was moderate, and the mean of the FEV1% deviation from the predicted value was 59.4 in the EMT group and 58.74 in the IMT group, as shown in Figure 1.
Subject characteristics
Tables 1 and 2 show the mean ± SD for age, weight, height, BMI, duration of smoking, amount of smoking in pack-years, chronicity of the disease, and medications for the EMT and IMT groups. There was no significant difference between the groups in mean age, weight, height, and BMI (p<0.05). About six patients withdrew from the study due to difficulties in transportation and another four patients reported acute exacerbation of COPD symptoms as a result of bacterial chest infections. Statistical analysis for these 10 patients showed that there was no significant difference between these patients and the other 40 patients who completed the study in all of the measured variables before starting the training program. The training program used in both groups had no relationship with their withdrawal from the study. The number of dropouts was equal in both groups (about five patients in each group) and therefore did not reflect an adverse outcome resulting from training program.
Effect of training on dependent variables
Mixed MANOVA revealed that there was a significant interaction between training and time (Wilks’ lambda =0.03; F (8.31) =106.64, p=0.0001). There was a significant main effect of time (Wilks’ lambda =0.007; F (8.31) =5,511.83, p=0.0001) and a significant main effect of training (Wilks’ lambda =0.06; F (8.31) =54.78, p=0.0001). Figure 2 shows the descriptive statistics of the dependent variables, the significance level of the comparison between groups before and after training, and the significance level of the comparison between pre- and post-training results in each group.
Within-group comparisons
Comparison between pretreatment and posttreatment results in the EMT group revealed that there was a significant increase in all variables of pulmonary functions after training compared with before training (p>0.01). In the IMT group there was a significant increase in FEV1, FVC, FEV1% from predicted values, and FVC% from predicted values after training compared with before training (p>0.0001), while there was no significant difference in FEV1/FVC before and after training (p=0.08).
Comparison between pre- and post-training results in the EMT group revealed that there was a significant difference in MEP (p=0.0001), while there was no significant increase in MIP after training compared with before training (p=0.42). There was no significant difference in pH before and after training (p=0.68). There was a significant decrease in PaCO2 (p=0.0001), and a significant increase in PaO2, HCO3, and SaO2% after training compared with pre-training values (p=0.0001). 6MWT showed a significant increase after training compared with pre-training values (p=0.0001).
Comparison between pre- and post-training results in the IMT group revealed that there was no significant difference in MEP (p=0.13), while there was a significant increase in MIP after training compared with before training (p=0.0001). There was no significant difference in pH before and after training (p=0.11). There was a significant decrease in PaCO2 (p=0.0001), and a significant increase in PaO2, HCO3, and SaO2% after training compared with pre-training values (p=0.0001). 6MWT showed a significant increase after training compared with pre-training values (p=0.0001) (Tables 3–5, Figure 2).
Comparison between EMT and IMT groups before training
There was no significant difference between the EMT and IMT groups in all dependent variables before training (p>0.05) as shown in Tables 3–5 and Figure 2.
Comparison between EMT and IMT groups after training
Pulmonary function test
Mixed MANOVA revealed that there was no significant interaction between treatment and time (Wilks’ lambda =0.91; F (5.34) =0.62, p=0.68). There was a significant main effect of time (Wilks’ lambda =0.07; F (5.34) =85.88, p=0.0001) and a significant main effect of treatment (Wilks’ lambda =0.48; F (5.34) =7.11, p=0.0001). Table 3 showed descriptive statistics relating to pulmonary functions as well as the significance level for comparisons between groups before and after treatment and the significance level of comparisons before and after treatment in each group.
There was no significant difference between the EMT and IMT groups for all variables of pulmonary functions before training (p>0.05); also, there was no significant difference between the groups after training (p>0.05) (Table 3).
Respiratory muscle strength
There was a significant increase in the MIP of the IMT group compared with the EMT group (p=0.0001), and a significant increase in the MEP of the EMT group compared with the IMT group (p=0.0001) (Tables 4 and 5, Figure 2).
