Back to Journals » Clinical Interventions in Aging » Volume 12
Association between butyrylcholinesterase K variant and mild cognitive impairment in the Thai community-dwelling patients
Authors Pongthanaracht N, Yanarojana S, Pinthong D, Unchern S, Thithapandha A, Assantachai P, Supavilai P
Received 17 March 2017
Accepted for publication 11 April 2017
Published 26 May 2017 Volume 2017:12 Pages 897—901
DOI https://doi.org/10.2147/CIA.S137264
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Richard Walker
Natsalil Pongthanaracht,1 Somchai Yanarojana,1 Darawan Pinthong,1 Supeenun Unchern,1 Amnuay Thithapandha,1 Prasert Assantachai,2 Porntip Supavilai1
1Department of Pharmacology, Faculty of Science, 2Department of Preventive and Social Medicine, Faculty of Medicine Siriraj Hospital, Mahidol University, Bangkok, Thailand
Objective: To study the association of the butyrylcholinesterase K variant (BChE-K) and the plasma BChE activity with mild cognitive impairment (MCI) in Thai community-dwelling patients.
Methods: One hundred patients diagnosed with MCI and 100 control subjects were recruited from the community-dwelling setting in Bangkok, Thailand. The genotype and allele distributions of the BChE-K were determined by polymerase chain reaction and subsequent DNA sequencing. The BChE activity was measured in plasma according to the Ellman’s method.
Results: The BChE-K allele frequencies in the Thai community-dwelling patients were in accordance with other ethnics. The BChE-K allele frequency in the control subjects (12%) was higher than that of MCI patients (5.5%), suggesting a protective role of BChE-K for MCI in the Thai community-dwelling patients. The BChE-K homozygotes were significantly associated with lower BChE activity.
Conclusion: Our results suggested that the BChE-K may be implicated as a protective factor for MCI in the Thai community-dwelling patients, although a further study with a large sample size is warranted to confirm this.
Keywords: butyrylcholinesterase K variant, butyrylcholinesterase activity, mild cognitive impairment, Thai community-dwelling patients
Introduction
Mild cognitive impairment (MCI) is a common problem in the elderly and a part of a cognitive continuum between normal aging and Alzheimer’s disease (AD).1 As the number of elderly people, as well as the prevalence of AD, is increasing rapidly due to the demographic changes in the country, identifying those who are likely to have clinical manifestation of AD in the earliest stage would benefit the patients, family and society as a whole.2,3 AD is the most common age-dependent neurodegenerative disorder noticeable by cognitive and memory deterioration, a variety of neuropsychiatric symptoms, behavioral disturbances and impairment of activities of daily living (ADL).4 In hippocampus and temporal cortex of AD patients, an increase in butyrylcholinesterase (BChE) and a reduction in acetylcholinesterase activity are observed.5 It is suggested that the increased level of BChE is associated with cognitive decline in dementias. Furthermore, BChE is associated with amyloid plaques and neurofibrillary tangles in AD.6,7 One of the most important challenges in research on cognitive impairment in the elderly is to identify genes that predispose to MCI and could be used to predict which subjects will progress to dementia. Recently, genetic variation is used as biomarkers for MCI and progression of MCI leading to dementia.8
One of the candidates that has been identified to date is the K variant of BChE (BChE-K). The K variant, a mutation at codon 539, leads to the replacement of an alanine by a threonine residue (A539T) and is associated with 30% reduction in enzyme activity.9 The BChE-K has an essential role in AD progression.10 The BChE-K is observed in various ethnic populations with homozygote frequencies between 2% and 32%.11–23 AD has a multifactorial pathogenesis and its challenge is the early diagnosis. Thus, genotyping BChE-K and measuring BChE activity in subjects with MCI will help identify potential biomarkers for MCI in easily accessible peripheral blood samples. The purpose of this study is to investigate the association of the BChE-K and plasma BChE activity with MCI in Thai community-dwelling patients.
