Back to Journals » Clinical Ophthalmology » Volume 9
Clinical performance of KeraSoft® IC in irregular corneas
Authors Su S, Johns L, Rah M , Ryan R, Barr J
Received 23 April 2015
Accepted for publication 30 July 2015
Published 22 October 2015 Volume 2015:9 Pages 1953—1964
DOI https://doi.org/10.2147/OPTH.S87176
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Stephanie Su,1 Lynette Johns,2 Marjorie J Rah,3 Robert Ryan,1 Joseph Barr3
1Visionary Eye Associates of Rochester, Rochester, NY, USA; 2Custom Lab Channel Business, Bausch & Lomb Incorporated, Wilmington, MA, USA; 3Medical Affairs – Vision Care, Bausch & Lomb Incorporated, Rochester, NY, USA
Purpose: This study evaluated the clinical performance of KeraSoft® IC (KIC) soft contact lenses in subjects with irregular corneas.
Patients and methods: This was a 12-month, prospective, open-label, observational study, which enrolled 43 subjects who were 18 years of age or older with irregular corneas. Subjects were fit according to the KIC Fitting Manual (kerasoftic.com). After achieving best fit according to the fitting manual, lenses were assessed for comfort, vision, centration, rotation, and movement. Subjects were instructed to wear their lenses between 8 and 16 hours each day. Assessments at the exit visit included logMAR visual acuity with high and low contrast, spherocylindrical overrefraction, slit-lamp findings, adverse events, and subjective outcomes.
Results: The average base curve was 8.17±0.32 mm (n=70 eyes), and the average diameter dispensed was 14.53±0.12 mm (n=70 eyes). From the baseline to 12 months, there was statistically significant improvement in logMAR visual acuity with high contrast (P=0.038), but no significant difference in low-contrast visual acuity was observed (P>0.05). Slit-lamp findings were ≤ grade 1 for the majority of subjects (89%). Two nonserious adverse events were reported for two of the 84 enrolled eyes (two subjects). At 12 months, subjects reported improvements from habitual baseline for comfort and vision, both upon insertion and just before removal of lenses.
Conclusion: Clinical outcomes at 12 months showed good visual, safety, and subjective outcomes for subjects with corneal irregularities who wore KeraSoft® IC soft contact lenses.
Keywords: irregular corneas, keratoconus, soft contact lenses, KeraSoft® IC, pellucid marginal degeneration, post penetrating keratoplasty
Introduction
Keratoconus is a progressive disorder that usually occurs in the second to third decade of life, affects both sexes and all ethnicities, and has an estimated prevalence in the general population of 54 per 100,000.1 In keratoconus, the cornea assumes a conical shape from the thinning of the corneal stroma and ectasia, which induces irregular astigmatism, myopia, and protrusion.2,3 Most patients eventually develop corneal scarring.1,2 Other corneal irregularities include pellucid marginal degeneration, which is characterized by inferior thinning 1–2 mm from the limbus, with ectasia occurring superiorly to the thinned area. Keratoglobus is defined by diffuse thinning and overall ectasia.3 Although early management may include spectacles, contact lenses are typically indicated as the disorder progresses, and severe cases may need keratoplasty.1
The standard correction of choice to date has been rigid gas permeable (RGP) contact lenses.4 Other contact lens options have also been introduced, such as soft spherical and soft toric lenses,5 hybrid lenses,6 scleral lenses, and piggyback lenses.4 In addition, preventative measures such as cross-linking have been used to potentially slow the progression of keratoconus.7,8 In advanced cases, such as when corneal scarring or contact lens use becomes intolerable or vision obtained is unsatisfactory, penetrating keratoplasty (PKP)2,3 or deep anterior lamellar keratoplasty9 may be indicated.
