Back to Journals » OncoTargets and Therapy » Volume 8
ABCB1 C3435T polymorphism is associated with leukemia susceptibility: evidence from a meta-analysis
Authors Ma L, Ruan L, Liu H, Yang H, Feng Y
Received 3 February 2015
Accepted for publication 18 March 2015
Published 5 May 2015 Volume 2015:8 Pages 1009—1015
DOI https://doi.org/10.2147/OTT.S82144
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Professor Daniele Santini
Limin Ma,1 Linhai Ruan,1 Hongchao Liu,2 Haiping Yang,1 Yanming Feng2
1Department of Hematology, The First Affiliated Hospital, 2Medical College, Henan University of Science and Technology, Luoyang, People’s Republic of China
Introduction and objective: Many studies have been conducted on the association between the adenosine triphosphate-binding cassette, subfamily B, member 1 (ABCB1) gene C3435T polymorphism and leukemia risk, however, the previously published findings remain controversial. Thus, a meta-analysis was carried out to accurately evaluate the effect of this polymorphism on leukemia susceptibility.
Methods: A computerized literature search was conducted of PubMed, Elsevier database, the China National Knowledge Infrastructure database, and Wanfang Database, to find published case–control studies exploring the relationship between ABCB1 C3435T polymorphism and leukemia risk. Odds ratios (ORs) with 95% confidence intervals (CIs) were applied to assess the strength of association.
Results: A total of 17 studies of 2,431 cases and 3,028 controls were included in this meta-analysis. The results of overall comparisons suggest that there is a significant association between ABCB1 C3435T polymorphism and leukemia risk under two genetic models (TT vs CC: OR=1.39, 95% CI=1.04–1.84, P=0.02; CT+TT vs CC: OR=1.20, 95% CI=1.06–1.36, P=0.004). In the subgroup analyses by ethnicity, age, and leukemia subtype, a significant association was found in Caucasian (CT vs CC: OR=1.22, 95% CI=1.03–1.45, P=0.02; TT vs CC: OR=1.34, 95% CI=1.10–1.64, P=0.004; CT+TT vs CC: OR=1.27, 95% CI=1.08–1.49, P=0.004), adult leukemia (CT vs CC: OR=1.46, 95% CI=1.17–1.83, P=0.001; CT+TT vs CC: OR=1.43, 95% CI=1.01–2.03, P=0.04), and lymphocytic leukemia (TT vs CC: OR=1.73, 95% CI=1.19–2.51, P=0.004; TT vs CC+CT: OR=1.62, 95% CI=1.10–2.38, P=0.01; CT+TT vs CC: OR=1.28, 95% CI=1.10–1.48, P=0.001).
Conclusion: The meta-analysis suggests that ABCB1 C3435T polymorphism is associated with increased risk of leukemia.
Keywords: adenosine triphosphate-binding cassette, subfamily B, member 1 gene, multidrug-resistance gene, meta-analysis
Introduction
Leukemia is a group of malignant diseases of the hematopoietic system, with an estimated 54,270 new cases and 24,450 deaths expected in the USA in 2015.1 Although the incidence of leukemia increases gradually, with age the most prominent risk factor, the etiology and mechanisms underlying the pathogenesis of leukemia are not yet fully understood.2 Epidemiological studies have found that advanced age, radiation, smoking, and exposure to chemical carcinogens are risk factors which contribute to the genesis of leukemia.3–5 However, only a small proportion of people exposed to these risk factors develop leukemia, suggesting that host genetic factors might play an important role.6 Recent genome-wide association studies have identified the presence of inherited genetic susceptibility to this disease.7–9 Therefore, multiple factors are considered implicated in the etiology of leukemia, including exogenous or endogenous exposure, genetic susceptibility, and chance.2
The human adenosine triphosphate-binding cassette, subfamily B, member 1 (ABCB1) gene, also named multidrug-resistance 1 (MDR1) gene, is located at 7q21.1, with 28 exons encoding a 170 kDa membrane transporter called P-glycoprotein (P-gp). The presence of a highly conserved adenosine triphosphate (ATP)-binding site in two homologous halves as well as the linker region makes this protein a member of the adenosine triphosphate-binding cassette (ABC) superfamily.10 P-gp acts as an efflux pump in an ATP-dependent fashion, transporting exogenous and endogenous substrates from the inside of cells to the outside. P-gp was first identified in human cancer cells as a protein responsible for resistance against many anticancer drugs. Subsequently, this