Back to Journals » Clinical Interventions in Aging » Volume 14
Cardioprotective effect of transcutaneous electrical acupuncture point stimulation on perioperative elderly patients with coronary heart disease: a prospective, randomized, controlled clinical trial
Authors Li H, Wu C, Yan C , Zhao S , Yang S, Liu P, Liu X , Wang M, Wang X
Received 8 April 2019
Accepted for publication 10 July 2019
Published 6 September 2019 Volume 2019:14 Pages 1607—1614
DOI https://doi.org/10.2147/CIA.S210751
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Zhi-Ying Wu
Huizhou Li1, Chuan Wu1, Caizhen Yan2, Shuang Zhao1, Shuhong Yang1, Peng Liu1, Xin Liu1, Mingjie Wang1, Xiuli Wang1
1Department of Anesthesiology, Third Hospital of Hebei Medical University, Shijiazhuang, People’s Republic of China; 2Department of Pharmacology, Hebei Medical University, Shijiazhuang, People’s Republic of China
Correspondence: Xiuli Wang
Department of Anesthesiology, Third Hospital of Hebei Medical University, No. 139 Ziqiang Road, Shijiazhuang, Hebei 050051, People’s Republic of China
Tel +86 3 118 860 2172
Email [email protected]
Purpose: The purpose of this study was to evaluate the effects of transcutaneous electrical acupoint stimulation (TEAS) on postoperative autonomic nervous system function and serum biomarkers in the elderly.
Patients and methods: A total of 122 American Society of Anesthesiologists class II or III patients with coronary heart disease undergoing spinal surgery were randomly divided into two groups: TEAS (received TEAS at Neiguan [PC6] and Ximen [PC4] for 30 minutes before anesthesia induction until the end of surgery) and control (received electrode plate at the same acupuncture points without any electrical stimulation). Serum was isolated for the measurement of concentration of high-sensitive troponin T (hs-cTnT), CRP, and CK. Heart rate (HR) and heart rate variability (HRV) including: total power (TP), low-frequency (LF) power, high-frequency (HF) power, and LF/HF ratio were used to assess autonomic nervous system function. The primary outcome was to evaluate whether TEAS changed the postoperative serum hs-cTnT. The secondary outcomes were to observe the effects of TEAS on HRV, circulating CK and CRP after surgery.
Results: Hs-cTnT, CRP, and CK concentrations were significantly higher on first, third and fifth day after surgery than those before anesthesia induction in both groups. Hs-cTnT concentration was significantly lower on the first and third day after surgery in TEAS group than in control group. Compared with 1 day before surgery, TP, LF, and HF decreased significantly and HR, LF/HF increased significantly on first, third, and fifth day after surgery in control group. Compared with control group, HR was significantly lower on the first, third, and fifth day after surgery, LF/HF decreased and TP, LF, HF were significantly higher on the first day after surgery in TEAS group.
Conclusion: TEAS at PC6 and PC4 could reduce postoperative serum hs-cTnT concentration and change HRV index to improve autonomic nervous system activity.
Keywords: transcutaneous electric acupuncture point stimulation, coronary artery disease, autonomic nervous system, high-sensitive troponin T, heart rate variability
Introduction
Although prone positioning of patients can induce negative effects on cardiac index and systolic and diastolic function, it is still necessary in certain clinical situations such as spinal or hemorrhoid surgery. A multi-center clinical study result showed that the incidence of 48-hour postoperative myocardial ischemia in high cardiovascular risk patients undergoing major non-cardiac surgery is 40%, and the incidence of 1-year postoperative cardiovascular events is 7%.1 In patients with ischemic heart disease, the negative changes of prone positioning were more significant.2 Therefore, intensive intraoperative care is important for surgical patients with coronary heart disease, and it would be meaningful to find an effective intervention which can improve perioperative cardiac function.