Methods
Participants were volunteers from the community-dwelling visit in the “Health Promotion and Common Disease Prevention in the Thai Elderly Project” being run in a community-dwelling setting in Bangkok, Thailand, as described by Muangpaisan et al.24 The study protocol was approved by the Mahidol University Ethics Committee 145/2004, and a written informed consent was obtained from each subject prior to their participation. Inclusion criteria were patients aged >50 years who had a subjective memory complaint and intact instrumental ADL (IADL) and were willing to participate. They had a Thai Mental State Examination (TMSE) score of 24 or more and showed no abnormality of judgment and insight during the structured interview.25 Exclusion criteria were patients who had dementia and depression diagnosed by the 4th edition of the Diagnostic and Statistical Manual criteria.26 They had a TMSE score of <24, illiteracy, communication problems, impairment in insight and judgment. The diagnosis of MCI was made according to the Petersen criteria: 1) impairment in one or more measures of memory, 2) clinician’s judgment based on a structured interview by using the Clinical Dementia Rating Scale score of 0.5, 3) the Global Deterioration Scale score of 3, but with normal performances in other cognitive domains and 4) no impairment in IADL.27–29 The control subjects had TMSE score of at least 24, no evidence of dementia or memory impairment on assessment, no diagnosis of major medical, neurological or psychiatric illness, no history of head injury and substance abuse and no impairment in ADL.
Genomic DNA was extracted from peripheral blood cells using a QIAamp DNA Blood Mini Kit according to the manufacturer’s instructions. BChE-K genotyping was performed as previously described.30 The BChE activity of plasma samples were measured according to the Ellman’s colorimetric method,31 with 5,5′dithio-bis 2 nitrobenzoic acid (DTNB) and butyrylthiocholine iodide as substrate.
The observed genotype and allele frequencies of BChE-K between MCI patients and controls were compared with the predicted values using chi-square (χ2) test. Odds ratio (OR) and confidence interval (CI) calculations were performed based on a crosstabs module included in the SPSS statistical software package version 17.0 (SPSS Inc., Chicago, IL, USA). Data were expressed as mean ± standard deviation. Analysis of variance was used to compare the differences among the groups and data were expressed as percentage. Two-tailed Student’s t-test was used to compare the differences between the two groups. P<0.05 was considered statistically significant.
Results
The demographic characteristics of the Thai community-dwelling patients are shown in Table 1. In this study, females are predominant; however, MCI patients and controls showed no statistical significant differences in gender, age, TMSE scores. Furthermore, there is no statistically significant difference of plasma BChE activity between the MCI patients and controls (P=0.4738).
The BChE-K genotype and allele frequencies for the Thai community-dwelling patients are summarized in Table 2. Distribution of the BChE-K genotype in MCI patients (χ2=1.8, degrees of freedom [df]=1, P=0.1796) and the controls (χ2=2.8, df=1, P=0.5959) were in Hardy–Weinberg equilibrium. It is noteworthy that 1% MCI patients and 2% controls were homozygous for the K allele (K/K). The BChE-K allelic frequency was found to be significantly different in MCI patients when compared with controls; 5.5% and 12%, respectively (χ2=5.357, df=1, P=0.02). The OR as an estimate of relative risk for MCI was 0.427 (95% CI=0.203–0.897, P<0.05), which indicated that BChE-K is a protective factor for MCI in the Thai community-dwelling patients. Among 200 subjects, included in this study were 141 females (controls =67, MCI patients =74) and 59 males (controls =33, MCI patients =26). Although females are predominant in the Thai community-dwelling female, both MCI and controls did not exhibit homozygous for the BChE-K. Homozygous for the BChE-K was found in MCI male patients (3.9%) and male controls (6.1%).
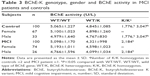
The values of plasma BChE activity did not exhibit significant difference between MCI patients and controls (Table 1). To determine whether there were genotype and gender differences in the influence of BChE activity, we stratified BChE activity according to genotype and gender. The plasma BChE activities in each genotype and gender are given in Table 3. The significant reduction in BChE activities was observed in the BChE-K homozygotes (K/K) of both MCI patients (55%) and controls (39% and 65%). However, comparison of BChE-K heterozygotes (wild type [WT]/K) with homozygotes for WT of BCh allele (WT/WT) with respect to their BChE activity and gender yielded no statistically significant difference. A reduction of BChE activity was observed only in males who are carrier homozygous for BChE-K.
