Back to Journals » International Journal of Nanomedicine » Volume 14
Biogenic selenium nanoparticles synthesized by Lactobacillus casei ATCC 393 alleviate intestinal epithelial barrier dysfunction caused by oxidative stress via Nrf2 signaling-mediated mitochondrial pathway
Authors Xu C , Qiao L, Ma L, Guo Y, Dou X, Yan S, Zhang B, Roman A
Received 21 December 2018
Accepted for publication 8 May 2019
Published 18 June 2019 Volume 2019:14 Pages 4491—4502
DOI https://doi.org/10.2147/IJN.S199193
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Mian Wang
Chunlan Xu, Lei Qiao, Li Ma, Yu Guo, Xina Dou, Shuqi Yan, Baohua Zhang, Alexandra Roman
The Key Laboratory for Space Bioscience and Biotechnology, School of Life Sciences, Northwestern Polytechnical University, Xi’an, Shaanxi, People’s Republic of China
Background: Selenium (Se) can exert antioxidative activity and prevent the body from experiencing oxidative injury. Biogenic Se nanoparticles (SeNPs) synthesized by probiotics possess relatively strong chemical stability, high bioavailability, and low toxicity, this makes them potential Se supplements. Previously, we demonstrated that SeNPs synthesized by Lactobacillus casei ATCC 393 can alleviate hydrogen peroxide (H2O2)-induced human and porcine intestinal epithelial cells’ oxidative damage. However, the antioxidant mechanism remains unclear.
Methods: The possible antioxidant mechanism and protective effect of SeNPs on intestinal epithelial permeability and mitochondrial function were evaluated by establishing an H2O2-induced oxidative damage model of human colon mucosal epithelial cells (NCM460) and conducting Nrf2 inhibitor interference experiments. Mitochondrial membrane potential (MMP), mitochondrial DNA content, adenosine triphosphate (ATP), ROS, and protein expression levels of Nrf2-related genes were determined. Mitochondrial ultrastructure was visualized by transmission electron microscopy.
Results: An amount of 4 μg Se/mL of SeNPs synthesized by L. casei ATCC 393 alleviated increase of ROS, reduced ATP and MMP, and maintained intestinal epithelial permeability in NCM460 cells challenged by H2O2. In addition, SeNPs improved the protein levels of Nrf2, HO-1, and NQO-1. Moreover, SeNPs attenuated the damage of mitochondrial ultrastructure caused by oxidative stress. Nrf2 inhibitor (ML385) abolished the regulatory effect of SeNPs on intracellular ROS production.
Conclusion: Data suggest that biogenic SeNPs synthesized by L. casei ATCC 393 can protect the intestinal epithelial barrier function against oxidative damage by alleviating ROS-mediated mitochondrial dysfunction via Nrf2 signaling pathway. Biogenic SeNPs are an attractive candidate for potential Se supplement agent in preventing oxidative stress-related intestinal disease by targeting mitochondria.
Keywords: Lactobacillus casei, selenium nanoparticles, mitochondrial, antioxidant, reactive oxygen species, intestinal barrier
Introduction
Nanotechnology is a vigorous research and innovation area which affects our daily lives in many ways. Nanoparticles have abundant applications in biomedicine, agriculture, and the environment etc, due to their unique physical and chemical properties.1–4 In recent years, biological synthesis of nanoparticles using biological entities such as microorganisms, algae, and plants have been regarded as “green processes” due to its rapidity, cost-effectiveness, and eco-friendly nature compared with traditional physical and chemical methods.5,6 Moreover, various non-metallic, metallic including metal oxide nanoparticles synthesized via biological technique exhibit various biological activities.7–10 Among the non-metallic nanoparticles, selenium nanoparticles (SeNPs) have attracted extensive attention due to their exclusive physicochemical properties including chemical stability, biocompatibility, and low toxicity. Biogenic SeNPs have wide applicability in the field of nanomedicine as antimicrobial, antioxidant, and anticancer agents.6,11 Furthermore, SeNPs are considered to be a potential and novel nutritional supplement for Se.12
The intestinal epithelial barrier function is the key component of intestines to prevent the invasion of pathogenic antigen. In order to maintain intestinal homeostasis, the intestinal epithelial barrier must be tightly regulated. If intestinal barrier dysfunction is not effectively controlled, it will result in inflammatory bowel disease (IBD). Mitochondria, as the core of cell viability and function, control many physiological metabolic processes.13,14 Mitochondria are crucial for the regulation of intestinal epithelial barrier function. Mitochondrial dysfunction could cause abnormal intestinal barrier function. Therefore, mitochondria may be a potential target to treat IBD.15 The “openness” of the mitochondrial permeability transition pore could cause the overproduction of ROS and release of Cytc,16 which could result in intestinal mucosal injury.17,18
The intestines are more susceptible to oxidative stress. Therefore, it is urgent to explore safe and effective antioxidants to alleviate intestinal disorders caused by oxidative stress. SeNPs are a highly effective antioxidant. SeNPs exert antioxidative effects in a variety of ways, including scavenging free radicals, activating antioxidative selenase activity such as GPx, and enhancing the activity of other key enzymes in the body’s antioxidant defense system.19 In previous studies, we found that biogenic SeNPs synthesized by Lactobacillus casei ATCC 393 had the ability to protect intestinal epithelium cells (IPEC-J2 and NCM460 cells) from oxidative injury.20 However, the potential antioxidative mechanism remains unclear.
