Back to Journals » Medical Devices: Evidence and Research » Volume 8
Characterizing the continuously acquired cardiovascular time series during hemodialysis, using median hybrid filter preprocessing noise reduction
Authors Wilson S, Bowyer A, Harrap S
Received 25 October 2014
Accepted for publication 24 November 2014
Published 8 January 2015 Volume 2015:8 Pages 65—70
DOI https://doi.org/10.2147/MDER.S76409
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Scott Wilson,1,2 Andrea Bowyer,3 Stephen B Harrap4
1Department of Renal Medicine, The Alfred Hospital, 2Baker IDI, Melbourne, 3Department of Anaesthesia, Royal Melbourne Hospital, 4University of Melbourne, Parkville, VIC, Australia
Abstract: The clinical characterization of cardiovascular dynamics during hemodialysis (HD) has important pathophysiological implications in terms of diagnostic, cardiovascular risk assessment, and treatment efficacy perspectives. Currently the diagnosis of significant intradialytic systolic blood pressure (SBP) changes among HD patients is imprecise and opportunistic, reliant upon the presence of hypotensive symptoms in conjunction with coincident but isolated noninvasive brachial cuff blood pressure (NIBP) readings. Considering hemodynamic variables as a time series makes a continuous recording approach more desirable than intermittent measures; however, in the clinical environment, the data signal is susceptible to corruption due to both impulsive and Gaussian-type noise. Signal preprocessing is an attractive solution to this problem. Prospectively collected continuous noninvasive SBP data over the short-break intradialytic period in ten patients was preprocessed using a novel median hybrid filter (MHF) algorithm and compared with 50 time-coincident pairs of intradialytic NIBP measures from routine HD practice. The median hybrid preprocessing technique for continuously acquired cardiovascular data yielded a dynamic regression without significant noise and artifact, suitable for high-level profiling of time-dependent SBP behavior. Signal accuracy is highly comparable with standard NIBP measurement, with the added clinical benefit of dynamic real-time hemodynamic information.
Keywords: continuous monitoring, blood pressure
Introduction
Despite strong focus on the identification and avoidance of intradialytic hypotension, the “usual” or “expected” profile of systolic blood pressure (SBP) during the delivery of routine maintenance hemodialysis (HD) remains unclear. In the absence of standardized guidelines, usual HD unit protocols involve noninvasive brachial cuff blood pressure (NIBP) measurements taken pre- and postdialysis, and every 30–60 minutes during treatment.1,2 The speed of intradialytic hemodynamic change may see these episodic measurements fail to sufficiently capture variability in the underlying parameters when they occur below a patient’s symptomatic threshold, making more continuous hemodynamic observation desirable.
For illustrative purposes, assuming a stable background heart rate of 70 beats per minute, a continuously monitored 4-hour treatment will generate in excess of 16,000 discrete SBP observations per individual dialysis. At its simplest, a continuously acquired SBP record is a unidimensional time-variant digital signal. Variability through the sequence of observations represents the present net sum of all physiologic influences acting cumulatively via both external stressor (treatment-related) and internal homeostatic feedback mechanisms.
Given requirements for arterial cannulation, it is inappropriate for this continuous time series to be repeatedly measured invasively in HD patients; however, both finger plethysmograph and pulse transit time-based technique can potentially provide continuous safe, noninvasive monitoring of real-time hemodynamics.3–5
In the clinical environment, the noninvasively acquired signal is subject to degradation by outliers and measurement artifact in addition to physiological and Gaussian-type noise. This should be appropriately minimized prior to interpretation of the underlying sequence. This common scenario (Figure 1) presents a challenge to extract a clinically relevant signal with sufficient detail to represent time-varying trends whilst maintaining adequate accuracy and depth of observations to prevent overfitting of the data.
The ideal solution to this problem would utilize signal preprocessing. The resultant dynamic regression series should retain the edge detection ability to accurately describe SBP peaks and troughs after removal of signal confounders. Traditionally used methods of noise reduction in hemodynamic time series involve manual editing and proximity-based techniques, using standard deviation or interquartile range difference thresholds.6,7 Outliers may be defined by the quantitative difference from the adjacent sequence, the “nearest neighbor”, or other parametric characteristic drawn from the original data set.8 Whilst these approaches are partially successful, they may be laborious and time-intensive for longer recordings, with a residual signal yield that remains obscured by underlying Gaussian-type noise. Median hybrid filters (MHFs) represent a class of nonlinear noise-exclusion algorithms typically used with two- and three-dimensional data sets; however, these have potential application in approximating the underlying physiological signal from a unidimensional hemodynamic time series. A MHF follows the central value of several concurrently running linear subfilters of various window lengths and can be represented by the function:
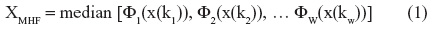
where: XMHF is the digitally filtered signal; φ is the mean value; x is the subject variable (blood pressure); k is the window length (number of observations); and w is the number of subfilter windows.