Blood gases
There was no significant difference in pH between the EMT and IMT groups (p=0.07). There was a significant decrease in PaCO2 in the IMT group compared with the EMT group (p=0.02), and a significant increase in PaO2 in the IMT group compared with the EMT group (p=0.005). Furthermore, there was a significant increase in HCO3 and SaO2% in the IMT group compared with the EMT groups (p=0.0001) (Tables 4 and 5, Figure 2).
Functional performance
There was a significant increase in 6MWT in the IMT group compared with the EMT group (p=0.0001) (Tables 4 and 5, Figure 2).
Correlation between functional performance, respiratory muscle strength, and blood gases
Table 6 shows that the correlation between 6MWT and respiratory muscle strength was a positive strong significant correlation for MIP (r=0.8, p=0.0001), and a moderate positive significant correlation for MEP (r=0.37, p=0.0001).
The correlation between 6MWT and blood gases was a weak positive nonsignificant correlation for pH (r=0.16, p=0.15), a moderate negative significant correlation for PaCO2 (r=−0.44, p=0.0001), a moderate positive significant correlation for PaO2 (r=0.52, p=0.0001), a moderate positive significant correlation for HCO3 (r=0.39, p=0.0001), and a moderate positive significant correlation for SaO2% (r=0.5, p=0.0001).
Discussion
Patients with COPD have a weakness in the inspiratory muscles, mainly the diaphragm, and so they use a greater percentage of their MIP during rest and daily activities.15 COPD patients suffer from diaphragmatic dysfunction. There are various causes for this: a mechanical disadvantage due to overinflation of the lungs, remodeling, oxidative stress, reduction of myosin filament due to a reduced protein production, and an increased apoptosis of muscle cells.22
The results reported in the present study after IMT showed a statistically significant increase in pulmonary function test, MIP, PaO2, PaCO2, HCO3, SaO2%, and distance walked in 6-min testing, with no effect on the MEP.
The results of the present study showed that there was a significant change in pulmonary function test parameters that may indicate an effect on airflow limitation. This was supported by Leelarungrayub et al23 who studied the effect of a simple prototype device in moderate to severe COPD patients for 6 weeks. The device consisted of six resistors of different diameters (2–7 mm). Patients showed an increase in FVC with no change in FEV1, FEV1%, and FVC%. The results differed from those of the present study and this may be attributed to a different training device and a more advanced degree of airway obstruction.
Another study conducted by Beaumont et al24 supports the present study. These authors demonstrated an improvement in FEV1 after respiratory muscle training for patients with COPD.
This study was contradicted by the study conducted by Ahmed et al25 who evaluated the effect of short-term IMT combined with chest physiotherapy on lung function in moderate hospitalized COPD patients. They concluded that an increase in FEV1/FVC could be seen after 4 weeks of training in contrast to the results of the present study in the IMT group.
Following IMT, a significant increase in the size of type II muscle fibers could occur. In addition, training may increase the shortening velocity of inspiratory muscles, allowing more time for expiration and reducing lung hyperinflation.3 This may facilitate lung emptying and explain the improvement in FEV1, FEV1%, FVC, and FVC% observed in the present study.
The resisted inspiratory training was conducted using threshold loading to unload the inspiratory muscles.26,27 The previous finding could account for the improved functional capacity, represented by an increase in the distance walked, and this was confirmed in the present study by the strong positive correlation between 6MWT and MIP (r=0.08). On the other hand, improved inspiratory muscle strength (about 14.7% increase) could attenuate metaboreflex due to reduced waste products, which reflexively inhibits sympathetic mediated vasoconstriction activity, thus sparing blood flow and oxygen transport for the lower limb muscles (an increase in 6MWD of about 25%) which could improve exercise capacity as seen in the present study.28 This positive correlation was also confirmed by Tudorache et al.29 Deeper and slower inspiration associated with increased inspiratory muscle strength would be responsible for enhanced oxygenation.30
As observed in the present study, SaO2% was increased from 86% to about 95% after training. Another mechanism that may explain the increase in SaO2% after IMT may be related to the improved thoracoabdominal asynchrony that was observed in COPD patients after the occurrence of inspiratory muscle fatigue.12 A study by Petrovic et al3 may explain another theory regarding the improvement observed in the present study after IMT. Their study concluded that the application of strength (80% of MIP) and endurance training (60% of MIP) in patients with COPD had reduced the dynamic hyperinflation as a result of improved breathing patterns during exercise. There was a reduction in breathing frequency as a fraction of minute ventilation and an increase in the inspiratory capacity as a ratio of total lung capacity.
