Back to Journals » International Journal of Nephrology and Renovascular Disease » Volume 12
Frailty Screening in Chronic Kidney Disease: Current Perspectives
Authors Worthen G , Tennankore K
Received 28 August 2019
Accepted for publication 21 November 2019
Published 5 December 2019 Volume 2019:12 Pages 229—239
DOI https://doi.org/10.2147/IJNRD.S228956
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Pravin Singhal
George Worthen,1 Karthik Tennankore2
1Department of Medicine, Nova Scotia Health Authority, Halifax, NS, Canada; 2Division of Nephrology, Nova Scotia Health Authority, Halifax, NS, Canada
Correspondence: Karthik Tennankore
Division of Nephrology, QEII - Dickson Building, Suite 5090 Dickson Building, 5820 University Avenue, Halifax, NS B3H 2Y9, Canada
Tel +1 902-473-2099
Fax +1 902-473-2675
Email [email protected]
Abstract: Frailty has been defined as a state of increased vulnerability as a consequence of deficit accumulation. Frailty screening has not yet been widely implemented into routine nephrology care. Patients with chronic kidney disease (CKD) are at high risk of being frail, and frailty has been associated with worse outcomes in this population. Standard management of CKD, including initiation of renal replacement therapies, may have decreased benefit or potentially cause harm in the presence of frailty, and a variety of interventions for modifying frailty in the CKD population have been proposed. The optimal means of screening for frailty in patients with kidney disease remains unclear. This review highlights the value of frailty screening in CKD by summarizing the outcomes associated with frailty and exploring proposed changes to the management of frail patients with CKD. Finally, we will propose a framework for how to implement frailty screening into standard nephrology care.
Keywords: geriatrics, sarcopenia, transplant, dialysis, conservative management
Introduction
Frailty has been defined as a state of increased vulnerability to stressors as a consequence of degeneration in multiple systems. There remain two schools of thought with regard to frailty: one which sees frailty as a physical phenotype characterized by sarcopenia and another that views frailty as an accumulation of deficits across a variety of domains.1 Regardless of which definition of frailty is used, it has been shown in the general population to be predictive of adverse outcomes, including falls, hospitalization, decreased quality of life, and mortality.2 Frailty is highly prevalent in patients at all stages of kidney disease, with as many as two-thirds of patients with end-stage renal disease (ESRD) being identified as frail.3 As in the general population, patients with kidney disease who are frail have been shown to be at increased risk of negative outcomes.4 Indeed, frailty is a significant contributor to morbidity and mortality in patients with kidney disease. However, despite a growing body of evidence suggesting that frailty is associated with worse outcomes, frailty screening has not been universally incorporated into routine nephrology care. Questions remain around the utility of frailty screening – how exactly does frailty affect patients with chronic kidney disease (CKD)? If a patient is identified as frail, are there interventions that can modify this? How can knowing a patient is frail inform decisions around their management? Are there effective and easily implementable screening tools available? In this review, we will 1) attempt to address these concerns by presenting an overview of the effects of frailty on outcomes in patients with CKD, 2) explore proposed strategies for managing frail patients with CKD, 3) provide suggestions for future study, and 4) propose a framework for incorporating frailty screening into standard nephrology care.
Is Frailty Associated with Poor Outcomes for Patients with CKD?
Before considering its use as a screening tool, it is important to establish the effect of frailty on subsequent health outcomes for patients with CKD. In general, in order to justify screening, a condition must be highly prevalent, it must have a significant effect on outcomes, and when the condition is identified, there must be a meaningful change to management.5 Using these criteria, frailty is certainly worth screening for.
Patients with CKD are at high risk of being frail. The prevalence of frailty for the elderly general population (ie, those greater than 65 years of age) is around 11%,6 whereas it is as high as 43% in predialysis patients and as high as 73% in the dialysis population, regardless of the numerical age. This, combined with the observation that the prevalence of frailty increases as glomerular filtration rate (GFR) drops, suggests a link between CKD and accelerated biological aging.4
Frail patients with CKD do worse than their nonfrail counterparts. In predialysis CKD, frailty is associated with faster disease progression,7 worse quality of life,8 and an increased risk of death.9 In the dialysis population, frail patients are three times as likely to fall,10 are up to twice as likely to be admitted to hospital, and experience a high rate of hospital readmission.11 As in the prerenal replacement population, dialysis patients have an increased risk of mortality, with some previous studies demonstrating a 1-year relative hazard (HR) of 2.24 (95% CI 1.6–3.15)12 and a 3-year HR of 2.7 (95% CI 1.02–7.07).13 This risk worsens as frailty becomes more severe.14
Frailty has a large impact on the probability of transplantation. As frailty severity worsens, the likelihood of being transplanted decreases.14 Frail patients on the waitlist are more likely to die or be removed from the waitlist.15 After transplantation, frail patients are twice as likely to experience delayed graft function and are at increased risk of prolonged and repeated hospitalizations.16 Ultimately, frail patients experience significantly higher mortality post-transplant compared with those who are not frail.17
How Should Our Management Change to Better Serve Frail Patients?