efflux transporter has been found in various normal human tissues, including in the intestinal epithelium, adrenal gland, placenta, kidney, liver, capillary endothelial cells of the brain, and testes. Physiological expression of P-gp in excretory tissues provides a cellular defense mechanism against potentially harmful compounds.10–12 ABCB1 is polymorphic and at least 50 single-nucleotide polymorphisms (SNPs) have been identified within ABCB1 gene locus.13 C3435T at exon 26 is the most widely investigated SNP of ABCB1 and has been associated with altered P-gp expression and activity in tissue studies including those on placenta, liver, and leukocytes.14–16 There is increasing evidence that genetic variants of ABCB1 affect P-gp activity and expression levels, and the alteration of P-gp transport activity results in decreased extrusion of harmful xenobiotics and cumulative cytotoxicity.17,18 The C3435T SNP in ABCB1 has been associated with the development of various cancers, including breast cancer, hepatocellular carcinoma, and non-Hodgkin lymphoma.19–21 Many case–control studies have been conducted to investigate whether the ABCB1 C3435T polymorphism is associated with leukemia risk but these have yielded controversial results. Therefore, we performed a meta-analysis to accurately evaluate the effect of ABCB1 C3435T polymorphism on leukemia susceptibility.
Materials and methods
Study identification
A systematic literature search of the PubMed, Elsevier, China National Knowledge Infrastructure, and Wanfang databases was conducted to identify studies that explored the relationship between ABCB1 C3435T polymorphism and leukemia risk. The search terms and keywords were as follows: “multidrug resistance gene” or “MDR1” or “ABCB1”, “polymorphism” or “variant” or “mutant”, and “leukemia” or “leukaemia” or “leukocythemia”. No language restriction was applied and the latest search was undertaken on April 30, 2014. The references cited in the eligible studies were also examined to find additional studies. Two reviewers examined the retrieved literature independently and disagreement was resolved by discussion.
Inclusion criteria
All studies included in this meta-analysis had to meet the following criteria. They had to: (1) be case–control studies assessing the relationship between ABCB1 C3435T polymorphism and leukemia risk; (2) have confirmed diagnosis in the case group; and (3) have genotype frequencies for both cases and controls available. Reviews, meta-analyses, case reports, and letters were excluded.
Data extraction
Two reviewers independently extracted information from each eligibility study and disagreements were addressed by discussion. The following data were extracted from each study: author(s), year of publication, country, ethnicity, source of controls, genotyping methods, sample size of cases and controls, genotype frequencies of the ABCB1 C3435T polymorphism for cases and controls, and Hardy–Weinberg equilibrium (HWE) of control group.
Statistical analysis
The HWE of the genotype distribution in the control groups was checked using the χ2 test and P-values <0.05 were designated as deviations from HWE. The strength of association between ABCB1 C3435T polymorphism and leukemia risk was assessed by odds ratios (ORs) with 95% confidence intervals (CIs) under the heterozygote model (CT vs CC), homozygote model (TT vs CC), dominant model (CT+TT vs CC), and recessive model (TT vs CC+CT). The significance of the combined OR was determined by the Z test. The Q-statistic test was used to evaluate the between-study heterogeneity. If P<0.05, indicating that there was significant heterogeneity, the random-effects model (DerSimonian–Laird) was applied to combine the data, otherwise, the fixed-effects model (Mantel–Haenszel) was selected. Subgroup analyses were performed by ethnicity, age, and leukemia subtype to find the source of heterogeneity. Funnel plots were produced to evaluate publication bias and sensitivity analysis was performed by omission of studies not in agreement with HWE to assess the stability of the results. All the tests were two-sided and P-values <0.05 were considered as statistically significant. The data analyses were performed using Review Manager software (v 5.2; The Cochrane Collaboration, Oxford, UK).