Aging is a physiological process associated with autonomic nervous system (ANS) dysfunction.3–5 Aged patients always have concurrent cardiovascular diseases, and general anesthesia can suppress autonomic function.6 The ANS plays an important role in response to various internal and external stimuli, which controls essential functions such as circulation, respiration, hormonal secretion, and thermoregulation.7 General anesthesia can attenuate stress response and inhibit sympathetic nerve activity, but it also inhibits vagus nerve activity.8,9 The decrease of vagus nerve activity can cause cardiovascular complications, such as atrial fibrillation,10 ventricular fibrillation,11 and heart failure,12 especially ventricular arrhythmia in patients with coronary heart disease.13 ANS dysfunction may complicate the perioperative course in surgical patients undergoing anesthesia and increase morbidity and mortality,14 which relates to the impairment of baroreceptor function caused by reduced parasympathetic activity.15
Previous studies have shown that transcutaneous electrical acupoint stimulation (TEAS) could regulate the function of ANS, enhance the activity of vagus nerve, and protect myocardial tissue.16 However, the effect of TEAS on ANS function and serum biomarkers in elderly patients with coronary heart disease remains unclear. The purpose of this study was to evaluate the effects of TEAS on postoperative ANS function and serum biomarkers in the elderly, and the results may provide a new direction for perioperative cardiac protection in the elderly.
Materials and methods
Design
We conducted a single-center, prospective, exploratory, randomized therapeutic clinical trial. This study was approved by the medical ethics committee of the Third Hospital of Hebei Medical University (approval number: KE2017-020-1) and conducted in accordance with the Helsinki Declaration. The study was registered at www.clinicaltrials.gov (identification number NCT03418194). Written informed consent was obtained from all patients.
Patients
A total of 137 patients with coronary heart disease planning to undergo spinal surgery (posterior decompression and fusion with internal fixation) from February 2018 to June 2018 at our university were screened for eligibility. As shown in Figure 1, among the patients enrolled, 15 patients were excluded because of refusal of surgery, excessive intraoperative blood loss, refusal of TEAS, and non-cooperation. The patients were randomly allocated to control or TEAS group using random number table (generated by SPSS, version 21.0 for Windows, [IBM Corporation, Armonk, NY, USA] seed=20,182,018). Patients, anesthesiologists, surgeons, and technicians in the biochemistry laboratory were blinded to the allocation.
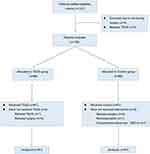 |
Figure 1 Flowchart of participations in the study.Abbreviation: TEAS, transcutaneous electrical acupoint stimulation. |
Inclusion criteria
Inclusion criteria for enrollment were as follows: 1) American Society of Anesthesiologists (ASA) physical status class II or III; 2) aged 65–80 years; 3) ischemic features (ischemic electrocardiography findings included any of the following: development of pathologic Q waves in any two contiguous leads that were ≥30 ms; development of ST segment elevation (≥2 mm in leads V1, V2, or V3 OR ≥1 mm in the other leads), ST segment depression (≥1 mm), or symmetric inversion of T waves ≥1 mm in at least two contiguous leads.
Exclusion criteria
The exclusion criteria were as follows: 1) congenital heart disease or a history of heart surgery, cardiac conduction block, frequent atrial/ventricular premature beat, atrial fibrillation and other serious arrhythmia; 2) serious liver or kidney disease; 3) use of anti-arrhythmic drug therapy over the past week; 4) temporary and permanent pacemaker implantation; 5) intraoperative blood loss >800 mL.
