Back to Journals » Clinical Ophthalmology » Volume 9
Argus II retinal prosthesis malrotation and repositioning with intraoperative optical coherence tomography in a posterior staphyloma
Received 17 September 2015
Accepted for publication 23 October 2015
Published 24 November 2015 Volume 2015:9 Pages 2213—2216
DOI https://doi.org/10.2147/OPTH.S96570
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
The supplementary video shows Argus II prosthesis repositioning.
Views: 357
Michael I Seider, Paul Hahn
Duke University Eye Center, Durham, NC, USA
Introduction: The Argus II retinal prosthesis may improve visual function in patients with severe vision loss from retinitis pigmentosa. Optimal centration of the electrode array over the macula is important to achieve optimal visual results. Argus tack malrotation is a novel entity that may be encountered during placement, especially in patients with posterior staphyloma.
Methods: Retrospective case review.
Results: During tacking of the electronics array a clockwise rotation occurred resulting in malposition. We hypothesize this was secondary to undue rotation or posterior pressure applied during tack insertion in conjunction with placement over a previously unrecognized posterior staphyloma. Intraoperative optical coherence tomography, because of the cross-sectional images provided, was helpful in visualizing the distance between the electronics array and the retina, which was difficult to assess using the surgical microscope alone. Repositioning was achieved by adjusting the tack without removal. The patient experienced an improvement in vision as a result of the surgery.
Conclusion: Malrotation may occur when tacking the Argus II prosthesis, and the presence of a posterior staphyloma may increase this risk. It is important to differentiate malrotation from tack misplacement – the former may be addressed with array unrotation or partial tack withdrawal and the latter may require tack removal and reinsertion. Also, intraoperative optical coherence tomography may be helpful in characterizing electronics array position during surgery.
Keywords: Argus, retinitis pigmentosa, retinal tack
Introduction
The Argus II Retinal Prosthesis System (Second Sight Medical Products Inc., Sylmar, CA, USA) may improve visual function in patients with end-stage retinitis pigmentosa.1 Visual outcomes likely depend on centration and apposition of the electronics array to the macula.2,3 A few cases of array malpositioning that were identified postoperatively have been reported.2 In some of these cases, a subsequent surgical procedure with tack withdrawal was performed to reposition the array and improve visual outcome.
A posterior staphyloma alters the normal contour of the posterior eye wall and may adversely influence proper centration and apposition of a semi-rigid array.1 We present a case of Argus II implantation in a patient with a posterior staphyloma who experienced intraoperative tack malrotation and who achieved array centration and partial apposition via guidance during surgery by feedback from direct visualization and intraoperative optical coherence tomography (OCT).
Results
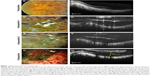
The Argus II epiretinal prosthesis was placed in the left eye of a 66-year-old man with bare light-perception vision in each eye from retinitis pigmentosa (Figure 1A). Preoperatively, spectral-domain OCT (Heidelberg Engineering, Heidelberg, Germany) demonstrated a posterior staphyloma (Figure 1B). During surgery, at the final moment of tack insertion, unintentional rotation of the array occurred (Figure 1C), resulting in the array partially overlying the optic nerve instead of being centered in the macula. The reason for this malrotation is unclear. We hypothesize this occurred because of an unintentional manual clockwise rotation of the forceps during insertion. It is also possible that excessive posterior pressure during tack placement resulted in tack rotation from over-compression of the tack spring. The position of the electronics array is determined by the orientation of the electronics cable as it enters the sclera, and it is also possible that array rotation occurred because of uneven tension on this cable due to an oblique sclerotomy.
In addition to poor centration, intraoperative OCT (Bioptigen Inc., Morrisville, NC, USA) revealed the electrode array to be elevated by the edge of the macular staphyloma and poorly apposed to the retinal surface (Figure 1D), which was not appreciated with the surgical microscope alone. At the time of surgery, prior to reviewing the surgical video, poor centration of the electronics array was attributed to tack misplacement rather than array malrotation. Attempts were made initially to withdraw and replace the tack. The tack was unable to be withdrawn with moderate force. As the tack was released from the tack forceps, the array was noted to unrotate, leaving the array still anchored by the tack and optimally centered over the macula (Figure 1E). We hypothesize this occurred due to the partial release of compressive tack pressure. Intraoperative OCT was then performed again demonstrating the array to be free from the staphyloma edge (Figure 1F). Three months postoperatively, the implant remained well centered in the macula and free from the staphyloma edge (Figure 1G) (Video S1). The array remained elevated up to 500 μm from the retinal surface postoperatively (Figure 1H). Despite the distance of the array from the retinal surface, the patient has done well with a significant subjective improvement in function following activation of the device. In addition, objective tests performed 1 year following surgery revealed improvements in visual function when the device was active. On square localization testing, the patient scored 7.5% (3/40) with the device turned off and 32.5% (13/40) with the device turned on. On direction of motion testing the patient scored 6.3% (5/80) with the device turned off and 12.5% (10/80) with the device turned on. The patient did not have appreciable improvements in grated visual acuity with device activation.
Conclusion
The presence of a posterior staphyloma may affect optimal implantation of the Argus II prosthesis.1 We suggest that the surgical plan for placement in patients with a posterior staphyloma account for curvature in the eye wall to maximize proximity of the semi-rigid array to the retinal surface. Intraoperative OCT enables cross-sectional visualization during surgery and may be helpful in visualizing axial proximity of the implant to the retina, which is otherwise difficult to assess using the surgical microscope alone. In addition, intraoperative OCT may permit identification of anatomic variations and/or pathology (posterior staphyloma, epiretinal membranes, residual posterior vitreous, etc), which may also affect surgical decision-making.
Also, unintended rotation of an array during implantation can occur. It is important to differentiate malrotation from tack misplacement – the former may be addressed with array unrotation or partial tack withdrawal and the latter may require tack removal and reinsertion. In cases of staphylomas, complete apposition of the Argus array to the retinal surface may not be possible although improvements in visual function may still be obtained in such patients, as Rizzo et al have also shown.1 Argus II electronics array positioning can be optimized by direct visualization and intraoperative OCT guidance. Our patient has achieved meaningful visual and functional gains, suggesting that the presence of a staphyloma and reduced array-retina contact should not be a contraindication to Argus II implantation.
Acknowledgment
This manuscript was supported by the Heed Ophthalmic Foundation.
Disclosure
Paul Hahn is a paid consultant for Second Sight Medical Products, Inc. (Sylmar, CA, USA). The authors report no other disclosures or pertinent conflicts of interest in this work.
References
Rizzo S, Belting C, Cinelli L, et al. The Argus II retinal prosthesis: 12-month outcomes from a single-study center. Am J Ophthalmol. 2014;157(6):1282–1290. | ||
de Juan E, Spencer R, Barale PO, da Cruz L, Neysmith J. Extraction of retinal tacks from subjects implanted with an epiretinal visual prosthesis. Graefes Arch Clin Exp Ophthalmol. 2013;251(10):2471–2476. | ||
Ahuja AK, Yeoh J, Dorn JD, et al. Factors affecting perceptual threshold in Argus II retinal prosthesis subjects. Transl Vis Sci Technol. 2013;2(4):1. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.