The optimal number of concordant subfilters is between three and seven.9 By taking advantage of the hierarchical reordering of underlying subfilters, MHFs can demonstrate signal edges10 whilst retaining representative signal accuracy by tracking the central tendency.11 In the context of continuously acquired SBP data, these attributes could facilitate time-based comparisons of SBP level changes, trajectory, and speed to assist clinical decision making.
Methods
This study complied with the National Health and Medical Research Council National Statement 2007 and the Australian Code for the Responsible Conduct of Research. A continuous beat-to-beat SBP data set from the complete intradialytic period was acquired using the Finometer® MIDI (Finapres Medical Systems BV, the Netherlands) applied to the third finger of the nonvascular access arm. The SBP time series was exported in *.csv format for off-line processing by the MHF.
The MHF substructure was designed to capture variations in short-term SBP trajectory over periods not exceeding 10 minutes duration. Seven simple moving average linear subfilters of progressive observation window size were constructed and defined in terms of total heartbeats. Window lengths were derived from the Fibonacci series, ensuring each individual moving average was of sufficiently independent length to remain nonfactorizable by hierarchically adjacent subfilters, eliminating the potential for oscillatory vibration bias where repeating shorter-term SBP cycles were mistaken for longer-term movements. Time-equivalence approximations for the MA subfilter lengths at normalized heart rates are shown in Table 1.
  | Table 1 Approximate time equivalence of moving average window lengths according to underlying heart rate |
The algorithm was applied to ensure the resulting postfilter series was equal in size (heartbeat observations) to the original raw signal file, to maintain integrity of the time series length.
MHF outputs were cross-validated against a simultaneously collected independent data set of brachial cuff NIBP measurements, using Bland–Altman analysis.12 NIBP readings were collected by trained dialysis nursing staff at pre- and postdialysis time points and approximately hourly through the intradialytic period, using a standardized Omron HM907 semioscillometric automated device (Omron Electronics, Kyoto, Japan). Validation of post-MHF signal integrity was performed using white noise regression periodogram analysis by Fisher’s kappa and Bartlett’s Kolmogorov–Smirnov methods.
Results
Figure 2 shows the example product of the MHF function across the intradialytic SBP traces recorded in two sample patients, superimposed on a graphical plot of the raw time series data file. The filter output demonstrates the exclusion of both extreme outliers and Gaussian-type noise to reveal a continuous linear representation of the underlying tracking trend in SBP.
  | Figure 2 Post-MHF product overlain against raw continuous intradialytic SBP data in two patients. |
Nonsustained runs of trend linearity occur; however, they appear inconsistent in terms of length, direction, and turning point. Changes in SBP trajectory and level appear rapidly but are neither captured nor conveyed sufficiently by the isolated NIBP measures. Figure 3 illustrates the point plot of standard intradialytic NIBP measurements, reflecting the current standard of blood pressure profiling in routine clinical practice. Figure 4 overlays the post-MHF continuous regression model using continuously acquired data from the same patient, to demonstrate the nonstationary, time-varying sequence with a heightened level of detail and wider (asymptomatic) SBP range.
Bland–Altman agreement (Figure 5) between the time-coincident MHF output and simultaneously recorded NIBP pairs demonstrates a mean difference of (0.95 mmHg) ±1.96 SD (5.88 mmHg), fulfilling international criteria for the validation of blood pressure measuring devices in adult humans.13
This agreement analysis demonstrates the clinical equivalence of the post-MHF continuously monitored SBP in accurately reflecting the concurrent NIBP measures, which currently form the basis of standard dialysis practice. Test statistics for Gaussian noise exclusion testing are shown in Table 2 and permit rejection of the null hypothesis that the post-MHF time series is consistent with Gaussian noise.
  | Table 2 Statistical white noise analysis of the MHF output product after application to the continuous intradialytic SBP trace |
Discussion
The clinical outcome associations between pre- and postdialysis, and interdialytic ambulatory blood pressure measurements have been extensively studied, with often paradoxical and inconsistent results between both individual reports and comparisons to the general population.14–18 The intradialytic period has been less comprehensively modeled, particularly from the perspective of a “normal” or “expected” SBP profile during routine HD treatment, in lieu of significant efforts focused on the timely identification and avoidance of acutely symptomatic hypotensive events. Such strategies are dependent upon the frequency of serial SBP measurement that, if intermittently gathered, necessitates linear interpolation between observations and resultant overfitting of the time series. Continuously acquired SBP data, processed online and in real-time, would abrogate the need for interpolative modeling by yielding a clinically relevant complex dynamic regression for potential integration into decision-making systems.