To determine whether biogenic SeNPs synthesized by L. casei ATCC 393 could alleviate oxidative stress-induced intestinal epithelial barrier dysfunction via Nrf2 signaling-mediated mitochondrial pathway, we established the oxidative damage model of human colon mucosal epithelial cells (NCM 460) induced by hydrogen peroxide (H2O2) and conducted an Nrf2 inhibitor interference experiment to detect intestinal epithelial permeability, the mitochondrial membrane potential (MMP), mitochondrial DNA (mtDNA) content, adenosine triphosphate (ATP) and ROS production, protein expression levels of Nrf2, NQO-1, and HO-1. The ultrastructure of mitochondria was visualized by transmission electron microscopy (TEM).
Materials and methods
Microorganism and culture conditions
L. casei ATCC 393 strain was kept in our laboratory. deMan, Rogosa and Sharpe (MRS) broth was purchased from Sigma-Aldrich Co. (St Louis, MO, USA). L. casei ATCC 393 used for biosynthesis of SeNPs was cultivated with MRS at 37°C without shaking.
Reagents
Sodium selenite, fluorescein isothiocyanate-dextran (FITC-dextran) with average molecular weight 4,000 and H2O2 were purchased from Sigma-Aldrich Co.. Nrf2 inhibitor (ML385) was purchased from MedChemExpress (Shanghai, People's Republic of China). All reagents for cell culture were purchased from Life Technologies (Thermo Fisher Scientific, Waltham, MA, USA). Mito-Tracker Green Kit, ATP Assay Kit, MMP Assay Kit, ROS detection Kit, and Cell Counting Kit-8 (CCK-8) were purchased from Beyotime Biotechnology (Shanghai, People's Republic of China). RIPA lysis buffer and bicinchoninic acid (BCA) protein assay kit were purchased from Solarbio Life Sciences Biotech Co. (Beijing, People's Republic of China). Nrf2, NQO-1, and HO-1 primary antibodies and corresponding secondary antibody were purchased from Abcam Biotechnology (Cambridge, MA, USA). Genomic DNA extraction Kit was purchased from OMEGA Bio-Tek (Norcross, GA, USA). ChamQTM SYBR® qPCR Master Mix Kit was purchased from Vazyme (NanJing, People's Republic of China).
Cell culture
NCM460 cells were purchased from Cell Resource Center, Shanghai Institute, Chinese Academy of Sciences (Shanghai, People's Republic of China). NCM460 cells were cultured in DMEM/high glucose medium with 10% FBS and 1% antibiotic mixture (100 U/mL of penicillin and 100 μg/mL streptomycin) at 37°C under a humidified atmosphere containing 5% CO2.
Preparation of SeNPs
SeNPs were prepared by the method established in our laboratory.20
Effect of SeNPs on cell viability of NCM460 exposed to H2O2
NCM460 cells (1×103 cells per well) were seeded into 96-well plates (Corning Incorporated, Corning, NY, USA) overnight to allow cells' attachment. Cells from SeNPs groups were cultivated with 4 μg Se/mL of SeNPs for 12 hours based on our previous study.20 Other groups were given an equal volume of FBS-free medium. Then, cells from H2O2-model and SeNPs protective groups were exposed to 500 μM H2O2 for 6 hours. Cell viability was evaluated with CCK-8 according to the manufacturer’s instructions.
Intestinal epithelial ATP levels
An amount of 2×105 NCM460 cells were seeded into 6-well plates (Corning Incorporated). After overnight cultivation, cells from SeNPs groups were cultivated with 4 μg Se/mL of SeNPs for 12 hours. Other groups were administered an equal volume of FBS-free medium. After that, cells from H2O2-model and SeNPs protective groups were exposed to 500 μM H2O2 for 6 hours. After treatment, intracellular ATP was measured with ATP Assay Kit according to the corresponding protocol using GloMax®-96 Microplate Luminometer (Bio-Rad Laboratories Inc., Hercules, CA, USA).
Mito-tracker green staining
Cell treatment methods were the same as described previously. Mito-Tracker Green is a mitochondria green fluorescent probe that can be used for mitochondrial-specific fluorescent staining of living cells. After treatment, cells were cultivated with 100 nM Mito-Tracker Green for 30 minutes at 37°C. After washing three times with serum-free medium, the cellular mitochondria were visualized by fluorescence microscope (Leica Microsystems, Wetzlar, Germany).