Discussion
In the present study, genotype and allele distributions of the BChE-K and the plasma BChE activity were investigated in Thai community-dwelling patients. The distributions of the genotype and allele frequencies of the BChE-K gene in both MCI patients and controls were in Hardy–Weinberg equilibrium. These results regarding the K allele and genotype distribution are in accordance with previous reports.11,12 In relation to the BChE polymorphism, our controls (12%) showed a frequency of the K allele and genotype distribution that is similar to British,13 Chinese,14 Finnish,15 Japanese,16 Korean,17 Northern Irish,18 Northern Spanish11 and the US19 populations (9%–18%). On the contrary, patients from Australia,20 the Canary Islands,12 Turkey21 and the USA22 have shown a higher K allele frequency (23%–32%), while patients from Brazil23 have a lower reported K allele frequency (2%). The K allele frequency shows a considerable ethnic variation.
The distribution of the BChE-K allelic frequency in the Thai community-dwelling patients was found to differ significantly in MCI patients (5.5%) compared with control (12%), suggesting that the BChE -K might be associated with a protective role for cognitive decline in MCI patients. So far, there has been no report on the association between the BChE-K and MCI in Thai population. However, the association of the BChE-K with AD is controversial. Some researchers18,32–35 found association between the BChE -K and AD, whereas others17,36 found no association. Alvarez-Arcaya et al11 and Bizzarro et al37 suggested a protective role of BChE -K. The correlation of BChE-K with MCI as well as AD is different between different ethnic groups. Thus, BChE-K may be either a risk or a protective factor in AD.38 Furthermore, the presence of the ε4 allele of apolipoprotein E (ApoE-ε4) is associated with an increased Aβ deposition and neurofibrillary tangle formation in the brain before the onset of dementia. The ApoE-ε4 allele is implicated as a risk factor associated with rate of cognitive decline in AD.35 Several studies have demonstrated a positive association between ApoE-ε4, BChE-K allele and AD,32–35 whereas others have shown no relationship between the presence of these genes and the disease.15,17,18 This discrepancy may also reflect different distribution of BChE-K variant in different ethnic populations. The association of BChE-K and ApoE-ε4 allele in Thai community-dwelling patients warrants further investigation.
In addition, the progression from MCI to AD is influenced by the BChE genotype and gender.39,40 In 2013, Lane and He41 reviewed the influence of BChE genotype and gender in AD. Male and female gender may differentially affect the progression of cognitive impairment, neurodegenerative processes and response to treatment. In Thai community-dwelling, females do not have homozygous for the BChE-K (Table 2), suggesting that females have higher possibility to develop MCI or AD than males. In agreement with AD epidemiology, females have higher risk than males of developing MCI.2 In our population, the protective effect of BChE-K was restricted to males. In MCI patients and controls, the BChE activity of the heterozygous for the BChE-K and the homozygous WT are not statistically different between males and females (Table 3). The homozygous for the BChE-K was associated with low BChE activity in males.
Genotyping for BChE-K is probably useful to predict which AD patients would favorably respond to BChE inhibitor. Thus, BChE inhibitor should not be prescribed to male AD patients who are homozygous for the BChE-K since they already have low BChE activity. However, Thai female AD patients are foreseeable to respond to BChE inhibitor since they have normal BChE activity.
Conclusion
The prevalence of MCI in the Thai community-dwelling patients was higher in females than males. The BChE-K may be implicated as a protective factor for MCI, although further study with a large sample size is warranted to confirm this.
Acknowledgments
This research work was supported by grants from the National Research Council of Thailand and Mahidol University. NP was supported by the Commission on Higher Education Staff Development Project of Thailand. Special thanks also go to all subjects who participated in the present study.
