Back to Journals » Clinical Interventions in Aging » Volume 14
Clinical study of the effects of deep brain stimulation on urinary dysfunctions in patients with Parkinson’s disease
Authors Zong H, Meng F, Zhang Y, Wei G, Zhao H
Received 6 February 2019
Accepted for publication 11 May 2019
Published 25 June 2019 Volume 2019:14 Pages 1159—1166
DOI https://doi.org/10.2147/CIA.S204368
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Zhi-Ying Wu
Huantao Zong,1 Fangang Meng,2 Yong Zhang,1 Guangzhu Wei,1 Huiqing Zhao3
1Department of Urology, Beijing Tiantan Hospital, Capital Medical University, Beijing, People’s Republic of China; 2Department of Neurosurgery, Beijing Tiantan Hospital, Capital Medical University, Beijing, People’s Republic of China; 3Department of Neurology, Beijing Tiantan Hospital, Capital Medical University, Beijing, People’s Republic of China
Purpose: To evaluate the effect of deep brain stimulation (DBS) on urinary dysfunctions in Parkinson’s patients.
Patients and methods: A total of 416 patients, diagnosed with Parkinson’s disease (PD) based on the UK Parkinson’s Disease Society Brain Bank Diagnostic Criteria, were enrolled in the study, including 307 males and 109 females. The effects of DBS treatment on urinary functions during urination and bladder storage of these patients were evaluated using testing and assessment scales, such as the American Urological Association Symptom Index (AUA-SI), Overactive Bladder Symptom Scores (OAB-SS), Quality Of Life Scale (QOL), and urodynamic tests. The data were statistically analyzed with the chi-square test and both independent-samples t-test and paired-samples t-test were used in this study.
Results: Symptoms of urinary dysfunctions, such as urinary frequency, urgency, and incontinence, in the patients with PD were notably relieved by DBS treatment (P<0.05), and the OAB-SS and bladder storage problems were greatly improved as well (P<0.05). Compared with those in male patients, DBS surgery significantly improved the AUA-SI, urinary symptom scores, and QOL in female PD patients (P<0.05), as well as other functional indicators related to the urinary tract, including the maximum urinary flow rate, detrusor pressure at peak flow, and residual urine volume in female PD patients (P<0.05).
Conclusion: DBS surgery is effective in improving urinary functions in PD patients, as primarily reflected by the alleviation of urinary symptoms such as urinary frequency, urgency, and incontinence. Female PD patients displayed better urinary function outcomes from DBS treatment than did male patients.
Keywords: Parkinson’s disease, urinary dysfunction, deep brain stimulation
Introduction
Parkinson’s disease (PD) is a common neurodegenerative disease that frequently develops in elderly patients, with an average onset age of approximately 60 years.1,2 In addition to motor symptoms, such as static tremors and muscle rigidity, patients with PD may also display urinary dysfunctions such as urinary frequency, urgency, and dysuria.Urinary problems may appear even in the prodromal phase of PD before the onset of motor symptoms and characterize for some patients an early symptom of the disease course.3
For PD patients who are not responsive to conservative treatments, deep brain stimulation (DBS) is often recommended by clinical researchers.4,5 The surgery has proven to be effective in treating motor system disorders in PD patients. However, the effectiveness of DBS in treating urinary dysfunctions in these patients has not been widely investigated.
This article reports the results from a prospective cohort study conducted by a group of medical doctors in urology, neurology, and neurosurgery. It included 416 patients, examined, and evaluated the effects of DBS treatment on the urinary functions of PD patients during the time of urination and the phase of bladder storage. The male and females patients were analyzed separately, and it focused not only on clinical results but also more on urodynamic results compared with previous studies. The assessment tools utilized in this study include American Urological Association Symptom Index (AUA-SI), Overactive Bladder Symptom Score (OAB-SS), Quality of Life Scale (QOL), and urodynamic testing scores. This study may provide a valuable clinical reference for the DBS treatment of PD patients.
