Back to Journals » Clinical Interventions in Aging » Volume 14
Aged kidneys are refractory to autophagy activation in a rat model of renal ischemia-reperfusion injury
Authors Diao C, Wang L, Liu H , Du Y, Liu X
Received 7 December 2018
Accepted for publication 26 January 2019
Published 1 March 2019 Volume 2019:14 Pages 525—534
DOI https://doi.org/10.2147/CIA.S197444
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Zhi-Ying Wu
Changhui Diao, Lei Wang, Hao Liu, Yang Du, Xiuheng Liu
Department of Urology, Renmin Hospital of Wuhan University, Wuhan, Hubei, People’s Republic of China
Background: Ischemia-reperfusion (I/R) injury is the most common cause of acute kidney injury (AKI). Numerous therapeutic approaches for I/R injury have been studied, including autophagy, particularly in animal models of renal I/R injury derived from young or adult animals. However, the precise role of autophagy in renal ischemia-reperfusion in the aged animal model remains unclear. The purpose of this study was to demonstrate whether autophagy has similar effects on renal I/R injury in young and aged rats.
Materials and methods: All rats were divided into two age groups (3 months and 24 months) with each group being further divided into four subgroups (sham, I/R, I/R+Rap (rapamycin, an activator of autophagy), I/R+3-MA (3-methyladenine, an inhibitor of autophagy)). The I/R+Rap and I/R+3-MA groups were intraperitoneally injected with rapamycin and 3-MA prior to ischemia. We then measured serum levels of urea nitrogen, creatinine and assessed damage in the renal tissue. Immunohistochemistry was used to assess LC3-II and caspase-3, and Western blotting was used to evaluate the autophagy-related proteins LC3-II, Beclin-1 and P62. Apoptosis and autophagosomes were evaluated by TUNEL and transmission electron microscopy, respectively.
Results: Autophagy was activated in both young and aged rats by I/R and enhanced by rapamycin, although the level of autophagy was lower in the aged groups. In young rats, the activation of autophagy markedly improved renal function, reduced apoptosis in the renal tubular epithelial cells and the injury score in the renal tissue, thereby exerting protective effects on renal I/R injury. However, this level of protection was not present in aged rats.
Conclusion: Our data indicated that the activation of autophagy was ineffective in aged rat kidneys. These discoveries may have major implications in that severe apoptosis in aged kidneys might be refractory to antiapoptotic effect induced by the activation of autophagy.
Keywords: autophagy, ischemia-reperfusion, renal, aged, apoptosis, rapamycin, 3-methyladenine
Introduction
Acute kidney injury (AKI) is a common clinical renal disease characterized by a sharp loss of renal function, leading to the accumulation of metabolic waste, water and electrolyte disorders, and a series of clinical symptoms such as edema, hypertension and hemorrhage.1,2 There are many causes of AKI, including ischemia-reperfusion injury, nephrotoxicity and sepsis; the most common cause is ischemia-reperfusion injury (I/R), which often occurs after severe trauma, partial nephrectomy, kidney transplantation and shock.3–5 AKI is associated with high morbidity and mortality and may contribute to the occurrence and progression of chronic kidney disease (CKD).6 Current research seeks to investigate protective treatment for renal ischemia-reperfusion injury, including ischemic preconditioning, drug pretreatment, postischemic treatment,5,7,8 and the regulation of autophagy.9–12 Autophagy is an evolutionarily conserved catabolic process characterized by the formation of bilayer membrane structures (autophagosomes). During the process of autophagy, defective organelles or pathogens are engulfed by autophagic vesicles with double-membrane structures and fused to lysosomes for degradation and recycling,9,13 in order to maintain the stability of the intracellular environment in the case of cellular ischemia and hypoxia, and to promote cell survival.9,12 An accumulating body of evidence suggests that autophagy plays a key role in renal I/R injury.9–12
In this study, we used rapamycin and 3-MA to modulate autophagy and explore the role of autophagy in a rat model of renal I/R injury. Rapamycin is a macrolide antibiotic, and can act as an inducer of autophagy by inhibiting mTOR (the mammalian target of rapamycin).9,14,15 3-MA is an inhibitor of phosphatidylinositol 3-kinase (PI3K), which blocks the formation of primary autophagic vacuoles and autophagosomes.16 Therefore, 3-MA can effectively block the initial formation of autophagy and downregulate autophagy activity.