The results of the present study were supported by Weiner et al21 who concluded that IMT had increased inspiratory muscle strength along with an improvement in the 6MWT in severe grades of COPD (FEV1 about 37% of the predicted value).
In addition, the results of the present study were supported by studies by Hill et al31 and Koppers et al32 who reported an increase of about 32 m in IMT groups compared to about 68 m in the present study.
It is noteworthy that the prognostic cut-off value for distance walked in 6MWT is about 350 m and the minimal clinically significant difference is about 54 m.33
The results of the present study were contradicted by Bavarsad et al34 who conducted IMT in patients with mild to very severe COPD for 8 weeks and their results showed a nonsignificant change in SaO2% after training. The study by Bavarsad et al34 may reveal different results from the present study as they used a flow-volumetric inspiratory exerciser (incentive spirometry) and their patients had a higher baseline SaO2% (about 92%).
As regards the results of the EMT group, there was a statistically significant increase in pulmonary function test and MEP, no change in MIP, an increase in 6-min walking distance, an increase in SaO2% and PaO2, and a reduction in PaCO2, but these changes were less than those observed in the IMT group, except for the pulmonary function test parameters. The results of the present study showed nonsignificant differences between both groups as regards pulmonary function test.
Mota et al18 supported the results of the present study, but they examined a group of severely obstructed COPD patients and they revealed a larger increase in the distance walked in 6 min (about 53 m as opposed to about 33 m in the present study). They explained the results in terms of: 1) improved expiratory muscle strength decreasing the oxygen consumption, 2) decreased sensations of dyspnea as a result of reduced output from motor neurons, and 3) reduction of air trapping in the thorax via improved abdominal muscle tone which could elevate the diaphragm and so decrease the air trapping inside the thorax.
The superiority of IMT over EMT may be based on the fact that recruitment of the strong expiratory muscles alone without strong inspiratory muscles is not enough to eliminate dyspnea associated with effort, as the potency of expiratory metaboreflex is lower and also its threshold is higher.19,35 It is noteworthy that in patients with COPD, recruitment of weak expiratory muscles to unload the diaphragm may decrease exercise tolerance by decreasing limb blood flow and increasing thoracoabdominal asynchrony.36,37
The positive moderate correlation (r=0.37) observed in the present study between MEP and 6MWD was confirmed by Chien et al12 who studied COPD patients with severe hypercapnia (PaCO2 =55 mmHg as opposed to about 48 mmHg in the present study), and these authors supposed that expiratory muscles would be recruited during exercise to decrease the sense of dyspnea so that the patient could walk longer.
The results of the EMT group observed in the present study were contradicted by Weiner et al38 who applied EMT in a group of severely obstructed, normocapnic (PaCO2 of 38 mmHg) patients with a normal baseline expiratory muscle strength (about 86 cm H2O as opposed to about 77 cm H2O in the present study). Their results revealed no changes in arterial blood gases, contrary to the present study, but they agreed with the present study as regards the increase in the distance walked.
Weiner et al continued their study10 to compare IMT and EMT in severely obstructed COPD patients and concluded that IMT was superior for increasing walking distance, but made no reference to the changes in arterial blood gases.
The results of the present study were contradicted by Nield et al39 who conducted an EMT experiment in the form of pursed-lip breathing and an expiratory retraining program with a mild intensity of about 10% of MEP, aiming only to prolong the expiratory phase without increasing the strength of the expiratory muscles. Their results concluded that this type of expiratory training could increase the MIP in contrast to the present study which showed that EMT did not change the MIP.
The results of the present study were confirmed by Weiner et al21 who concluded that IMT did not improve the strength of the expiratory muscles and so rehabilitation programs for these patients should include EMT. A review of the literature conducted by Neves et al19 revealed that there was no increase in the walking distance after EMT.