Materials and methods
Selection of the subjects: PD patients with urinary dysfunctions
Directed by neurological experts, PD patients were selected following the UK Parkinson’s Disease Society Brain Bank Diagnostic Criteria:6
Step 1: diagnosis of clinical symptoms of PD
Reduced movements: casual movements are slow to initiate, and the speed and amplitude of repetitive movements have gradually decreased, accompanied by the development of at least one of the following symptoms: muscle rigidity; resting tremor (4–6 Hz); inability to stand steadily (the following possible causes were excluded: primary vision impairment, vestibular dysfunction, or disorders in the cerebellum or proprioception).
Step 2: Exclusion criteria: PD patients with the following problems were excluded from the study: repeated history of stroke accompanied by “stepwise” progression of PD symptoms; repeated history of brain damage; medical history of encephalitis; oculogyric crisis; patient began receiving neuroleptic treatment when symptoms of PD appeared; more than 1 family member is suffering from PD; continuous alleviation of symptoms; more than 3 years since disease onset with strict unilateral involvement; progressive supranuclear palsy; cerebellar disease symptoms; early involvement of severe autonomic nervous system damage; early appearance of severe dementia, accompanied by memory, language or behavioral disorders; pyramidal tract symptoms (positive Babinski sign); intracranial tumors or communicating hydrocephalus was detected on CT scan; unresponsive to levodopa (L-DOPA) treatments at high dosages (excluding cases of malabsorption); and history of MPTP (1-Methyl-4-phenyl-1,2,3,6-tetrahydropyridine, an opioid analgesic derivative) usage.
Step 3: Supporting diagnostic criteria: PD patients (from Step 1) with 3 or more of the following symptoms were confirmed to have Parkinson’s: unilateral onset of the disease; detectable resting tremor; gradually progressive symptoms; continued asymmetrical progression with onset side remaining as the side with more severe disease; excellent treatment response to dopamine (70–100%); severe dyskinesia caused by L-DOPA treatment; the therapeutic effect of levodopa lasted for at least 5 years; and clinical course of the disease has been at least 10 years.
Clinical PD diagnosis is determined by patients meeting the diagnostic criteria in Step 1 and matching 3 symptoms in Step 3 diagnostic criteria but not meeting any exclusion criteria in Step 2.
Diagnosis of urinary dysfunction in PD patients by urologists
Patients with PD symptoms accompanied by clinical manifestation of urinary dysfunctions, including dysuria, urinary frequency or urgency, elevated residual urine, urinary incontinence, nocturia, and urinary retention, were selected.
PD patients were excluded from the study if they exhibited one or more of the following symptoms of urinary dysfunction: history of diabetes, history of cerebrovascular diseases or neurological diseases other than PD; repeated urinary tract infections, urethral stricture, or bladder stones; previous surgical procedures in the prostate, bladder, uterus or pelvis; abnormal results in prostate-specific antigen (PSA) screening tests and/or prostate imaging examinations; as well as patients with possible prostate cancers.
General health information and the status of the urinary functions of the enrolled patients
A total of 416 patients with PD were enrolled, including 307 males and 109 females, with an average age of 61.2±5.9 years and average disease course of 7.7±4.1 years (range 4.9–10.4 years). The course of accompanying urinary symptoms lasted from 3 months to 10.2 years, averaging 5.1±4.7 years. All patients were receiving levodopa treatment along with rehabilitation therapies and psychotherapies, and they also received appropriate care instructions. All patients had urinary dysfunction to varying degrees. Nocturia is the most prominent urinary symptom among all the patients with the highest incidence (265/416, 63.7%), followed by urinary frequency (232/416, 55.8%), residual urine (182/416, 43.8%), urinary urgency (158/416, 38.0%), dysuria (150/416, 36.1%), and urinary incontinence (74/416, 17.8%). All patients underwent hematuria and urinary tract ultrasonography, and male patients were also subjected to the PSA test.