Overview
Frailty is prevalent in patients with kidney disease, and being frail carries with it an increased risk of negative outcomes; however, screening for frailty is only worthwhile if it changes our management. Frail patients with CKD are a distinct population with a unique risk profile, and as such the management of their CKD is different than in fit patients. There are also potential therapeutic options for improving a patient’s frailty severity itself. Although frailty most commonly follows a downward trajectory, there is growing evidence in the general population to suggest that a patient’s frailty severity can be improved with intervention.18
CKD Patients
Impaired physical function, sarcopenia, and an increased risk of falls are hallmarks of the frailty syndrome. Throughout all stages of CKD, these factors may be accelerated. The interplay between frailty, aging, and CKD has been termed “senescent nephropathy” – a state characterized by a synergistic decline in physical and renal function, proposed to be caused by increased levels of inflammation associated with each condition.19 In addition to CKD itself, it is possible that the treatments offered to frail CKD patients may also accelerate declining health. Strict targets for blood pressure, glycemic control, and salt and protein intake, while proven to be beneficial in fit patients with CKD, may, in fact, accelerate the decline of frail patients.20 Frailty carries with it a similar risk of mortality and morbidity as CKD,9 and as such, initiation of treatment should include careful consideration of what impact the proposed therapy may have on the patient’s frailty status and severity. In addition, therapies with expected benefits in the longer term may not occur in a timeframe that is relevant for the frail and elderly patients and only add potential harms.
Given the interplay between frailty, aging, and CKD, therapies designed to modify frailty would be expected to slow the progression of kidney disease. As frail patients with CKD are uniquely vulnerable to geriatric complications (including falls and polypharmacy),10,21 they would benefit from a geriatric assessment, either incorporated directly into standard nephrology care or through a formal geriatric referral.22 This assessment would more clearly delineate the patient’s deficits, allowing more targeted and individualized therapy.
Another promising option for modifying frailty is exercise therapy. Exercise therapy has been shown in the general frail population to improve mobility, independence, quality of life, and bone mineral density and to decrease falls. Importantly, even a modest amount of exercise in severely frail patients has been shown to improve outcomes.23 Dialysis patients, in general, live a sedentary lifestyle,24 contributing to sarcopenia, dynapenia, and further physical and functional limitation. Moreover, reduced physical activity in elderly hemodialysis patients has been associated with an increased risk of mortality.25 Exercise interventions in the CKD population have shown a variety of benefits, including improved function, increased muscle mass,26 as well as reduced systemic inflammation.27 In particular, exercise has been shown to improve lower-extremity strength and gait speed;26 impairment in both are strongly associated with mortality.28 There have been trends toward improved survival with increased exercise in this population;25 however, this remains an area for further study. While it is expected that exercise would have a greater benefit in those whose major determinants of frailty severity are on the basis of functional impairment or reduced activity, the benefits of physical exercise may extend to other domains of frailty including cognition, mood, and social function.29
ESRD – General
The severity of a patient’s frailty should inform decisions about management as patients approach ESRD. An individualized approach to managing ESRD should be undertaken in frail patients, and their values and goals should be elicited. Dialysis and transplant represent significant stressors on a patient’s reserve, and therefore should only be expected to benefit patients who are adequately robust.30 As a patient’s frailty severity progresses, focus should shift to potentially modifying care to less invasive treatment options (ie, incremental dialysis or conservative management). The severity of a patient’s frailty should be expected to impact significantly on their experience of different renal replacement strategies.
Dialysis
While life sustaining, dialysis is associated with significant morbidity. Cramping, pruritus, intradialytic hypotension, access-site complications, and postdialysis fatigue are major symptoms that can all significantly have an impact on a patient’s quality of life.31 Although data are limited, frail patients appear to be more vulnerable to complications,32 likely owing to their diminished reserve.
Frail patients who are started on HD appear to do so at a higher GFR than their nonfrail counterparts. The reason for this is not clear: perhaps some frailty-associated symptoms are misinterpreted by the patient and provider as uremic symptoms, or perhaps by virtue of being frail, these patients are less tolerant of uremia and experience it at a higher GFR. Regardless, this is a worrisome trend, as there has been a suggestion that dialysis itself may accelerate the progression of frailty.33 The exact mechanism by which this occurs is not clear but appears to be multifactorial. Dialysis initiation is associated with decreased physical activity, which may accelerate the process of senescent nephropathy. Intradialytic hypotension has also been implicated, as it appears to have deleterious effects on cardiovascular and cerebral functioning,33 adding to deficit burden.
Residual kidney function (RKF) may also be impacted through HD. Frequent hemodialysis has been shown to accelerate the loss of RKF,34 and RKF has been shown to be protective against a variety of negative outcomes, including mortality.35 A loss in RKF may also lead to worsened intradialytic hypotension by increasing the amount of ultrafiltration that is needed through dialysis. These increased ultrafiltration targets may lead to further intradialytic hypotension, and further worsen cardiovascular, cerebral, and residual kidney function. In light of this, efforts to maintain RKF are likely to slow the progression of frailty, although to our knowledge this has yet to be demonstrated.
One proposed strategy for limiting frailty progression is that of incremental hemodialysis. Incremental hemodialysis refers to HD regimens that are shorter or less frequent than standard three times per week maintenance therapy and are increased over time to accommodate a further decline in RKF. This strategy has been shown to slow the loss of RKF in all comers.36 By starting frail patients on shorter duration and/or less frequent treatments, the physiologic stress of dialysis is decreased, leading to reduced postdialysis recovery time, less interference with social and family life, and ultimately improved quality of life,33 in addition to limiting intradialytic hypotension and subsequent organ dysfunction. While this may be one strategy, the benefits of this approach must be weighed against the risks of potentially inadequate dialytic clearance and ensuing complications.