MMP (∆ψm) assay
MMP of experimental cells was detected by MMP Assay Kit with JC-1 staining. Cell treatment methods were the same as described previously. After that, cells were washed three times with phosphatic buffer solution (PBS) and stained with JC-1 for 20 minutes. Then, cells were washed three times with JC-1 buffer after JC-1 solution was discarded. Finally, images were captured with a Laser Scanning Confocal Microscope (Leica sp5, Leica Microsystems). Green monomeric JC-1 and red aggregated JC-1 were detected at an emission wavelength of 530 nm and 590 nm, respectively. Moreover, MMP was also detected by Multi-function Microplate Reader (Bio-Rad Laboratories Inc.) with JC-1 staining in the same manner as described previously.
ROS detection and quantification
Cell treatment methods were the same as described previously. After treatment, intracellular ROS were measured by ROS detection Kit. Briefly, cells were incubated with 10 μM 2ʹ,7ʹ-dichlorodihydrofluorescein diacetate (DCFH-DA) for 20 minutes. Then, cells were washed three times with FBS-free medium to remove DCFH-DA. The levels of intracellular ROS were determined by FACSCalibur Flow Cytometry (BD Biosciences, San Jose, CA, USA) at an excitation wavelength of 480 nm and emission wavelength of 515 nm. In addition, intracellular ROS were observed by Fluorescence Microscope (Leica Microsystems).
Measurement of intestinal epithelial mtDNA copy number
mtDNA copy number was determined according to a previously reported method with slight modification.21 Primer sequences for real-time PCR assay were as follows: mt D-Loop sense 5ʹ- GATCGTACATAGCACATATCATGTC-3ʹ, anti-sense 5ʹ-GGTCCTGAAGTAAGAACCAGATG-3ʹ; β-actin sense 5ʹ- CCCCTCCTCTCTTGCCTCTC-3ʹ, anti-sense 5ʹ- AAAAGTCCTAGGAAAATGGCAGAAG-3ʹ.21 Firstly, total DNA was extracted from NCM460 cells by genomic DNA extraction Kit. Real-time PCR was performed with a ChamQTM SYBR® qPCR Master Mix Kit. The PCR amplification system had 20 µL reaction volume consisting of 1 µL DNA, 10 µL 2×ChamQ SYBR qPCR Master Mix, 0.5 µL each of sense and anti-sense primers, and 8 µL ddH2O. Then, PCR reaction was performed on CFX96 TouchTM Real-Time PCR Detection System (Bio-Rad Laboratories Inc.) according to the following conditions: 95°C for 10 seconds, 50 cycles including 95°C for 10 seconds, 55°C for 30 seconds, and 72°C for 30 seconds. The relative mtDNA copy number was analyzed by 2−∆∆Ct method.
Intestinal epithelial permeability analysis
An amount of 1×105 NCM460 cells per well were seeded into 24-well transwell plates with a pore size of 0.4 μm (Corning Incorporated). Cell treatment methods were the same as detailed previously. After treatment, transendothelial electrical resistance (TER) was determined by Millicell Electrical Resistance System (Millipore, Billerica, MA, USA). Moreover, effects of SeNPs on the paracellular passage of FITC-dextran (4 kDa) in NCM460 exposed to H2O2 were evaluated. Briefly, after treatment, 100 μL of FITC-dextran (2.2 μg/mL) was added to the upper chamber of transwells and co-incubated for 2 hours. After that, 100 μL of culture medium at the apical side and basolateral side were collected, respectively. Concentration of FITC-dextran in medium was detected by Multi-function Microplate Reader (Bio-Rad Laboratories Inc.) at an excitation wavelength of 490 nm and an emission wavelength of 520 nm.
Ultrastructure of mitochondria observed by TEM
An amount of 5×105 NCM460 cells were seeded into a 25 cm2 flask and cultivated until they reached 80%~90% confluence. Then, cells were exposed to H2O2 for 6 hours after treatment with 4 μg Se/mL of SeNPs for 12 hours. After that, cells were collected and washed three times with PBS, then fixed with 2.5% glutaraldehyde overnight at 4°C. After samples were pretreated according the standard procedures including staining, dehydration, and embedding, and slicing ultra-thin sections, the ultrastructure of mitochondria was observed on a Techni 20 transmission electron microscope.
Expression levels of Nrf2 signaling-associated proteins analyzed by Western blot
Cell treatment methods were the same as described previously. After treatment, cells were washed three times with PBS. Total protein was extracted by homogenizing cells with 200 µL RIPA lysis buffer. Protein concentrations were measured by the BCA protein Assay Kit. Samples containing approximately 40 μg protein were boiled with 5× loading buffer at 95°C for 5 minutes and separated by 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis, and then transferred to polyvinylidene difluoride membrane (Millipore, Massachusetts, USA). After being blocked with 5% skim milk blocking buffer for 2 hours at room temperature, the membranes were incubated with the primary antibodies for Nrf2, HO-1, NQO-1, and β-actin at 4°C overnight. Subsequently, the membranes were washed three times with Tris buffered saline with Tween 20, and then incubated with secondary antibodies for 1 hour at room temperature. After that, the immunoreactive bands were visualized by Clarity Western ECL substrate Kit (BioRad Laboratories Inc.). The gray values of the bands were analyzed by the built-in software.
