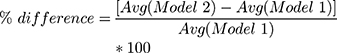Back to Journals » Medical Devices: Evidence and Research » Volume 14
Advanced Capnography Sampling Lines May Improve Patient Comfort and Compliance
Authors Blonder Y , Mazor E, Jew K
Received 20 November 2020
Accepted for publication 1 February 2021
Published 18 February 2021 Volume 2021:14 Pages 37—42
DOI https://doi.org/10.2147/MDER.S292526
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Video abstract of "New capnography sampling lines improve patient comfort and safety" [ID 292526].
Views: 630
Yedidia Blonder,1 Eytan Mazor,1 Korinne Jew2
1Research & Development, Medtronic, Inc., Jerusalem, Israel; 2Clinical Research and Medical Science, Medtronic, Boulder, CO, USA
Correspondence: Korinne Jew
Medtronic, 6135 Gunbarrel Ave., Boulder, CO, 80301, USA
Tel +1 303 305 2509
Email [email protected]
Purpose: Capnography monitoring in non-intubated patients requires the use of an end-tidal carbon dioxide (EtCO2) sampling line composed of a nasal or oral/nasal cannula connected to tubing that goes behind the ears to secure it in place. Some patients find wearing sampling lines to be uncomfortable, which can lead to compliance issues with monitoring. To address this important issue, we developed advanced sampling lines, designed to ameliorate the primary factors impacting patient tolerance.
Patients and Methods: A clinical evaluation was conducted to assess patient comfort level and wearing experience with the advanced sampling lines compared to the original sampling lines. Subjects were asked to wear the predicate line and the advanced line for 72 hours each, with individual testing periods separated by at least 48hrs. Subjects were asked to complete questionnaires assessing comfort and smell of the sampling lines at designated intervals throughout the trial process. In addition, a clinician assessed subjects’ skin during and after wearing each sampling line to determine if any skin irritation and disruption was evident.
Results: Repeated measures analysis demonstrated improved patient comfort with the advanced sampling line compared to the original line over the course of the wearing period (p< 0.05). Additionally, scores indicate that the smell of the advanced lines was perceived as less noticeable than the original line over time. No incidents of skin redness or irritation were reported for either sampling line.
Conclusion: The enhancements to the newly designed sampling lines improve the user experience, related to both line comfort and smell, which may increase patient compliance with monitoring.
Keywords: capnography, supplemental oxygen, end-tidal carbon dioxide, cannula, filter line
Plain Language Summary
Capnography monitoring, which measures a patient’s respiratory rate and the amount of carbon dioxide exhaled in each breath, is used to evaluate patient ventilatory status. This monitoring requires the use of a sampling line that is placed on a patient’s face, to allow measurement of the patient’s breath. In some cases, the sampling line can cause patient discomfort, which can lead to patients removing the monitor against clinicians’ wishes. The purpose of our study was to compare the comfort and wearing experience of an advanced sampling line, compared to an original sampling line. Participants wore each sampling line for 72 hours, and completed questionnaires focused on the comfort and smell of the two sampling lines. We found that compared to the original sampling line, participants rated the advanced sampling line as more comfortable with a less noticeable smell. Together, these results suggest that overall patient experience is improved using the advanced sampling line, which could increase patient compliance when undergoing capnography monitoring.
Introduction
Capnography is considered the reference gold standard measurement for respiratory rate monitoring1 and requires the patient to be connected to a bedside monitor via a cannula sampling end-tidal carbon dioxide (EtCO2) in each exhaled breath. Capnography sampling lines incorporate either a nasal or an oral/nasal cannula to sample exhaled breath and, in some models, deliver oxygen. In general, monitoring devices can interfere with patient movement and cause discomfort which may, in turn, impact clinician–patient interaction. Therefore, comfort of monitoring technologies should be maximized to ensure patient compliance and continuous monitoring.