Peritoneal dialysis (PD) may be a preferential option for frail patients. In frail patients who are unable to manage their PD independently, having a family member or caregiver assist them may be an alternative to standard in-centre HD. Importantly, there does not appear to be a difference in mortality between dialysis modalities in the elderly,37 and the risk of PD treatment failure is not increased in the context of functional impairment.38 Unfortunately, the limited data comparing HD and PD in the elderly found no significant difference in the quality of life, except with regard to treatment satisfaction, which favored PD.39 In addition, caregivers may experience impaired quality of life and burnout.40 However, for the right patient in the right clinical setting, assisted PD offers an option for home dialysis in the presence of frailty. Just as in hemodialysis, incremental prescriptions of PD may make the treatment more easily tolerated in the frail and elderly. There is evidence in the general population that incremental peritoneal dialysis protects RKF, reduces health-care costs, and improves patient's quality of life, without increasing mortality risk.41 As with incremental HD, more study is necessary to determine the benefits and risks in patients who are frail.
Finally, there are much less robust data on the benefit of home hemodialysis (either conventional, frequent, or intensive) in frail patients. Similar to PD, home hemodialysis allows greater involvement of family, more freedom of the travel burden of in-center hemodialysis, and more flexibility in dialysis prescription. An important future consideration is an evaluation of the impact of home hemodialysis on frailty and outcomes of frail patients who initiate home hemodialysis.
Transplant
Undergoing a kidney transplant is a significant physiologic stress to a patient’s reserve. By definition, as frailty severity increases, a patient’s ability to endure and recover from this stressor is reduced, which may explain the increase in postoperative complications and mortality in frail patients.16,17 While it is known that certain baseline comorbid conditions are associated with a low likelihood of surviving the transplant waitlist (and an even lower likelihood of survival to net benefit),42 it is currently unclear at what degree of frailty the risks of transplantation outweigh the benefits. This is further complicated by the finding that transplant may be one of the few “interventions” that could improve a patient’s frailty status and reverse deficit accumulation. Although frailty initially worsens post-transplant, it has been shown to improve as early as 3 months post in a number of patients.43 In addition, by 9 months post-transplant, a similar survival benefit is seen in frail and fit patients alike,44 perhaps owing to an improvement in frailty as a consequence of kidney transplantation. If this hypothesis is confirmed in future studies, it could dramatically change practice around kidney transplantation in the setting of frailty. It would be expected that a certain degree of baseline frailty would make the likelihood of a health benefit post-transplant very low. However, at less severe degrees of baseline frailty, kidney transplantation could be a means of halting the process of senescent nephropathy, both by returning renal function and by reversing a patient’s frailty.
Conservative Care
As frailty advances, and the risks and benefits of more invasive options for renal replacement therapy shift, symptomatic management of kidney disease without dialytic support must be explored with patients and families.45 Most discussions of conservative management focus on trading the increased quantity of life offered by dialysis for prolonged freedom and improved quality of life. Conceptually, however, at a certain degree of frailty, it can be expected that a patient’s quality and quantity of life would not improve with renal replacement therapy. Defining this population is difficult; it remains unclear at what degree of frailty a patient’s expected prognosis is unlikely to be improved by dialysis.
Regardless of the uncertainty with regard to overall prognosis, the burden of dialysis is higher in patients who are frail, and as such, the clinician must be able to communicate to patients what they can reasonably expect from conservative management. While data are limited, patients who opt for conservative management spend less time in hospital and are 4 times more likely to die at home or in hospice care.46 Those who opt for conservative management may live as long as 12 months past a time where dialysis would have otherwise been started.47 One-year survival appears to be similar between frail patients started on dialysis and those being managed conservatively; however, data are limited and may be subject to recognized bias.48
Improved quality of life for patients who are conservatively managed relies on adequate symptom control. Management of symptoms associated with ESRD requires a multidisciplinary approach and oftentimes involves referral to palliative care. A practical guide for the management of these symptoms has been presented elsewhere.46
How Can We Screen for Frailty in Patients with CKD?
There are a variety of options to choose from when deciding on a frailty screening tool. Importantly, frailty has been shown to be predictive of outcomes regardless of the method of assessment; therefore, the fact that frailty is being screened for is likely more important than which method is used.22 That being said, there are specific criteria that make certain assessment tools preferred for the purposes of screening. A good screening test must be safe, inexpensive, and easily administered. The results must be valid, reproducible, and allow detection at a time where intervention can be implemented. And finally, a test must be appropriately sensitive to catch a large number of patients with the condition, but also adequately specific to minimize false-positives.5 In this light, there are specific characteristics of frequently cited frailty assessment tools that make them more or less attractive for use as a screening test (Table 1).
 |
Table 1 Comparison of Select Existing Frailty Assessment Tools, Weighing Their Merits as Potential Screening Tools in Patients with CKD |
The most frequently cited tool for assessing frailty is the Fried phenotype (FP). Using the FP, a patient is described as frail in the presence of three of the following: weight loss, impaired hand-grip strength, decreased energy level, impaired walk speed, and physical inactivity.49 The presence of one or two of the above characteristics defines a patient as pre-frail. The original frailty phenotype required a combination of patient reports and directly observed physical tests, which can be cumbersome to perform. The FP has been modified frequently in an attempt to reduce the burden of data collection; however, this has come at the expense of generalizability.50 Despite these potential limitations, the FP is the most commonly used method of frailty assessment in patients with CKD.4
Similar to the FP, the short physical performance battery (SPPB) offers a validated tool for determining a patient’s physical fitness. The SPPB is comprised of three physical assessments: standing balance, gait speed, and a chair stand test. Like the FP, its strengths lie in its objectivity. Another potential strength of the SPPB is that it provides a range of scores, from 0 (worst performance) to 12 (best performance), allowing some quantification of a patient’s level of frailty. Importantly, in patients with CKD, the SPPB has been shown to be reliable,51 associated with disease progression,52 and to be predictive of mortality.53 Limitations of the SPPB are similar to the FP – they both require directly observed and potentially cumbersome physical tests and both rely solely on physical characteristics to define a patient as frail. Of note, however, gait speed alone has been shown to correlate best with the FP54 and has been shown to be highly predictive of mortality, hospitalization, and functional impairment.55 Despite requiring physical testing, it is also relatively easy to assess.
