Back to Journals » Clinical Ophthalmology » Volume 13
Central Serous Chorioretinopathy: Pathogenesis and Management
Authors Semeraro F , Morescalchi F , Russo A , Gambicorti E, Pilotto A , Parmeggiani F , Bartollino S , Costagliola C
Received 26 June 2019
Accepted for publication 21 October 2019
Published 2 December 2019 Volume 2019:13 Pages 2341—2352
DOI https://doi.org/10.2147/OPTH.S220845
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Francesco Semeraro,1 Francesco Morescalchi,1 Andrea Russo,1 Elena Gambicorti,1 Andrea Pilotto,2 Francesco Parmeggiani,3 Silvia Bartollino,4 Ciro Costagliola4
1Department of Medical and Surgical Specialties, Radiological Sciences and Public Health, University of Brescia, Brescia, Italy; 2Department of Clinical and Experimental Sciences, Neurology Unit, University of Brescia, Brescia, Italy; 3Department of Morphology, Surgery and Experimental Medicine, University of Ferrara, Ferrara, Italy; 4Department of Medicine and Health Science “V. Tiberio”, University of Molise, Campobasso, Italy
Correspondence: Silvia Bartollino
Department of Medicine and Health Science “V. Tiberio”, University of Molise, Via de Santis, Campobasso 86100, Italy
Email [email protected]
Abstract: Central serous chorioretinopathy (CSC) is a common retina disease and has a relative high recurrence rate, etiology, and pathogenesis of which remains largely ambiguous. The effects on the retina are usually self-limited, although some people are left with permanent vision loss due to progressive and irreversible photoreceptor damage or retinal pigment epithelium atrophy. There have been a number of interventions used in CSC, including, but not limited to, laser treatment, photodynamic therapy (PDT), intravitreal injection of anti-vascular endothelial growth factor agents, and subthreshold lasers. It is not clear whether there is a clinically important benefit to treating acute CSC, which often resolves spontaneously as part of its natural history. Of the interventions studied to date, PDT and micropulse laser treatment appear the most promising.
Keywords: central serous chorioretinopathy, acute central serous chorioretinopathy, chronic central serous chorioretinopathy
Introduction
Central serous chorioretinopathy (CSC) was first identified and described in 1866 by von Graefe, who referred to the disease as “recurrent central retinitis.”
It is a common ocular disease characterized by decompensation of the retinal pigment epithelium (RPE), which results in neurosensory retinal detachment, serous pigment epithelium detachment (PED), and RPE atrophy. It is usually unilateral and predominantly affects young or middle-aged (25 to 50 years) adults, with men being affected more frequently than women.1
In its typical presentation, CSC appears as a localized serous detachment of the neurosensory retina involving the region of the macula without subretinal blood or lipid exudates. The margins of the serous detachment are sloping and merge gradually into the normally attached retina of the posterior pole. Occasionally, it is associated with a single or multiple serous PED.
Patients affected by CSC often complain of blurred vision (usually only in one eye), typically perceived as a dark spot or scotoma in the central visual field, with associated image distortion (metamorphopsia). Other common symptoms include micropsia (reduction of the apparent size of objects), mild dischromatopsia (abnormal color perception), and reduced contrast sensitivity. However, CSC may also be asymptomatic.2
The visual acuity of people affected by CSC varies widely; Amsler grid testing reveals distortion of the straight lines, which may appear blurred due to a central scotoma. A small relative defect of the afferent pupillary reflex is also occasionally present.
Disease Course and Prognosis
The natural course of CSC is often self-limiting, and spontaneous resolution and complete fluid reabsorption often occurs. The visual prognosis is good in 90–95% of cases and visual acuity returns to normal within a few months once the fluid has resolved. The visual distortion often reduces as the eye heals, but in some patients some visual abnormalities may persist even after the fluid has dissipated.3 Lasting visual symptoms can include localized distortion, reduced color discrimination and decreased night vision (likely caused by a disruption of the retinal microarchitecture), subretinal fibrosis, and scarring or atrophy of the RPE.4–6 Poor visual recovery may be associated with advanced age, multiple recurrences, persistent neurosensory retinal detachment, retinal PED, or severe forms of the disease that cause bullous retinal detachment. This variant of CSC is an acute form that may develop spontaneously or following corticosteroid therapy, organ transplantation, hemodialysis, or pregnancy. Bullous retinal detachment is characterized by a serous or serofibrinous subretinal exudation, and is thought to be due to an exaggerated breakdown of the permeability of the choriocapillaris, accompanied by single or multiple areas of RPE decompensation, which allows fluid to gain access to the subretinal space.7,8 The bullous retinal detachment usually resolves slowly after many weeks or months of blurred vision.
In most cases, CSC is acute (aCSC), with only a single episode of illness occurring in the patient’s lifetime; however, it can be recurrent, and it can become chronic (cCSC). A recurrence of aCSC may occur within 12 months in 30–50% of patients; it may then resolve again spontaneously or continue to cause some vision disturbances. The chronic form, also known as Type II CSC, occurs in approximately 5% of cases.
The definition of cCSC may be somewhat ambiguous because it relies on a temporal criterion (the duration of the serous retinal detachment), and on the presence of extended RPE changes. The duration threshold above which CSC is considered to be chronic varies among different studies, from 3 to 6 months.9,10 Diffuse, rather than focalized, abnormality of the RPE is exhibited in cCSC, which produces persistent subretinal fluid. The serous detachment in these cases tends to be shallower, rather than dome shaped, in comparison with aCSC. Persistence of serous retinal detachment in cCSC is associated with progressive RPE decompensation and deterioration of visual acuity.11,12
Diagnosis
Diagnosis of CSC usually begins with a standard examination of the retina, which shows central shallow serous retinal detachment; this is confirmed by optical coherence tomography (OCT), fluorescein and/or indocyanine green (ICG) angiography, and optical coherence tomography angiography (OCTA). The visual acuity of the affected eye is usually slightly or moderately reduced due to the decreased focal length caused by the raised retina; this phenomenon makes the eye more hyperopic than before.