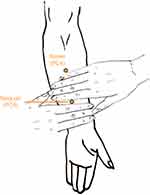
TEAS administration
Two acupuncture points were selected as the target points: Neiguan (PC6) and Ximen (PC4). As shown in Figure 2, PC6 is located on the anterior forearm 2 cun (≈40 mm) above the wrist crease between the tendons of palmaris longus and flexor carpi radialis muscles in the ventral midline of the forearm. PC4 is located 3 cun (≈60 mm) above PC6. TEAS was performed by an independent anesthesiologist who has been trained in the use of TEAS. On the day of surgery, after entering the operating room, all patients received standard monitoring including electrocardiogram (ECG), pulse oxygen saturation, respiratory rate, PetCO2, bispectral index (BIS), and invasive blood pressure (BP). TEAS group received electrical stimulation at bilateral PC6 and PC4. An electrical acupuncture point stimulator and a transcutaneous, pre-gelled silver/silver chloride surface disposable electrode were used (Instrument model G6805, SMIF, Shanghai, People's Republic of China; Shanghai Shenfeng Medical & Health Co., Ltd, Shanghai, People's Republic of China). One electrode was attached to the PC6 skin surface and connected to the positive wire of the stimulator. Another electrode was attached to PC4 skin surface and connected to the negative wire. The stimulus was kept from 30 minutes, with a dense-and-disperse frequency at 4/20 Hz before anesthesia induction until the end of surgery. The electric intensity was adjusted to maintain a slight twitching of the ring or middle finger. Patients in control group received identical electrode placement without any electrical stimulation. Every patient was told that they might or might not feel electrical stimulation.
Anesthetic and surgical procedure
The method of anesthesia was standard for the two groups. General anesthesia was induced using 0.2–0.5 μg/kg sufentanil, 0.03–0.05 mg/kg midazolam, 0.15–0.3 mg/kg etomidate, and 0.15–0.3 mg/kg cisatracurium. A reinforced catheter was inserted 2 minutes after patients received cisatracurium. Ventilation frequency was set to 12 times per minute, inhaled oxygen concentration was set to 100%, oxygen flow was set to 2 L/min, and PetCO2 was maintained between 35~45 mmHg. The entire course of anesthesia was maintained using 3–6 mg/(kg×h) propofol and 0.1–0.3 μg/(kg×min) remifentanil, which kept the change of heart rate (HR) and BP within 20% of the initial levels and sustained the BIS value range from 40–55. All patients’ surgical procedures were performed by the same experienced surgical team. All patients took prescribed, guideline-directed medication after surgery, including antibiotics and analgesics.
Data collection and biochemistry measurements
As shown in Figure 3, 5-minute of ECGs were performed for all patients 1 day before surgery. CV200 ECG monitor (Beijing Gushanfeng biomedical technology co., Ltd) was used at first, third, and fifth day after surgery. All patients underwent ECG in the morning, 7:00–9:00 am in supine position. R–R intervals were derived from the monitor. To evaluate whether TEAS at PC6 and PC4 changed the postoperative serum high-sensitive troponin T (hs-cTnT), and to observe the effects of TEAS on heart rate variability (HRV) and measure levels of CK and CRP after surgery, we collected 3 mL of venous blood once a day before the induction of anesthesia and on first, third and fifth day after surgery. The blood samples were centrifuged separately and the supernatant was collected and stored at −80°C. Serum concentration of hs-cTnT was quantified using the hs-cTnT assay (Hoffman-La Roche Ltd., Basel, Switzerland) on the Elecsys 2010/cobas e411 immunoanalyzer. CRP was determined by a particle concentrated turbidimetric immunoassay (Siemens Healthcare Diagnostics Inc, Newark, DE, USA). CK was measured using spectrophotometry (Biomate; Thermo Fisher Scientific, Inc., Waltham, MA, USA). Then, HRV analyses including total power (TP), low-frequency (LF) power, high-frequency (HF) power, and LF/HF ratio were performed by Kubios HRV Standard analysis software (Version 3.0.0, Biosignal Analysis and Medical Imaging Group at the Department of Applied Physics, University of Kuopio, Kuopio, Finland). The primary outcome was to evaluate whether TEAS at PC6 and PC4 changed the postoperative serum hs-cTnT. The secondary outcomes were to observe the effects of TEAS on HRV, circulating CK and CRP after surgery.