The goals of fitting contact lenses for patients with keratoconus include providing good visual acuity (VA) and acceptable comfort, while maintaining the integrity of the ocular surface. Optimal fit for contact lenses can be challenging as the disease advances. Modifying the contact lens fit to reduce the potential for corneal scarring is controversial.10 Baseline factors that have been associated with corneal scarring in keratoconus include corneal staining, contact lens wear, Fleischer’s ring, a steeper cornea, and increasing age.10 In some cases, patients have developed corneal scarring secondary to increased corneal staining associated with the contact lens modality (RGP).11 Therefore, providing patients with a good contact lens fit may aid in minimizing contact lens-induced scarring, as well as delay the need for keratoplasty.12 Patients with irregular astigmatism after refractive surgery can have complications relating to the fit of the contact lenses due to the surgically modified curvature changes of the cornea. Patients may have suboptimal contact lens fits with too much movement or be intolerant to the comfort because of altered corneal nerves.13
A customizable quarterly replacement soft contact lens (KeraSoft® IC; Art Optical Contact Lens, Inc., Grand Rapids, MI, USA) has been developed to address the unique challenges of patients with a variety of irregular corneal conditions, including keratoconus and conditions due to postgraft and postrefractive surgery. The lens is designed to fit all stages of keratoconus and other irregular corneas, and has an adjustable periphery, allowing the lens to fit many corneal shapes. The objective of this study was to evaluate the clinical performance, particularly comfort and vision, of KeraSoft® IC (KIC) soft contact lenses in Efrofilcon A silicone hydrogel material.
Material and methods
Study design
This was a 12-month, prospective, open-label study conducted at a single site from 2011 to 2013. This protocol was approved by Southwest Independent Institutional Review Board, and all subjects provided informed consent. The study was conducted in accordance with good clinical practice guidelines and the tenets of the Declaration of Helsinki and adhered to ethical standards and practices for responsible conduct of clinical research.
Eligibility criteria
To be eligible for entry into the study, subjects were required to be 18 years of age or older and have full legal capacity to volunteer; have read, understood, and signed the informed consent form; and provided Health Insurance Portability and Accountability Act authorization. Subjects had keratoconus, as defined by corneal irregularity indicative of corneal ectasia determined by topography, keratometry, or retinoscopy, and at least one slit-lamp sign consistent with the diagnosis of keratoconus, including Vogt’s striae, Fleischer’s ring, or central/paracentral corneal scarring in at least one eye, or have an irregular cornea due to pellucid marginal degeneration, postrefractive surgery, or post-PKP. Subjects needed to be correctable to 20/50 or better (distance, high contrast) in each eye with either spectacles or contact lenses. Subjects had to agree to wear study lenses daily for at least 8 hours each day for 12 months and to meet the protocol specified visit schedule.
Ineligibility was based on the following criteria: presence of systemic disease affecting ocular health, use of any systemic or topical medications that could affect ocular physiology or lens performance, spherical correction >+45.00 D or <−45.00 D in either eye, refractive astigmatism of greater than 13.00 D in either eye, was not correctable to 20/50 or better in each eye with contact lenses, greater than grade 2 slit-lamp exam findings, any atypical scar or neovascularization within the central 4 mm of the cornea for a subject who had keratoconus or was postoperative. Enrollment was also precluded if the subject was aphakic, currently participating in any other clinical research study or had participated in any other clinical research study within 2 weeks prior to enrollment, was an employee of the investigative site, or was pregnant or currently breast-feeding.
Test article
The KIC lenses in this study were composed of a silicone hydrogel material, Efrofilcon A. The front surface of the lens has an aspheric or aspheric toric design, and the adjustable periphery is designed to fit most corneal shapes. The base curve is available from 7.40 to 9.40 mm in 0.2 mm increments. The lens diameter begins at 14.5 mm and is customizable in 0.5 mm increments from 14.0 to 15.5 mm. There is a single laser marking at the 6 o’clock position for assessing rotation (the right lens has a single line and the left lens has an interrupted laser marking). The lens specifications are shown in Table 1.
  | Table 1 KeraSoft® product specifications |
Study procedures
Subjects meeting eligibility criteria participated in a screening visit, dispensing visit, and follow-up visits with the dispensed lenses at 2 weeks, 3 months, 6 months, and 12 months.