Results
Characteristics of studies
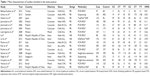
In accordance with the inclusion criteria, a total of 17 publications with 2,431 leukemia cases and 3,028 controls were included in the meta-analysis presented here.22–38 The main characteristics of included studies are summarized in Table 1. All of the 17 studies were case–control studies, 14 of which were population-based controls and the remaining three were hospital-based controls. All the cases had a confirmed diagnosis of leukemia and the controls were mainly matched for age and sex. Among the included studies, six studies were conducted in an Asian population,25,28,31,34,36,38 nine studies in a Caucasian population,22–24,26,29,30,32,33,35 and two studies in a mixed population.27,37 The distribution of genotypes among the control groups in four studies was not in agreement with HWE.22,33,35,36
Meta-analysis results
Table 2 lists the main results of this meta-analysis. When all the eligible studies were pooled into the meta-analysis, a significant association between ABCB1 C3435T polymorphism and leukemia risk was observed under two genetic models (TT vs CC: OR=1.39, 95% CI=1.04–1.84, P=0.02; CT+TT vs CC: OR=1.20, 95% CI=1.06–1.36, P=0.004) (Table 2). Subgroup analyses were also performed to explore the effect of ethnicity, age, and leukemia subtype. When stratified by ethnicity, a significantly increased risk was observed in Caucasians (CT vs CC: OR=1.22, 95% CI=1.03–1.45, P=0.02; TT vs CC: OR=1.34, 95% CI=1.10–1.64, P=0.004; CT+TT vs CC: OR=1.27, 95% CI=1.08–1.49, P=0.004) (Table 2). Significant associations were also found in adult leukemia (CT vs CC: OR=1.46, 95% CI=1.17–1.83, P=0.001; CT+TT vs CC: OR=1.43, 95% CI=1.01–2.03, P=0.04) (Table 2, Figure 1) and lymphocytic leukemia (TT vs CC: OR=1.73, 95% CI=1.19–2.51, P=0.004; TT vs CC+CT: OR=1.62, 95% CI=1.10–2.38, P=0.01; CT+TT vs CC: OR=1.28, 95% CI=1.10–1.48, P=0.001) (Table 2, Figure 2).
Sensitivity analysis and publication bias
Sensitivity analysis was performed by excluding the studies in which the distribution of genotypes in the control groups was not in agreement with HWE. The results show that the statistical significance was not changed, suggesting that the results of this meta-analysis are relatively stable and credible. Four funnel plots were produced to assess the publication bias and the shapes of funnel plots reveal no evidence of obvious asymmetry, indicating there is no statistical evidence of publication bias (Figure 3).
  | Figure 3 Funnel plot for the publication bias under dominant model (CT+TT vs CC). |
Discussion
At least 50 SNPs have been described in all 28 exons of the ABCB1 gene and the C3435T (rs1045642) polymorphism is a synonymous variant with no effect on amino-acid change at codon 1142 (Ile1142Ile) in the second ATP-binding domain.13,17 Studies have shown that 3435T is associated with lower ABCB1 expression levels and subsequently reduces P-gp activity and alters substrate specificity.14,39,40 P-gp is not only expressed in tumor cells, but also in cells of several normal tissues. It was detected in the apical membranes of excretory tissues, such as the intestine, kidney, and liver, suggesting its role in the elimination of xenobiotics.41,42 Besides, P-gp is also expressed in peripheral leukocytes and hematopoietic stem cells from which acute myeloid leukemia originates, which indicates that ABCB1 may play an important role in the etiology of leukemia.43,44 Functional polymorphisms in ABCB1 may cause various human malignancies including leukemia. However, the exact biological mechanism for the association of ABCB1 polymorphisms with leukemia risk still requires explanation through further studies.