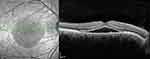
Use of an Amsler grid can be helpful to confirm suspected macular edema in less overt cases and may help localize the involved area of the visual field.13–15 The morphologic changes that occur in eyes with aCSC are formation of a limited bubble of central serous retinal detachment (Figure 1), RPE detachment, and RPE abnormalities.16,17 The outer retinal layers, which include the photoreceptor layer, intermediate line, and inner segment/outer segment (IS-OS) junction, are well-visualized and preserved; occasionally, thickened and hyperreflective tips of detached photoreceptors may be noted. Less frequent findings include focal protrusions of the RPE layer, focal defects in the RPE at the leaking site, minutes fibrinous exudate in the subretinal space, and subretinal precipitates.15,18,19 Lastly, an increased choroidal thickness associated with an increased choroidal vascular component is well detectable with enhanced depth imaging (EDI) OCT, predominantly in aCSC forms.20–23 The anatomical resolution of subretinal fluid is concomitant with the normalization of the choroidal thickness.24
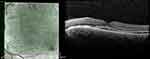
The chronic form of CSC exhibits diffuse, rather than focalized, abnormality of the RPE, producing persistent subretinal fluid. The serous detachment in these cases tends to be shallower rather than dome shaped in comparison with aCSC. Changes in the RPE include low to flat PED, the presence of micro-rips, hypertrophy and atrophy, thickening of the deeper and thinning of the inner coroidal layers, and thinned posterior surfaces of the detached retina with hyperreflective detached photoreceptor tips. In persistent cases, OCT reveals thinning of the outer retinal layers corresponding to the photoreceptor layers, with poor visualization of the IS-OS junction (Figure 2).14
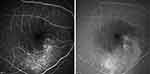
Fluorescein and ICG angiography allow observation of the fluid leakage from the choroidal vessels, the fluid invasion into the subretinal space, and the choroidal blood vessels. Typically, fluorescein angiography reveals one or more fluorescent “pinpoints” indicating fluid leakage. An expanding point of fluorescein leakage with late pooling into a serous detachment (the classic “smokestack pattern”) is visible in 10–15% of cases (Figure 3). If neuroretinal detachment persists, it tends to move toward the lower part of the macula leading to depigmentation and secondary decompensation of the inferior RPE, resulting in the clinical appearance of gravitational atrophy of the RPE.25
Indocyanine green angiography reveals focal delays and hyperpermeability of the choroidal circulation, revealing a delayed initial filling of choriocapillaris arteries in the early phase, and dilation of the choriocapillaris veins leaking fluid in the subsequent phases. Use of ICG angiography demonstrates that CSC is likely caused by a primary dysfunction of the choriocapillaris; the fluid build-up seems to occur due to small breaks in the RPE, secondary to choriocapillaris disease.26
OCTA reveals areas of focal hypo- and hyper-perfusion in the choriocapillaris. Though, due to the inability to detect plasma flow, OCTA is not suitable to detect leakage points in CSC with confidence. However, OCTA reliably detects choroidal neovascularization (CNV) in CSC even in the absence of exudative activity and may, therefore, represent an important supplement in the diagnosis of CSC.24,27–29 Particularly, OCTA imaging revealed that eyes with CNV complicating CSC exhibited significantly lower choroidal vascular component compared to those with CSC without CNV.30,31
Etiology and Pathophysiology
Stress Burden and Personality Traits
As previously discussed, CSC most often affects men who are actively working, often as freelancers, managers, entrepreneurs, or workers burdened by job responsibilities. Psychological stress is considered one of the most important risk factors for developing CSC. In 1986, Yannuzzi32 observed a strong association between CSC and the “Type-A” personality pattern, or rather, the personality traits of having a competitive drive, a sense of urgency, an aggressive nature, and a hostile temperament. In a later report, impulsiveness, emotional instability, overachievement, and competitiveness were discussed as personality features observed in CSC patients.33 More recently, the possible association between CSC and the “narcissistic personality” (characterized by an extreme gratification of self) was suggested.34 During the past two decades, several independent studies have shown that CSC occurs more frequently in men with higher emotional distress35 a lower frustration tolerance threshold33,36 and poor coping strategies for stressful events.37,38 The presence of recent psychological stress has been largely demonstrated as one of most important CSC triggers.33,39–41 Resolution of the stressor may be followed by symptomatic improvement in some cases.42 However, it is still a topic of debate whether stress minimization (e.g., by psychological intervention or using beta-blocker therapy) could prevent recurrences or progression to chronic CSC. Interestingly, Type-A behavior and stress are strongly linked to psychopharmacological medication use43,44 and sleeping disturbances,45 which have also been suggested as possible independent risk factors for CSC.41 Although the exact mechanisms are still poorly understood, the link between CSC, personality traits, and stress is proposed to be mediated through higher levels of circulating stress hormones, particularly corticosteroids and catecholamine.
Glucocorticoid Use
Many reports have suggested that high blood levels of glucocorticoids (also called glucocorticosteroids, corticosteroids, or steroids), mineralocorticoids, and occasionally testosterone are involved in the pathogenesis of CSC. There is extensive evidence of an important effect of the cortisol hormone on the capillary permeability of the choroid, which may contribute to the underlying pathogenesis of CSC.
Clinical studies on patients with Cushing syndrome, which is characterized by abnormally high levels of cortisol, found concurrent CSC in 5–10% of cases. The increased risk of CSC during pregnancy is also likely driven by increased levels of endogenous corticosteroids, as ocular dysfunction usually appears during the third semester, but generally resolves spontaneously after delivery. Furthermore, abnormal cortisol elevation may explain the association between CSC and erectile dysfunction, which has been recently suggested by the authors of a large case-control study.46
Endogenous cortisol levels are usually higher in patients with CSC47 and the rate of urinary cortisol excretion is increased.48 Exogenous systemic corticosteroids (e.g., cortisone) are commonly used to treat inflammation and allergies, but nasal corticosteroid sprays, eye drops, and topical creams can also trigger CSC, aggravate it, and cause relapses. Above all, inhalant/nasal, oral, and injectable steroids seemed to be significant risk factors.41 Steroid-induced CSC has less male predilection than idiopathic CSC, and frequently has a bilateral presentation. Among solid organ transplant patients receiving long-term corticosteroids, those with renal transplants appear to be at a higher risk, possibly due to underlying renal disease, hypertension, microangiopathy, and previous exposure to hemodialysis, all of which may modify choroidal hemodynamics.
The strong association between steroid use and CSC may be identified by the RPE and choriocapillary receptor reactivity during systemic corticosteroid treatment. Daruich et al49 proposed that excess activation of the mineralocorticoid receptor (MR) in the choroidal endothelial cells, either by aldosterone or glucocorticoids with a high affinity for the MRs, induces upregulation of the vasodilator potassium channel KCa2.3 that modulates smooth muscle relaxation in the choroidal vessels. Systemic treatment with corticosteroids may damage the posterior blood-retinal barrier, altering the permeability of the choriocapillaris and RPE, resulting in focal areas of increased permeability that lead to subretinal fluid accumulation.38 In contrast, mineralocorticoid antagonists reverse the upregulation of KCa2.3 channels in the choriod; this finding supports the use of MR antagonists in CSC treatment.50
Catecholamines
Compared to healthy subjects, CSC patients present with a significant sympathetic overactivation and decreased parasympathetic activity, as observed by blood pressure and heart rate variability.51 This may explain the association between CSC and hypertension, as confirmed by a recent meta-analysis.41 The use of sympathomimetic agents, such as pseudoephedrine, oxymetazoline, ephedra (found in bodybuilding/dietary products), and MMDA (3-methoxy-4,5-methylenedioxyamphetamine, an illicit amphetamine) has been associated with CSC.52,53
The development of CSC has also been noted after the use of phosphodiesterase-5 inhibitors (such as sildenafil), but there is conflicting evidence regarding CSC resolution after treatment discontinuation.54 The autonomic nervous system modulation of the choroidal blood flow may play an important role in CSC pathogenesis, and it is consistent with the damage induced by epinephrine on RPE cells in vitro.