Previous studies have demonstrated different protective treatments to protect renal I/R injury. However, most of these treatments have failed in clinical practice, especially in aged patients.17 An important reason for these findings is that healthy young animals were usually utilized in most of the experiments to develop treatments. However, in clinical practice, donors and patients are often elder and also suffering from comorbidities.7,18 So far, most studies of autophagy in renal I/R injury adopted healthy young animals and ignored the role of aged animals. In our present study, we investigated whether autophagy can be activated in both young and aged rat models of renal I/R injury and whether the regulation of autophagy has similar effects on the kidney of young and aged rats.
Materials and methods
Animals and models
Male Sprague Dawley rats were divided into 2 groups according to age, young (3-month-old) and aged (24-month-old) (n=24 per group). All SD rats were purchased from the first clinical college experimental animal center of Wuhan University and kept at 20°C–22°C under a 12 hours light/dark cycle. All procedures were performed in accordance with the guidelines for the care and use of laboratory animals published by the National Institutes of Health. In brief, animals were anesthetized an intraperitoneal injection of pentobarbital (45 mg/kg). Then, rats were placed on a thermostatic surgical plate to maintain a core temperature of 37°C. For surgery, a midline incision was adopted, and the right kidney was resected. The left renal pedicle was clamped for 45 minutes (or not, depending on the experimental group). All rats were sacrificed 24 hours after reperfusion.
In total, 24 male SD rats from each group were randomly divided into 4 subgroups (n=6 per group): in the sham group, the left renal artery was not clamped; in the I/R group, the left renal artery was clamped for 45 minutes, followed by reperfusion for 24 hours; in the I/R+Rap group, rats received an intraperitoneal injection of rapamycin (Selleckchem, Houston, TX, USA, 1 mg/kg) 15 minutes before renal artery clipping, as previously described;9 in the I/R+3-MA group, rats received an intraperitoneal injection of 3-MA (Selleckchem, 30 mg/kg) 15 minutes before renal artery clamping, as previously described.9 Experimental procedures were consistent with experimental protocols approved by the Laboratory Animal Welfare & Ethics Committee of Renmin Hospital of Wuhan University.
Specimen collection and preservation
The left kidney was removed under general anesthesia and either fixed in 10% phosphate-buffered formalin, 4% glutaraldehyde or immediately stored at −80°C for further examination.
Renal function
Renal function was assessed by measuring serum levels of blood urea nitrogen (BUN) and creatinine (Cr). At 24 hours after reperfusion, blood samples were collected and analyzed with commercial creatinine and urea testing kits (Nanjing Jian cheng Bioengineering Institute, Nanjing, People’s Republic of China) in accordance with the manufacturer’s instructions.
Histological evaluation
According to the standard procedure, kidney tissue was fixed in 4% paraformaldehyde, embedded in paraffin, and then cut into 4 μm-thick sections. Hematoxylin and eosin staining was then performed. The results were examined by two experienced pathologists using blind methods. Morphological changes were assessed by two experienced renal pathologists who were unaware of group assignments. Pathological scores (0–5 points) were used to assess the extent of tubular damage.9
Immunohistochemistry
Immunohistochemical staining of LC3-II and caspase-3 protein were detected using a commercial chemical kit. Renal tissues, which had been fixed with 10% neutral buffered formalin, were embedded in paraffin and sectioned into 4 μm slices. The sections were then deparaffinized and hydrated. Afterward the antigen was thermally repaired and 3% H2O2 was used to block endogenous peroxidase. In order to minimize non-specific staining, 4% goat serum was used to block the sections. Samples were incubated with LC3-II and caspase-3 primary antibody (1:200 Signaling Technology, Danvers, MA, USA) overnight at 4°C. After incubation with secondary antibody at 37°C for 30 minutes, each section was treated with freshly prepared DAB color solution. Then, hematoxylin was used for counterstaining. Each sample was randomly selected from five different fields (×400) and professional image analysis software IP win32 (Acromag, Inc., Wixom, MI, USA) was used to evaluate the staining using ratio of integrated optical density (IOD).