While compliance with capnography monitoring may be improved when patients are educated on the importance of monitoring, research has shown that EtCO2 sampling lines are not well tolerated by some patients, which can result in non-compliance.2 Although not reported in the literature, clinician and patient feedback obtained via observation and informal interviews suggests that compliance issues stem primarily from discomfort at tubing–skin interfaces and a noticeable odor from the plastics used in manufacturing of the sampling lines (Medtronic Internal Data). Customer complaint data compiled between 2011 and 2015 indicate that issues of smell and skin irritation made up 82% of total complaints related to capnography sampling lines during this time period (Internal Medtronic data). To address these issues, the MicrostreamTM Advance sampling lines (Medtronic, Boulder, CO, USA), which are composed of new materials and incorporate design modifications to significantly enhance comfort and mitigate skin irritation, while minimizing odor, were created. While design changes may improve patient comfort, the more important outcome is patient safety which may be enhanced with increased compliance to monitoring.
A clinical evaluation was performed to assess patient comfort while wearing the device (specifically at patient–cannula interfaces), smell, and the tube fit around the ears, neck, and face (Figure 1). The primary objective of this study was to compare the comfort levels between the current sampling lines and advanced filter lines.
 |
Figure 1 Capnography sampling line with nasal prong oxygen delivery and oral EtCO2 capture. |
Patients and Methods
Previous Generation Sampling Lines
The Smart CapnoLine® H Plus EtCO2 sampling line (Medtronic, Boulder, CO, USA) was selected for testing as representative of existing technology. It has a large oral scoop designed to provide highly effective oral and nasal EtCO2 sampling. In addition, a unique oxygen delivery system within the cannula provides comfortable oxygen delivery for the patient and is designed to reduce the oxygen drying effect on sensitive mucus membranes. This original sampling line includes a dryer which reduces the sampled gas moisture content and is located near the oral-nasal cannula, sometimes coming into contact with the patient’s face.
Advanced Sampling Lines
The newly developed sample line portfolio is made with new materials and a novel configuration designed to enhance patient comfort and compliance. The oral/nasal carbon dioxide (CO2) filter line, selected as representative of the new advance portfolio, differs from the original device in the following ways:
- The dryer has been moved away from the face to improve comfort. To increase the usable life of the line, the dryer length was increased (from 4 to 6cm) because drying capacity is proportional to length. This modification resulted in a >25% increase in drying capacity and in turn, will extend the usable life of the dryer/sampling line (Internal Validation data).
- The cannula, connectors, and tubing are composed of phthalate-free polyvinyl chloride (PVC) compound with reduced rigidity, replacing di-(2-ethylhexyl) phthalate (DEHP) and diisononyl phthalate (DINP) plasticizers. The compound used to make the advanced sampling line yields a Shore durometer reading that is 25% lower than the material used to make the older lines; the lower score indicating less resistance to indentation and, therefore, a softer material. The softer DEHP-free PVC material was used to improve the feel of the skin–cannula interface on the face as well as the scent of the sampling line. The inner geometry of the oxygen delivery tubing was modified to include three “ribs” which reduce the impact of kinking by preserving airflow (Figure 2). This change was incorporated to support compliance with ISO 13544-2 and, more importantly, to ensure a continuous oxygen supply is maintained when lines are kinked. When lines are kinked, the new ribbed geometry minimizes the reduction in air flow to below 25% (Internal Medtronic data).
 |
Figure 2 Internal geometry of current and new capnography sampling lines. |
All other components are identical to the original design. The advanced oral/nasal CO2 filter line was utilized for testing as representative of performance enhancements made to the newly designed lines.
This study was conducted at the Israeli Skin Research Institute in accordance with the Helsinki Declaration and Good Clinical Practice. The study protocol and informed consent form (ICF) were reviewed and approved by the Maayaney Hayeshua Medical Center Ethics Committee, which serves as the ethics review board for the Institute. All subjects received an oral and written explanation of the study from the principal investigator and signed the ICF. Thirty healthy subjects, aged 50–83 years, provided written informed consent and were enrolled in the study. Older subjects were of interest as their skin is often more fragile and prone to breakdown than younger individuals. Subjects were required to wear each of the sampling lines for 72 hours while performing their normal daily activities. Subjects were asked to sleep with the sampling line and not to remove it for an extended period during the test. On a given day, subjects could remove the cannula for three-short and four-long breaks, 10 and 30 minutes per break, respectively. After wearing the first sampling line for the 72-hour study period, subjects were asked to repeat the process again with the other sampling line.