 |
Figure 3 Experimental flow of the study.Abbreviations: ECG, electrocardiogram; TEAS, transcutaneous electrical acupoint stimulation. |
Statistical analysis
In our previous study, hs-cTnT on first day after surgery was 35.72±11.26 (mean ± SD, ng/L). We assumed that the application of TEAS produced a protective effect in reducing the hs-cTnT after surgery, ie, about 15% reduction (30 ng/L expected) compared to the control group. A power analysis showed that a minimum sample size of 55 patients in each group was needed to provide 80% power of test and 5% marginal error. The sample size calculation was performed using PASS software (PASS 14; NCSS, LLC, Kaysville, UT, USA [ncss.com/software/pass]). Taking the potential exclusion of patients into account, we enrolled 137 patients into the trial.
All statistical analyses were performed with SPSS 21 software. Normally distributed data were described as means ± SD. Log transformations were used to correct the non-normally distributed data of HRV. Categorical data were described as numbers (percentage) and analyzed with the chi-squared test. Comparison between the two groups was performed with Student’s t-test. Comparison of repeated measures data was done with two-way repeated-measures ANOVA. Two-tailed probability value of P<0.05 was considered as statistically significant.
Results
There were no significant differences between control group and TEAS group with regard to sex, age, body mass index, ASA class, concurrent diseases, anesthesia time, operation time, and blood loss (Table 1) (P>0.05).
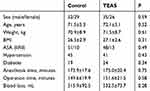 |
Table 1 General information of patients (n=122) |
As shown in Table 2, there were no significant differences with regard to hs-cTnT, CRP, and CK concentration between control group and TEAS group before anesthesia induction. Overall, the serum hs-cTnT, CRP, and CK concentrations increased significantly on the first, third, and fifth day after surgery in the two groups (P<0.05). Although the differences did not have a statistical significance on the fifth day (P>0.05), hs-cTnT concentrations in the TEAS group were significantly lower than that in control group on the first and third day after surgery (P<0.05). The postoperative values for CRP and CK were not statistically different in the two groups.
 |
Table 2 Serum markers' concentration in the two groups (n=61, ±s) |
As shown in Table 3, there were no significant differences with regard to TP, LF, HF, LF/HF, and HR values between control group and TEAS group on 1 day before surgery. Compared with 1 day before surgery, TP, LF, and HF were significantly lower whereas HR and LF/HF were significantly higher on the first, third, and fifth day after surgery in the control group. Compared with 1 day before surgery, HR increased significantly on the first and third day after surgery in TEAS group. Compared with control group, HR decreased significantly on the first, third, and fifth day after surgery, LF/HF decreased significantly on the first day after surgery, and TP, LF, and HF increased significantly on the first day after surgery in the TEAS group.
 |
Table 3 HRV measurements of the patients in the two groups (n=61, ±s) |
Discussion
To our knowledge, this was the first study which has demonstrated clinical myocardial protection using TEAS treatment in elderly patients with coronary heart disease undergoing spinal surgery. In the present study, we focused on the cardiac protection of elderly patients with coronary heart disease undergoing spinal surgery, because for these people, prone positioning worsens their perioperative insufficient cardiac function.2 In addition, the improvement of ANS function was observed.
TEAS is a non-invasive therapy similar to acupuncture, which can have effects on the nervous system and regulates the function of organs. The effect of TEAS is similar to electrical acupuncture and hand needle acupuncture.17–19 There is evidence showing that stimulus at PC6 could adjust ANS20,21 and protect myocardial tissue.16 Therefore, these two acupuncture points were chosen for electrical stimulation.