TUNEL staining
Apoptosis was assessed according to the TUNEL method using the in situ cell death assay fluorescein kit (Shanghai Yisheng Biotechnology Co. Ltd, Shanghai, People’s Republic of China). Then 100 μL of proteinase K solution, at a concentration of 20 μg/mL, was added to each sample and incubated for 20 minutes at room temperature. After washing with PBS, 100 μL 1× equilibration buffer was added dropwise and incubated for 30 minutes at room temperature. TdT incubation buffer was then added to the tissue, incubated at 37°C for 60 minutes, and the nucleus was counterstained in the dark. Apoptotic cells were then observed under a 400× fluorescence microscope; apoptotic cells were bright red and were quantified. Renal tubular apoptotic cells were counted in 10 fields and then averaged.
Transmission electron microscopy
Renal tissue samples were harvested under general anesthesia after 24 hours of reperfusion. Approximately 1 mm3 of renal tissue was obtained from each animal, including a portion of renal cortex and outer medulla, and fixed in 4% glutaraldehyde for 24 hours at 4°C. The tissues were then rinsed, fixed, dehydrated, soaked, embedded, and sectioned at a thickness of 60–90-nm. Then an experienced pathologist who was unaware of the treatment observed autophagosome ultrastructure under a transmission electron microscope (Hitachi, Ltd., Tokyo, Japan).
Western blotting
Total proteins were extracted from renal issue using M-PER Mammalian Protein Extraction Reagent (Thermo Fisher Scientific, Waltham, MA, USA) and quantified using a Protein Assay kit (Bio-Rad, Hercules, CA, USA) according to the manufacturer’s protocol. Then, equal amounts of protein (40 μg/lane) were separated by electrophoresis on 10% sodium dodecyl sulfate-polyacrylamide gels and transferred to polyvinylidene fluoride membranes. The membranes were blocked with 5% non-fat milk in Tris-buffered saline and Tween 20 buffer for 2 hours at room temperature and then incubated with the following primary antibodies: LC3-I (1:500, DF7421; Affinity Biosciences, Cincinnati, OH, USA); LC3-II (1:1,000, AB128025; Abcam, Cambridge, UK); Beclin-1 (1:1,000, AB207612; Abcam); P62 (1:1,000, AB109012; Abcam); β-actin (1:200, AB207612; Abcam). After washing with TBST buffer, HRP-labeled secondary antibody (Wuhan Dr Bioengineering Co., Ltd. 1:2,000) was added. After incubation at 37°C for 2 hours, the secondary antibody was washed off and positive staining was imaged.
Statistical analysis
All data are described as mean and standard error of the mean (mean ± SEM). Comparisons between groups were performed by one-way ANOVA. All data analyses were carried out with GraphPad Prism 5 statistical software (GraphPad Software, Inc., La Jolla, CA, USA). P<0.05 was considered to be statistically significant.
Results
Renal function
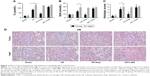
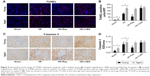
Renal function in the sham group was similar between young and aged rats. After I/R injury, the creatinine and urea nitrogen levels of the I/R group were significantly increased, compared with the sham group both in young and aged rats (P<0.05). Furthermore, levels of BUN and Cr were higher in aged rats than in young rats. In the young group, the application of rapamycin significantly improved renal function compared with the I/R group; 3-MA markedly (P<0.05). However, in the aged group, renal function was similar between the I/R group, I/R+Rap group and I/R+3-MA group (P>0.05) (Figure 1A and B).
Histological changes
Renal morphology was similar in the sham group in young and aged rats. Both in the young and aged I/R group, after 45 minutes ischemia and 24 hours reperfusion, renal tissue damage was obvious, including tubular epithelial cell swelling, brush border disappearance, tubular dilatation, cell shedding, cast formation and glomerular vascular congestion. (Figure 1D). Quantitative analysis showed that the kidney injury scores of the I/R group were significantly increased compared with sham group (P<0.05), and that the kidney injury score was more significant in the aged rats (Figure 1C and D). Compared with the I/R group, the application of rapamycin significantly improved renal tissue damage and reduced the injury score in the young group (P<0.05); a brush border of proximal renal tubular epithelial cells appeared and both cell swelling and cell shedding were reduced. The inhibition of autophagy remarkably increased renal tissue damage and increase the injury score in young groups of rats. However, in the aged groups, the protective effect of rapamycin did not exist. There was no significant difference between the I/R group, I/R+Rap group and I/R+3-MA group (P>0.05), which was consistent renal function findings.