Effect of Nrf2 inhibitor on cell viability and ROS production
Nrf2 inhibitor (ML385) was dissolved in 100% DMSO to prepare a stock solution, and then diluted with DMEM/high glucose medium containing 0.5% DMSO.22 Firstly, NCM460 cells were exposed to different concentrations of ML385 (0.2, 0.5, 1, 2, 5, 10, 15 μM) for 12 hours. After that, cell viability was determined by CCK-8. Moreover, NCM460 cells were cultivated with 4 µg/mL of SeNPs and/or 5 µM ML385 for 12 hours, followed by treatment with 500 μM H2O2 for 6 hours. Then, the effect of ML385 on the protein expression levels of Nrf2 were evaluated by Western blot analysis. In addition, intracellular ROS levels in each experimental group were measured by the ROS detection Kit with DCFH-DA staining.
Statistical analysis
All experimental data were statistically analyzed by SPSS v.19.0 statistical software (IBM Corporation, Armonk, NY, USA) and are shown as mean ± standard error of mean (SEM). All assays were performed in at least three independent experiments. The statistical significance was calculated by one-way ANOVA or Student’s t-test. Differences were considered to be significant at P<0.05.
Results
Establishment of H2O2-induced cell injury model
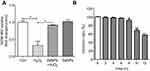
Results showed that treatment with 500 μM H2O2 for 12 hours significantly affected the viability of NCM460 cells with inhibitory ratio of 56% (Figure 1A). Compared to the H2O2 model group, pretreatment with 4 μg Se/mL of SeNPs significant alleviated the toxic effects of 500 μM H2O2 on NCM460 cells. However, exposure to 500 μM H2O2 for 6 hours did not exhibit an inhibitory effect on the growth of NCM460 cells (Figure 1B). Therefore, exposure to 500 μM H2O2 for 6 hours at 37°C was chosen as the conditions to induce NCM460 cells' oxidative damage in the following experiments.
SeNPs alleviates H2O2-induced intestinal epithelial barrier dysfunction
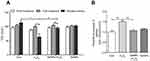
As shown in Figure 2A, treatment with SeNPs before exposure to H2O2 did not affect the TER of cells. However, H2O2 obviously caused a reduction in TER after NCM460 cells were exposed to H2O2 for 6 hours, which suggests that H2O2-induced oxidative stress destroyed the intestinal epithelial barrier function and the models were successfully established. Pretreatment with SeNPs significantly attenuated the reduction of TER caused by H2O2. Moreover, compared with the normal control, FITC-dextran fluxes across NCM460 cells in H2O2-model group increased 1.49-fold. In contrast, pretreatment with SeNPs significantly alleviated the H2O2-induced increase of FITC-dextran permeation (Figure 2B).
SeNPs inhibits H2O2-induced ROS production
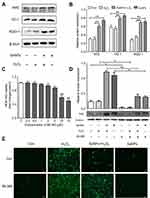
Effect of SeNPs on redox state of NCM460 cells challenged by oxidative stress was evaluated by detecting intracellular ROS production. DCFH can be oxidized to DCF by ROS, which stimulates the green fluorescence and indicates ROS levels. Results showed that H2O2 caused the increase of ROS production when compared with control group. However, pretreatment with SeNPs obviously alleviated the increase of ROS caused by H2O2 (Figure 3A–C).
SeNPs attenuates H2O2-caused mitochondrial dysfunction
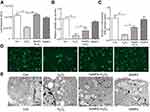
Intracellular ATP levels were determined after treatment with H2O2 and/or SeNPs. Results showed that exposure to H2O2 significantly reduced intracellular ATP levels when compared with the normal control group, which indicated that H2O2 resulted in mitochondrial dysfunction. Pretreatment with SeNPs could alleviate the above-mentioned phenomenon (Figure 4A). As shown in Figure 4B, mtDNA copy number of the H2O2-model group decreased rapidly. However, mtDNA copy number in SeNPs pretreatment group was higher than that in H2O2-model group. Moreover, pretreatment with SeNPs effectively inhibited decrease of mitochondria number caused by H2O2 (Figure 4C and D).
Ultrastructure of mitochondria observed by TEM
Compared with the normal control, the ultrastructure of mitochondria in H2O2-induced oxidative damage model group exhibited apparent change: vacuoles were found; the swell was obvious; the mitochondrial cristae were vague; the density of matrix was low; minority of mitochondrial membrane was not intact. However, pretreatment with SeNPs alleviated H2O2-induced destruction of mitochondria. In control and SeNPs protective groups, the mitochondria had a column or mesh shape; the mitochondrial cristae were clear; the density of matrix was normal; the mitochondrial membrane was intact (Figure 4E).