In the aforementioned discussion of previous studies, the results were conflicting may be due to different training protocols, a limited number of studies, and small sample sizes in each study. The present study could act as a trial to shed light on the underlying mechanism and illustrate the importance of adding IMT and/or EMT in pulmonary rehabilitation programs for patients with COPD. Both IMT and EMT programs were effective in improving pulmonary function test, cardiopulmonary endurance represented by distance walked, and oxygenation. Improvements favored IMT with no difference between both types of training as regards deviations of FVC, FEV1, FEV1% from the predicted values, and FVC% from the predicted value.
Conclusion
It is essential to implement both IMT and EMT in pulmonary rehabilitation programs for male patients with moderate airway obstruction, in order to achieve improvements in pulmonary function test, respiratory muscle strength, blood oxygenation, and 6-min walking distance.
Strengths of the study
This study implemented a safe, double-blinded, and effective respiratory muscle training program for patients with chronic obstructive lung disease. This was the first study to compare the effects of IMT and EMT, with 40 patients and a sample size power of 0.87. All the patients had a baseline weakness in the inspiratory muscles (as represented by a reduction in the MIP) and a weakness in the expiratory muscle strength (as represented by a reduction in the MEP), and as a result, they showed a greater percentage of improvement in their respiratory muscle strength, arterial blood gases, 6MWT, and pulmonary function test after the training program. There was a high compliance rate from the patients except for the withdrawals (10 from 50 patients).
Limitations
The study included only male COPD patients with moderate airway obstruction. The study did not include a control group or a patient group receiving both IMT and EMT. Further studies are needed to compare IMT, EMT, and both types of training. In addition, further studies are needed to compare IMT, EMT, and pursed-lip breathing, which is a type of simple expiratory training that aims to prevent early collapse of the alveoli during expiration, thus decreasing the dynamic load of hyperinflation on the respiratory muscles.
Further reading
- Alison M. Inspiratory muscle training for managing breathlessness. Nursing in Practice. 2005:60–64.
- Battaglia E, Fulgenzia, Ferrero M. Rational of the combined use of inspiratory and expiratory devices in improving inspiratory pressure and maximal expiratory pressure of patients with chronic obstructive pulmonary disease. Arch Phys Med Rehabil. 2009;90:913–918.
- Berry MJ, Adair NE, Sevensky KS, Quinby A, Lever HM. Inspiratory muscle training and whole–body reconditioning in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2006;153(6 Pt 1):1812–1816.
- Casanova C, Celli BR, Barria P, et al; Six Minute Walk Distance Project (ALAT). The 6-minute walk distance in healthy subjects: reference standards from seven countries. Eur Respir J. 2011;37(1):150–156.
- Brooks D, Solway S, Weinacht K, Wang D, Thomas S. Comparison between indoor and outdoor 6-minute walk test among individuals with chronic obstructive pulmonary disease. Arch Phys Med Rehabil. 2003;84(6):873–876.
- Fabbri LM, Rabe KF. From COPD to chronic systemic inflammatory syndrome. Lancet. 2007;370(9589):797–799.
- Shahin B, Germain M, Kazem A, et al. Benefits of short inspiratory muscle training on exercise capacity, dyspnea, and inspiratory fraction in COPD patients. Int J Chron Obstruct Pulmon Dis. 2008;3(3):423–427.
Acknowledgments
The author would like to thank Dr Yosri Mohamed Akl, professor of Pulmonary Medicine, Faculty of Medicine, Cairo University, who has been the study collaborator.
Disclosure
The author reports no conflicts of interest in this work.