Analysis of the urinary functions in PD patients after DBS surgery
Of the 416 patients, 220 patients received DBS surgery due to unsatisfying outcomes of motor-symptom treatments with conservative therapies. The general backgrounds of the two groups of patients with or without DBS were considered comparable (Table 1). Assessment parameters, including AUA-SI, OAB-SS, QOL, and subjective urodynamic tests, were used to conduct a systematic evaluation of the urinary functions of these patients.
 | Table 1 General health information of the two groups of patients with and without DBS |
PD patients who continued to show urinary dysfunctions after DBS were prescribed an α-receptor blocker to relieve symptoms during urination and tolterodine to relieve symptoms during bladder storage.
Ethics
The study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of Beijing Tiantan Hospital affiliated with Capital Medical University. All patients were aware of the entire study process and agreed to participate in the research. All patients signed an informed consent form prior to receiving any treatment or examination.
Statistical analysis
All the data were processed with SPSS 11.5 software (SPSS Inc., USA). The general health information, AUA-SI, OAB-SS, QOL, and urodynamic test data of the two groups of patients are expressed as the means ± standard deviation (X ± SD). The chi-square test and t-test were used to perform the statistical analysis, and P<0.05 was considered statistically significant.
Results
The impact of DBS surgery on urinary functions of the PD patients
Compared with patients who did not receive DBS, urinary frequency, urgency, and incontinence were all improved in patients who received DBS (P<0.05); however, symptoms of nocturia, residual urine feeling, and dysuria were not improved significantly (Tables 2 and 3). Compared with their preoperative values, the postoperative OAB-SS and urinary symptom scores during bladder storage in patients were significantly improved after DBS (P<0.05). Although the AUA-SI, symptom score during urination, and QOL were somewhat improved after DBS, the differences were not statistically significant (Table 4). Finally, the urodynamics-related parameters, such as the maximum urinary flow rate (Qmax), detrusor pressure at peak flow (Pdet), bladder capacity, and post-void residual urine volume, tested after DBS treatment were not significantly different from their corresponding preoperative values (Table 4).
 | Table 2 Incidence of urinary dysfunctions in PD patients with and without DBS |
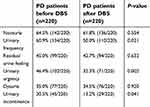 | Table 3 Incidence of urinary dysfunctions in PD patients before and after DBS |
 | Table 4 Comparison of the assessment scales of urinary functions and urodynamic test results in PD patients before and after DBS |
Effect of DBS on urinary functions in male patients with PD
The postoperative OAB-SS and urinary symptom scores during bladder storage in male patients treated with DBS were significantly improved after the surgery (P<0.05), while the AUA-SI score, symptoms during urination, and QOL of the male patients were slightly improved after DBS but not significantly different (Table 5). Compared with those before DBS, the related parameters, such as Qmax, Pdet, bladder volume, and residual urine volume, did not display any statistically significant differences in males after DBS (Table 5).
 | Table 5 Comparison of urinary functional assessment parameters and urodynamic test results in male PD patients before and after DBS |
The effect of DBS on urinary functions in female patients with PD
The OAB-SS and bladder storage symptoms of female PD patients were markedly improved after DBS surgery (P<0.01), and the post-DBS AUA-SI, urinary symptoms, and QOL were also improved significantly (P<0.05) (Table 6). Consistently, the related urodynamic test parameters, such as the postoperative Qmax, Pdet, and residual urine, showed notable improvements in female patients as well (P<0.05) (Table 6).
 | Table 6 Comparison of the urinary assessment scales and urodynamic test results in female PD patients before and after DBS treatment |
Discussion
The results of our study demonstrated that PD patients experienced improved urinary frequency, urgency, and urinary incontinence after DBS, but without significant relief in symptoms including nocturia, residual urine, and dysuria. DBS markedly improved OAB-SS and bladder storage symptoms compared with those before DBS; however, there were no statistically significant changes in AUA-SI, urination symptoms, and QOL of the patients after the procedure. A difference in response to the DBS treatment existed between the male and female patients with PD. The female patients showed an across-the-board improvement in all urinary functions after DBS surgery, including the AUA-SI, OAB-SS, symptoms during bladder storage and urination, and QOL. Furthermore, other urinary-related postoperative functional parameters, such as Qmax, Pdet, and residual urine volume, in the female patients were significantly improved as well compared with their corresponding preoperative values.