Sleep Disturbances
Shift work and sleep disturbances have recently been suggested as important independent risk factors for CSC.40,41 Cortisol and catecholamine levels are controlled by circadian regulation and can be deregulated in shift workers, thereby contributing to CSC pathophysiology. Other reasons for sleep disturbance were previously identified in CSC patients, including obstructive sleep apnea (OSA). Early reports suggested that early diagnosis and management of OSA could eventually reduce the risk of CSC and preserve ocular function. Although several studies have shown a high prevalence of OSA in CSC patients, there is still insufficient evidence to support OSA screening in patients with CSC.52,55–57
Despite the strong association between sleep disturbance and CSC,41 there have been no studies evaluating melatonin levels and rhythms in CSC patients. Melatonin production is suppressed by light through melanopsin ganglion cell activation during the day, and is typically reduced in shift workers.58 Restoring sleep by reducing stress or by using oral melatonin may, therefore, have potential beneficial effects in patients with CSC.59
Other Factors
Infection with Helicobacter (H.) pylori may be a predisposing factor for CSC. It appears that the presence of these bacteria is correlated with the visual prognosis and severity of the clinical manifestations of the disease. Though the mechanism is still poorly understood, infection with H. pylori allegedly creates an increased susceptibility of the retinal tissue to oxidative stress generated by the inflammation.
Patients suffering from kidney diseases, such as membranoproliferative glomerulonephritis type II, may develop retinal abnormalities similar to CSC; this is likely caused by choroidal deposits of the same material that originally damaged the glomerular basement membrane in the kidneys.
Genetic risk factors may also play a role in CSC, possibly via the complement system; however, only very rare familial cases have been reported thus far.60 Chronic CSC is associated with genetic variants in ARMS2 and CFH, indicating a genetic overlap between CSC and AMD, otherwise alleles in ARMS2 and CFH that determines risk of AMD, may be protective for cCSC.
Chorioretinal Changes
Advances in imaging techniques, particularly ICG angiography, OCT and OCTA have led to a better understanding of the pathophysiology of CSC.61 Clinical evidence from multimodal imaging, such as choroidal congestion and hyperpermeability with localized areas of choroidal nonperfusion, suggests that choroidal dysfunction is an important underlying cause of RPE dysfunction and subretinal fluid leakage in CSC.62 The vascular hyperpermeability and congestion belong to the pachychoroid vascular changes that are believed to underlie the development of focal alterations in the RPE, leading to the formation of PED.30,63
Indocyanine green angiography has demonstrated lobular choroidal ischemia and venous congestion with consequent leakage of fluid from the choriocapillaris. The relative ischemia and increased hydrostatic pressure in the choroidal network create secondary damage in the RPE that leads to the breakdown of the external blood-retinal barrier, causing fluid accumulation in the subretinal space.64 The subretinal fluid originates from the choriocapillaris, not the vitreous, thus the photoreceptors receive sufficient nourishment that allows them to survive even during prolonged detachment. For this reason, the patient’s vision is usually totally recovered, even though the disease can last more than a month. However, if the retina is detached for a long period, or if there is a bullous retinal detachment with wide separation of the photoreceptors from the source of oxygen, which is the choriocapillaris, ischemic damage of the photoreceptors may occur. This damage subsequently leads to damage of the RPE via gravitational epitheliopathy. In cCSC, large areas of the RPE decompensate, and central vision deteriorates. The detached retina may also induce release of vascular endothelial growth factor (VEGF) into the subretinal space, thereby delaying spontaneous resolution and, in some cases, lead to a subretinal choroidal neovascularization.4,65
Therapy
The prognosis for resolution and visual recovery in patients with aCSC is usually good, thus observation and patient reassurance is considered the best treatment course. Positive converging lenses can be used to improve visual acuity until CSC resolves. Most affected patients recover spontaneously with improvement of visual acuity, reattachment of the sensory retina, and improvement of the other symptoms within 3–4 months.3 Conversely, approximately 20–30% of patients will develop one or more recurrences of aCSC, and 5% will develop cCSC. A significant number of these patients develop a limited area of RPE atrophy and depigmentation after resolution of the affected retinal area.
For patients with CSC, it is important to suspend, or replace with an alternative, any current medications containing steroids, including creams, sprays, and eye drops. In patients treated with corticosteroids, only suspension of this therapy will be effective in reducing the bullous retinal detachment.
Faster regression of subretinal exudation may be obtained using laser photocoagulation applied to close focal sites of RPE leakage, or by photodynamic therapy (PDT) applied to the areas of choroidal vascular hyperpermeability.
However, a recent meta-analysis concerning the therapeutic interventions for aCSC analyzed 25 randomized clinical trials (RCTs) concluded that it is not completely clear whether a clinically important benefit exists to treating aCSC since it often resolves spontaneously.66 Nevertheless, treatment should be considered if resorption does not occur within 3–4 months and in cases of aCSC recurrence in eyes that sustained a permanent visual deficit from a previous episode. Treatment is also suggested in bilateral cases of aCSC, in cases with reduced visual acuity in the fellow eye, and in patients that absolutely require prompt restoration of their vision owing to occupational necessity. The majority of ophthalmologists recommend treatment of cCSC.10,67–71
Before the advent of photodynamic therapy, the only treatment option for patients with aCSC was conventional argon laser focal photocoagulation applied to close focal sites of RPE leakage. It consists of light laser treatment that may be applied directly to the point of angiographic leakage if it is located no more than 400 µm from the fovea. Laser treatment consists of a few spots (3–5 impacts) of small diameter (100 µm), low intensity (100–150 mWatt, so as to achieve a slight whitening of the retina), and short pulse duration (100 mSec).72 The laser treatment induces scar tissue formation on the RPE, while coagulating the cluster of diseased RPE cells. It is plausible that the scar restores the RPE barrier to further subretinal fluid accumulation, allowing the pump function of the surrounding healthy epithelium to return the fluid to the choriocapillaris.73 Argon laser treatment of the leakage point of the RPE accelerates visual recovery, even though it does not likely improve the final visual outcome significantly. Only selected cases of aCSC may be treated with conventional argon laser therapy; for many cases the leak is too close to the central macula where photocoagulation would leave a blind spot, or the leakage is too widespread, and its source is difficult to identify. More recently, navigated laser photocoagulation (Navilas® System; GmbH, Teltow, Germany) has improved accuracy and comfort for retinal laser photocoagulation for various retinovascular diseases.74,75 In particular, navigated laser photocoagulation, due to its ability of eye tracking and laser planning on the fluorescein angiography on the same device, can perform precise targeted treatments thereby reducing iatrogenic retinal damage.76
Photodynamic therapy was introduced as an off-label treatment for both aCSC and cCSC approximately 10 years ago and is applied to the areas of choroidal vascular hyperpermeability. PDT utilizes verteporfin (trade name, Visudyne; Novartis AG, Switzerland), a photosensitive dye that accumulates in abnormal blood vessels. When stimulated by a nonthermal red laser light with a wavelength of 689 nm, the presence of oxygen produces many oxygen radicals resulting in local damage to the endothelium and blockage of the abnormal vessels.77,78 After the dye infusion, a laser fluence of 50 J/cm2 is directed at the surface of the damaged areas to activate the verteporfin, which triggers a healing mechanism both in the damaged choroidal vessels and RPE. Exploiting the mechanism of causing oxidative damage to the abnormal vessels of the choriocapillaris, PDT induces the absorption of the subretinal fluid and choroidal vascular remodeling with reduction of choroidal hyperpermeability.79 The efficacy of this treatment, however, is questionable since PDT is suspected of causing choriocapillaris closure and RPE alterations with subsequent damage to the photoreceptors. In addition, PDT may also stimulate the upregulation of VEGF and choroidal neovascularization.80,81 In recent years, various adjustments to the fluence and verteporfin dosage have gained good quality evidence demonstrating a low incidence of adverse effects with this CSC treatment. To reduce PDT toxicity, half-dose PDT, low-dose PDT, and reduced-fluence PDT were introduced.82 The standard dose of administered verteporfin is 6 mg/m2; a reduced dose of 3 mg/m2 (half-dose PDT) has been proposed to treat CSC.10,12,83 A lower dose of 2 mg/m2 (⅓- dose PDT) was tested, but appeared less effective.84 Reduced or half-fluence PDT consists of reducing of the laser fluence to 25 J/cm2 instead of the standard 50 J/cm2.68 Half-dose and half-fluence PDT have shown little difference in efficacy, and no difference in safety when compared to treatment with the standard/full doses. Nicolò et al85 performed a retrospective study comparing half-fluence and half-dose PDT, and concluded that half-dose PDT seems to induce a longer duration of effects in patients with cCSC than half-fluence PDT.