References
Sasaki M, Kurosawa H, Kohzuki M. Effects of inspiratory and expiratory muscle training in normal subjects. J Jpn Phys Ther Assoc. 2005;8(1):29–37. | ||
Vimal G, Kolek V, Jaskova J. Respiratory muscle assessment in acute exacerbation of chronic obstructive pulmonary disease and its role as a potential biomarker. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2012;156(4):302–311. | ||
Petrovic M, Reiter M, Zipko H, Pohl W, Wanke T. Effect of inspiratory muscle training on dynamic hyperinflation in patients with COPD. Int J Chron Obstruct Pulmon Dis. 2012;7:797–805. | ||
Geddes EL, O’Brien K, Reid WD, Brooks D, Crowe J. Inspiratory muscle training in adults with chronic obstructive pulmonary disease: an update of a systemic review. Respir Med. 2008;102(12):1715–1729. | ||
Dumitru L, Iliescu A, Dinu H, et al. Disability in COPD and chronic heart failure, is the skeletal muscle the final common pathway. Maedica (Buchar). 2013;8(2):206–213. | ||
Romer LM, Polkey MI. Exercise induced respiratory muscle fatigue implications for performance. J Appl Physiol (1985). 2008;104(3):879–888. | ||
Berteanu M, Iliescu A, Dumitru L. Chest physiotherapy. In: Tratat de Reabilit are pulmonarasubredactia. Tudorache V, Lovin S, Friesen M, editors, Mirton, Timisoara; 2009:189–208. | ||
Tateishi Y, Eguchi Y, Tohyama Y. Impaired muscle oxygenation during incremental cycle exercise in COPD patients compared with age matched healthy subjects. Open Rehabil J. 2011;4:32–41. | ||
Ramirez-Sarmiento A, Orozco–Levi M, Guell R, et al. Inspiratory muscle training in patients with chronic obstructive pulmonary disease: structural adaptation and physiological outcomes. Am J Respir Critical Care Med. 2002;166(11):1491–1497. | ||
Weiner P, McConnell A. Respiratory muscle training in chronic obstructive pulmonary disease: inspiratory, expiratory or both? Curr Opin Pulm Med. 2005;11(2):140–144. | ||
Chen S, Wu YT, Lin JJ, Lee CN, Huang CY, Chiang LL. The correlations of the six-minute walk test and respiratory functions in chronic obstructive pulmonary disease patients with chronic hypercapnia. J Ex Clin Med. 2014;4(1):47–51. | ||
Chien JY, Ruan SY, Huang YC, Yu CJ, Yang PC. Asynchronous thoraco-abdominal motion contributes to decrease 6-minute walk test in patients with COPD. Respir Care. 2013;58(2):320–326. | ||
Ghofraniha L, Sani ZD, Vakilian F, et al. The six-minute walk test for the evaluation of pulmonary disease. JCTM. 2015;3(2):284–287. | ||
Lotters F, Van Tol B, Kwakkel G, et al. Effects of controlled inspiratory muscle training patients with COPD: a meta–analysis. Eur Respir J. 2002;20(3):570–576. | ||
La-Ciuga H, Rosenbek JC, Davenport PW, Sapienza CM. Functional outcomes associated with expiratory muscle strength training: narrative review. JRRD. 2014;51(4):535–546. | ||
Gosselink R, De Voss J, Heuvel VD, Segers J, Decramer M, Kwakkel G. Impact of inspiratory muscle training in patients with COPD: what is the evidence? Eur Respir J. 2011;37(2):416–425. | ||
Ramos EMC, Vanderlei LCM, Ramos D, Teixeira LM, Pitta F, Veloso M. Influence of pursed – lip breathing on heart rate variability and cardiorespiratory parameters in subject with chronic obstructive pulmonary disease. Braz J Physical Ther. 2009;13(4):2288–2293. | ||
Mota S, Guell R, Barreiro E, et al. Clinical outcomes of expiratory muscle training in severe COPD Patients. Respire Med. 2007;101(3):516–524. | ||
Neves LF, Reis MH, Plentz RD, Matte DL, Coronel CC, Sbruzzi G. Expiratory and expiratory plus inspiratory muscle training improves respiratory muscle strength in subjects with COPD: systematic review. Respir Care. 2014;59(9):1381–1388. | ||
Rupple GL. Blood gases and related tests. In: Manual of pulmonary function testing. 2009. 9th ed Elsevier. 157–175. | ||