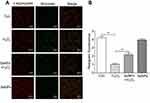
SeNPs weakens the depolarization of MMP of intestinal epithelial cells
In order to explore how SeNPs protect mitochondria against H2O2-induced oxidative damage, firstly, levels of MMP in each experimental group were determined using JC-1. Compared with the normal control, MMP in H2O2-induced oxidative damage model group was significantly decreased. However, pretreatment with SeNPs significantly weakened the decrease of MMP caused by H2O2 (Figure 5A and B).
Effect of SeNPs on Nrf2 activation and its downstream genes
In the current study, we investigated the relationship of the protective effect of SeNPs on intestinal barrier dysfunction caused by oxidative stress with Nrf2 signaling pathway due to the crucial role of Nfr2 in redox homeostasis. Results showed that SeNPs activated Nrf2 signaling and significantly improved HO-1 and NQO-1 protein levels compared with the H2O2 model group (Figure 6A and B).
Nrf2 inhibitor abolishes the antioxidant effects of SeNPs
Results showed that 5 μM ML385 as Nrf2 inhibitor did not affect cell viability (Figure 6C). However, the expression levels of Nrf2 were significantly downregulated in NCM460 cells treated with 5 μM ML385. Therefore, 5 μM was chosen as the concentration of ML385 interference experiment. SeNPs pretreatment activated Nrf2 signaling compared with the normal and oxidative stress groups (Figure 6D). Moreover, inhibition of Nrf2-signaling pathway abolished the regulatory effect of SeNPs on ROS overproduction caused by H2O2 (Figure 6E).
Discussion
Oxidative stress contributes to many pathological conditions and diseases.23 ROS, as highly reactive molecules, can damage cell structures and alter cell functions. Mitochondria are the key organelles for energy metabolism and ROS production.24 The plasticity of mitochondrial structure and function is a crucial feature for maintaining cellular homeostasis. Mitochondrial structure damage can affect mitochondrial function, and result in ROS accumulation and activation of inflammatory bodies, which may eventually lead to intestinal inflammation. IBD patients and experimental colitis mouse model exhibited mitochondrial stress and mitochondrial dysfunction in their intestinal epithelial cells.25 Previous studies indicated that increase of paracellular permeability was observed in dinitrophenol-induced uncouplers of oxidative phosphorylation of intestinal epithelial cell model, characterized by lower TER.26,27 mtDNA encodes respiratory chain complexes Ⅰ, Ⅲ, Ⅳ, and Ⅴ. In addition, mitochondria possess the ability to modulate crucial cellular functions including ROS production and stress signal transduction.28,29 In the current study, we found that H2O2-induced oxidative stress leaded to the drastic production of ROS, the depletion of ATP, and the reduction of MMP and mtDNA in normal human colon mucosal epithelial cells. These results indicated that H2O2 caused mitochondrial damage and dysfunction. Conversely, pretreatment with SeNPs alleviated these adverse effects. Moreover, SeNPs ameliorated H2O2-induced increase in FITC-dextran fluxes across NCM460 cells and H2O2-induced decrease of TER. These results indicate that SeNPs synthesized by L. casei ATCC 393 exhibit therapeutic potential against oxidative stress and preserve intestinal epithelial cell survival, mitochondrial function, and further maintain intestinal epithelial barrier integrity.