Despite its effectiveness, PDT is not practical in most countries owing to economic reasons. Transpupillary thermotherapy (TTT) is a less expensive therapeutic option; it uses a lower temperature than that of conventional laser photocoagulation, which minimizes permanent retinal and choroidal damage. Recently, this therapy was introduced to accelerate the resolution of CSC.86,87 The mechanism by which TTT leads to the repair of RPE damage is not yet completely understood.88
Similarly, subthreshold micropulse laser (SML) therapy is a new treatment that controls the thermal elevation induced by conventional continuous wave laser photocoagulation. Treatment with an SML reduces the thermal elevation by “chopping” a continuous wave beam into a train of repetitive short pulses, which allows the tissue to cool between pulses, thereby reducing thermal buildup. The rise in temperature in the target tissue remains sub-lethal, but it is able to create a stress response which induces anti-angiogenic effects. Therapy with an SML consists of using a grid to apply micropulsed laser burst to the retinal tissue that cause an approximate 7-°C increase in temperature with each 100-microsecond micropulse.89–91 This therapy has been applied to the retina using different lasers with different wavelengths (810, 532, and 577 nm), and it seems that thermal elevation of the retina induces the release of a group of molecules known as heat shock proteins (HSPs),90 which are involved in repairing RPE cells and in reducing the inflammatory cascade.92 Recently, in comparison to half-dose PDT, the application of 577-nm SML treatment resulted in a slightly greater reduction of the subretinal fluid in patients with chronic CSC.93
A similar mechanism of action (sub-lethal thermal effect) has been proposed by Russo et al94 using an infrared laser light of 689 nm (the same laser utilized for PDT), and has shown promising clinical results. They evaluated the photothermal effect of near-infrared 689-nm laser treatment (689-LT) versus half-dose verteporfin PDT (hd-PDT) for the treatment of cCSC. Both treatment groups showed significant improvement in BCVA, as well as reductions in central retinal and subfoveal choroidal thickness. Although hd-PDT led to a faster reduction in central retinal thickness, no significant differences were recorded between the groups for any other measured parameter at any time point. The authors thereby concluded that both hd-PDT and 689-LT were effective for treating cCSC, and speculated that the thermal effect stimulating the expression of HSPs may potentially explain the favorable effect of the non-lethal continuous wave 689-LT. Specifically, this near-infrared laser treatment, delivering 95 J/cm2 via an intensity application of 805 mW/cm2 over 118 seconds, produces a photothermal effect that heats the RPE, potentially eliciting the production of HSPs as a form of TTT.
Additionally, intravitreal injections of anti-VEGF drugs have been used, off-label, for the treatment of aCSC. This group of drugs effectively reduces choroidal hyperpermeability by blocking vascular leakage. Choroidal hyperpermeability is often associated with increased expression of VEGF, although high VEGF levels are not detected in the aqueous humor of CSC patients. Many reports have demonstrated that therapy with anti-VEGF drugs is effective, with good functional short-term outcomes for treatment of acute and chronic CSC.95,96 However, during the middle-long term period (six months), low-fluence PDT treatment is more effective than intravitreal anti-VEGF therapy in reducing the subretinal fluid in patients affected by CSC.97 Semeraro et al82 compared intravitreal bevacizumab, a recombinant humanized monoclonal antibody that inhibits the biologic activity of VEGF and prevents binding of VEGF to its receptors on the surface of endothelial cells, with low-fluence PDT in patients with cCSC. In a series of 22 patients, no statistically significant difference could be identified between the two treatment groups after 9 months of follow-up; therefore, they concluded that intravitreal bevacizumab may be a valid treatment option for treating cCSC.82 A recent meta-analysis of 6-month studies showed that patients affected by aCSC who were treated with anti-VEGF drug injections had significantly better BCVA than the observation group at the 1-month follow-up appointment; however, no significant difference was observed for longer follow-up times.98
Finally, numerous orally administered drugs have been proposed to treat CSC, including acetazolamide and other diuretics, mineralocorticoid antagonists (spironolactone and eplerenone), 5-alpha reductase inhibitors (finasteride), aspirin, beta-blockers, vitamins, and nonsteroidal anti-inflammatory medications.99 All these drugs were found to be mostly effective in animal and in vitro studies, but none have been shown to have unequivocal benefits in humans.6,82
Supporting the hypothesis of the role of mineralocorticoids on the pathogenesis of CSC, oral spironolactone and eplerenone have recently been proposed as a treatment option for CSC.100–102 Eplerenone acts as a competitive antagonist of the mineralocorticoid receptors that may be active in modulating local permeability in cCSC patients. In three case series treatment with spironolactone and eplerenone resulted in a significant reduction in central macular thickness and submacular fluid in patients with non-resolving cCSC.100–102 These drugs seemed to be beneficial and well-tolerated in half of the patients, and a complete resolution of the subretinal fluid 6 months after treatment initiation occurred.49 In non-randomized studies, treatment with carbonic anhydrase inhibitors (e.g., acetazolamide) has been shown to reduce the duration of CSC, but without significant effect on final visual recovery.103,104 Treatment with finasteride, an inhibitor of dihydrotestosterone synthesis, was evaluated in a small series of 5 patients affected by cCSC. Macular thickness and subretinal fluid accumulation diminished during the treatment period, but CSC unfortunately recurred immediately upon planned cessation of the medication105.
Conclusion
Central serous chorioretinopathy is an enigmatic disease that is still not completely understood due to its multifactorial etiology, complex pathogenesis, and wide systemic associations. No single treatment has provided overwhelming evidence of efficacy in published RCTs.66 However, the majority of these studies possess many limitations, specifically a relatively small number of participants and short follow-up periods.
Furthermore, it is not still clear whether there it is clinically beneficial to treat aCSC since it often resolves spontaneously as part of its natural progression. Some studies have suggested that early intervention (within 3 months) results in better visual outcomes, other than a timely treatment did not induce a significant difference in long-term visual acuity.106–109 Currently, approximately 79.1% of ophthalmologists tend to observe cases of aCSC, whereas for chronic cases, 66.7% use PDT, with the majority preferring half-fluence PDT.110 Based on our knowledge regarding CSC pathogenesis, it seems that the best treatment approach for this disease is individualized therapy. Factors that may prompt earlier treatment include the following: recurrent CSC, the patient’s demands, a massive serous detachment, or a history of poor response in the fellow eye to initial observation.111
A greater understanding of the pathophysiology of CSC could be provided by future studies using newer imaging techniques, such as optical coherence tomography angiography, which could provide more information as to the reactivity of the choriocapillaris to drugs and other therapies that affect choroidal vessel permeability.112
Method of Literature Search
Articles providing data on central serous chorioretinopathy were found by searching PubMed and Google Scholar with the following MESH terms: “central serous chorioretinopathy.” Additional search terms included “central serous retinopathy, central serous choroidopathy.” Case reports were considered only if they provided novel information beyond that already covered in reviews. Publications cited in articles selected by searches were also retrieved. Only articles in English were reviewed, and the full publication was retrieved. No restrictions were applied to the date of publications, which span from 1900 to 2017.