Author contributions
DP, NP, PA, PS, SU and SY participated in design experiments and recruitment of subjects and interpreted data. NP conducted genotyping, enzyme activity experiments and statistical analysis. AT, NP and PS were involved in the preparation of the manuscript. All authors read and approved the final manuscript, contributed toward data analysis, drafting and critically revising the paper and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
Gauthier S, Reisberg B, Zaudig M, et al. Mild cognitive impairment. Lancet. 2006;367:1262–1270. | ||
Alzheimer’s Association. 2016 Alzheimer’s disease facts and figures. Alzheimers Dement. 2016;12(4):459–509. | ||
National Statistical Office (NSO). 2014 Survey of Older Persons in Thailand. Bangkok: National Statistical Office; 2014. | ||
Querfurth HW, LaFerla FM. Alzheimer’s disease. N Eng J Med. 2010;362:329–344. | ||
Perry E, McKeith I, Ballard C. Butyrylcholinesterase and progression of cognitive deficits in dementia with Lewy bodies. Neurology.2003;60:1852–1853. | ||
Mesulam MM, Geula C. Butyrylcholinesterase reactivity differentiates the amyloid plaques of aging from those of dementia. Ann Neurol. 1994;36:722–727. | ||
Gómez-Ramos P, Bouras C, Morán MA. Ultrastructural localization of butyrylcholinesterase on neurofibrillary degeneration sites in the brains of aged and Alzheimer’s disease patients. Brain Res. 1994;640:17–24. | ||
Reitz C, Mayeux R. Use of genetic variation as biomarkers for mild cognitive impairment and progression of mild cognitive impairment to dementia. J Alzheimers Dis. 2010;19:229–251. | ||
Bartels CF, Jensen FS, Lockridge O, et al. DNA mutation associated with the human butyrylcholinesterase K-variant and its linkage to the atypical variant mutation and other polymorphic sites. Am J Hum Genet. 1992;50(5):1086–1103. | ||
Holmes C, Ballard C, Lehmann D, et al. Rate of progression of cognitive decline in Alzheimer’s disease: effect of butyrylcholinesterase K gene variation. J Neurol Neurosurg Psychiatr. 2005;76(5):640–643. | ||
Alvarez-Arcaya A, Combarros O, Llorca J, et al. The butyrylcholinesterase K variant is a protective factor for sporadic Alzheimer’s disease in women. Acta Neurol Scand. 2000;102(6):350–353. | ||
Deniz-Naranjo MC, Munoz-Fernandez C, Alemany-Rodriguez MJ, et al. Butyrylcholinesterase, ApoE and Alzheimer’s disease in a population from the Canary Islands (Spain). Neurosci Lett. 2007;427(1):34–38. | ||
Evans RT, Wardell J. On the identification and frequency of the J and K cholinesterase phenotypes in a Caucasian population. J Med Genet. 1984;21(2):99–102. | ||
Lee DW, Liu HC, Liu TY, Chi CW, Hong CJ. No association between butyrylcholinesterase K-variant and Alzheimer’s disease in Chinese. Am J Med Genet. 2000;96(2):167–169. | ||
Hiltunen M, Mannermaa A, Helisalmi S, et al. Butyrylcholinesterase K variant andapolipoprotein E4 genes do not act in synergy in Finnish late-onset Alzheimer’s disease patients. Neurosci Lett. 1998;250(1):69–71. | ||
Yamamoto Y, Yasuda M, Mori E, Maeda K. Failure to confirm a synergistic effect between the K-variant of the butyrylcholinesterase gene and the epsilon 4 allele of the apolipoprotein gene in Japanese patients with Alzheimer’s disease. J Neurol Neurosurg Psychiatry. 1999;67(1):94–96. | ||
Ki CS, Na DL, Kim JW, Kim HJ, Kim DK, Yoon BK. No association between the genes for butyrylcholinesterase K variant and apolipoprotein E4 in late-onset Alzheimer’s disease. Am J Med Genet. 1999;88:113–115. | ||
McIlroy SP, Crawford VL, Dynan KB, et al. Butyrylcholinesterase K variant is genetically associated with late onset Alzheimer’s disease in Northern Ireland. J Med Genet. 2000;37(3):182–185. | ||
Bartels CF, Jensen FS, Lockridge O, et al. DNA mutation associated with the human butyrylcholinesterase K-variant and its linkage to the atypical variant mutation and other polymorphic sites. Am J Hum Genet. 1992;50(5):1086–1103. | ||