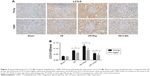
Expression of LC3-II by immunohistochemistry
Both in the young sham-operated group and the aged sham-operated group, the expression level of autophagy-related protein LC3-II was low. Compared with the sham group, the levels of LC3-II in the I/R group were significantly upregulated (Figure 2), which demonstrated that I/R injury could induce autophagy. Both in young and aged rats, compared with I/R, the expression of LC3-II increased after the application of rapamycin (P<0.05). Moreover, after I/R or rapamycin administration, the expression levels of LC3-II were significantly higher in the young group compared to the aged group (P<0.05). On the other hand, after the application of 3-MA, the expression of LC3-II decreased (P<0.05). Furthermore, the level of LC3-II was significantly lower in the aged group than in the young group (P<0.05).
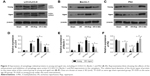
Expression of autophagy-related proteins by Western blot
In the sham group, Western blotting revealed low expression of autophagy-related proteins both in young and aged groups, including LC3-II and Beclin-1. Compared with the sham group, the expression of LC3-II and Beclin-1 in the I/R group was significantly upregulated (P<0.05), and the expression of P62 was significantly decreased both in the young group and aged group (P<0.05) (Figure 3). Both in the young group and aged group, the expression of the autophagy-related proteins LC3-II and Beclin-1, increased significantly after application with rapamycin compared with I/R groups (P<0.05), and the expression of P62 decreased significantly (P<0.05). On the other hand, after the application of 3-MA, the expression of LC3-II and Beclin-1 decreased, and the expression of P62 increased. Moreover, under the same interventions, the aged groups showed lower expression level of LC3-II and Beclin-1 but higher expression level of P62 compared with young groups, either in the I/R groups, I/R+Rap groups or I/R+3-MA groups (P<0.05).
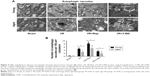
Autophagosomes in rats of different ages
Autophagosomes possessed a bilateral membrane structure under electron microscopy, and contained lysosome components and proteins that undergoing degradation (Figure 4). The number of autophagy bodies was similar in the sham group when compared between young and aged rats. After renal I/R injury, the number of autophagosomes increased in both young and aged groups (P<0.05). Moreover, the number of autophagosomes was significantly higher in the young I/R group than in aged I/R groups. After rapamycin administration, the number of autophagosomes increased in both the young group and aged groups (P<0.05), and the number of autophagosomes was significantly higher in the young group (P<0.05). Similarly, after 3-MA administration, the number of autophagosomes decreased significantly in both the young group and aged groups (P<0.05), and the number of autophagosomes was significantly smaller in the aged group (P<0.05).
Cellular apoptosis
Cellular apoptosis was examined using TUNEL staining (Figure 5) and evaluated by the immunohistochemical expression of caspase-3. The aged sham-operated rats had more apoptotic cells than young sham-operated rats (P<0.05). Renal I/R injury significantly upregulated the number of apoptotic cells in the kidneys of both young and aged rats. Moreover, there was a higher number of apoptotic cells in aged rats than in young rats. Compared with the I/R group, the application of rapamycin significantly reduced the number of apoptotic cells in young rats (P<0.05). Meanwhile, the inhibition of autophagy remarkably increased the number of apoptotic cells (P<0.05). However, in the aged rat model, there was no significant difference in the number of apoptotic cells when compared among the I/R group, I/R+Rap group and I/R+3-MA group (P>0.05).
The expression level of caspase-3 was lower in the young sham-operated group than in the aged sham-operated group. I/R injury significantly upregulated the expression of caspase-3 in the kidneys of both young and aged rats. Moreover, the expression level was higher in aged rats than in young rats. Compared with the I/R group, the application of rapamycin significantly downregulated the expression of caspase-3 in young rats, while the inhibition of autophagy remarkably upregulated the expression of caspase-3 (P<0.05). However, in the aged rat group, there was no significant difference in the expression of caspase-3 when compared among the I/R group, I/R+Rap group and I/R+3-MA group (P>0.05).