The broadening of the definition of frailty from impaired physical function to an accumulation of deficits across a variety of domains has led to the creation of a more global and complete frailty assessment, the frailty index (FI). A frailty index can be created with any combination of potential deficits. For this reason, an FI can be tailored to a specific population, with the list of potential deficits being reflective of the deficit burden most frequently experienced by those being assessed. Despite the variability in components that make up an FI, so long as the list of deficits is sufficiently robust, it has been shown to agree with the FP56 and be similarly associated with negative outcomes.57 The major limitation of an FI as a screening tool lies in the requirement for robust data, making it a relatively time-consuming alternative. In addition, operationalizing of a frailty index for use as a screening tool may require the creation of thresholds that undermine the continuous nature of the value. Nevertheless, an FI has been shown to be predictive of death and hospitalization in the CKD population.9
The Groningen frailty indicator (GFI) represents another multidimensional method of assessing frailty. This assessment tool consists of 15 questions across 8 domains, including mobility, vision, hearing, nutrition, comorbidity, cognition, psychosocial, and physical fitness. The GFI is attractive as a frailty screening test for a variety of reasons – the absence of physical testing makes it easy to implement, and unlike most other assessment tools, it offers a more robust examination of a patient’s psychosocial status. Unfortunately, in the general population, the GFI has been shown to have limited sensitivity, making it less optimal for an initial screen.58 In the CKD population, while the GFI was similarly predictive of death and hospitalization as other methods, it failed to identify specific geriatric deficits, most commonly of a physical nature.59 This may be due to the means by which physical impairment is screened for in the GFI, ie, with a single question, asking the patient to rate their own fitness from 0 to 10.
The multidimensional prognostic index (MPI) has been developed in an effort to predict the longevity of hospitalized adults. A patient’s frailty status is determined through eight individual assessments including function (activities of daily living and instrumental activities of daily living), polypharmacy, mental status, nutrition, risk of pressure sores, comorbidity, and social circumstances. Deficits in each domain are graded as 0 (none), 0.5 (minor), or 1 (major), and then averaged.60 A score greater than 0.66 is indicative of frailty and has been shown to be associated with increased hospital mortality and length of stay in the general population as well as in a number of specific disease states.61 While the MPI has shown excellent predictive abilities for mortality in the general population,62 it does require fairly robust data collection. In the elderly CKD population, addition of the MPI to the estimated GFR drastically improved prediction of 1- and 2-year mortality,63,64 and chronic dialysis patients have been shown to have higher MPI scores than the global geriatric population.65 However, the predictive abilities of the MPI have only been validated in hospitalized patients with kidney disease and would require further validation before generalization to the outpatient CKD population.
Finally, in an effort to produce a simple yet global frailty assessment for screening purposes, a clinical frailty scale (CFS) has been proposed. Frailty is again defined as an accumulation of deficits, with cognition, physical fitness, and comorbidity being assessed, specifically with regard to how they affect a patient’s function. The CFS presents eight categories with increasing degrees of frailty and a ninth category for individuals who are terminally ill and asks the clinician to place their patient along this spectrum. This simplicity is the greatest strength of the CFS when compared to other methods of assessing frailty. In addition, like the SPPB and FI, the CFS is graduated and allows for monitoring of changes in frailty severity over time. The CFS has been shown to have similar predictive characteristics as the FP in the general population66 and is associated with death in patients with predialysis CKD67 as well as those on dialysis.14 Worsened severity of frailty as measured by the CFS has also been shown to increase the risk of mortality.14 The major limitations of the CFS are that it lacks robust validation data in CKD4 and is a subjective tool. However, when assessed in the CKD population, the CFS agreed with the FP better than the SPPB and FI, suggesting that the CFS may be a valuable option for accurate screening of frailty when it is not practical to perform a physical assessment.54
How Can Frailty Screening Be Incorporated into the Care of Patients with CKD?
The specifics of how to screen for frailty in patients with CKD have been discussed recently in the literature. The 2016 European renal best practice (ERBP) guideline on the management of older patients with CKD suggests frailty screening in all older adults who are not otherwise at risk of imminently dying or at low risk for progression to ESRD. Impaired functional status and frailty are seen as interchangeable in this recommendation although they do note that frailty scores may “provide additional information during assessment and shared decision-making on the planning of patients.” A specific screening test is not endorsed; however, they recommend that after an initial assessment, functional status be reassessed every 6–8 weeks for dialysis patients, and at every visit for ambulatory patients. They list exercise therapy and dietary interventions as potential means of modifying frailty and present a schema wherein the presence of frailty prompts a shift in focus to conservative management and advanced care planning.22
A more recent proposal suggests frailty screening be performed at first contact with an elderly (age 65 and older) CKD patient. They propose modifying nephroprotective strategies for frail patients, such as using more lenient blood pressure and glucose targets. In addition, they recommend targeted therapy toward the patient’s frailty, such as exercise in sarcopenic patients. While they stress the importance of an individualized approach for all frail patients, they suggest that the presence of frailty should lead to a delayed dialysis start or conservative management. Of note, they recommend that fit patients be reassessed for the development of frailty but give no specific recommendation for repeated assessments in patients after they have already screened as frail (likely due to a lack of sufficient data to guide practice).20
Building on these previous proposals, we present a putative framework for incorporating frailty screening into general nephrology practice (Figure 1). We differ from previous recommendations, suggesting that all CKD patients be screened, regardless of age, as the prevalence of frailty remains high in patients under 65.4 To ensure that frail patients are not missed, we recommend screening at first encounter, and then similarly to the ERBP, we recommend repeat screening. An optimal interval for screening has not been established; however, reassessing CKD patients at each ambulatory visit and every 6–8 weeks in dialysis patients is very reasonable. Beyond standard screening, we propose additional assessments. We recommend consideration toward screening after major health events (ie, prolonged or nonelective hospitalizations). Furthermore, determining the need to include frailty in the waitlist eligibility assessment is an internationally recognized priority for kidney transplantation,68 and, as such, frailty assessment could be considered as a standard part of the kidney transplant workup. The benefits of frequent frailty screening are two-fold: it will increase the likelihood of diagnosing incident frailty as well as allow monitoring of the progression of frailty over time. This will facilitate reevaluating frailty interventions as well as offer prognostic information.