At the screening visit, high-contrast logMAR VA with high ambient illumination (HCHI) and low-contrast (10% contrast) VA with high ambient illumination (LCHI) were measured with patients usual visual correction (baseline). All VA testing was performed using an iMac monitor with Posture Video Analysis Tool (PVAT) software, a simulated 20 ft lane, and with high ambient illumination (4 lamp 32 W fluorescent). Subjects rated visual quality and comfort on an analog scale. In addition, hours of wear with habitual lenses were captured. Subjects completed a consumer-based questionnaire based on their habitual correction (contact lenses/spectacles). Corneal topography was captured. Slit-lamp assessment was performed, including limbal and bulbar redness, corneal and conjunctival staining, contact lens-induced conjunctival impression with fluorescein, epithelial edema, microcysts, neovascularization, infiltrates, and scarring. The severity of slit-lamp findings was graded from 0 to 4, with 0 representing no findings; extent of slit-lamp findings was graded in clock hours. A spherocylindrical refraction was performed through which HCHI and LCHI VAs were measured. Subjects were fit with study lenses according to the KIC Fitting Manual that uses a dynamic assessment technique called MoRoCCo VA. The acronym MoRoCCo VA represented the steps involved in the lens assessment process: movement, rotation, centration, comfort, and visual acuity. An optimal fit demonstrated up to 2.0 mm movement, less than 10° rotation, minimal decentration, comfort, and stable vision between blinks. A spherocylindrical overrefraction was performed, and HCHI and LCHI VAs were measured.
At the dispensing visit, HCHI and LCHI VAs were measured with the study lenses, and spherocylindrical overrefraction with HCHI and LCHI VAs were performed. Contact lens fit was assessed, and replacement lenses were ordered if needed (either due to fit or overrefraction), with a maximum of three orders (one initial and two reorders) per patient throughout the study. Subjects were instructed to wear their lenses between 8 and 16 hours each day with the final dispensed lenses. Patients were instructed to use a multipurpose solution (Biotrue; Bausch & Lomb Incorporated, Rochester, NY, USA). If patients were sensitive to the multipurpose solution, they were then instructed to use a peroxide care system (ClearCare; Alcon, Fort Worth, TX, USA). In some cases, a daily cleaner was required (Opti-Free Daily Cleaner; Alcon, Fort Worth, TX, USA).
At the 3-, 6-, 9-month, and exit visits, subjects completed a consumer-based questionnaire based on their experience with the study lenses. Subjects also rated visual quality and comfort of the study lenses on an analog scale. The following tests were performed: HCHI and LCHI VAs with the study contact lenses, as well as a spherocylindrical overrefraction with HCHI and LCHI VAs. The surfaces of the lenses were evaluated for lens wettability and surface deposition. Limbal and bulbar redness were assessed; lenses were removed, then the following were performed: corneal topography, corneal and conjunctival staining with fluorescein, and spherocylindrical refraction through which HCHI and LCHI VAs were measured.
Statistical methods
Subjective responses were analyzed comparing habitual lens experience to end of study assessments for comfort, vision, and the consumer-based subjective questionnaire. With regard to the questionnaire, the P-values were based on the unfavorable responses to comfort, vision, handling, and performance of the habitual lens and KIC after 12 months, and they were calculated using the McNemar’s tests. A category that is found to be statistically significant demonstrates that KIC is more favorable than habitual lens. Differences at the α<0.05 level were considered to be statistically significant.
Results
Demographics and baseline characteristics
Demographic data and baseline eye characteristics are summarized in Table 2. Of the 43 subjects (84 eyes) enrolled, 88.4% were white, 55.8% were male, and the average age was 44.7 years. The habitual correction worn by subjects was as follows: 34.9% spectacles, 22.1% soft contact lens, 12.8% hybrid contact lens, 23.3% gas permeable contact lens, and 7.0% piggyback lens. Baseline refractive characteristics by keratometry category are detailed in Table 3. Of the 43 enrolled subjects, one had a history of cataract extraction, one had a PKP, and two had laser-assisted in situ keratomileusis (LASIK). Representative topography examples are shown in Figure 1, including those for keratoconus (top), post-LASIK (middle), and post-PKP. A total of 36 subjects completed the study, and seven subjects discontinued the study due to reasons such as lack of motivation (one), lost to follow-up (two), and unacceptable distance VA (four).
  | Table 2 Subject demographics and baseline eye characteristics |
  | Table 3 Baseline refractive characteristics by keratometry categories |
Endpoints
Visual acuity
Distance high- and low-contrast logMAR VAs are shown in Table 4. The mean distance HCHI logMAR VA with study lenses at the 12-month visit was 0.14±0.14 (approximate equivalent of 20/27 Snellen). The mean distance VA with LCHI at the 12-month visit was 0.40±0.16 logMAR (approximately equivalent of 20/50 Snellen). The improvement in distance HCHI logMAR from habitual VA was statistically significant at the 12-month visit (−0.058±0.20; P=0.038). The change in distance LCHI logMAR from habitual VA was not statistically significant at the 12-month visit (−0.01±0.18; P=0.481).