Weiner P, Magadle R, Beckerman M, Weiner M, Berar-Yanay N. Comparison of specific expiratory, inspiratory and combined muscle training programs in COPD. Chest. 2003;124(4):1357–1364. | ||
Scheibe N, Sosnowski N, Pinkhasik A, Vonderbank S, Bastian A. Sonographic evaluation of diaphragmatic dysfunction in COPD patients. Int J Chron Obstruct Pulmon Dis. 2015;10(1):1925–1930. | ||
Leelarungrayub J, Pinkaew D, Puntumetakul R, Klaphajone J. Effects of a simple prototype respiratory muscle trainer on respiratory muscle strength, quality of life, dyspnea, and oxidative stress in COPD patients: a preliminary study. Int J COPD. 2017;12:1415–1425. | ||
Beaumont M, Mialon P, Le Ber-Moy C, et al. Inspiratory muscle training during pulmonary rehabilitation in chronic obstructive pulmonary disease: a randomized trial. Chron Respir Dis. 2015;12(4):305–312. | ||
Ahmed H, Justine M, Othman Z, Mohan V, Mirza FT. Outcomes of short term inspiratory muscle training (IMT) combined with chest physiotherapy in hospitalized COPD patients. Bangladesh J Med Sci. 2013;12(4):398–404. | ||
Weiner P, Weiner M. Inspiratory muscle training may increase peak inspiratory flow in chronic obstructive pulmonary disease. Respiration. 2006;73:151–156. | ||
Petrovic M, Werner I, Reiter M, et al. Effects of inspiratory muscle training on dynamic hyperinflation in patients with COPD. Eur Respir J. 2009;34:678s. | ||
Dempsey JA, Amann M, Romer LM, Miller JD. Respiratory system determinants of peripheral fatigue and endurance performance. Med Sci Sports Exerc. 2008;40(3):457–461. | ||
Tudoroche V, Oancea C, Miladiescu O. Clinical relevance of maximal inspiratory pressure: determination in COPD exacerbation. Int J Chron Obstruct Pulmon Dis. 2010;5:119–123. | ||
Pomidori L, Campigotto F, Amatya TM, Bernardi L, Cogo A. Efficacy and tolerability of yoga breathing in patients with chronic obstructive pulmonary disease: a pilot study. J Cardiopulm Rehabil Prev. 2009;29(2):133–137. | ||
Hill K, Jenkins SC, Philippe DL, et al. High intensity inspiratory muscle training in COPD. Eur Respir J. 2006;27(6):1119–1128. | ||
Koppers RJ, Vos PJ, Boot CR, Folgering HT. Exercise performance improves in patients with COPD due to respiratory muscle endurance training. Chest. 2006;129(4):886–892. | ||
Bourdin A, Burgel PR, Chanez P, Garcia G, Perez T, Roche N. Recent advances in COPD: Pathophysiology, respiratory physiology and clinical aspects, including co morbidities. Eur Respir Rev. 2009;18(114):198–212. | ||
Bavarsad MB, Shariati A, Idani E, Zaheri H, Latifi SM. Inspiratory muscle training and arterial blood oxygen saturation in patients with chronic obstructive pulmonary disease. Jundisha Pur J Chronic Dis Care. 2015;4(1):e26623. | ||
Griffiths LA, McConnell AK. The influence of inspiratory and expiratory muscle training upon rowing performance. Eur J Appl physiol. 2007;99(5):457–466. | ||
Vogiatzis I, Stratatkos G, Athanasopoulos D, et al. Chest wall volume regulation during exercise in COPD patients with Gold Stage II to IV. Eur Respir J. 2008;32(1):42–52. | ||
Aliverti A, Stevenson N, Dellaca RL, Lo Mauro A, Pedotti A, Calverley PM. Regional chest wall volumes during exercise in chronic obstructive pulmonary disease. Thorax. 2004;59(3):210–216. | ||
Weiner P, Magadle R, Beckerman M, Weiner M, Berar-Yanay N. Specific expiratory muscle training in COPD. Chest. 2003;124(2):468–473. | ||
Nield MA, Soo Hoo GW, Roper JM, Santiago S. Efficacy of pursed lip Breathing: a breathing pattern retraining strategy for dyspnea reduction. J Cardiopulm Rehabil Prev. 2007;27(4):237–244. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.



 , mean; p-value, level of significance.
, mean; p-value, level of significance.