Antioxidant supplementation can effectively regulate the redox state of the body and maintain cellular homeostasis. Se is a much more potent antioxidant than vitamins E, C, and A.30 Se compounds protect mtDNA as well as telomere length and function.31 However, traditional Se supplements usually have relatively low bioavailability and high toxicity. Therefore, it is urgent to explore an innovative system for transforming carriers of Se.11 The effect of nanoscale Se on alleviating oxidative stress is well-known. Moreover, there is no significant medical side effect as a therapeutic agent.32,33 Therefore, Nano-Se has the potential to be applied in biomedical fields. The advantage of SeNPs in a zero oxidation state is that they possess low toxicity, high bioavailability and biocompatibility compared to other oxidation states.34 Various microorganisms such as Bacillus lichenifrrmis,35 Lactobacillus acidophillus,36 and Zooglea ramigera37 are able to convert toxic Se+IV and (or) Se+VI into nontoxic Se with the form of SeNPs. We have confirmed that probiotics L. casei ATCC 393 could synthesize SeNPs. Moreover, biogenic SeNPs by L. casei ATCC 393 possess strong antioxidant activity and low cytotoxicity compared with selenomethionine and sodium selenite, and can effectively protect human and porcine intestinal epithelial cells against H2O2- or Diquat-induced injury.20 Currently, biogenic SeNPs by L. casei ATCC 393 ameliorated H2O2-induced mithochondrial dysfunction in normal human colon mucosal epithelial cells. Nanoparticles usually pass through the intestinal epithelium in paracellular or transcellular pathway.38 Intestinal epithelial cells possess the ability to transfer mineral elements NPs. Endocytosis is an important start of transcellular transport. Internalization of NPs is a process of energy-dependence. The particle size of SeNPs is closely related to their biological activities.39 The absorption of NPs with a size of 100 nm in gastrointestinal tract is 15–250 times higher than that of large NPs.40 The size of biogenic SeNPs applied in the study was 50~80 nm.20
Exposure to acute or chronic oxidative stress can lead to cell damage, and impairs the normal physiological function of various organs, resulting in a variety of diseases. However, cells can protect themselves from serious damage by activating systems that scavenge free radicals to withstand oxidative stress. Nrf2-Keap1 is an important scavenger system-related signaling pathway that mediates a protective effect on cells challenged by oxidative stress.41,42 Nrf2 protects gastrointestinal cells from oxidative stress and subsequent inflammation through several mechanisms: 1) up-regulation of antioxidant and xenobiotic enzymes; 2) amelioration of inflammation by downregulation of NFκB; 3) stimulation of ABC transporters and MRP2.43 In homeostatic conditions, Nrf2 is not functionally activated, and exits in the form of Nrf2-Keap1 complex. However, when cells are subjected to oxidative stress, Keap1 is degraded from Nrf2-Keap1 complex, and then Nrf2 is released and transferred to the nucleus, and then combines with antioxidant response element, thereby regulating the expression of antioxidant-related genes and finally exerting its antioxidant defense function. Biogenic SeNPs protect cells against oxidative stress via activation of Nrf2 signaling, and up-regulation of antioxidant enzymes.44 Our results are consistent with the previously mentioned study. Up-regulation of Nrf2 and its downstream target genes was observed in cells treated with SeNPs under H2O2-induced oxidative stress. Those results demonstrated that Nrf2-mediated signaling pathway was closely related to cellular ROS production and the regulatory effect of biogenic SeNPs on intestinal epithelial barrier dysfunction caused by oxidative stress.
Conclusion
In summary, biogenic SeNPs synthesized by L. casei ATCC 393 exhibited considerably strong antioxidative activity to alleviate oxidative stress-caused intestinal epithelial barrier dysfunction. Moreover, as summarized in Figure 7, the potential mechanism of antioxidant properties may be associated with the regulatory effect of SeNPs on mitochondrial function via Nrf2-signaling pathway. These results suggest that SeNPs synthesized by probiotics are alternative supplements of Se for prevention and treatment of oxidative stress-related intestinal disorders. Moreover, the present study enlightens us that mitochondria-targeting SeNPs may be applied to regulate intestinal barrier dysfunction so as to avoid intestinal diseases in humans challenged by oxidative stress.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (no 31672435), Seed Foundation of Innovation and Creation for Graduate Students in Northwestern Polytechnical University (no ZZ2018046), and the Key Research and Development Program of Shaanxi Province (no 2018NY-102).
Author contributions
All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Gu WY, Wu CT, Chen JZ, Xiao Y. Nanotechnology in the targeted drug delivery for bone diseases and bone regeneration. Int J Nanomedicine. 2013;8:2305–2317. doi:10.2147/IJN.S44393
2. Pugazhendhi A, Shobana S, Nguyen DD, et al. Application of nanotechnology (nanoparticles) in dark fermentative hydrogen production. Int J Hydrogen Energy. 2019;44:1431–1440. doi:10.1016/j.ijhydene.2018.11.114
3. Saratale RG, Saratale GD, Shin HS, et al. New insights on the green synthesis of metallic nanoparticles using plant and waste biomaterials: current knowledge, their agricultural and environmental applications. Environ Sci Pollut Res. 2018;25:10164–10183. doi:10.1007/s11356-017-9912-6