The impact of DBS on urinary functions of PD patients
DBS, applying high-frequency electrical stimulation to deep brain structures, has now provided an effective therapeutic option for treatment of various neurological and psychiatric disorders. DBS targeting the internal segment of the globus pallidus, subthalamic nucleus, and thalamus is used to treat symptoms of movement disorders via neurochemistry modulatiion, such as PD, dystonia, and tremor. However, numerous questions still remain on the neurophysiological basis for the therapeutic mechanisms of action of DBS. It is speculated by various studies that DBS may bring about changes in neurogenesis and microenvironment, and then activate the neuronal network by changes of synapses.7–9 DBS-induced synaptic events could change the balance of neurotransmitters within the stimulated brain network and then rescue memory and synaptic activity, and synaptic degeneration may perhaps be the earliest sign of neurodegeneration.10–12 In the context of PD, urinary dysfunctions may far precede the classic central nervous system and motor symptoms, which may be the earliest sign for diagnosis and timely treatment for PD.
From a physiological perspective, the pathogenesis of PD is closely related to the developing mechanisms of urinary dysfunctions.13 The most prominent pathological changes in patients with PD are the degeneration or loss of pigmented neurons localized in the basal ganglia and brainstem, with the most significant loss of the dopaminergic neurons occurring in the pars compacta of the substantia nigra. Urinary functions are controlled and regulated to various degrees by a series of structures in the central nervous system, including the cerebral cortex, basal ganglia, hypothalamus and the reticular formation of the brainstem. Thus, the degeneration and loss of neurons in these structures may lead to urinary dysfunctions in PD patients.
The DBS procedure consists of implanting neuron-stimulating electrodes at specifically targeted locations in patients’ brains through precise stereotactic positioning and performing high-frequency electrical stimulation of the hypothalamic nuclei of the patients to excite the nuclei, thereby improving motility, controlling seizures, and relieving pain in PD patients.14 The nuclei of the brain, such as the subthalamic nucleus, are involved in the regulation of urinary functions. DBS treatments stimulate these nuclei to also improve urinary functions, offering one of the possible reasons for the relief of urinary symptoms in the group of PD patients who received DBS.15 Additionally, the stimulation of the hypothalamic nuclei can increase the release of vasopressin through the hypothalamic-neuronal pituitary system, which in turn promotes the reabsorption of water molecules in the renal collecting duct and in the distal convoluted tubule, consequently reducing the quantity of urine production.16 The regulatory control of urination, together with the reduction in urine production, can result in significant improvements in urinary frequency in male PD patients.
The pudendal nerve innervates the external urinary sphincter, and its excitation triggers the contraction of the external urinary sphincter to stop urination.17 This effect is under strict control and regulation by the consciousness of our brains. DBS may excite specific nuclei in the brain that exert strong control of the pudendal nerves, which could be the main reason for the alleviation of urinary frequency, urinary urgency, and urinary incontinence observed in PD patients after DBS.
Meanwhile, some previous studies revealed that DBS maybe useful for urinary dysfunctions caused by nervous system disease, such as PD. Based on the study conducted by Winge K (2007) which 16 PD patients included, symptoms of overactive bladder decreased after DBS, while the urodynamic parameters did not change significantly after DBS.18 While the study by Mordasini L (2014) found that low-frequency DBS may cause detrusor overactivity, which offered a contrary result.19 However, both of them were small sample studies. Another study by Winge K (2012) revealed that nocturia was significantly improved after DBS, which differed from our study.20 The study by Roy HA (2018) found that DBS did slightly increase maximal capacity, and the study by Mock S (2016) revealed that there were notable improvements in QOL for LUTS post-DBS in the subthalamic nucleus target.21,22 Overall, the number of patients included in these studies was limited, and most of the research focused more on clinical results. And, our study focused not only on clinical results but also on urodynamic results compared with previous studies.