Discussion
Acute and severe trauma, such as acute kidney injury, is a common clinical disease associated with high morbidity and mortality. For severe trauma, the prognosis is different for young or old patients, and physicians in trauma and intensive care units have noticed differences in terms of response and prognosis for severe trauma when compared between children and adults.19 Some lines of evidence have also shown that age is an important factor which can influence the prognosis of liver transplantation and liver resection.20 Similarly, older patients are more sensitive to acute renal impairment.17 Our present study successfully established a model of renal ischemia-reperfusion (I/R) injury in young and aged rats, and the relationship between I/R injury and autophagy was studied. Based on changes in renal function and tissue damage scores, the results confirmed that the activation of autophagy significantly improved renal function and attenuated tissue damage induced by I/R injury in young rats but not elderly rats.
Interestingly, renal I/R injury activated autophagy both in young rats and aged rats compared to the sham-operated group and this activation could be enhanced or suppressed by rapamycin or 3-MA, respectively. These features could be demonstrated clearly by changes in the expression of autophagy-related proteins. Previous reports have found that autophagy-related proteins (such as LC3-II and Beclin-1) are elevated in renal I/R injury.21 Phosphatidylethanolamine converts LC3-I to LC3-II, which is involved in the construction of autophagosomes and promotes their fusion with lysosomes.9,22 Beclin-1 has been reported to play a key role in the formation of autophagosomes, enhancing autophagosome and lysosome fusion.10,23 Our results were consistent with the previous studies.9,10,21–23 In addition, the autophagy protein P62 is involved in the autophagy process and is degraded by autophagy, which is inversely related to autophagy activity.9 High expression levels of LC3-II and Belin-1 and low expression of P62 in the I/R+Rap group indicated that rapamycin activated autophagy, while low expression of LC3-II and Beclin-1 and high expression levels of P62 in the I/R+3-MA group demonstrated that 3-MA inhibited autophagy. These results were also verified by electron microscopy. However, in aged groups, the expression of autophagy-related proteins was lower and the number of autophagic vesicles was smaller compared with young rats under the same interventions. This may be the underlying reason for the loss of protective effect against autophagy in aged rats.
Apoptosis is an important factor in the development of acute renal failure after I/R injury. Caspase-3 is an important factor in the apoptosis of renal tubular epithelial cells and plays a key role in renal I/R injury. Studies have shown that caspase-3 is positively associated with kidney injury.24 Compared with the I/R group, the levels of BUN and Cr were decreased in the I/R+Rap group and the pathological score of renal injury was also decreased. Given that the number of TUNEL-positive cells was also decreased in this group, it is highly apparent that rapamycin can effectively inhibit apoptosis in young rats, which is consistent with previous studies.24–26 Unfortunately, in the aged rat model, the activation of autophagy had no effect on apoptosis after renal I/R, which may be the reason for the lack of a protective effect against renal I/R injury. These features were also demonstrated by the expression of caspase-3 and the number of apoptotic cells.
Mitochondria play a critical role in the aging process of animals, and studies suggest that mitochondrial volume increases in male rodents with aging, even in the absence of ischemia.27 With aging, mitophagy also decreases and excessive oxygen free radicals are generated, which will therefore aggravate I/R damage,28 leading to increased mitochondrial permeability transition pore opening (MPT) and increased levels of cell death.29 Previous studies have investigated protective measures for I/R injury and found that age is an important factor for increasing the susceptibility of tissues and organs to I/R injury.7,18,30 Our study firstly focused on the relationship between autophagy and renal I/R injury in young rats and in aged rats. According to our experiments, the enhancement of autophagy had no protective effect on I/R injury in aged rats. Moreover, with aging, the level of autophagy declined, which was consistent with the results of other studies.18
In aged rat kidneys, although autophagy could be activated, it was not protective against apoptosis. Collectively, these data allow us to make several bold speculations. Of course, body and cell aging were the most important factors. As the body ages, the number of intracellular damaged organelles or misfolded proteins increases. However, in our study, we found that the level of autophagy declined with aging, which was consistent with other studies.15 On the other hand, with aging, the secretion of hormones is known to decrease. Studies have shown that a decline in androgen levels would increase the susceptibility of tissue to I/R, and that the provision of an appropriate amount of androgen could reduce I/R injury.28–31 Interestingly, age-related estrogen deficiency also can also increase tissue susceptibility to I/R injury.28 Of course, changes in other hormones, such as glucocorticoids, can also affect autophagy activity.12
There are some limitations to our study which need to be considered. Firstly, we only studied male rats and did not consider effects in the female gender. Further studies are now required to investigate the specific mechanisms underlying renal I/R injury in aged rats.