4. Kumar PKSM, Ponnusamy VK, Deepthi KR, et al. Controlled synthesis of Pt nanoparticle supported TiO2 nanorods as efficient and stable electrocatalysts for the oxygen reduction reaction. J Mater Chem A. 2018;6:23435–23444. doi:10.1039/C8TA07380E
5. Shah M, Fawcett D, Sharma S, Tripathy SK, Poinern GEJ. Green synthesis of metallic nanoparticles via biological entities. Materials. 2015;8:7278–7308. doi:10.3390/ma8115377
6. Wadhwani SA, Shedbalkar UU, Singh R, Chopade BA. Biogenic selenium nanoparticles: current status and future prospects. Appl Microbiol Biotechnol. 2016;100:2555–2566. doi:10.1007/s00253-016-7300-7
7. Sisubalan N, Sri Ramkumar VS, Pugazhendhi A, et al. ROS-mediated cytotoxic activity of ZnO and CeO2 nanoparticles systhesized using the Rubia cordifolia L.leaf extract on MG-63 human osteosaarcoma cell lines. Environ Sci Pollut Res. 2018;25:10482–10492. doi:10.1007/s11356-017-0003-5
8. Suganthy N, Ramkumar VS, Pugazhendhi A, Benelli G, Archunan G. Biogenic synthesis of gold nanoparticles from Terminalia arjuna bark extract: assessment of safety aspects and neuroprotective potential via antioxidant, anticholinesterase and antiamyloidogenic effects. Environ Sci Pollut Res. 2018;25:10418–10433. doi:10.1007/s11356-017-9789-4
9. Pugazhendhi A, Prabhu R, Muruganantham K, Shanmuganathan R, Natarajan S. Anticancer, antimicrobial and photocatalytic activities of green synthesized magnesium oxide nanoparticles (MgONPs) using aqueous extract of Sargassum wightii. J Photochem Photobiol B. 2019;190:86–97. doi:10.1016/j.jphotobiol.2018.11.014
10. Vasantharaj S, Sathiyavimal S, Senthikumar P, LewisOscar F, Pugazhendhi A. Biosynthesis of iron oxide nanoparticles using leaf extract of Ruellia tuberosa: antimicrobial properties and their applications in photocatalytic degradation. J Photochem Photobiol B. 2019;192:74–82. doi:10.1016/j.jphotobiol.2018.12.025
11. Hosnedlova B, Kepinska M, Skalickova S, et al. Nano-selenium and its nanomedicine applications: a critical review. Int J Nanomedicine. 2018;13:2107–2128. doi:10.2147/IJN.S157541
12. Skalickova S, Milosavljevic V, Cihalova K, Horky P, Richtera L, Adam V. Selenium nanoparticles as a nutritional supplement. Nutrition. 2017;33:83–90. doi:10.1016/j.nut.2016.05.001
13. Anand SK, Singh J, Gaba A, Tikoo SK. Effect of bovine adenovirus 3 on mitochondria. Vet Res. 2014;45:45. doi:10.1186/1297-9716-45-45
14. Chan DC. Mitochondria: dynamic organelles in disease, aging, and development. Cell. 2006;125(7):1241–1252. doi:10.1016/j.cell.2006.06.010
15. Wang A, Keita AV, Phan V, et al. Targeting mitochrondria-derived reactive oxygen species to reduce epithelial barrier dysfunction and colitis. Am J Pathol. 2014;184(9):2516–2527. doi:10.1016/j.ajpath.2014.05.019
16. Niklison-Chirou MV, Dupuy F, Pena LB, et al. Microcin J25 triggers cytochrome c release through irreversible damage of mitochondrial protein and lipids. Int J Biochem Cell B. 2010;42(2):273–281. doi:10.1016/j.biocel.2009.11.002
17. Handa O, Majima A, Onozawa Y, et al. The role of mitochondria-derived reactive oxygen species in the pathogenesis of non-steroidal anti-inflammatory drug-induced small intestinal injury. Free Radic Res. 2014;48:1095–1099. doi:10.3109/10715762.2014.928411
18. Wu B, Iwakiri R, Ootani A, Fujise T, Tsunada S, Fujimoto K. Platelet activating factor promotes mucosal apoptosis via FasL mediating caspase-9 active pathway in rat small intestine after ischemia-reperfusion. Faseb J. 2003;17(9):1156–1158. doi:10.1096/fj.02-0499fje
19. Urbankova L, Horky P, Skladanka J, et al. Antioxidant status of rats’ blood and liver affected by sodium selenite and selenium. Peer J. 2018;6:e4862. doi:10.7717/peerj.4862
20. Xu C, Qiao L, Guo Y, Ma L, Cheng Y. Preparation, characteristics and antioxidant activity of polysaccharides and proteins-capped selenium nanoparticles synthesized by Lactobacillus casei ATCC 393. Carbohyd Polym. 2018;195:576–585. doi:10.1016/j.carbpol.2018.04.110
21. Huang Q, Xu W, Bai KW, et al. Protective effects of leucin on redox status and mitochondrial-related gene abundance in the jejunum of intrauterine growth-retarded piglets during early weaning period. Arch Anim Nutr. 2017;71(2):93–107. doi:10.1080/1745039X.2017.1279712
22. Zhang J, Tong W, Sun H, et al. Nrf2-mediated neuroprotection by MANF against 6-OHDA-induced cell damage via PI3K/AKT/GSK3beta pathway. Exp Gerontol. 2017;100:77–86. doi:10.1016/j.exger.2017.10.021
23. Birben E, Sahiner UM, Sackesen C, Erzurum S, Kalayci O. Oxidative stress and antioxidant defense. World Allergy Organ J. 2012;5(1):9–19. doi:10.1097/WOX.0b013e3182439613
24. Hock MB, Kralli A. Transcriptional control of mitochondrial biogenesis and function. Annu Rev Physiol. 2009;71:177–203. doi:10.1146/annurev.physiol.010908.163119
25. Novak EA, Mollen KP. Mitochondrial dysfunction in inflammatory bowel disease. Front Cell Dev Biol. 2015;3:62. doi:10.3389/fcell.2015.00062
26. Nazli A, Yang PC, Jury J, et al. Epithelia under metabolic stress perceive commensal bacteria as a threat. Am J Pathol. 2004;164:947–957. doi:10.1016/S0002-9440(10)63131-8
27. Nazli A, Wang A, Steen O, et al. Enterocyte cytoskeletal changes are crucial for enhanced translocation of nonpathogenic Escherichia coli across metabolically stressed gut epithelium. Infect Immun. 2006;74:192–201. doi:10.1128/IAI.74.1.192-201.2006
28. Tait SWG, Green DR. Mitochondria and cell signaling. J Cell Sci. 2012;125(4):807–815. doi:10.1242/jcs.099234
29. Galluzzi L, Kepp O, Kroemer G. Mitochondria: master regulators of danger signaling. Nat Rev Mol Cell Biol. 2012;13:780–788. doi:10.1038/nrm3479
30. Baraboi VA, Shestakova EN. Selenium: the biological role and antioxidant activity. Ukr Biokhim Zh (1999). 2004;76(1):23–32.