Spherocylindrical refractive changes from baseline to exit visit
The mean absolute change in spherocylindrical refractive error between baseline and the 12-month visit was 0.80±1.10 D (n=70 eyes). The majority of eyes (91%, 64/70) showed an absolute change within 0.00–1.99 D (Figure 2).
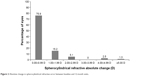  | Figure 2 Absolute change in spherocylindrical refractive error between baseline and 12-month visits. |
Final dispensed lens specifications
The final dispensed lens information, stratified by keratometry category, is shown in Table 5. The average base curve was 8.17±0.32 mm (n=70 eyes), with a range from 7.40 to 8.60 mm. The average diameter dispensed was 14.53±0.12 mm (n=70 eyes), with a range from 14.0 to 15.5 mm. The average sphere power was −2.411±3.743 D (n=70 eyes), and the average cylinder power was −3.121±1.724 D (n=70 eyes). The number of trials needed to achieve the final dispensed lens was as follows: 1 (22%), 2 (40%), 3 (30%), and 4 (7%). The peripheries ranged from 81.4% standard to 10.0% flat and 8.6% steep.
  | Table 5 Final dispensed contact lens information stratified by keratometry category |
Lens material wettability and deposition
Lens wettability was optimal with no wetting defects in 23% of lenses. In 58% of evaluated lenses, one wetting defect was observed that was less than 0.5 mm. In 2%, a grade of moderate wettability meant that there were multiple wetting defects in 0.5 mm zone. Lastly, 17% of lenses had extreme wetting defects that were greater than 0.5 mm. The material demonstrated no deposits in 45% of lenses; light deposition in 43%, medium deposition in 10%, and only 2% demonstrated heavy deposition.
Safety
Slit-lamp findings at the exit visit were absent (grade 0) in the majority of eyes. Assessments of grade 0 and grade 1–2, respectively, were determined for limbal injection (57% and 43%), bulbar injection (61% and 39%), corneal neovascularization (57% and 43%), corneal infiltrates (97% and 3%), and contact lens-induced conjunctival impression severity (84% and 16%). Likewise, slit-lamp findings at the habitual baseline visit were either absent (grade 0) in the majority of eyes or were grade 1–2, respectively, for assessments of limbal injection (95% and 5%), bulbar injection (89% and 11%), corneal neovascularization (97% and 2%), corneal infiltrates (100% grade 0), and contact lens-induced conjunctival impression severity (99% and 1%). At 12 months, there was no epithelial edema or epithelial microcysts observed in any of the eyes (100%), and the extent of contact lens-induced conjunctival impression (measured in clock hours) with grade 0 in 81% of eyes and grade 2 (2 o’clock hours) or higher for 19% compared with the habitual baseline of grade 0 in 97% eyes and 12 o’clock hours in 1 eye (3%). The severity and extent of staining for the cornea and conjunctiva are shown in Table 6.
  | Table 6 Corneal and conjunctival staining at the 12-month visit |
Over the course of the study, two adverse events were reported (two of 84 eyes). One subject had mild graft rejection in the left eye and presented with slit-lamp findings of superior edema 1+ and punctate epithelial keratopathy superiorly in the graft, which resolved with treatment. This event was deemed unrelated to the study lens, but the patient ultimately discontinued the study due to unacceptable distance VA. One subject had a superior temporal peripheral epithelial defect in the left eye, which resolved with treatment. The patient continued the study and had a logMAR VA of −0.12 at resolution; it was undetermined if the event was related to the lens.