Conclusion
The present study found that autophagy could be activated by renal I/R in both young and aged rat models and could be regulated by rapamycin and 3-MA, respectively. Enhancement of autophagy had significant protective effects on renal I/R injury in young rats but did not in aged rats. These findings demonstrated that the kidneys of older rats were refractory to protection induced by autophagy activation. The lack of protective effects related to the activation of might be associated with severe apoptosis and a reduction in the level of autophagy in aged rats.
Data availability
The data used to support the findings of this study are available from the corresponding authors upon request.
Acknowledgments
This study was supported by the Wuhan Morning Light Plan of Youth Science and Technology (No. 2017050304010281), Hubei Province Health and Family Planning Scientific Research Project (No. WJ2017M025 and No. WJ2017Z005), Natural Science Foundation of Hubei Province (No. 2017CFB181), and Research Project of Wuhan University (No. 2042017kf0097).
Disclosure
The authors report no conflicts of interest in this work.
References
Matsumoto T, Urushido M, Ide H, et al. Small heat shock protein Beta-1 (HSPB1) is upregulated and regulates autophagy and apoptosis of renal tubular cells in acute kidney injury. PLoS One. 2015;10(5):e0126229. | ||
Mehta RL, Burdmann EA, Cerdá J, et al. Recognition and management of acute kidney injury in the International Society of nephrology 0 by 25 global snapshot: a multinational cross-sectional study. Lancet. 2016;387(10032):2017–2025. | ||
Kunzendorf U, Haase M, Rölver L, Haase-Fielitz A. Novel aspects of pharmacological therapies for acute renal failure. Drugs. 2010;70(9):1099–1114. | ||
Venkatachalam MA, Weinberg JM, Kriz W, Bidani AK. Failed tubule recovery, AKI-CKD transition, and kidney disease progression. J Am Soc Nephrol. 2015;26(8):1765–1776. | ||
Xie Y, Xiao J, Fu C, Zhang Z, Ye Z, Zhang X. Ischemic preconditioning promotes autophagy and alleviates renal ischemia/reperfusion injury. Biomed Res Int. 2018;2018:1–11. | ||
Basile DP, Anderson MD, Sutton TA. Pathophysiology of acute kidney injury. Compr Physiol. 2012;2(2):1303–1353. | ||
Chen H, Xing B, Wang L, Weng X, Chen Z, Liu X. Aged kidneys are refractory to ischemic postconditioning in a rat model. Ren Fail. 2014;36(10):1575–1580. | ||
Sandal S, Bansal P, Cantarovich M. The evidence and rationale for the perioperative use of loop diuretics during kidney transplantation: a comprehensive review. Transplant Rev. 2018;32(2):92–101. | ||
Zhang YL, Zhang J, Cui LY, Yang S. Autophagy activation attenuates renal ischemia-reperfusion injury in rats. Exp Biol Med. 2015;240(12):1590–1598. | ||
Zhang LX, Zhao HJ, Sun DL, Gao SL, Zhang HM, Ding XG. Niclosamide attenuates inflammatory cytokines via the autophagy pathway leading to improved outcomes in renal ischemia/reperfusion injury. Mol Med Rep. 2017;16(2):1810–1816. | ||
Tang C, Han H, Yan M, et al. PINK1-PRKN/PARK2 pathway of mitophagy is activated to protect against renal ischemia-reperfusion injury. Autophagy. 2018;14(5):880–897. | ||
Xie Y, Jiang D, Xiao J, et al. Ischemic preconditioning attenuates ischemia/reperfusion-induced kidney injury by activating autophagy via the SGK1 signaling pathway. Cell Death Dis. 2018;9(3):338. | ||
Glick D, Barth S, Macleod KF. Autophagy: cellular and molecular mechanisms. J Pathol. 2010;221(1):3–12. | ||