Panegyres PK, Mamotte CDS, Vasikaran SD, Wilton S, Fabian V, Kakulas BA. Butyrylcholinesterase K variant and Alzheimer’s disease. J Neurol. 1999;246(5):369–370. | ||
Babaoglu MO, Ocal T, Bayar B, Kayaalp SO, Bozkurt A. Frequency and enzyme activity of the butyrylcholinesterase K-variant in a Turkish population. Eur J Clin Pharmacol. 2004;59(12):875–877. | ||
Grubbera JM, Saundersa AM, Crane-Gatherum AR, et al. Analysis of association between Alzheimer disease and the K variant of butyrylcholinesterase (BCHE-K). Neurosci Lett. 1999;269(2):115–119. | ||
Alcantara VM, Chautard-Freire-Maria EA, Picheth G, Vieira MM. Frequency of the CHE1*Kallele of serum cholinesterase in a sample from southern Brazil. Hum Hered. 1999;40(6):386–390. | ||
Muangpaisan W, Assantachai P, Intalapaporn S, Pisansalakij D. Quality of life of the community-based patients with mild cognitive impairment. Geriatr Gerontol Int. 2008;8(2):80–85. | ||
Train the Brain Forum Committee. Thai Mental State Examination (TMSE). Siriraj Hosp Gaz. 1993;45:661–674. | ||
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 4th ed. Washington, DC: American Psychiatric Association; 1994. | ||
Petersen RC, Doody R, Kurz A, et al. Current concepts in mild cognitive impairment. Arch Neurol. 2001;58(12):1985–1992. | ||
Hughes CP, Berg L, Danziger WL, Coben LA, Martin RL. A new clinical scale for the staging of dementia. Br J Psychiatry. 1982;140:566–572. | ||
Reisberg B, Ferris SH, de Leon MJ, Crook T. The global deterioration scale for assessment of primary degenerative dementia. Am J Psychiatry. 1982;139(9):1136–1139. | ||
Jenese LR, Nielsen LR, Schwartz M. Detection of the plasma cholinesterase K variant by PCR using amplification-created restriction site. Hum Herd. 1996;46(1):26–31. | ||
Ellman GL, Courtney KD, Andres V Jr, Feather-Stone RM. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol. 1961;7:88–95. | ||
Lehmann DJ, Johnston C, Smith AD. Synergy between the genes for butyrylcholinesterase K variant and apolipoprotein E4 in late-onset confirmed Alzheimer’s disease. Hum Mol Genet. 1997;6(11):1933–1936. | ||
Tilley L, Morgan K, Grainger J, et al. Evaluation of polymorphisms in the presenilin-1 gene and the butyrylcholinesterase gene as risk factors in sporadic Alzheimer’s disease. Eur J Hum Genet. 1999;7(6):659–663. | ||
Wiebusch H, Poirier J, Sevigny P, Schappert K. Further evidence for a synergistic association between APOE ε4 and BCHE-K in confirmed Alzheimer’s disease. Hum Genet. 1999;104:158–163. | ||
Raygani AV, Zahrai M, Soltanzadeh A, Doosti M, Javadi E, Pourmotabbed T. Analysis of association between butyrylcholinesterase K variant and apolipoprotein E genotypes in Alzheimer’s disease. Neurosci Lett. 2004;371(2–3):142–146. | ||
Singleton AB, Smith G, Gibson AM, et al. No association between the K variant of the butyrylcholinesterase gene and pathologically confirmed Alzheimer’s disease. Hum Mol Genet. 1998;7(5):937–939. | ||
Bizzarro A, Guglielmi V, Lomastro R, et al. BChE K variant is decreased in Alzheimer’s disease not in fronto-temporal dementia. J Neural Transm (Vienna). 2010;117(3):377–383. | ||
Podoly E, Shalev DE, Shenhar-Tsarfaty S, et al. The butyrylcholinesterase K variant confers structurally derived risks for Alzheimer pathology. J Biol Chem. 2009;284(25):17170–17179. | ||
Holmes C, Ballard C, Lehman D, et al. Rate of progression of cognitive decline in Alzheimer’s disease: effect of butyrylcholinesterase K gene variation. J Neurol Neurosurg Psychiatry. 2005;76(5):640–643. | ||
Ferris S, Nordberg A, Soininen H, Darreh-Shori T, Lane R. Progression from mild cognitive impairment to Alzheimer’s disease: effects of sex, butyrylcholinesterase genotype, and rivastigmine treatment. Pharmacogenet Genomics. 2009;19(8):635–646. | ||
Lane RM, He Y. Butyrylcholinesterase genotype and gender influence Alzheimer’s disease phenotype. Alzheimers Dement. 2013;9(2):e17–e73. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.