Subjective outcomes
Lens wear
At the exit visit, the typical hourly wear time per day reported by subjects was as follows: ≤6 hours (3%, 1/36), 7–10 hours (22%, 8/36), 11–14 hours (28%, 10/36), and 15–18 hours (47%, 17/36). In comparison, the habitual hourly wear time at baseline was as follows: ≤6 hours (4%, 1/28), 7–10 hours (14%, 4/28), 11–14 hours (36%, 10/28), and 15–18 hours (46%, 13/28). The average habitual wearing time was 14.0±3.1 (n=28) hours. At the exit visit, the average wearing time was 13.4±3.7 (n=36) hours (P>0.05). At the exit visit, the typical days of wear time per week were as follows: 3–4 days a week (11%, 4/36) and 5 or more days a week (89%, 32/36). In comparison, the habitual weekly wear times were as follows: for specific occasions only (14%, 4/28), 3–4 days a week (11%, 3/28), and 5 or more days a week (75%, 21/28).
Daily experience with lenses
The questionnaire queried subjects about their comfort and vision experiences with the contact lenses upon insertion, at the beginning of the day, at work or in education settings, and at the end of the day (Table 7). When reviewing the favorable responses at the 12-month visit with the study lenses, the majority of subjects (94%, 34/36) reported a comfortable experience upon insertion for responses of “so comfortable I can’t feel them” (19%), “very comfortable” (39%), and “quite comfortable” (36%). With the habitual correction, 71% (20/28) of subjects reported a comfortable experience upon insertion with responses of “so comfortable I can’t feel them” (7%), “very comfortable” (39%), and “quite comfortable” (25%). Concerning vision upon insertion of the study lenses, most subjects at the 12-month visit (89%, 32/36) reported good vision, with responses of excellent (19%), very good (39%), or quite good (31%). In comparison, with the habitual correction, 79% (22/28) of subjects reported good vision upon insertion, with responses of excellent (18%), very good (21%), or quite good (39%).
Subjects were also queried about the lens wear experience just before removing them at the end of the day. At the 12-month visit, the majority of subjects (81%, 29/36) reported a comfortable experience just before removal of the study lenses: responses of extremely comfortable (14%), very comfortable (22%), and quite comfortable (44%). When compared with the habitual correction, only 29% (8/28) of subjects reported a comfortable experience just before removal of lenses, with no responses of extremely comfortable, very comfortable (11%), and quite comfortable (18%). Concerning vision just before removal of the study lenses, most subjects at the exit visit (81%, 29/36) reported good vision, with responses of excellent (8%), very good (31%), or quite good (42%). For habitual baseline correction, 50% of subjects (14/28) reported good vision before removal, with responses of excellent (4%), very good (14%), or quite good (32%).
Difficulty with vision
The questionnaire included questions about difficulty with vision during dim/low lighting conditions, driving at night, working on a computer or with hand held device, and socializing in the evening or at night. In general, subjects typically indicated less difficulty when performing these tasks or activities at 12 months with the study lenses compared with habitual baseline correction (Figure 3).
Lens satisfaction during activities
Subjects also responded to questions about lens satisfaction during various activities, which included cooking, reading, cinema/theater, extreme sports, DIY (do it yourself)/gardening, and shopping. The percentage of subjects who indicated they were able to wear lenses satisfactorily for the various activities are shown in Figure 4. In general, more subjects reported being able to wear lenses satisfactorily at 12 months compared with the habitual baseline, with the exception of extreme sports for which there were no respondents at baseline or 12 months, and crafts which had a similar rate of response at both time points.
Discussion
The KIC lens addresses the unique challenges of people with a variety of irregular corneal conditions, including keratoconus, as well as, corneal irregularity due to postgraft and postrefractive surgery. This 12-month prospective study evaluated the clinical performance with the KIC lens, particularly comfort and vision. Outcomes from the study demonstrated good safety, effectiveness, and comfortable lens wear experience in this study population. High-contrast VA demonstrated improvement and low-contrast VAs were comparable at the habitual baseline and 12 months visits. Slit-lamp findings were typically absent or low grade, and there were very few adverse events, suggesting the lens was well tolerated by subjects. Subjects reported greater comfort and satisfaction, and less difficulty with tasks with the study lens at 12 months than with the habitual correction at baseline. Subjective responses typically showed improvements from baseline, which is of note particularly in patients who were previously unable to use contact lens corrective wear (34.9% habitually wore spectacles).