Prior to starting the study, subjects received training on sampling line use (wearing procedure), study instructions, and a Likert-scale questionnaire to rate their comfort and wearing experience. A cross-over design was used in which subjects were randomly assigned to the original or advanced oral/nasal CO2 filter line and, after completing a test cycle, switched to the other device to complete another test cycle. The subjects were periodically monitored throughout the study for proper sampling line usage. At the end of each test cycle, subjects completed questionnaires to assess to their comfort level and wearing experience.
Scoring Assessment
Each subject scored their comfort level and wearing experience (parameters listed below) using a Likert scale from 1 (not comfortable at all) to 7 (very comfortable).
- General comfort – Represents the overall subject experience interfacing with the device
- Comfort around the ears – Represents the interface with ears
- Comfort on the face – Represents the interface with face and cheeks
- Comfort in the nose – Represents the interface with nose, nostrils, and upper lip
- Smell – Represents the perception of the smell intensity and odor
The scoring was performed at the following time intervals: 5, 30, 90, 180, 360 minutes; 12, 24, 36 and 72 hours. Data from the different time intervals were grouped into the following time frames: initial use (0 to 1.5 hr), short term use (3 to 6 hr), first day (12–24 hr), second day (36–48 hr), and third day (60–72 hr). The subjects had a 48-hour break between model testing to prevent carryover effects. Additional qualitative data were gathered using open-ended questions at each scoring point. Additional remarks regarding comfort and smell were collected from open-ended questions and coded as either positive or negative utilizing key words such as “good”, “OK”, “comfortable”, etc. for positive and “discomfort”, “disturbance”, “pressure” “bad smell”, “nausea”, “headaches”, etc., for negative.
Additional clinical observations were performed by an expert physician for each subject before, during, and after wearing the device to assess the skin for reaction and presence of eczema using the Eczema Area and Severity Index (EASI).3 The EASI tool is used to assess the extent and severity of atopic dermatitis (AD) or eczema at four different body sites (head/neck, trunk, and upper and lower extremities) and diagnoses for erythema, induration/papulation, excoriation and lichenification. Only the face and neck were of interest in this study. The intensity of redness, thickness/swelling, scratching, and lichenification was scored from 0 to 3 (0 = Absent/None, 1 = Mild, 2 = Moderate, 3 = Severe).
Statistical Analysis
Differences between the comfort scores of the cannulas were assessed using an ANOVA test using a general linear model with a pairwise comparison, and adjustment for multiple comparisons using the Bonferroni method. An additional non-parametric test was performed to support the results by using Mood’s Median Test for a categorical factor (Device) and a continuous response (Comfort), due to chance of low confidence that the data for both groups have similar shaped distributions. The use of a non-parametric test to analyze Likert scale scores is conservative, and peer-reviewed articles suggest that parametric analysis may be used for Likert scales even for a small sample size and an abnormal distribution of the results.4 It was decided to include both parametric and non-parametric analyses and evaluate any differences between methods. There were no significant differences in the results between the parametric and the non-parametric analyses. The percentage difference for each subject’s scoring between the advanced sampling line (Model 2) and original sampling line (Model 1) was calculated to represent a specific user’s interpretation of the scoring scale used, giving equal weight to those recording scores lower on the scale.
Results
The study included 30 subjects (19 females) with a mean age of 62.5 years. Results demonstrate that the advanced oral/nasal CO2 filter line was more comfortable than the original design (Table 1). Subjects reported significantly improved comfort compared to the original sampling line around ears by hour 36 (Table 2). The comfort score for the nose interface improved significantly after the first 24 hours (Table 2). The scores indicated that the smell was perceived as noticeably better than the predicate over the course of testing. At no point during the study period was the original line scored higher in comfort than the advanced line (Table 2). In most testing time segments, the advanced line had a double-digit percent improvement over the predicate (Table 3).
 |
Table 1 Overall Differences in Comfort Score Means Between Original and Advanced Sampling Lines Across All Time Periods |
 |
Table 2 Statistical Comparison Using ANOVA General Linear Model and Comparison with Bonferroni Simultaneous Tests for Differences of Means |
 |
Table 3 Overall Percentage Change in Comfort Scores Between the Original (Model 1) and Advanced (Model 2) Sampling Lines Across Time Periods |
No subjects experienced a skin reaction, as scored with the Eczema Area and Severity Index, with either sampling line during the study periods as assessed by an expert clinician.