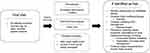 |
Figure 1 Proposed framework for incorporating frailty screening into standard nephrology practice. |
We agree with previous recommendations that it is more important that frailty be screened for than the most ideal screening tool be used. However, as stated above, there are characteristics that make certain tools more attractive for clinical practice. We prefer the use of tools that are easy to implement and graded. A walk-test has been shown to agree best with the FP,54 and although it has not been used this way, as it provides a numerical value, it could be developed for use in a graded fashion. The CFS (notwithstanding its limitations) has also been shown to agree well with the FP54 and may be a tool that is easier to operationalize.
In all patients, screening positive for frailty should prompt a more thorough assessment to look for modifiable deficits, as well as careful consideration of how their current CKD management may affect their degree of frailty. In patients approaching ESRD (GFR < 15 mL/min), the presence of frailty should prompt an exploration of patient values, including a frank discussion of expectations for renal replacement strategies and prognosis. Conservative management should be presented as part of the continuum of kidney care.
Future Areas of Study
Much work is needed to further explore the interplay between frailty and kidney disease. Perhaps, most paramount is gaining a better understanding of how frailty changes with time, as this will aid in creating and assessing potential frailty interventions. Frequent frailty assessments using graded scoring systems should help with this.
Expanding our understanding of frailty severity would have additional benefits. By further studying how outcomes change as frailty worsens, it may be possible to identify degrees of frailty at which different renal replacement therapy options offer little benefit. This would allow frail patients with ESRD to make better informed decisions; patients with milder degrees of frailty might opt for incremental dialysis or conservative care, while patients with more severe frailty could instead focus on symptom management and comfort and be spared unnecessary suffering.
With regard to frailty assessment tools, deciding on a preferred method for frailty assessment for use in future studies would certainly help make data more uniform and allow easier comparison of trials. However, this may come at the expense of missing frail patients, as different assessment methods appear to capture different aspects of frailty.69
Developing targeted interventions for improving frailty severity is a promising area of study. Specific interventions to further investigate include the use of geriatric referral and exercise. Furthermore, determining the effect of kidney transplantation on frailty severity (using other frailty assessment tools) would be valuable. Given the strong relationship between frailty and adverse outcomes, monitoring changes in frailty as a surrogate marker of more established objective outcomes (hospitalization, morbidity, mortality) may facilitate future research.
One final area of promising research is the development of prognostic models to help inform discussions at the time of renal replacement therapy. To date, validated tools have been developed but focus primarily on measures of comorbidity and age.70,71 Frailty measures may compare favorably in prognostication or may be added to these measures to improve their predictive capabilities. Improving prognostic certainty would help inform discussions with patients and families around conservative management.
Conclusion
Frailty is highly prevalent in patients with kidney disease and is an important determinant in their outcomes and prognosis. Screening for frailty in patients with CKD will lead to changes in treatment for patients who are frail, both through modification of standard CKD management and by informing discussions around renal replacement therapy. All patients with CKD should undergo frailty screening, with frequent reassessments. Assessment should ideally be done using a method that grades the severity of frailty in order to monitor progression over time, as well as the efficacy of frailty modifying therapies. Directed therapies for modifying frailty in patients with kidney disease, such as exercise and transplantation, are an exciting and promising area of future research.
Disclosure
Dr. Tennankore has received unrestricted grant funding for research from Astellas, Canada, and Otsuka. Dr. Tennankore has participated in Advisory boards for AstraZeneca and Otsuka. The authors report no other conflicts of interest in this work.