Ethics Statement
The study has been conducted in accordance with the Declaration of Helsinki. This study obtained the approval of the Ethic Committee “Provinciale di Brescia” Approval Number NP2246. The authors confirm that all patients or participants have provided written informed consent for their details to be used in this manuscript.
Acknowledgment
The authors alone are responsible for the content and writing of the paper.
Disclosure
Dr Andrea Pilotto reports personal fees from BioMarin, Chiesi, Nutricia, UCB, Zambon and Z-cube S.r.l.; grants from VItaflo Germany and Zambon Italy, Italian Ministry of Health, outside of the submitted work. The authors report no other conflicts of interest in this work.
References
1. Spaide RF, Campeas L, Haas A, et al. Central serous chorioretinopathy in younger and older adults. Ophthalmology. 1996;103(12):2070–2079. doi:10.1016/S0161-6420(96)30386-2
2. Wang M, Munch IC, Hasler PW, Prünte C, Larsen M. Central serous chorioretinopathy. Acta Ophthalmol (Copenh). 2008;86(2):126–145. doi:10.1111/j.1600-0420.2007.00889.x
3. Liegl R, Ulbig MW. Central serous chorioretinopathy. Ophthalmologica. 2014;232(2):65–76. doi:10.1159/000360014
4. N V B, Gürlü VP, Esgin H. Long-term macular function in eyes with central serous chorioretinopathy. Clin Experiment Ophthalmol. 2005;33(4):369–372. doi:10.1111/j.1442-9071.2005.01027.x
5. Mudvari SS, Goff MJ, Fu AD, et al. The natural history of pigment epithelial detachment associated with central serous chorioretinopathy. Retina. 2007;27(9):1168–1173. doi:10.1097/IAE.0b013e318156db8a
6. Loo RH, Scott IU, Flynn HW, et al. Factors associated with reduced visual acuity during long-term follow-up of patients with idiopathic central serous chorioretinopathy. Retina. 2002;22(1):19–24. doi:10.1097/00006982-200202000-00004
7. Sahu DK, Namperumalsamy P, Hilton GF, de Sousa NF. Bullous variant of idiopathic central serous chorioretinopathy. Br J Ophthalmol. 2000;84(5):485–492. doi:10.1136/bjo.84.5.485
8. Otsuka S, Ohba N, Nakao K. A long-term follow-up study of severe variant of central serous chorioretinopathy. Retina. 2002;22(1):25–32. doi:10.1097/00006982-200202000-00005
9. Yannuzzi LA. Central serous chorioretinopathy: a personal perspective. Am J Ophthalmol. 2010;149(3):361–363. doi:10.1016/j.ajo.2009.11.017
10. Chan W-M, Lai TYY, Lai RYK, Tang EWH, Liu DTL, Lam DSC. Safety enhanced photodynamic therapy for chronic central serous chorioretinopathy: one-year results of a prospective study. Retina. 2008;28(1):85–93. doi:10.1097/IAE.0b013e318156777f
11. Taban M, Boyer DS, Thomas EL, Taban M. Chronic central serous chorioretinopathy: photodynamic therapy. Am J Ophthalmol. 2004;137(6):1073–1080. doi:10.1016/j.ajo.2004.01.043
12. Reibaldi M, Cardascia N, Longo A, et al. Standard-fluence versus low-fluence photodynamic therapy in chronic central serous chorioretinopathy: a nonrandomized clinical trial. Am J Ophthalmol. 2010;149(2):307–315.e2. doi:10.1016/j.ajo.2009.08.026
13. Ojima Y, Hangai M, Sasahara M, et al. Three-dimensional imaging of the foveal photoreceptor layer in central serous chorioretinopathy using high-speed optical coherence tomography. Ophthalmology. 2007;114(12):2197–2207. doi:10.1016/j.ophtha.2007.02.015
14. Song IS, Shin YU, Lee BR. Time-periodic characteristics in the morphology of idiopathic central serous chorioretinopathy evaluated by volume scan using spectral-domain optical coherence tomography. Am J Ophthalmol. 2012;154(2):366–375.e4. doi:10.1016/j.ajo.2012.02.031
15. Kon Y, Iida T, Maruko I, Saito M. The optical coherence tomography–ophthalmoscope for examination of central serous chorioretinopathy with precipitates. Retina. 2008;28(6):864–869. doi:10.1097/IAE.0b013e3181669795
16. Tsujikawa A, Hirami Y, Sasahara M, et al. Alterations of retinal pigment epithelium in central serous chorioretinopathy. Clin Exp Ophthalmol. 2007;35:225–230. doi:10.1111/j.1442-9071.2006.01447.x
17. Fujimoto H, Gomi F, Wakabayashi T, Sawa M, Tsujikawa M, Tano Y. Morphologic changes in acute central serous chorioretinopathy evaluated by fourier-domain optical coherence tomography. Ophthalmology. 2008;115(9):1494–1500.e2. doi:10.1016/j.ophtha.2008.01.021
18. Shanmugam MP, Bhende M. Retinal pigment epithelial tears associated with idiopathic central serous chorioretinopathy. Indian J Ophthalmol. 2000;48(4):315–317.