Hs-cTnT is a serum biomarker with high specificity and sensitivity which can be detected in blood after myocardial injury. Even minor postoperative cTnT elevation may increase morbidity and mortality in patients with myocardial injury.22–24 CRP is a non-specific biomarker of inflammation which has been described as a risk factor for cardiovascular disease.25 CK is an indicator that reflects the amplitude of operation injury in spinal surgery.26 In this study, our data indicated that TEAS reduced the extent of hs-cTnT elevation for 3 days, although hs-cTnT and CRP concentration increased significantly after spinal surgery. Similar to a previous study,27 our study suggests that TEAS could attenuate myocardial injury by reducing cTnT. However, in contrast to this previous study, our study indicates that application of TEAS could not reduce CRP level. The reasonable explanation is probably the even older age of patients enrolled in our study..
HRV refers to the beat-to-beat variation in HR intervals, which can reflect the balance of the ANS tone.28,29 In this study, the effects of TEAS on HRV were evaluated using TP, LF, HF, and LF/HF, and the change of the postoperative HR was analyzed at the same time. TP mainly reflects the total ANS activity. LF reflects sympathetic and vagus nerve activity and mainly measures sympathetic nerve activity; HF reflects the activity of the vagus nerve activity and LF/HF ratio measures the balances of sympathetic and vagus nerve activity.30 In our study, the HRV index changes after surgery indicated that general anesthesia suppressed ANS function, which was consistent with a previous study.6 Although the application of TEAS at PC6 and PC4 did not reverse the suppression totally, it improved ANS activity, especially on the first day after surgery. Compared with control group, the improvement of HF and decrease of LF and LF/HF indicate that vagus nerve activity was well protected on the first day after surgery, the sympathetic and vagus equilibrium shifted in the vagus direction. This indicates that the protection of ANS may only have lasted for 1 day and that TEAS intervention may be not strong enough to afford complete protection to ANS of the elderly with coronary heart disease.
The mechanism of effect of TEAS on HRV and serum hs-cTnT is still unclear. Stimulation of P5-6 reduced vagally evoked activity of cardiovascular NTS cells31 and improved cardiac function by increasing left ventricular diastolic and systolic function in rat myocardial cells.32 In addition, stimulation of PC6 could also improve the blood flow of coronary artery33 and increase Gs-alpha protein in cardiomyocytes.34 PC4 synergistically treats heart diseases combined with PC6.35 These may partly explain the mechanism of cardioprotective effect of TEAS.
There is no doubt that this study has several limitations. First, the number of patients was relatively small. Second, we only studied elderly patients undergoing spinal surgery. Third, among all enrolled patients, the withdrawal of eight patients after randomization and the lack of an intention-to-treat approach is a potential source of bias. Therefore further supporting studies are needed to confirm the effects of TEAS on postoperative HRV and biomarkers in patients receiving other surgeries or patients of other age groups.
Conclusion
Application of TEAS at PC6 and PC4 could reduce postoperative serum hs-cTnT concentration and change HRV index to improve ANS activity. Our results suggest that TEAS is an effective and simple approach which can be used clinically to reduce perioperative heart ischemic injury.
Data sharing statement
The datasets generated and/or analyzed during the current study are not publicly available due to provisions for the preservation of relevant raw data in our hospital, but are available from the corresponding author on reasonable request.