References
1. Morley JE, Vellas B, Abellan van Kan G, et al. Frailty consensus: a call to action. J Am Med Dir Assoc. 2013;14(6):392–397. doi:10.1016/j.jamda.2013.03.022
2. Vermeiren S, Vella-Azzopardi R, Beckwée D, et al. Frailty and the prediction of negative health outcomes: a meta-analysis. J Am Med Dir Assoc. 2016;17(12):
3. Kojima G. Prevalence of frailty in end-stage renal disease: a systematic review and meta-analysis. Int Urol Nephrol. 2017;49(11):1989–1997. doi:10.1007/s11255-017-1547-5
4. Chowdhury R, Peel NM, Krosch M, Hubbard RE. Frailty and chronic kidney disease: a systematic review. Arch Gerontol Geriatr. 2017;68:135–142. doi:10.1016/J.ARCHGER.2016.10.007
5. Herman C. What makes a screening exam “good”? AMA J Ethics. 2006;8(1):34–37. doi:10.1001/virtualmentor.2006.8.1.cprl1-0601
6. Collard RM, Boter H, Schoevers RA, Oude Voshaar RC. Prevalence of frailty in community-dwelling older persons: a systematic review. J Am Geriatr Soc. 2012;60(8):1487–1492. doi:10.1111/j.1532-5415.2012.04054.x
7. Roshanravan B, Khatri M, Robinson-Cohen C, et al. A prospective study of frailty in nephrology-referred patients with CKD. Am J Kidney Dis. 2012;60(6):912–921. doi:10.1053/J.AJKD.2012.05.017
8. Mansur H, Colugnati FA, dos Santos Grincenkov FR, Bastos M. Frailty and quality of life: a cross-sectional study of Brazilian patients with pre-dialysis chronic kidney disease. Health Qual Life Outcomes. 2014;12(1):27. doi:10.1186/1477-7525-12-27
9. Vezza C, Vettoretti S, Caldiroli L, Bergamaschini L, Messa P, Cesari M. Use of the frailty index in older persons with chronic kidney disease. J Am Med Dir Assoc. 2019;20:1179–1180. doi:10.1016/J.JAMDA.2019.04.015
10. McAdams-DeMarco MA, Suresh S, Law A, et al. Frailty and falls among adult patients undergoing chronic hemodialysis: a prospective cohort study. BMC Nephrol. 2013;14(1):224. doi:10.1186/1471-2369-14-224
11. Kang SH, Do JY, Lee SY, Kim JC. Effect of dialysis modality on frailty phenotype, disability, and health-related quality of life in maintenance dialysis patients. PLoS One. 2017;12(5). doi:10.1371/journal.pone.0176814
12. Johansen KL, Chertow GM, Jin C, Kutner NG. Significance of frailty among dialysis patients. J Am Soc Nephrol. 2007;18:2960–2967. doi:10.1681/ASN.2007020221
13. McAdams-DeMarco MA, Law A, Salter ML, et al. Frailty as a novel predictor of mortality and hospitalization in individuals of all ages undergoing hemodialysis. J Am Geriatr Soc. 2013;61(6):896–901. doi:10.1111/jgs.12266
14. Alfaadhel TA, Soroka SD, Kiberd BA, Landry D, Moorhouse P, Tennankore KK. Frailty and mortality in dialysis: evaluation of a clinical frailty scale. Clin J Am Soc Nephrol. 2015;10(5):832–840. doi:10.2215/CJN.07760814
15. Cheng XS, Lentine KL, Koraishy FM, Myers J, Tan JC. Implications of frailty for peritransplant outcomes in kidney transplant recipients. Curr Transplant Rep. 2019;6(1):16–25. doi:10.1007/s40472-019-0227-z
16. McAdams-DeMarco MA, Law A, Salter ML, et al. Frailty and early hospital readmission after kidney transplantation. Am J Transplant. 2013;13(8):2091–2095. doi:10.1111/ajt.12300
17. McAdams-DeMarco MA, Law A, King E, et al. Frailty and mortality in kidney transplant recipients. Am J Transplant. 2015;15(1):149–154. doi:10.1111/ajt.12992
18. Puts MTE, Toubasi S, Andrew MK, et al. Interventions to prevent or reduce the level of frailty in community-dwelling older adults: a scoping review of the literature and international policies. Age Ageing. 2017;46(3):383–392. doi:10.1093/ageing/afw247
19. Kooman JP, Dekker MJ, Usvyat LA, et al. Inflammation and premature aging in advanced chronic kidney disease. Am J Physiol Physiol. 2017;313(4):F938–F950. doi:10.1152/ajprenal.00256.2017
20. Ostuni M, Musso CG. Usefulness of frailty evaluation for handling chronic kidney disease elderly patients: a review and original proposal. Int Urol Nephrol. 2019;51(3):461–465. doi:10.1007/s11255-018-2061-0
21. Triantafylidis LK, Hawley CE, Perry LP, Paik JM. The role of deprescribing in older adults with chronic kidney disease. Drugs Aging. 2018;35(11):973–984. doi:10.1007/s40266-018-0593-8
22. Farrington K, Covic A, Nistor I, et al. Clinical Practice Guideline on management of older patients with chronic kidney disease stage 3b or higher (eGFR< 45 mL/min/1.73 m2): a summary document from the European Renal Best Practice Group. Nephrol Dial Transplant. 2017;32(1):9–16. doi:10.1093/ndt/gfw411
23. Greco A, Paroni G, Seripa D, Addante F, Dagostino MP, Frailty AF. Disability and physical exercise in the aging process and in chronic kidney disease. Kidney Blood Press Res. 2014;39(2–3):164–168. doi:10.1159/000355792
24. Avesani CM, Trolonge S, Deleaval P, et al. Physical activity and energy expenditure in haemodialysis patients: an international survey. Nephrol Dial Transplant. 2012;27(6):2430–2434. doi:10.1093/ndt/gfr692
25. Stack AG, Molony DA, Rives T, Tyson J, Murthy BVR. Association of physical activity with mortality in the US dialysis population. Am J Kidney Dis. 2005;45(4):690–701. doi:10.1053/J.AJKD.2004.12.013