19. Wang M, Sander B, La Cour M, Larsen M. Clinical characteristics of subretinal deposits in central serous chorioretinopathy. Acta Ophthalmol Scand. 2005;83(6):691–696. doi:10.1111/j.1600-0420.2005.00582.x
20. Maruko I, Iida T, Sugano Y, Ojima A, Sekiryu T. Subfoveal choroidal thickness in fellow eyes of patients with central serous chorioretinopathy. Retina. 2011;31(8):1603–1608. doi:10.1097/IAE.0b013e31820f4b39
21. Maruko I, Iida T, Sugano Y, Furuta M, Sekiryu T. One-year choroidal thickness results after photodynamic therapy for central serous chorioretinopathy. Retina. 2011;31(9):1921–1927. doi:10.1097/IAE.0b013e31822bf6b1
22. Agrawal R, Chhablani J, Tan K-A, Shah S, Sarvaiya C, Banker A. Choroidal vascularity index in central serous chorioretinopathy. Retina. 2016;36(9):1646–1651. doi:10.1097/IAE.0000000000001040
23. Sahoo NK, Maltsev DS, Goud A, Kulikov AN, Chhablani J. Choroidal changes at the leakage site in acute central serous chorioretinopathy. Semin Ophthalmol. 2019;34(5):380–385. doi:10.1080/08820538.2019.1635171
24. Kim DY, Joe SG, Yang HS, Lee JY, Kim J-G, Yoon YH. Subfoveal choroidal thickness changes in treated idiopathic central serous chorioretinopathy and their association with recurrence. Retina. 2015;35(9):1867–1874. doi:10.1097/IAE.0000000000000557
25. Yamada K, Hayasaka S, Setogawa T. Fluorescein-angiographic patterns in patients with central serous chorioretinopathy at the initial visit. Ophthalmologica. 1992;205(2):69–76. doi:10.1159/000310315
26. Maltsev DS, Kulikov AN, Chhablani J. Topography-guided identification of leakage point in central serous chorioretinopathy: a base for fluorescein angiography-free focal laser photocoagulation. Br J Ophthalmol. 2018;102(9):1218–1225. doi:10.1136/bjophthalmol-2017-311338
27. Warrow DJ, Hoang QV, Freund KB. Pachychoroid pigment epitheliopathy. Retina. 2013;33(8):1659–1672. doi:10.1097/IAE.0b013e3182953df4
28. Rochepeau C, Kodjikian L, Garcia M-A, Mathis T. Optical coherence tomography angiography quantitative assessment of choriocapillaris blood flow in central serous chorioretinopathy. Am J Ophthalmol. 2019;201:82–83. doi:10.1016/j.ajo.2019.01.014
29. Matet A, Daruich A, Hardy S, Behar-Cohen F. Patterns of choriocapillaris flow signal voids in central serous chorioretinopathy: an optical coherence tomography angiography study. Retina. 2018. doi:10.1097/IAE.0000000000002271
30. Bousquet E, Bonnin S, Mrejen S, Krivosic V, Tadayoni R, Gaudric A. Optical coherence tomography angiography of flat irregular pigment epithelium detachment in chronic central serous chorioretinopathy. Retina. 2018;38(3):629–638. doi:10.1097/IAE.0000000000001580
31. Kim R-Y, Chung DH, Kim M, Park Y-H. Use of choroidal vascularity index for choroidal structural evaluation in central serous chorioretinopathy with choroidal neovascularization. Retina. 2019. doi:10.1097/IAE.0000000000002585
32. Yannuzzi LA. Type A behavior and central serous chorioretinopathy. Trans Am Ophthalmol Soc. 1986;84:799–845.
33. Spahn C, Wiek J, Burger T, Hansen L. Psychosomatic aspects in patients with central serous chorioretinopathy. Br J Physiol Opt. 2003;87(6):704–708. doi:10.1136/bjo.87.6.704
34. Carlesimo SC, Piazzi G, Leone C, Di Santo L, Coccanari dè Fornari MA. Masuda’s Central Serous Chorioretinopathy (C.S.C.R.) and its somatic investment in Narcissism: our observations on new psychiatric nosography. Clin Ter. 2014;165(1):27–30. doi:10.7471/CT.2014.1657
35. Conrad R, Weber NF, Lehnert M, Holz FG, Liedtke R, Eter N. Alexithymia and emotional distress in patients with central serous chorioretinopathy. Psychosomatics. 2007;48(6):489–495. doi:10.1176/appi.psy.48.6.489
36. Conrad R, Bodeewes I, Schilling G, Geiser F, Imbierowicz K, Liedtke R. Central serous chorioretinopathy and psychological stress. Ophthalmologe. 2000;97(8):527–531. doi:10.1007/s003470070059
37. Lahousen T, Painold A, Luxenberger W, Schienle A, Kapfhammer H-P, Ille R. Psychological factors associated with acute and chronic central serous chorioretinopathy. Nord J Psychiatry. 2016;70(1):24–30. doi:10.3109/08039488.2015.1041156
38. Conrad R, Geiser F, Kleiman A, Zur B, Karpawitz-Godt A. Temperament and character personality profile and illness-related stress in central serous chorioretinopathy. Sci World J. 2014;2014:1–7. doi:10.1155/2014/631687
39. Gelber GS, Schatz H. Loss of vision due to central serous chorioretinopathy following psychological stress. Am J Psychiatry. 1987;144(1):46–50. doi:10.1176/ajp.144.1.46
40. Bousquet E, Dhundass M, Lehmann M, et al. Shift work: a risk Factor for central serous chorioretinopathy. Am J Ophthalmol. 2016;165:23–28. doi:10.1016/j.ajo.2016.02.012
41. Liu B, Deng T, Zhang J. Risk factors for central serous chorioretinopathy. Retina. 2016;36(1):9–19. doi:10.1097/IAE.0000000000000837
42. Spahn C, Wiek J, Burger T. Operationalisierte psychodynamische Diagnostik (OPD) bei patienten mit chorioretinopathia centralis serosa. Psychother Psychosom Med Psychol. 2004;54(2):52–57. doi:10.1055/s-2003-812610
43. Tittl MK, Spaide RF, Wong D, et al. Systemic findings associated with central serous chorioretinopathy. Am J Ophthalmol. 1999;128(1):63–68. doi:10.1016/S0002-9394(99)00075-6
44. Mansuetta CC, Mason JO, Swanner J, et al. An association between central serous chorioretinopathy and gastroesophageal reflux disease. Am J Ophthalmol. 2004;137(6):1096–1100. doi:10.1016/j.ajo.2004.01.054
45. Eom Y, Oh J, Kim S-W, Huh K. Systemic factors associated with central serous chorioretinopathy in Koreans. Korean J Ophthalmol. 2012;26(4):260. doi:10.3341/kjo.2012.26.4.260
46. Tsai D-C, Huang -C-C, Chen S-J, et al. Increased risk of erectile dysfunction among males with central serous chorioretinopathy - a retrospective cohort study. Acta Ophthalmol (Copenh). 2013;91(7):666–671. doi:10.1111/j.1755-3768.2012.02528.x
47. Garg SP, Dada T, Talwar D, Biswas NR. Endogenous cortisol profile in patients with central serous chorioretinopathy. Br J Physiol Opt. 1997;81(11):962–964. doi:10.1136/bjo.81.11.962