Given the volume of data acquired, the extraction of a robust and analyzable signal from a continuous noninvasive beat-to-beat data set requires an automated algorithmic process. To make this signal clinically meaningful, both erratic impulse artifact and the frequency band of Gaussian-type noise should be excluded. The proposed MHF algorithm creates a novel framework to potentially characterize and quantify the profile and prognostic significance of the spectrum of intradialytic SBP behavior.
First conceived as a novel class of noise removal for digital image processing applications, the MHF combines the most favorable properties of linear and nonlinear techniques without the requirement to modify data points by either substitution or interpolation.19 Predominantly used in multidimensional data sets, the method has been demonstrated to yield better resolution of image details with reduced computational burden when compared with conventional median or nearest neighbor averaging filters.11 Having found diverse applications in aeronautics, visual image processing, financial analysis, and telecommunications,9,20–22 there has been limited application of the technique to biological signal processing in medicine or physiological time series research.
A single report describes an MHF algorithm studied in the clinical setting to remove blink noise from electrooculogram traces where equivalency, but not superiority, was demonstrated between the proposed filter and both simpler median and more complex weighted variants.23 It was noted that each type of algorithm the authors trialed performed to a high standard of clinical relevance. Significant differences exist in both the quality of the raw data and level of signal decontamination required between the typical electrooculogram and clinical beat-to-beat SBP data, however. Blink noise is a transient interruption characterized by a sharp rise and fall, as opposed to the mixed Gaussian-type noise and impulsive outlier phenomena seen in cardiovascular data sets. Importantly, the neurological study saw negligible levels of baseline Gaussian-type noise as the “raw” signal underwent analog preprocessing prior to the application of trial algorithms.
Overlaying the postfilter continuous trace with the brachial cuff NIBP record reveals a deeper level of complexity and detail by filling in the time spaces between usual observations. This “filling of the blanks” with analyzable data highlights trends and extremes of SBP variation, which if not opportunistically sampled, are simply not identified by conventional means. In previous studies of intradialytic SBP patterns, this degree of volatility has not been recognized and warrants more rigorous examination, particularly in light of the excess cardiovascular burden in the HD population24 and the increasingly recognized associations between SBP variability and outcome.25,26
Given the impracticality of direct intra-arterial measurement, the clinical importance of a representative continuous SBP trace should be viewed in comparison with current standards of pressure measurement in routine practice. International protocols for the validation of blood pressure measuring devices in adults wholly accept an intradevice discrepancy of up to 5 mmHg and partially, a discrepancy of up to 10 mmHg for machines providing intermittent measures.13 In the context of the Bland–Altman characteristics and a focus on intradialytic SBP trend-tracking, the clinical relevance of any absolute variation between the MHF product and current standards of automated brachial NIBP measurement is negligible. Furthermore, the real-world consequence of any potential time point pressure discrepancy is likely to be mitigated by the sheer number of observations afforded by the continuous data stream.
Conclusion
The proposed novel algorithm represents the first application of MHF preprocessing to continuously acquired cardiovascular time series data. Through exclusion of outliers and Gaussian-type noise, a detailed dynamic profile of intradialytic SBP trends can be captured for analysis and comparison between any nominal time points. In the clinical HD setting, this method reveals an increased level of detail over the intermitted NIBP cuff readings of standard practice, without the need to interpolate data points, allowing for the more complete description of both underlying SBP movement and assessment of the magnitude and direction of volatility over time.
Disclosure
Professor Harrap has been in receipt of research funding from Servier. The authors report no further conflicts of interest.