Acknowledgment
This work was supported by Key Discipline Tracking Project of Hebei Provincial Health and Family Planning Commission (GL-2016-29) and Hebei Excellent Talent Fund.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Lurati Buse GA, Schumacher P, Seeberger E, et al. Randomized comparison of sevoflurane versus propofol to reduce perioperative myocardial ischemia in patients undergoing noncardiac surgery. Circulation. 2012;126(23):2696–2704. doi:10.1161/CIRCULATIONAHA.112.126144
2. Shimizu M, Fujii H, Yamawake N, Nishizaki M. Cardiac function changes with switching from the supine to prone position: analysis by quantitative semiconductor gated single-photon emission computed tomography. J Nucl Cardiol. 2015;22(2):301–307. doi:10.1007/s12350-014-0058-3
3. El Beheiry H, Mak P. Effects of aging and propofol on the cardiovascular component of the autonomic nervous system. J Clin Anesth. 2013;25(8):637–643. doi:10.1016/j.jclinane.2013.07.004
4. Voss A, Schroeder R, Heitmann A, Peters A, Perz S, Hernandez AV. Short-term heart rate variability–influence of gender and age in healthy subjects. PLoS One. 2015;10(3):e0118308. doi:10.1371/journal.pone.0118308
5. Ogliari G, Mahinrad S, Stott DJ, et al. Resting heart rate, heart rate variability and functional decline in old age. CMAJ. 2015;187(15):E442–E449. doi:10.1503/cmaj.150462
6. Matchett G, Wood P. General anesthesia suppresses normal heart rate variability in humans. Chaos. 2014;24(2):023129. doi:10.1063/1.4882395
7. Wehrwein EA, Orer HS, Barman SM. Overview of the anatomy, physiology, and pharmacology of the autonomic nervous system. Compr Physiol. 2016;6(3):1239–1278. doi:10.1002/cphy.c150037
8. Shin S, Lee JW, Kim SH, Jung YS, Oh YJ. Heart rate variability dynamics during controlled hypotension with nicardipine, remifentanil and dexmedetomidine. Acta Anaesthesiol Scand. 2014;58(2):168–176. doi:10.1111/aas.12233
9. Heid F, Kauff DW, Lang H, Kneist W. Impact of inhalation vs. intravenous anaesthesia on autonomic nerves and internal anal sphincter tone. Acta Anaesthesiol Scand. 2015;59(9):1119–1125. doi:10.1111/aas.12535
10. Chen PS, Chen LS, Fishbein MC, Lin SF, Nattel S. Role of the autonomic nervous system in atrial fibrillation: pathophysiology and therapy. Circ Res. 2014;114(9):1500–1515. doi:10.1161/CIRCRESAHA.114.303772
11. Shen MJ, Zipes DP. Role of the autonomic nervous system in modulating cardiac arrhythmias. Circ Res. 2014;114(6):1004–1021. doi:10.1161/CIRCRESAHA.113.302549
12. Florea VG, Cohn JN. The autonomic nervous system and heart failure. Circ Res. 2014;114(11):1815–1826. doi:10.1161/CIRCRESAHA.114.302589
13. Bigger JT
14. Lankhorst S, Keet SW, Bulte CS, Boer C. The impact of autonomic dysfunction on peri-operative cardiovascular complications. Anaesthesia. 2015;70(3):336–343. doi:10.1111/anae.12904
15. La Rovere MT, Christensen JH. The autonomic nervous system and cardiovascular disease: role of n-3 PUFAs. Vascul Pharmacol. 2015;71:1–10. doi:10.1016/j.vph.2015.02.005
16. Shi L, Fang J, Zhao J, et al. Comparison of the therapeutic effects of acupuncture at PC6 and ST36 for chronic myocardial ischemia. Evid Based Complement Alternat Med. 2017;2017:7358059. doi:10.1155/2017/7358059
17. Zhou J, Peng W, Xu M, Li W, Liu Z. The effectiveness and safety of acupuncture for patients with alzheimer disease: a systematic review and meta-analysis of randomized controlled trials. Medicine. 2015;94(22):e933. doi:10.1097/MD.0000000000000874
18. Lu Z, Dong H, Wang Q, Xiong L. Perioperative acupuncture modulation: more than anaesthesia. Br J Anaesth. 2015;115(2):183–193. doi:10.1093/bja/aev227
19. Ng MC, Jones AY, Cheng LC. The role of Acu-TENS in hemodynamic recovery after open-heart surgery. Evid Based Complement Alternat Med. 2011;2011:301974. doi:10.1155/2011/196190