Assessment of the qualitative responses shows that subjects reported more negative assessments of the original sampling line, including discomfort around the mouth and nose (Original line: Oral = 84 comments, Nose= 63 comments vs Advanced line: Oral= 59 comments, Nose=37 comments). Subjects gave 4 times as many negative remarks regarding smell with the original sampling line compared to the advanced (110 remarks vs 27 remarks).
Discussion
The main objective of this study was to assess if the design changes made to the advanced oral/nasal CO2 filter line, which are also incorporated into the portfolio of non-intubated filter lines, improved patient comfort compared to the original design. The design changes included softer tubes extruded from compounds with diminished smell, and the movement of the dryer away from the patient’s face. A softer tube decreases the friction between the cannula and the patient’s cheeks, ears and neck. Using a different compound with less volatile components gradually reduces the smell intensity inhaled by the patient during use, especially when oxygen is being delivered via the cannula. Improvements in comfort may make wearing the device more tolerable, resulting in greater compliance with capnography monitoring.
Overall, the subjects reported that advanced oral/nasal CO2 filter line was more comfortable than original filter line. No skin reaction was observed after using either cannula. Additionally, the advanced oral/nasal CO2 filter lines had significantly better smell. Based on the subjects’ qualitative remarks, we conclude that softer tubes and the removal of the dryer from the face had a noticeable impact on patient comfort.
The study was limited by a small sample size of healthy subjects. As subjects were not monitored continuously during their wearing of the devices, it is possible that the testing protocol was not strictly followed. However, records indicate that valid data were collected from subjects in most cases (ie, 27 of 30 valid cases for the predicate line and 26 of 30 valid cases for the advance line).
Conclusions
The enhancements to the newly designed sampling lines improve both the user experience, which could impact patient compliance, and the performance of the sampling line itself. Increased compliance with monitoring may improve patient safety and so too will the improved kink resistance of the sampling lines by maintaining an uninterrupted oxygen supply to patients.
Abbreviations
AD, atopic dermatitis; ANOVA, analysis of variance; CO2, carbon dioxide; DEHP, di-(2-ethylhexyl) phthalate; DINP, diisononyl phthalate; EASI, eczema area and severity index; EtCO2, end-tidal carbon dioxide; ICF, informed consent form; ISO, international organization for standardization; PVC, polyvinyl chloride.
Acknowledgments
We would like to acknowledge the technical contributions of Raz Levin (Medtronic).
Funding
The study was funded by Medtronic, Inc.
Disclosure
The authors are or have been employees of Medtronic, Inc. The authors report no other conflicts of interest in this work.
References
1. Liu G-Z, Wu D, Mei Z-Y, Zhu Q-S, Wang L. Automatic detection of respiratory rate from electrocardiogram, respiration induced plethysmography and 3D acceleration signals. J Cent South Univ. 2013;20(9):2423–2431. doi:10.1007/s11771-013-1752-z
2. Miller KM, Kim AY, Yaster M, et al. Long-term tolerability of capnography and respiratory inductance plethysmography for respiratory monitoring in pediatric patients treated with patient-controlled analgesia. Paediatr Anaesth. 2015;25(10):1054–1059. doi:10.1111/pan.12702
3. Hanifin JM, Thurston M, Omoto M, Cherill R, Tofte SJ, Graeber M. The eczema area and severity index (EASI): assessment of reliability in atopic dermatitis. EASI Evaluator Group. Exp Dermatol. 2001;10(1):11–18. doi:10.1034/j.1600-0625.2001.100102.x
4. Norman G. Likert scales, levels of measurement and the “laws” of statistics. Adv Health Sci Educ Theory Pract. 2010;15(5):625–632. doi:10.1007/s10459-010-9222-y
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.