Maiuri MC, Zalckvar E, Kimchi A, Kroemer G. Self-eating and self-killing: crosstalk between autophagy and apoptosis. Nat Rev Mol Cell Biol. 2007;8(9):741–752. | ||
Cai Z, Yan LJ. Rapamycin, autophagy, and Alzheimer’s disease. J Biochem Pharmacol Res. 2013;1(2):84–90. | ||
Klionsky DJ, Cuervo AM, Seglen PO. Methods for monitoring autophagy from yeast to human. Autophagy. 2007;3(3):181–206. | ||
van den Akker EK, Hesselink DA, Manintveld OC, et al. Ischemic postconditioning in human DCD kidney transplantation is feasible and appears safe. Transpl Int. 2014;27(2):226–234. | ||
Jankauskas SS, Pevzner IB, Andrianova NV, et al. The age-associated loss of ischemic preconditioning in the kidney is accompanied by mitochondrial dysfunction, increased protein acetylation and decreased autophagy. Sci Rep. 2017;7:44430. | ||
Calkins CM, Bensard DD, Moore EE, et al. The injured child is resistant to multiple organ failure: a different inflammatory response? J Trauma. 2002;53(6):1058–1063. | ||
Clavien PA, Selzner M, Rüdiger HA, et al. A prospective randomized study in 100 consecutive patients undergoing major liver resection with versus without ischemic preconditioning. Ann Surg. 2003;238(6):843–852. | ||
Lo S, Yuan SS, Hsu C, et al. LC3 over-expression improves survival and attenuates lung injury through increasing autophagosomal clearance in septic mice. Ann Surg. 2013;257(2):352–363. | ||
Livingston MJ, Dong Z. Autophagy in acute kidney injury. Semin Nephrol. 2014;34(1):17–26. | ||
Zhong Y, Wang QJ, Li X, et al. Distinct regulation of autophagic activity by Atg14L and rubicon associated with beclin 1-phosphatidylinositol-3-kinase complex. Nat Cell Biol. 2009;11(4):468–476. | ||
Kunduzova OR, Escourrou G, Seguelas MH, et al. Prevention of apoptotic and necrotic cell death, caspase-3 activation, and renal dysfunction by melatonin after ischemia/reperfusion. FASEB J. 2003;17(8):872–874. | ||
Padanilam BJ. Cell death induced by acute renal injury: a perspective on the contributions of apoptosis and necrosis. Am J Physiol Renal Physiol. 2003;284(4):F608–F627. | ||
Lee HT, Park SW, Kim M, et al. Interleukin-11 protects against renal ischemia and reperfusion injury. Am J Physiol Renal Physiol. 2012;303(8):F1216–F1224. | ||
Cury DP, Dias FJ, Sosthenes MC, et al. Morphometric, quantitative, and three-dimensional analysis of the heart muscle fibers of old rats: transmission electron microscopy and high-resolution scanning electron microscopy methods. Microsc Res Tech. 2013;76(2):184–195. | ||
Garvin AM, Aurigemma NC, Hackenberger JL, Korzick DH. Age and ischemia differentially impact mitochondrial ultrastructure and function in a novel model of age-associated estrogen deficiency in the female rat heart. Pflugers Arch. 2017;469(12):1591–1602. | ||
Hool LC. What cardiologists should know about calcium ion channels and their regulation by reactive oxygen species. Heart Lung Circ. 2007;16(5):361–372. | ||
Adam T, Sharp S, Opie LH, Lecour S. Loss of cardioprotection with ischemic preconditioning in aging hearts: role of sirtuin 1. J Cardiovasc Pharmacol Ther. 2013;18(1):46–53. | ||
Rubio-Gayosso I, Ramirez-Sanchez I, Ita-Islas I, et al. Testosterone metabolites mediate its effects on myocardial damage induced by ischemia/reperfusion in male Wistar rats. Steroids. 2013;78(3):362–369. |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.