26. Roshanravan B, Gamboa J, Exercise WK. CKD: skeletal muscle dysfunction and practical application of exercise to prevent and treat physical impairments in CKD. Am J Kidney Dis. 2017;69(6):837–852. doi:10.1053/J.AJKD.2017.01.051
27. Viana JL, Kosmadakis GC, Watson EL, et al. Evidence for anti-inflammatory effects of exercise in CKD. J Am Soc Nephrol. 2014;25:2121–2130. doi:10.1681/ASN.2013070702
28. Roshanravan B, Robinson-Cohen C, K V P, et al. Association between physical performance and all-cause mortality in CKD. J Am Soc Nephrol. 2013;24:822–830. doi:10.1681/ASN.2012070702
29. Tarazona-Santabalbina FJ, Gómez-Cabrera MC, Pérez-Ros P, et al. A multicomponent exercise intervention that reverses frailty and improves cognition, emotion, and social networking in the community-dwelling frail elderly: a randomized clinical trial. J Am Med Dir Assoc. 2016;17(5):426–433. doi:10.1016/j.jamda.2016.01.019
30. Kiberd BA, Tennankore KK, West K. Eligibility for the kidney transplant wait list: a model for conceptualizing patient risk. Transplant Res. 2014;3(1):2. doi:10.1186/2047-1440-3-2
31. Flythe JE, Hilliard T, Castillo G, et al. Symptom prioritization among adults receiving in-center hemodialysis: a mixed methods study. Clin J Am Soc Nephrol. 2018;13(5):735–745. doi:10.2215/CJN.10850917
32. Chao C-T, Chiang C-K, Huang J-W, Hung K-Y. Self-reported frailty among end-stage renal disease patients: a potential predictor of dialysis access outcomes. Nephrology. 2017;22(4):333–334. doi:10.1111/nep.12961
33. Basile C, Casino FG, Aucella F. Incremental hemodialysis, a valuable option for the frail elderly patient. J Nephrol. 2019;1–10. doi:10.1007/s40620-019-00611-4
34. Daugirdas JT, Greene T, M V R, et al. Effect of frequent hemodialysis on residual kidney function. Kidney Int. 2013;83(5):949–958. doi:10.1038/KI.2012.457
35. Mathew AT, Fishbane S, Obi Y, Kalantar-Zadeh K. Preservation of residual kidney function in hemodialysis patients: reviving an old concept. Kidney Int. 2016;90(2):262–271. doi:10.1016/j.kint.2016.02.037
36. Garofalo C, Borrelli S, De Stefano T, et al. Incremental dialysis in ESRD: systematic review and meta-analysis. J Nephrol. 2019:1–14. doi:10.1007/s40620-018-00577-9.
37. Pavasini R, Guralnik J, Brown JC, et al. Short physical performance battery and all-cause mortality: systematic review and meta-analysis. BMC Med. 2016;14(1):215. doi:10.1186/s12916-016-0763-7
38. Tennankore K, Zhao J, Karaboyas A, et al. The association of functional status with mortality and dialysis modality change: results from the Peritoneal Dialysis Outcomes and Practice Patterns Study (PDOPPS). Perit Dial Int. 2019;39(2):103–111. doi:10.3747/pdi.2018.00094
39. Iyasere OU, Brown EA, Johansson L, et al. Quality of life and physical function in older patients on dialysis: a comparison of assisted peritoneal dialysis with hemodialysis. Clin J Am Soc Nephrol. 2016;11(3):423–430. doi:10.2215/CJN.01050115
40. Belasco A, Barbosa D, Bettencourt AR, Diccini S, Sesso R. Quality of life of family caregivers of elderly patients on hemodialysis and peritoneal dialysis. Am J Kidney Dis. 2006;48(6):955–963. doi:10.1053/J.AJKD.2006.08.017
41. Auguste BL, Bargman JM. Incremental peritoneal dialysis: new ideas about an old approach. Semin Dial. 2018;31(5):445–448. doi:10.1111/sdi.12712
42. Kiberd BA, AlBugami MM, Panek R, Tennankore K. Contraindications to kidney transplantation: uneven grounds? Transplant Res. 2015;4:2. doi:10.1186/s13737-015-0024-x
43. McAdams-DeMarco MA, Isaacs K, Darko L, et al. Changes in frailty after kidney transplantation. J Am Geriatr Soc. 2015;63:2152–2157. doi:10.1111/jgs.2015.63.issue-10
44. Reese PP, Shults J, Bloom RD, et al. Functional status, time to transplantation, and survival benefit of kidney transplantation among wait-listed candidates. Am J Kidney Dis. 2015;66(5):837–845. doi:10.1053/J.AJKD.2015.05.015
45. Farrington K, Covic A, Aucella F, et al. Clinical practice guideline on management of older patients with chronic kidney disease stage 3b or higher (eGFR <45 mL/min/1.73 m 2). Nephrol Dial Transplant. 2016;31(suppl2):ii1–ii66. doi:10.1093/ndt/gfw356
46. Raghavan D, Holley JL. Conservative care of the elderly CKD patient: a practical guide. Adv Chronic Kidney Dis. 2016;23(1):51–56. doi:10.1053/J.ACKD.2015.08.003
47. Brown MA, Collett GK, Josland EA, Foote C, Li Q, Brennan FP. CKD in elderly patients managed without dialysis: survival, symptoms, and quality of life. Clin J Am Soc Nephrol. 2015;10(2):260–268. doi:10.2215/CJN.03330414
48. Foote C, Kotwal S, Gallagher M, Cass A, Brown M, Jardine M. Survival outcomes of supportive care versus dialysis therapies for elderly patients with end-stage kidney disease: a systematic review and meta-analysis. Nephrology. 2016;21(3):241–253. doi:10.1111/nep.12586
49. Fried LP, Tangen CM, Walston J, et al. Frailty in older adults: evidence for a phenotype. J Gerontol a Biol Sci Med Sci. 2001;56(3):M146–M157. doi:10.1093/gerona/56.3.M146