Given the potential influence of this functional polymorphism on cancer susceptibility, many molecular epidemiological studies have been conducted to investigate the association between ABCB1 C3435T polymorphism and leukemia risk. However, the results from different studies are controversial, which may be owing to different genetic backgrounds in individual studies. To synthetically evaluate the effect of ABCB1 C3435T polymorphism on susceptibility to leukemia, we performed a meta-analysis of 17 case–control studies that assessed the relationship between ABCB1 C3435T polymorphism and leukemia risk. In this meta-analysis, in overall comparisons under a homozygote model and dominant model, we found that the ABCB1 3435TT genotype significantly increases leukemia risk. When stratified by ethnicity, the results indicate that the individuals with a TT genotype in the C3435T of ABCB1 had significantly increased risk of leukemia among Caucasian populations, but not in other ethnicities. An explanation for this is that the racial differences in leukemia incidence can partly be attributed to differences in genotype frequencies between different populations at ABCB1 loci. In the subgroup analysis by age, we found significant association between ABCB1 C3435T polymorphism and increased risk in adult leukemia in the heterozygote model and dominant model, but not in children with leukemia under any comparison models. When restricting the analysis to the subtype of leukemia, significant association was found in lymphocytic leukemia in the homozygote model, recessive model, and dominant model, but not in myeloid leukemia in any comparison model, which indicates that clinical type might have a critical effect on the association.
The results of this study show that ABCB1 C3435T polymorphism might modify susceptibility to leukemia, which is consistent with the meta-analysis reported by Qian et al.45 Since our study combined a total of 2,431 cases and 3,028 controls from 17 case–control studies, our results are more convincing.
Limitations
Some limitations of this meta-analysis should be taken into consideration and caution is needed when the results are interpreted. First, non-differential misclassification bias is possible, as the controls were not uniformly defined. Population-based controls and hospital-based controls have different risks of evolving leukemia. Second, our analyses were based on estimates not adjusted for other risk factors such as folate-intake status, lifestyle, and environmental exposures, which might influence the combined results. Third, although subgroup analyses were performed by ethnicity, age, and leukemia subtype to find the sources of heterogeneity, significant heterogeneity still existed in some subgroups. The heterogeneity may have been caused by different lifestyles, exposure to different risk factors, and the different levels of exposure to risk factors. In addition, due to the limited original data, potential gene–gene and gene–environment interactions which have an important impact on leukemia risk were not evaluated in this study.
Conclusion
This meta-analysis found that ABCB1 C3435T polymorphism is associated with an increased risk of leukemia and is likely a risk factor facilitating leukemia development. However, well-designed case–control studies with larger sample size focusing on more ethnicities or leukemia subtypes are required to validate our findings in the future.
Disclosure
The authors report no conflicts of interest in this work.
References
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2015. CA Cancer J Clin. 2015;65(1):5–29. | ||
Inaba H, Greaves M, Mullighan CG. Acute lymphoblastic leukaemia. Lancet. 2013;381(9881):1943–1955. | ||
Finch SC. Radiation-induced leukemia: lessons from history. Best Pract Res Clin Haematol. 2007;20(1):109–118. | ||