References
Kooman J, Basci A, Pizzarelli F, et al. EBPG guideline on haemodynamic instability. Nephrol Dial Transplant. 2007;22 Suppl 2:ii22–ii44. | |
K/DOQI Workgroup. K/DOQI clinical practice guidelines for cardiovascular disease in dialysis patients. Am J Kidney Dis. 200545(4 Suppl 3):S16–S153. | |
Gesche H, Grosskurth D, Küchler G, Patzak A. Continuous blood pressure measurement by using the pulse transit time: comparison to a cuff-based method. Eur J Appl Physiol. 2012;112(1):309–315. | |
Guelen I, Westerhof BE, Van Der Sar GL, et al. Finometer, finger pressure measurements with the possibility to reconstruct brachial pressure. Blood Press Monit. 2003;8(1):27–30. | |
Schutte AE, Huisman HW, Van Rooyen JM, Oosthuizen W, Jerling JC. Sensitivity of the Finometer device in detecting acute and medium-term changes in cardiovascular function. Blood Press Monit. 2003;8(5):195–201. | |
Task Force of the European Society of Cardiology and the North American Society of Pacing Electrophysiology. Heart rate variability: Standards of measurement, physiological interpretation, and clinical use. Circulation. 1996;93(5):1043–1065. | |
Wax DB, Beilin Y, Hossain S, Lin HM, Reich DL. Manual editing of automatically recorded data in an anesthesia information management system. Anesthesiology. 2008;109(5):811–815. | |
Porta A, Bari V, Bassani T, et al. Entropy-based complexity of the cardiovascular control in Parkinson disease: comparison between binning and k-nearest-neighbor approaches. Conf Proc IEEE Eng Med Biol Soc. 2013;2013:5045–5048. | |
Ganguli R. Noise and outlier removal from jet engine health signals using weighted FIR median hybrid filters. Mech Syst Signal Process. 2002;16(6):967–978. | |
Astola J, Heinonen P, Neuvo Y. Linear median hybrid filters. IEEE Trans Circuits Syst. 1989;36(11):1430–1438. | |
Fried R, Bernholt T, Gather U. Repeated median and hybrid filters. Comput Stat Data Anal. 2006;50(9):2313–2338. | |
Bland JM, Altman DG. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet. 1986; 327(8476):307–310. | |
O’Brien E, Atkins N, Stergiou G, et al; Working Group on Blood Pressure Monitoring of the European Society of Hypertension. European Society of Hypertension International Protocol revision 2010 for the validation of blood pressure measuring devices in adults. Blood Press Monit. 2010;15(1):23–38. | |
Heerspink HJ, Ninomiya T, Zoungas S, et al. Effect of lowering blood pressure on cardiovascular events and mortality in patients on dialysis: a systematic review and meta-analysis of randomised controlled trials. Lancet. 2009;373(9668):1009–1015. | |
Agarwal R, Peixoto AJ, Santos SF, Zoccali C. Pre- and postdialysis blood pressures are imprecise estimates of interdialytic ambulatory blood pressure. Clin J Am Soc Nephrol. 2006;1(3):389–398. | |
Moriya H, Oka M, Maesato K, et al. Weekly averaged blood pressure is more important than a single-point blood pressure measurement in the risk stratification of dialysis patients. Clin J Am Soc Nephrol. 2008;3(2):416–422. | |
Stidley CA, Hunt WC, Tentori F, et al; Medical Directors of Dialysis Clinic Inc. Changing relationship of blood pressure with mortality over time among hemodialysis patients. J Am Soc Nephrol. 2006;17(2):513–520. | |
Rohrscheib MR, Myers OB, Servilla KS, et al; DCI Medical Directors. Age-related blood pressure patterns and blood pressure variability among hemodialysis patients. Clin J Am Soc Nephrol. 2008;3(5):1407–1414. | |
Nieminen A, Heinonen P, Neuvo Y. A new class of detail-preserving filters for image processing. IEEE Trans Pattern Anal Mach Intell. 1987;9(1):74–90. | |
Pitas I, Venetsanopoulos AN. Nonlinear Digital Filters. Principles and Applications. New York, NY: Springer; 1990. | |
Ko S-J, Lee YH. Center weighted median filters and their applications to image enhancement. IEEE Trans Circuits Syst. 1991;38(9):984–993. | |
Arce GR, Foster RE. Detail-preserving ranked-order based filters for image processing. IEEE Trans Acoust. 1989;37(1):83–98. | |
Martinez M, Soria E, Magdalena R, Serrano López AJ, Vila J. Application of FIR median hybrid filters to remove blinks in electrooculograms. Presented at: 7th WSEAS International Conference on Applied Computer Science; November 21–23, 2007; Venice, Italy. | |
Sarnak MJ, Levey AS. Cardiovascular disease and chronic renal disease: a new paradigm. Am J Kidney Dis. 2000;35(4 Suppl 1):S117–S131. | |
Rothwell PM, Howard SC, Dolan E, et al. Prognostic significance of visit-to-visit variability, maximum systolic blood pressure, and episodic hypertension. Lancet. 2010;375(9718):895–905. | |
Muntner P, Shimbo D, Tonelli M, Reynolds K, Arnett DK, Oparil S. The relationship between visit-to-visit variability in systolic blood pressure and all-cause mortality in the general population: findings from NHANES III, 1988 to 1994. Hypertension. 2011;57(2):160–166. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.