48. Haimovici R, Rumelt S, Melby J. Endocrine abnormalities in patients with central serous chorioretinopathy. Ophthalmology. 2003;110(4):698–703. doi:10.1016/S0161-6420(02)01975-9
49. Daruich A, Matet A, Dirani A, et al. Oral mineralocorticoid-receptor antagonists: real-life experience in clinical subtypes of nonresolving central serous chorioretinopathy with chronic epitheliopathy. Transl Vis Sci Technol. 2016;5(2):2. doi:10.1167/tvst.5.2.2
50. Yang D, Eliott D. Systemic mineralocorticoid antagonists in the treatment of central serous chorioretinopathy. Semin Ophthalmol. 2017;32(1):36–42. doi:10.1080/08820538.2016.1228418
51. Tewari HK, Gadia R, Kumar D, Venkatesh P, Garg SP. Sympathetic–parasympathetic activity and reactivity in central serous chorioretinopathy: a case–control study. Invest Ophthalmol Vis Sci. 2006;47(8):3474. doi:10.1167/iovs.05-1246
52. Pierce KK, Lane RG. Central serous chorioretinopathy associated with the use of ephedra. Retin Cases Brief Rep. 2009;3(4):376–378. doi:10.1097/ICB.0b013e31818ad3ce
53. Michael JC, Pak J, Pulido J, de Venecia G. Central serous chorioretinopathy associated with administration of sympathomimetic agents. Am J Ophthalmol. 2003;136(1):182–185. doi:10.1016/S0002-9394(03)00076-X
54. Fraunfelder FW, Fraunfelder FT. Central serous chorioretinopathy associated with sildenafil. Retina. 2008;28(4):606–609. doi:10.1097/IAE.0b013e31815ec2c8
55. Grover DP. Obstructive sleep apnea and ocular disorders. Curr Opin Ophthalmol. 2010;21(6):454–458. doi:10.1097/ICU.0b013e32833f00dc
56. Kloos P, Laube I, Thoelen A. Obstructive sleep apnea in patients with central serous chorioretinopathy. Graefes Arch Clin Exp Ophthalmol. 2008;246(9):1225–1228. doi:10.1007/s00417-008-0837-0
57. Leveque TK, Yu L, Musch DC, Chervin RD, Zacks DN. Central serous chorioretinopathy and risk for obstructive sleep apnea. Sleep Breath. 2007;11(4):253–257. doi:10.1007/s11325-007-0112-3
58. Arendt J. Shift work: coping with the biological clock. Occup Med (Chic Ill). 2010;60(1):10–20. doi:10.1093/occmed/kqp162
59. Gramajo AL, Marquez GE, Torres VE, et al. Therapeutic benefit of melatonin in refractory central serous chorioretinopathy. Eye. 2015;29(8):1036–1045. doi:10.1038/eye.2015.104
60. Park DW, Schatz H, Gaffney MM, Mcdonald HR, Johnson RN, Schaeffer D. Central serous chorioretinopathy in two families. Eur J Ophthalmol. 1998;8(1):42–47. doi:10.1177/112067219800800110
61. Hayashi K, Hasegawa Y, Tokoro T. Indocyanine green angiography of central serous chorioretinopathy. Int Ophthalmol. 1986;9(1):37–41. doi:10.1007/BF00225936
62. Guyer DR, Yannuzzi LA, Slakter JS, Sorenson JA, Ho A, Orlock D. Digital indocyanine green videoangiography of central serous chorioretinopathy. Arch Ophthalmol. 1994;112(8):1057–1062. doi:10.1001/archopht.1994.01090200063023
63. Chung Y-R, Kim JW, Choi S-Y, Park SW, Kim JH, Lee K. Subfoveal choroidal thickness and vascular diameter in active and resolved central serous chorioretinopathy. Retina. 2018;38(1):102–107. doi:10.1097/IAE.0000000000001502
64. Prünte C, Flammer J. Choroidal capillary and venous congestion in central serous chorioretinopathy. Am J Ophthalmol. 1996;121(1):26–34. doi:10.1016/S0002-9394(14)70531-8
65. Kanyange ML, De Laey JJ. Long-term follow-up of central serous chorioretinopathy (CSCR). Bull Soc Belge Ophtalmol. 2002;284:39–44.
66. Salehi M, Wenick AS, Law HA, Evans JR, Gehlbach P. Interventions for central serous chorioretinopathy: a network meta-analysis. Cochrane Database Syst Rev. 2015;12:CD011841. doi:10.1002/14651858.CD011841.pub2
67. Zhao M, Célérier I, Bousquet E, et al. Mineralocorticoid receptor is involved in rat and human ocular chorioretinopathy. J Clin Invest. 2012;122(7):2672–2679. doi:10.1172/JCI61427
68. Khosla PK, Rana SS, Tewari HK, Azad RU, Talwar D. Evaluation of visual function following argon laser photocoagulation in central serous retinopathy. Ophthalmic Surg Lasers. 1997;28(8):693–697.
69. Ober MD, Yannuzzi LA, Do DV, et al. Photodynamic therapy for focal retinal pigment epithelial leaks secondary to central serous chorioretinopathy. Ophthalmology. 2005;112(12):2088–2094. doi:10.1016/J.OPHTHA.2005.06.026
70. LIM SJ, ROH MI, KWON OW. Intravitreal bevacizumab injection for central serous chorioretinopathy. Retina. 2010;30(1):100–106. doi:10.1097/IAE.0b013e3181bcf0b4
71. Smretschnig E, Ansari-Shahrezaei S, Hagen S, Glittenberg C, Krebs I, Binder S. Half-fluence photodynamic therapy in chronic central serous chorioretinopathy. Retina. 2013;33(2):316–323. doi:10.1097/IAE.0b013e318280769c
72. Robertson DM, Ilstrup D. Direct, indirect, and sham laser photocoagulation in the management of central serous chorioretinopathy. Am J Ophthalmol. 1983;95(4):457–466. doi:10.1016/0002-9394(83)90265-9
73. Spitznas M. Pathogenesis of central serous retinopathy: a new working hypothesis. Graefes Arch Clin Exp Ophthalmol. 1986;224(4):321–324. doi:10.1007/BF02150023
74. Chhablani J, Rani PK, Mathai A, Jalali S, Kozak I. Navigated focal laser photocoagulation for central serous chorioretinopathy. Clin Ophthalmol. 2014;8:1543–1547. doi:10.2147/OPTH.S67025
75. Müller B, Tatsios J, Klonner J, Pilger D, Joussen AM. Navigated laser photocoagulation in patients with non-resolving and chronic central serous chorioretinopathy. Graefes Arch Clin Exp Ophthalmol. 2018;256(9):1581–1588. doi:10.1007/s00417-018-4031-8
76. Mastropasqua L, Di Antonio L, Toto L, Mastropasqua A, Di Iorio A, Carpineto P. Central serous chorioretinopathy treated with navigated retinal laser photocoagulation: visual acuity and retinal sensitivity. Ophthalmic Surg Lasers Imaging Retina. 2015;46(3):349–354. doi:10.3928/23258160-20150323-09
77. Schmidt-Erfurth U, Hasan T. Mechanisms of action of photodynamic therapy with verteporfin for the treatment of age-related macular degeneration. Surv Ophthalmol. 2000;45(3):195–214. doi:10.1016/S0039-6257(00)00158-2
78. Schmidt-Erfurth U, Michels S, Barbazetto I, Laqua H. Photodynamic effects on choroidal neovascularization and physiological choroid. Invest Ophthalmol Vis Sci. 2002;43(3):830–841.