The results from the present study were consistent with those reported in a previously reported study. In a retrospective study of 94 eyes, fit with KIC and 77 eyes fit with Rose-K® (Menicon Co., Ltd., Nagoya, Japan) RGP lenses, Fernandez-Velazquez14 found that best-corrected VA with lenses (BCLCVA) was statistically insignificant when comparing habitual lenses to KIC. In addition, in the KIC group, BCLCVA were similar between types of ectasia (P=0.19) as well as in mild and moderate keratoconus (P=0.45). Rathi et al4 also noted a similar experience with mild and moderate keratoconus. Relative to corneal staining, Fernandez-Velazquez14 noted staining was greater with the Rose-K lens, than with the KIC lens. Slit-lamp findings from the present study found minimal corneal staining (grade 2 or less) with the KIC lens. Wearing time in the present study averaged a little over 13 hours, which was slightly higher than the wearing time of 11 hours observed by Fernandez-Velazquez.14
Of note in the present study was the trend toward improved functionality in daily activities reported by subjects. This improvement was also observed in the subjective responses indicating improvements in comfort and satisfaction with lens wear. Moreover, a higher percentage of subjects at 12 months reported having no difficulty with night time activities such as driving at night (30% vs 11%) and socializing (43% vs 18%) when compared with habitual baseline contact lens correction. Compared with habitual baseline, a higher percentage of subjects at 12 months reported satisfactorily wearing the KIC lens for activities of cooking, reading, cinema/theater, DIY/gardening, and shopping. Understanding the lifestyle of the patient and assessing visual needs can aid in lens selection.
This study provides an assessment of the patient experience with the KIC lens, of which little has been published to date. Comfort remains a consideration for patients with keratoconus, particularly given the progressive nature of the disorder, the dependence on contact lenses for best vision in moderate to advanced stages, and the potential for corneal scarring. A factor potentially contributing to comfort with this lens may be the soft silicone hydrogel material, as some studies have shown improved comfort with soft contact lens options.5,15 The KIC lens appeared to be well tolerated by subjects based on the low incidence of adverse events, slit-lamp findings, and subjective satisfaction and comfort. In addition, the ability to customize the lens to fit the patient in diameter, base curve, and periphery may allow for a more comfortable fit. In a study of 130 patients with keratoconus, Lim and Vogt16 reported that the main reasons for PKP were contact lens intolerance (83%), frequent contact lens displacement (8.5%), and unsatisfactory VA despite good contact lens fit (8.5%). A customizable, soft contact lens option may provide a comfortable modality for patients and possibly delay the need for comfort-related surgical intervention.
As with many contact lens modalities for keratoconus, it is important to allow time for the patient to adjust to the lens. In the present study, for over 60% of subjects two or less trials were needed to achieve the final dispensed lens, following the standardized MoRoCCo VA technique for fitting (KeraSoft® Fitting Manual). Although some subjects required three or four trials, fewer trials were needed as familiarity with lens fitting practices increased throughout the study. In our experience, clinicians should be cautious about changing/refitting lenses too often or too quickly. Patients should be allowed time for adaptation to the lens, then to assess the need for refitting. During this study, the mean spherocylindrical refraction changed by 0.80 D suggesting either corneal unmolding or advancing of the ectasia. In our opinion, within 3 months of wear was found to be an optimal timeframe for adjustments and evaluation of fit, comfort, and VA.
Although an increase in mild staining occurred with this lens from the baseline, moderate to severe staining was not observed in either the baseline or final visit category. One possible explanation may be that approximately 35% of participants enrolled in the study were not wearing any form of contact lens correction at baseline. In addition, multipurpose solution was dispensed to the patients initially, and some of the patients were changed to ClearCare® for sensitivity reasons. Further studies may be needed to determine the optimal solution compatibility with the Efrofilcon A material.
To date, Efrofilcon A is one of the only latheable silicone hydrogel materials available for custom soft contact lenses. In this study, lenses were replaced quarterly. The material itself has some limitations of wettability and deposition. In extreme cases, 19% of lenses had moderate to severe wetting defects, and 12% had moderate to severe deposition.
There were some additional limitations to this study that should be noted, such as the lack of an active comparator. Forty-three patients were selected in this case series in which the habitual baseline provided a real-world approach for assessment of differences between habitual lens wear versus the study lens. In addition, there are limited reports about patient experience with this modality, and this study provided a comprehensive assessment using an extensive questionnaire. To our knowledge, this is the first published report of prospective outcomes describing vision, safety, and subjective outcomes with the KIC lens. Future studies could include comparisons with other types of keratoconus treatment modalities. Another area of clinical interest would be the safety of the KIC for post-crosslinking, or perhaps in the prevention of comorbid scarring.