Gender is likely an independent factor influencing DBS-induced improvement of urinary functions in PD patients
It can be seen from our study that the improvement in urinary dysfunctions after DBS in female PD patients was superior to that in males. In female patients, DBS surgery not only alleviated problems during the bladder storage phase but also improved symptoms during urination. However, the regulatory control of urination in the higher and lower levels of the central nervous system is identical between males and females, and the specifically targeted nuclei by DBS stimulation in the brain are also the same.23 Thus, why did such a gender-specific response exist in patients with PD?
Urination control in males mainly lies in the membranous portion of the urethra, whereas the control in females resides inside the mid to distal parts of the urethra. Although the male membranous urethra and female mid-distal urethra are very similar in terms of anatomy, tissue structure, embryonic development, and innervation, the morphology of the male urethral sphincter is different from that of females due to the presence of the male prostate.24–26 This morphological difference between males and females forms the anatomical basis for the functional difference in the urinary sphincter between genders. As a result of the position and existence of the prostate in males, as well as the morphological and functional differences in the urethral sphincter between genders, the response to DBS treatment with respect to urinary functions of PD patients appeared to be more gender-specific, with broader improvements in females. The male patients who received DBS were 61.3±5.9 years old with a prostate volume of 37.8±6.8 cm3. Hyperplasia in the prostate can also cause urinary dysfunctions, which could not be ruled out in our study. Therefore, it is especially important to continue to monitor the urinary functions of these patients in the follow-up visits after DBS, as treating the prostate with medications and/or surgical procedures may become necessary.
Conclusion
In summary, urinary dysfunctions are important non-motor symptoms in patients with PD. DBS has been proven to be effective in improving urinary frequency, urgency, and incontinence, but it did not display much effect on symptoms such as nocturia, residual urine, and dysuria. The outcomes of DBS treatment in improving urinary dysfunctions in female patients with PD are far superior to those in male patients, as symptoms during urination and bladder storage were both significantly improved in the females. However, case-controlled multicenter studies with larger sample sizes are still needed to further confirm these findings.
Acknowledgments
This study was funded by the Beijing Municipal Administration of Hospitals Clinical Medicine Development of Special Funding Support (ZYLX201820) and the Capital City Award for Featured Applied Clinical Research and Achievement Popularization (Z151100004015163).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Keener AM, Paul KC, Folle A, et al. Cognitive impairment and mortality in a population-based Parkinson’s disease cohort. J Parkinsons Dis. 2018;8:353–362. doi:10.3233/JPD-171257
2. Tanguy A, Jönsson L, Ishihara L. Inventory of real world data sources in Parkinson’s disease. BMC Neurol. 2017;17:213. doi:10.1186/s12883-017-0985-0
3. Sakakibara R, Tateno F, Yamamoto T, et al. Urological dysfunction in synucleinopathies: epidemiology, pathophysiology and management. Clin Auton Res. 2018;28:83–101. doi:10.1007/s10286-017-0480-0
4. Nazzaro JM, Pahwa R, Lyons KE. Symptomatic, non-infectious, non-hemorrhagic edema after subthalamic nucleus deep brain stimulation surgery for Parkinson’s disease. J Neurol Sci. 2017;383:42–46. doi:10.1016/j.jns.2017.10.003
5. James ND, McMahon SB, Field-Fote EC, Bradbury EJ. Neuromodulation in the restoration of function after spinal cord injury. Lancet Neurol. 2018;17:905–917. doi:10.1016/S1474-4422(18)30287-4