79. Chan W-M, Lam DSC, Lai TYY, Tam BSM, Liu DTL, Chan CKM. Choroidal vascular remodelling in central serous chorioretinopathy after indocyanine green guided photodynamic therapy with verteporfin: a novel treatment at the primary disease level. Br J Ophthalmol. 2003;87(12):1453–1458. doi:10.1136/bjo.87.12.1453
80. Lai TYY, Chan W-M, Lam DSC. Transient reduction in retinal function revealed by multifocal electroretinogram after photodynamic therapy. Am J Ophthalmol. 2004;137(5):826–833. doi:10.1016/j.ajo.2003.11.079
81. Tzekov R, Lin T, Zhang K-M, et al. Ocular changes after photodynamic therapy. Invest Ophthalmol Vis Sci. 2006;47(1):377. doi:10.1167/iovs.05-0838
82. Semeraro F, Romano MR, Danzi P, Morescalchi F, Costagliola C. Intravitreal bevacizumab versus low-fluence photodynamic therapy for treatment of chronic central serous chorioretinopathy. Jpn J Ophthalmol. 2012;56(6):608–612. doi:10.1007/s10384-012-0162-3
83. Chan W-M, Lai TYY, Lai RYK, Liu DTL, Lam DSC. Half-dose verteporfin photodynamic therapy for acute central serous chorioretinopathy. Ophthalmology. 2008;115(10):1756–1765. doi:10.1016/j.ophtha.2008.04.014
84. Uetani R, Ito Y, Oiwa K, Ishikawa K, Terasaki H. Half-dose vs one-third-dose photodynamic therapy for chronic central serous chorioretinopathy. Eye. 2012;26(5):640–649. doi:10.1038/eye.2012.66
85. Nicoló M, Eandi CM, Alovisi C, et al. Half-fluence versus half-dose photodynamic therapy in chronic central serous chorioretinopathy. Am J Ophthalmol. 2014;157(5):1033–1037.e2. doi:10.1016/j.ajo.2014.01.022
86. Hussain N, Khanna R, Hussain A, Das T. Transpupillary thermotherapy for chronic central serous chorioretinopathy. Graefes Arch Clin Exp Ophthalmol. 2006;244(8):1045–1051. doi:10.1007/s00417-005-0175-4
87. Shukla D, Kolluru C, Vignesh TP, Karthikprakash S, Kim R. Transpupillary thermotherapy for subfoveal leaks in central serous chorioretinopathy. Eye. 2008;22(1):100–106. doi:10.1038/sj.eye.6702449
88. Kawamura R, Kawamura R, Ideta H, et al. Transpupillary thermotherapy for atypical central serous chorioretinopathy. Clin Ophthalmol. 2012;6:175. doi:10.2147/OPTH.S28239
89. Vujosevic S, Bottega E, Casciano M, Pilotto E, Convento E, Midena E. Microperimetry and fundus autofluorescence in diabetic macular edema. Retina. 2010;30(6):908–916. doi:10.1097/IAE.0b013e3181c96986
90. Luttrull JK, Chang DB, Margolis BWL, Dorin G, Luttrull DK. Laser resensitization of medically unresponsive neovascular age-related macular degeneration. Retina. 2015;35(6):1184–1194. doi:10.1097/IAE.0000000000000458
91. Luttrull JK. Low-intensity/high-density subthreshold diode micropulse laser for central serous chorioretinopathy. Retina. 2016;36(9):1658–1663. doi:10.1097/IAE.0000000000001005
92. Kregel KC. Heat shock proteins: modifying factors in physiological stress responses and acquired thermotolerance. J Appl Physiol. 2002;92(5):2177–2186. doi:10.1152/japplphysiol.01267.2001
93. Scholz P, Altay L, Fauser S. Comparison of subthreshold micropulse laser (577 nm) treatment and half-dose photodynamic therapy in patients with chronic central serous chorioretinopathy. Eye. 2016;30(10):1371–1377. doi:10.1038/eye.2016.142
94. Russo A, Turano R, Morescalchi F, et al. Comparison of half-dose photodynamic therapy and 689 nm laser treatment in eyes with chronic central serous chorioretinopathy. Graefes Arch Clin Exp Ophthalmol. 2017;255(6):1141–1148. doi:10.1007/s00417-017-3626-9
95. Schaal KB, Hoeh AE, Scheuerle A, Schuett F, Dithmar S. Intravitreal bevacizumab for treatment of chronic central serous chorioretinopathy. Eur J Ophthalmol. 2009;19(4):613–617. doi:10.1177/112067210901900415
96. Lim JW, Kim MU, Shin M-C. Aqueous humor and plasma levels of vascular endothelial growth factor and interleukin-8 in patients with central serous chorioretinopathy. Retina. 2010;30(9):1465–1471. doi:10.1097/IAE.0b013e3181d8e7fe
97. Bae SH, Heo JW, Kim C, et al. A randomized pilot study of low-fluence photodynamic therapy versus intravitreal ranibizumab for chronic central serous chorioretinopathy. Am J Ophthalmol. 2011;152(5):784–92.e2. doi:10.1016/j.ajo.2011.04.008
98. Lu HQ, Wang EQ, Zhang T, Chen YX. Photodynamic therapy and anti-vascular endothelial growth factor for acute central serous chorioretinopathy: a systematic review and meta-analysis. Eye. 2016;30(1):15–22. doi:10.1038/eye.2015.208
99. Moisseiev E, Holmes AJ, Moshiri A, Morse LS. Finasteride is effective for the treatment of central serous chorioretinopathy. Eye (Lond). 2016;30(6):850–856. doi:10.1038/eye.2016.53
100. Bousquet E, Beydoun T, Zhao M, Hassan L, Offret O, Behar-Cohen F. Mineralocorticoid receptor antagonism in the treatment of chronic central serous chorioretinopathy: a pilot study. Retina. 2013;33(10):2096–2102. doi:10.1097/IAE.0b013e318297a07a
101. Herold TR, Prause K, Wolf A, Mayer WJ, Ulbig MW. Spironolactone in the treatment of central serous chorioretinopathy – a case series. Graefes Arch Clin Exp Ophthalmol. 2014;252(12):1985–1991. doi:10.1007/s00417-014-2780-6
102. Singh RP, Sears JE, Bedi R, et al. Oral eplerenone for the management of chronic central serous chorioretinopathy. Int J Ophthalmol. 2015;8(2):310–314. doi:10.3980/j.issn.2222-3959.2015.02.17
103. Pikkel J, Beiran I, Ophir A, Miller B. Acetazolamide for central serous retinopathy. Ophthalmology. 2002;109(9):1723–1725. doi:10.1016/S0161-6420(02)01157-0
104. Ross A, Ross AH, Mohamed Q. Review and update of central serous chorioretinopathy. Curr Opin Ophthalmol. 2011;22(3):166–173. doi:10.1097/ICU.0b013e3283459826
105. Forooghian F, Meleth AD, Cukras C, Chew EY, Wong WT, Meyerle CB. Finasteride for chronic central serous chorioretinopathy. Retina. 2011;31(4):766–771. doi:10.1097/IAE.0b013e3181f04a35
106. Kim M, Lee S-C, Lee S-J. Intravitreal ranibizumab for acute central serous chorioretinopathy. Ophthalmologica. 2013;229(3):152–157. doi:10.1159/000345495
107. Seong HK, Bae JH, Kim ES, Han JR, Nam WH, Kim HK. Intravitreal bevacizumab to treat acute central serous chorioretinopathy: short-term effect. Ophthalmologica. 2009;223(5):343–347. doi:10.1159/000224782
108. Chung Y-R, Seo EJ, Lew HM, Lee KH. Lack of positive effect of intravitreal bevacizumab in central serous chorioretinopathy: meta-analysis and review. Eye. 2013;27(12):1339–1346. doi:10.1038/eye.2013.236
109. Lim JW, Ryu SJ, Shin MC. The effect of intravitreal bevacizumab in patients with acute central serous chorioretinopathy. Korean J Ophthalmol. 2010;24(3):155. doi:10.3341/kjo.2010.24.3.155
110. Mehta PH, Meyerle C, Sivaprasad S, Boon C, Chhablani J. Preferred practice pattern in central serous chorioretinopathy. Br J Physiol Opt. 2017;101(5):587–590. doi:10.1136/bjophthalmol-2016-309247
111. Fine HF, Ober MD, Hariprasad SM. Current concepts in managing central serous chorioretinopathy. Ophthalmic Surg Lasers Imaging Retina. 2014;45(1):9–13. doi:10.3928/23258160-20131220-01
112. Chan SY, Wang Q, Wei WB, Jonas JB. Optical coherence tomographic angiography in central serous chorioretinopathy. Retina. 2016;36(11):2051–2058. doi:10.1097/IAE.0000000000001064
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.