31. Ferguson LR, Karunasinghe N, Zhu S, et al. Selenium and it’s role in the maintenance of genomic stability. Mutat Res. 2004;33(1):100–110.
32. Sadeghian S, Kojouri GA, Mohebbi A. Nanoparticles of selenium as species with stronger physiological effects in sheep in comparison with sodium selenite. Biol Trace Elem Res. 2012;146(3):302–308. doi:10.1007/s12011-011-9266-8
33. Kojouri GA, Sharifi S. Preventing effects of nano-selenium particles on serum concentration of blood urea nitrogen, creatinine, and total protein during intense exercise in donkey. J Equine Vet Sci. 2013;33(8):597–600. doi:10.1016/j.jevs.2012.09.008
34. Wang H, Zhang J, Yu H. Elemental selenium at nano size possesses lower toxicity without compromising the fundamental effect on sele-noenzymes: comparison with selenomethionine in mice. Free Radic Biol Med. 2007;42(10):1524–1533. doi:10.1016/j.freeradbiomed.2006.10.038
35. Sonkusre P, Singh Cameotra S. Biogenic selenium nanoparticles inhibit Staphylococcus aureus adherence on different surfaces. Colloids Surf B Biointerfaces. 2015;136:1051–1057. doi:10.1016/j.colsurfb.2015.10.052
36. Srivastava N, Mukhopadhyay M. Biosynthesis and structural characterization of selenium nanoparticles mediated by zooglea ramigera. Powder Technol. 2013;244:26–29. doi:10.1016/j.powtec.2013.03.050
37. Yang J, Huang K, Qin S, Wu X, Zhao Z, Chen F. Antibacterial action of selenium-enriched probiotics against pathogenic Escherichia coli. Dig Dis Sci. 2009;54:246–254. doi:10.1007/s10620-008-0361-4
38. des Rieux A, Fievez V, Garinot M, Schneider Y-J, Préat V. Nano-particles as potential oral delivery systems of proteins and vaccines: a mechanistic approach. J Control Release. 2006;116(1):1–27. doi:10.1016/j.jconrel.2006.08.013
39. Torres SK, Campos VL, León CG, et al. Biosynthesis of selenium nanoparticles by pantoea agglomerans and their antioxidant activity. J Nanopart Res. 2012;14(11):1236. doi:10.1007/s11051-012-1236-3
40. Bergin IL, Witzmann FA. Nanoparticle toxicity by the gastrointes-tinal route: evidence and knowledge gaps. Int J Biomed Nanosci Nanotechnol. 2013;3(1–2):163–210. doi:10.1504/IJBNN.2013.054515
41. Suzuki T, Yamamoto M. Stress-sensing mechanisms and the physiological roles of the Keap1-Nrf2 system during cellular stress. J Biol Chem. 2017;292:16817–16824. doi:10.1074/jbc.R117.800169
42. Itoh K, Tong KI, Yamamoto M. Molecular mechanism activating Nrf2-Keap1 pathway in regulation of adaptive response to electrophiles. Free Radic Biol Med. 2004;36:1208–1213. doi:10.1016/j.freeradbiomed.2004.02.075
43. Yanaka A. Role of Nrf2 in protection of the gastrointestinal tract against oxidative stress. J Clin Biochem Nutr. 2018;63(1):18–25.
44. Song D, Cheng YY, Li X, et al. Biogenic nanoselenium particles effectively attenuate oxidative stress-induced intestinal epithelial barrier injury by activating the Nrf2 antioxidant pathway. ACS Appl Mater Interfaces. 2017;9(17):14724–14740. doi:10.1021/acsami.7b03377
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.