6. Homayoun H. Parkinson disease. Ann Intern Med. 2018;169:ITC33–ITC48. doi:10.7326/AITC201809040
7. Stone SS, Teixeira CM, Devito LM, et al. Stimulation of entorhinal cortex promotes adult neurogenesis and facilitates spatial memory. J Neurosci. 2011;31:13469–13484. doi:10.1523/JNEUROSCI.3100-11.2011
8. Chen YC, Shi L, Zhu GY, et al. Effects of anterior thalamic nuclei deep brain stimulation on neurogenesis in epileptic and healthy rats. Brain Res. 2017;1672:65–72. doi:10.1016/j.brainres.2017.07.021
9. Gajera CR, Emich H, Lioubinski O, et al. LRP2 in ependymal cells regulates BMP signaling in the adult neurogenic niche. J Cell Sci. 2010;123:1922–1930. doi:10.1242/jcs.065912
10. McIntyre CC, Anderson RW. Deep brain stimulation mechanisms: the control of network activity via neurochemistry modulation. J Neurochem. 2016;139(Suppl 1):338–345. doi:10.1111/jnc.13649
11. Chiken S, Nambu A. mechanism of deep brain stimulation: inhibition, excitation, or disruption? Neuroscientist. 2016;22:313–322. doi:10.1177/1073858415581986
12. Gondard E, Teves L, Wang L, et al. Deep brain stimulation rescues memory and synaptic activity in a rat model of global ischemia. J Neurosci. 2019;39:2430–2440. doi:10.1523/JNEUROSCI.1222-18.2019
13. Madan A, Ray S, Burdick D, Agarwal P., et al. Management of lower urinary tract symptoms in Parkinson’s disease in the neurology clinic. Int J Neurosci. 2017;127:1136–1149. doi:10.1080/00207454.2017.1327857
14. Lee DJ, Dallapiazza RF, De Vloo P, et al. Current surgical treatments for Parkinson’s disease and potential therapeutic targets. Neural Regen Res. 2018;13:1342–1345. doi:10.4103/1673-5374.235220
15. Gao Y, Liao L, Blok BF. A resting-state functional MRI study on central control of storage: brain response provoked by strong desire to void. Int Urol Nephrol. 2015;47:927–935. doi:10.1007/s11255-015-0978-0
16. Crowley WR. Neuroendocrine regulation of lactation and milk production. Compr Physiol. 2015;5:255–291. doi:10.1002/cphy.c140029
17. Peters KM, Feber KM, Bennett RC. Sacral versus pudendal nerve stimulation for voiding dysfunction: a prospective, single-blinded, randomized, crossover trial. Neurourol Urodyn. 2005;24:643–647. doi:10.1002/nau.20174
18. Winge K, Nielsen KK, Stimpel H, et al. Lower urinary tract symptoms and bladder control in advanced Parkinson’s disease: effects of deep brain stimulation in the subthalamic nucleus. Mov Disord. 2007;22:220–225. doi:10.1002/mds.21253
19. Mordasini L, Kessler TM, Kiss B, et al. Bladder function in patients with dystonia undergoing deep brain stimulation. Parkinsonism Relat Disord. 2014;20:1015–1017. doi:10.1016/j.parkreldis.2014.05.016
20. Winge K, Nielsen KK. Bladder dysfunction in advanced Parkinson’s disease. Neurourol Urodyn. 2012;31:1279–1283. doi:10.1002/nau.22237
21. Roy HA, Pond D, Roy C, et al. Effects of pedunculopontine nucleus stimulation on human bladder function. Neurourol Urodyn. 2018;37:726–734. doi:10.1002/nau.23321
22. Mock S, Osborn DJ, Brown ET, et al. The impact of pallidal and subthalamic deep brain stimulation on urologic function in parkinson’s disease. Neuromodulation. 2016;19:717–723. doi:10.1111/ner.12446
23. Kitta T, Mitsui T, Kanno Y, et al. Brain-bladder control network: the unsolved 21st century urological mystery. Int J Urol. 2015;22:342–348. doi:10.1111/iju.12721
24. Wen JG, Lu YT, Cui LG, et al. Bladder function development and its urodynamic evaluation in neonates and infants less than 2 years old. Neurourol Urodyn. 2015;34:554–560. doi:10.1002/nau.22626
25. Kerdraon J, Peyronnet B, Gamé X, et al. Pathophysiology of detrusor underactivity in the elderly. Prog Urol. 2017;27:402–412. doi:10.1016/j.purol.2017.04.005
26. Hester AG, Kretschmer A, Badlani G. Male incontinence: the etiology or basis of treatment. Eur Urol Focus. 2017;3:377–384. doi:10.1016/j.euf.2017.12.002
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