20. Sun J, Li X, Yang C, et al. Transcutaneous electrical acupuncture stimulation as a countermeasure against cardiovascular deconditioning during 4 days of head-down bed rest in humans. Acupunct Med. 2015;33(5):381–387. doi:10.1136/acupmed-2014-010730
21. Wang G, Tian Y, Jia S, Zhou W, Zhang W. Pilot study of acupuncture point laterality: evidence from heart rate variability. Evid Based Complement Alternat Med. 2013;2013:476064. doi:10.1155/2013/476064
22. Devereaux PJ, Biccard BM, Sigamani A, et al. Association of postoperative high-sensitivity troponin levels with myocardial injury and 30-day mortality among patients undergoing noncardiac surgery. Jama. 2017;317(16):1642–1651. doi:10.1001/jama.2017.4360
23. Devereaux PJ, Chan MT, Alonso-Coello P, et al. Association between postoperative troponin levels and 30-day mortality among patients undergoing noncardiac surgery. Jama. 2012;307(21):2295–2304. doi:10.1001/jama.2012.5502
24. Kavsak PA, Walsh M, Srinathan S, et al. High sensitivity troponin T concentrations in patients undergoing noncardiac surgery: a prospective cohort study. Clin Biochem. 2011;44(12):1021–1024. doi:10.1016/j.clinbiochem.2011.05.017
25. Libiszewski M, Drozda R, Bialecki J, et al. Assesment of inflammatory response intensity in early postoperative period in patients after hernioplasty operated on with classic stoppa method and videoscopic TEP method. Pol Przegl Chir. 2011;83(9):497–501. doi:10.2478/v10035-011-0077-6
26. Lombao Iglesias D, Bago Granell J, Vilor Rivero T. Validity of creatine kinase as an indicator of muscle injury in spine surgery and its relation with postoperative pain. Acta Orthop Belg. 2014;80(4):545–550.
27. Ni X, Xie Y, Wang Q, et al. Cardioprotective effect of transcutaneous electric acupoint stimulation in the pediatric cardiac patients: a randomized controlled clinical trial. Paediatr Anaesth. 2012;22(8):805–811. doi:10.1111/j.1460-9592.2012.03822.x
28. Pumprla J, Howorka K, Groves D, Chester M, Nolan J. Functional assessment of heart rate variability: physiological basis and practical applications. Int J Cardiol. 2002;84(1):1–14.
29. Task force of the european society of cardiology and the North American society of pacing and electrophysiology. Heart rate variability: standards of measurement, physiological interpretation and clinical use. Circulation. 1996;93(5):1043–1065.
30. Rajendra Acharya U, Paul Joseph K, Kannathal N, Lim CM, Suri JS. Heart rate variability: a review. Med Biol Eng Comput. 2006;44(12):1031–1051. doi:10.1007/s11517-006-0119-0
31. Tjen ALSC, Fu LW, Guo ZL, Longhurst JC. Modulation of neurally mediated vasodepression and bradycardia by electroacupuncture through opioids in nucleus tractus solitarius. Sci Rep. 2018;8(1):1900. doi:10.1038/s41598-018-19672-9
32. Wang S, Ren L, Jia L, et al. Effect of acupuncture at neiguan (PC 6) on cardiac function using echocardiography in myocardial ischemia rats induced by isoproterenol. J Tradit Chin Med. 2015;35(6):653–658.
33. Syuu Y, Matsubara H, Kiyooka T, et al. Cardiovascular beneficial effects of electroacupuncture at neiguan (PC-6) acupoint in anesthetized open-chest dog. Jpn J Physiol. 2001;51(2):231–238.
34. Gao J, Fu W, Jin Z, Yu X. A preliminary study on the cardioprotection of acupuncture pretreatment in rats with ischemia and reperfusion: involvement of cardiac beta-adrenoceptors. J Physiol Sci. 2006;56(4):275–279. doi:10.2170/physiolsci.RP006606
35. Wang Q, Liang D, Wang F, et al. Efficacy of electroacupuncture pretreatment for myocardial injury in patients undergoing percutaneous coronary intervention: a randomized clinical trial with a 2-year follow-up. Int J Cardiol. 2015;194:28–35. doi:10.1016/j.ijcard.2015.05.043
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.