Björk J, Johansson B, Broberg K, Albin M. Smoking as a risk factor for myelodysplastic syndromes and acute myeloid leukemia and its relation to cytogenetic findings: a case-control study. Leuk Res. 2009;33(6):788–791. | ||
Lichtman MA. Obesity and the risk for a hematological malignancy: leukemia, lymphoma, or myeloma. Oncologist. 2010;15(10):1083–1101. | ||
Mullighan CG. Genetic variation and the risk of acute lymphoblastic leukemia. Leuk Res. 2010;34(10):1269–1270. | ||
Sherborne AL, Hosking FJ, Prasad RB, et al. Variation in CDKN2A at 9p21.3 influences childhood acute lymphoblastic leukemia risk. Nat Genet. 2010;42(6):492–494. | ||
Wang Y, Chen J, Li J, et al. Association of three polymorphisms in ARID5B, IKZF1 and CEBPE with the risk of childhood acute lymphoblastic leukemia in a Chinese population. Gene. 2013;524(2):203–207. | ||
Speedy HE, Di Bernardo MC, Sava GP, et al. A genome-wide association study identifies multiple susceptibility loci for chronic lymphocytic leukemia. Nat Genet. 2014;46(1):56–60. | ||
Schinkel AH. The physiological function of drug-transporting P-glycoproteins. Semin Cancer Biol. 1997;8(3):161–170. | ||
Sun J, He ZG, Cheng G, Wang SJ, Hao XH, Zou MJ. Multidrug resistance P-glycoprotein: crucial significance in drug disposition and interaction. Med Sci Monit. 2004;10(1):RA5–RA14. | ||
Hodges LM, Markova SM, Chinn LW, et al. Very important pharmacogene summary: ABCB1 (MDR1, P-glycoprotein). Pharmacogenet Genomics. 2011;21(3):152–161. | ||
Breier A, Barancík M, Sulová Z, Uhrik B. P-glycoprotein – implications of metabolism of neoplastic cells and cancer therapy. Curr Cancer Drug Targets. 2005;5(6):457–468. | ||
Hoffmeyer S, Burk O, von Richter O, et al. Functional polymorphisms of the human multidrug-resistance gene: multiple sequence variations and correlation of one allele with P-glycoprotein expression and activity in vivo. Proc Natl Acad Sci U S A. 2000;97(7):3473–3478. | ||
Kurata Y, Ieiri I, Kimura M, et al. Role of human MDR1 gene polymorphism in bioavailability and interaction of digoxin, a substrate of P-glycoprotein. Clin Pharmacol Ther. 2002;72(2):209–219. | ||
Lamba J, Strom S, Venkataramanan R, et al. MDR1 genotype is associated with hepatic cytochrome P450 3A4 basal and induction phenotype. Clin Pharmacol Ther. 2006;79(4):325–338. | ||
Marzolini C, Paus E, Buclin T, Kim RB. Polymorphisms in human MDR1 (P-glycoprotein): recent advances and clinical relevance. Clin Pharmacol Ther. 2004;75(1):13–33. | ||
Brambila-Tapia AJ. MDR1 (ABCB1) polymorphisms: functional effects and clinical implications. Rev Invest Clin. 2013;65(5):445–454. | ||
Baldissera VD, de Mattos AA, Coral GP, et al. Evaluation of the C3435T polymorphism in the MDR1 gene in patients with hepatocellular carcinoma. Ann Hepatol. 2012;11(6):899–906. | ||
Wang Z, Wang T, Bian J. Association between MDR1 C3435T polymorphism and risk of breast cancer. Gene. 2013;532(1):94–99. | ||
Kim HN, Kim NY, Yu L, et al. Polymorphisms in DNA repair genes and MDR1 and the risk for non-Hodgkin lymphoma. Int J Mol Sci. 2014;15(4):6703–6716. | ||
Jamroziak K, Młynarski W, Balcerczak E, et al. Functional C3435T polymorphism of MDR1 gene: an impact on genetic susceptibility and clinical outcome of childhood acute lymphoblastic leukemia. Eur J Haematol. 2004;72(5):314–321. | ||
Jamroziak K, Balcerczak E, Cebula B, Janus A, Mirowski M, Robak T. No influence of 3435C>T ABCB1 (MDR1) gene polymorphism on risk of adult acute myeloid leukemia and P-glycoprotein expression in blast cells. Ther Drug Monit. 2006;28(5):707–711. | ||
Jamroziak K, Balcerczak E, Smolewski P, et al. MDR1 (ABCB1) gene polymorphism C3435T is associated with P-glycoprotein activity in B-cell chronic lymphocytic leukemia. Pharmacol Rep. 2006;58(5):720–728. | ||
Hattori H, Suminoe A, Wada M, et al. Regulatory polymorphisms of multidrug resistance 1 (MDR1) gene are associated with the development of childhood acute lymphoblastic leukemia. Leuk Res. 2007;31(12):1633–1640. | ||