50. Theou O, Cann L, Blodgett J, Wallace LMK, Brothers TD, Rockwood K. Modifications to the frailty phenotype criteria: systematic review of the current literature and investigation of 262 frailty phenotypes in the survey of health, ageing, and retirement in Europe. Ageing Res Rev. 2015;21:78–94. doi:10.1016/J.ARR.2015.04.001
51. Ortega-pérez de Villar L, Martínez-Olmos FJ, Junqué-Jiménez A, et al. Test-retest reliability and minimal detectable change scores for the short physical performance battery, one-legged standing test and timed up and go test in patients undergoing hemodialysis. Reboldi G, ed. PLoS One. 2018;13(8):e0201035. doi:10.1371/journal.pone.0201035
52. Lattanzio F, Corsonello A, Abbatecola AM, et al. Relationship between renal function and physical performance in elderly hospitalized patients. Rejuvenation Res. 2012;15(6):545–552. doi:10.1089/rej.2012.1329
53. Nastasi AJ, McAdams-DeMarco MA, Schrack J, et al. Pre-kidney transplant lower extremity impairment and post-kidney transplant mortality. Am J Transplant. 2018;18(1):189–196. doi:10.1111/ajt.14430
54. Nixon AC, Bampouras TM, Pendleton N, Mitra S, Dhaygude AP. Diagnostic accuracy of frailty screening methods in advanced chronic kidney disease. Nephron. 2019;141(3):147–155. doi:10.1159/000494223
55. Kutner NG, Zhang R, Huang Y, Gait Speed PP, Mortality H. Functional status change among hemodialysis patients: a US renal data system special study. Am J Kidney Dis. 2015;66(2):297–304. doi:10.1053/J.AJKD.2015.01.024
56. Theou O, Walston J, Rockwood K. Operationalizing Frailty Using the Frailty Phenotype and Deficit Accumulation Approaches. Vol. 41. Karger Publishers;2015:66–73. doi:10.1159/000381164
57. Rockwood K, Andrew M, Mitnitski A Unconventional views of frailty a comparison of two approaches to measuring frailty in elderly people; 2007. Available from: http://biomedgerontology.oxfordjournals.org/.
58. Clegg A, Rogers L, Young J. Diagnostic test accuracy of simple instruments for identifying frailty in community-dwelling older people: a systematic review. Age Ageing. 2015;44(1):148–152. doi:10.1093/ageing/afu157
59. Meulendijks FG, Hamaker ME, Boereboom FTJ, Kalf A, NPJ V, van Munster BC. Groningen frailty indicator in older patients with end-stage renal disease. Ren Fail. 2015;37(9):1419–1424. doi:10.3109/0886022X.2015.1077315
60. Pilotto A, Ferrucci L, Franceschi M, et al. Development and validation of a multidimensional prognostic index for one-year mortality from comprehensive geriatric assessment in hospitalized older patients. Rejuvenation Res. 2008;11(1):151–161. doi:10.1089/rej.2007.0569
61. Volpato S, Bazzano S, Fontana A, Ferrucci L, Pilotto A. Multidimensional prognostic index predicts mortality and length of stay during hospitalization in the older patients: a multicenter prospective study. J Gerontol a Biol Sci Med. 2015;70(3):325–331. doi:10.1093/gerona/glu167
62. Pilotto A, Rengo F, Marchionni N, et al. Comparing the prognostic accuracy for all-cause mortality of frailty instruments: a multicentre 1-year follow-up in hospitalized older patients. Vina J, ed. PLoS One. 2012;7(1):e29090. doi:10.1371/journal.pone.0029090
63. Pilotto A, Sancarlo D, Franceschi M, et al. A multidimensional approach to the geriatric patient with chronic kidney disease. J Nephrol. 2010;23(SUPPL. 15)S5–10.
64. Pilotto A, Sancarlo D, Aucella F, et al. Addition of the multidimensional prognostic index to the estimated glomerular filtration rate improves prediction of long-term all-cause mortality in older patients with chronic kidney disease. Rejuvenation Res. 2012;15:82–88. doi:10.1089/rej.2011.1210
65. Aucella F, Stoico L, Cicchella A, et al. Comprehensive geriatric assessment in the hemodialysis elderly population. J Nephrol. 2012;25(SUPPL.19):85–89. doi:10.5301/jn.5000223
66. Rockwood K, Song X, MacKnight C, et al. A global clinical measure of fitness and frailty in elderly people. CMAJ. 2005;173(5):489–495. doi:10.1503/cmaj.050051
67. Pugh J, Aggett J, Goodland A, et al. Frailty and comorbidity are independent predictors of outcome in patients referred for pre-dialysis education. Clin Kidney J. 2016;9:324–329. doi:10.1093/ckj/sfv150
68. Segall L, Nistor I, Pascual J, et al. Criteria for and appropriateness of renal transplantation in elderly patients with end-stage renal disease. Transplantation. 2016;100(10):e55–e65. doi:10.1097/TP.0000000000001367
69. Cesari M, Gambassi G, Abellan van Kan G, Vellas B. The frailty phenotype and the frailty index: different instruments for different purposes. Age Ageing. 2014;43(1):10–12. doi:10.1093/ageing/aft160
70. Bansal N, Katz R, De Boer IH, et al. Development and validation of a model to predict 5-year risk of death without ESRD among older adults with CKD. Clin J Am Soc Nephrol. 2015;10(3):363–371. doi:10.2215/CJN.04650514
71. Couchoud C, Labeeuw M, Moranne O, et al. A clinical score to predict 6-month prognosis in elderly patients starting dialysis for end-stage renal disease. Nephrol Dial Transplant. 2009;24(5):1553–1561. doi:10.1093/ndt/gfn698
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