Conclusion
After 12 months of KeraSoft® IC lens wear, clinical outcomes showed good VA, safety, and subjective outcomes in this study population. Visual outcomes were similar for the habitual baseline and 12-month visit. The safety outcomes of this study showed very few adverse events, and minimal corneal inflammation or staining responses. Subjects reported improvements in comfort and vision, and less difficulty with vision at exit visit compared with habitual baseline. Study outcomes demonstrated that the KeraSoft® IC provided a comfortable option for patients with various stages of keratoconus and other irregular corneas.
Disclosure
Lynette Johns is a paid consultant for the Custom Lab Channel Business, Bausch & Lomb Incorporated, Wilmington, MA, USA. Marjorie Rah is an employee of Bausch & Lomb Incorporated, Rochester, NY, USA. During this study, Joseph Barr was a full-time employee of Bausch & Lomb Incorporated, Rochester, NY, USA. The authors report no other conflicts of interest in this work.
References
Kennedy RH, Bourne WM, Dyer JA. A 48-year clinical and epidemiological study of keratoconus. Am J Ophthalmol. 1986;101:267–273. | ||
Rabinowitz Y. Keratoconus. Surv Ophthalmol. 1998;42:297–319. | ||
Krachmer J, Feder RS, Belin MW. Keratoconus and related noninflammatory corneal thinning disorders. Surv Ophthalmol. 1984;28(4):293–322. | ||
Rathi V, Mandathara PS, Dumpati S. Contact lens in keratoconus. Indian J Ophthalmol. 2013;61:410–415. | ||
Jinabhai A, Radhakrishnan H, Tromans C, O’Donnell C. Visual performance and optical quality with soft lenses in keratoconus patients. Ophthalmic Physiol Opt. 2012;32:100–116. | ||
Vreugdenhil W, Rijneveld WJ, Eggink FA. Fitting therapeutic softperm contact lenses in the event of corneal pathology with highly irregular astigmatism. J Br Contact Lens Assoc. 1994;17(2):59–61. | ||
Cummings A, McQuaid R, Mrochen M. Newer protocols and future in collagen cross-linking. Indian J Ophthalmol. 2013;61:425–427. | ||
Vinciguerra P, Albe E, Trazza S, et al. Refractive, topographic, tomographic, and aberrometric analysis of keratoconic eyes undergoing corneal cross-linking. Ophthalmology. 2009;116:369–378. | ||
Cassidy D, Beltz J, Jhanji V, Loughnan MS. Recent advances in corneal transplantation for keratoconus. Clin Exp Optom. 2013;96:165–172. | ||
Barr J, Zadnik K, Wilson BS, et al. Factors associated with corneal scarring in the Collaborative Longitudinal Evaluation of Keratoconus (CLEK) study. Cornea. 2000;19:501–507. | ||
Korb DR, Finnemore VM, Herman JP. Apical changes and scarring in keratoconus as related to contact lensfitting techniques. J Am Optom Assoc. 1982;53(3):199–205. | ||
Smiddy WE, Hamburg TR, Kracher GP, Stark WJ. Keratoconus. Contact lens or keratoplasty? Ophthalmology. 1988;95(4):487–492. | ||
Alió JL, Belda JL, Artola A, Garciá-Lledó M, Osman O. Contact lens fitting to correct irregular astigmatism after corneal refractive surgery. J Cataract Refract Surg. 2002;28(10):1750–1757. | ||
Fernandez-Velazquez F. KeraSoft® IC compared to Rose-K in the management of corneal ectasias. Cont Lens Anterior Eye. 2012;35(4):175–179. | ||
Johnson T, Schnider CM. Clinical performance and patient preferences for hydrogel versus RGP lenses: a crossover study. Int Contact Lens Clin. 1991;18:130–135. | ||
Lim N, Vogt U. Characteristics and functional outcomes of 130 patients with keratoconus attending a specialist contact lens clinic. Eye. 2002;16(1):54–59. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.