Urayama KY, Wiencke JK, Buffler PA, Chokkalingam AP, Metayer C, Wiemels JL. MDR1 gene variants, indoor insecticide exposure, and the risk of childhood acute lymphoblastic leukemia. Cancer Epidemiol Biomarkers Prev. 2007;16(6):1172–1177. | ||
Leal-Ugarte E, Gutiérrez-Angulo M, Macías-Gómez NM, et al. MDR1 C3435T polymorphism in Mexican children with acute lymphoblastic leukemia and in healthy individuals. Hum Biol. 2008;80(4):449–455. | ||
Liu RR. The role of multidrug resistance gene 1 polymorphism to prognosis of adult acute lymphoblastic leukemia patients. Central South University. 2008;5:1–48. | ||
Semsei AF, Erdélyi DJ, Ungvári I, et al. Association of some rare haplotypes and genotype combinations in the MDR1 gene with childhood acute lymphoblastic leukaemia. Leuk Res. 2008;32(8):1214–1220. | ||
Rocha V, Porcher R, Fernandes JF, et al. Association of drug metabolism gene polymorphisms with toxicities, graft-versus-host disease and survival after HLA-identical sibling hematopoietic stem cell transplantation for patients with leukemia. Leukemia. 2009;23(3):545–556. | ||
Rao DN, Anuradha C, Vishnupriya S, et al. Association of an MDR1 gene (C3435T) polymorphism with acute leukemia in India. Asian Pac J Cancer Prev. 2010;11(4):1063–1066. | ||
Penna G, Allegra A, Alonci A, et al. MDR-1 polymorphisms (G2677T and C3435T) in B-chronic lymphocytic leukemia: an impact on susceptibility and prognosis. Med Oncol. 2011;28(4):1549–1554. | ||
Bektaş-Kayhan K, Küçükhüseyin Ö, Karagöz G, et al. Is the MDR1 C3435T polymorphism responsible for oral mucositis in children with acute lymphoblastic leukemia? Asian Pac J Cancer Prev. 2012;13(10):5251–5255. | ||
Lü H, Du ZZ, Wang W, et al. [Relationship between genetic polymorphism of multidrug resistance 1 gene and the risk of childhood acute lymphocytic leukemia.] Zhonghua Er Ke Za Zhi. 2012;50(9):692–696. Chinese. | ||
Gréen H, Falk IJ, Lotfi K, et al. Association of ABCB1 polymorphisms with survival and in vitro cytotoxicity in de novo acute myeloid leukemia with normal karyotype. Pharmacogenomics J. 2012;12(2):111–118. | ||
Singh O, Chan JY, Lin K, Heng CC, Chowbay B. SLC22A1-ABCB1 haplotype profiles predict imatinib pharmacokinetics in Asian patients with chronic myeloid leukemia. PLoS One. 2012;7(12):e51771. | ||
Vivona D, Bueno CT, Lima LT, et al. ABCB1 haplotype is associated with major molecular response in chronic myeloid leukemia patients treated with standard-dose of imatinib. Blood Cells Mol Dis. 2012;48(2):132–136. | ||
Zhai X, Wang H, Zhu X, et al. Gene polymorphisms of ABC transporters are associated with clinical outcomes in children with acute lymphoblastic leukemia. Arch Med Sci. 2012;8(4):659–671. | ||
Song P, Lamba JK, Zhang L, et al. G2677T and C3435T genotype and haplotype are associated with hepatic ABCB1 (MDR1) expression. J Clin Pharmacol. 2006;46(3):373–379. | ||
Kimchi-Sarfaty C, Oh JM, Kim IW, et al. A “silent” polymorphism in the MDR1 gene changes substrate specificity. Science. 2007;315(5811):525–528. | ||
Terao T, Hisanaga E, Sai Y, Tamai I, Tsuji A. Active secretion of drugs from the small intestinal epithelium in rats by P-glycoprotein functioning as an absorption barrier. J Pharm Pharmacol. 1996;48(10):1083–1089. | ||
Tanigawara Y. Role of P-glycoprotein in drug disposition. Ther Drug Monit. 2000;22(1):137–140. | ||
Elliott JI, Raguz S, Higgins CF. Multidrug transporter activity in lymphocytes. Br J Pharmacol. 2004;143(7):899–907. | ||
Pawlik A, Baskiewicz-Masiuk M, Machalinski B, Kurzawski M, Gawronska-Szklarz B. Involvement of C3435T and G2677T multidrug resistance gene polymorphisms in release of cytokines from peripheral blood mononuclear cells treated with methotrexate and dexamethasone. Eur J Pharmacol. 2005;528(1–3):27–36. | ||
Qian X, Cao S, Yang G, et al. Variant genotypes of MDR1 C3435T increase the risk of leukemia: evidence from 10 case-control studies. Leuk Lymphoma. 2012;53(6):1183–1187. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.