Back to Journals » Patient Preference and Adherence » Volume 12
A patient-reported, non-interventional, cross-sectional discrete choice experiment to determine treatment attribute preferences in treatment-naïve overactive bladder patients in the US
Authors Athavale A , Gooch K, Walker D , Suh M, Scaife J , Haber A, Hadker N, Dmochowski R
Received 29 June 2018
Accepted for publication 19 September 2018
Published 12 October 2018 Volume 2018:12 Pages 2139—2152
DOI https://doi.org/10.2147/PPA.S178668
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Johnny Chen
Amod Athavale,1 Katherine Gooch,2 David Walker,2 Marissa Suh,1 Jillian Scaife,1 Ali Haber,1 Nandini Hadker,1 Roger Dmochowski3
1Trinity Partners LLC, Waltham, MA, USA; 2Astellas Pharma Global Development, Inc., Northbrook, IL, USA; 3Urologic Surgery, Vanderbilt University, Nashville, TN, USA
Purpose: Many pharmacotherapeutic treatment options are available for the symptoms of overactive bladder (OAB), each offering varying efficacy, safety, and tolerability profiles that must be carefully considered when selecting treatment. The objective of the present study was to characterize pharmacotherapy treatment preferences of individuals with symptoms of OAB and to examine how preferences differ by both patient characteristics and disease burden metrics.
Patients and methods: Patient preferences for OAB treatment attributes were examined using a discrete choice experiment (DCE). Attributes were identified through literature review, clinical relevance, and input from patients. Eligible respondents were required to be ≥18 years of age, have a self-reported physician OAB diagnosis or have self-reported symptoms of OAB, and be naïve to pharmacotherapy or invasive OAB treatments. A hierarchical Bayesian random-effects-only model was used to estimate the mean relative preference weights and mean relative importance scores of treatment attributes. Multivariable linear regression models with backward selection were used to analyze the differences in relative importance scores by demographic characteristics and disease burden-related metrics.
Results: In total, 514 individuals participated in the study. Most respondents were <65 years of age (66.0%), female (68.5%), and reported moderate/severe OAB symptoms (64.2%). Overall, respondents placed the greatest importance on drug delivery method, with a preference for oral and patches over injectables, followed by efficacy defined as reduced daytime micturition and out-of-pocket cost. Multivariable linear regression analyses revealed that females were less likely to select injectables, that symptom control of incontinence was the most important to respondents who reported greater work productivity loss, and that out-of-pocket cost was the most important to respondents with moderate/severe OAB.
Conclusion: OAB treatment preferences vary depending on individual patient characteristics and disease severity. Overall, drug delivery method, reduced daytime micturition, and out-of-pocket costs were the most important treatment attributes among treatment-naïve individuals with symptoms of OAB. Preferences for OAB treatment were also found to vary depending on patient demographics and disease comorbidities, which has previously not been examined.
Keywords: overactive bladder, discrete choice experiment, treatment preferences
Introduction
Overactive bladder (OAB) is a common condition, affecting both men and women, with as many as 35.6% of men and women ≥40 years of age reporting OAB symptoms in the US.1 The International Continence Society (ICS) defines OAB as urgency, with or without urge incontinence, usually with frequency and nocturia, in the absence of infection or significant urological pathology.2 Individuals affected by OAB are burdened by the frequent occurrence of these symptoms, which may limit everyday activities.3 Furthermore, feelings of anxiety, frustration, or embarrassment surrounding their OAB may cause psychological stress, all of which negatively impact the quality of life (QoL).3 Despite the substantial burden associated with OAB symptoms, OAB is largely an undertreated condition. In a large retrospective database study in the US, only 24.4% of patients with OAB filled a prescription for an OAB medication in 2007.4
Oral pharmacotherapy is the mainstay treatment for OAB; common therapies include muscarinic antagonists (antimuscarinics) and the β3-adrenoreceptor agonist mirabegron, both with distinct profiles in terms of treatment effectiveness in symptom reduction, tolerability, and side effects. Additionally, patients must also consider different drug delivery methods when selecting treatments, for example, onabotulinumtoxinA injection into the bladder for OAB treatment. Patients place great value on the benefits and risks associated with treatments when making treatment decisions, which may vary based on a variety of patient-related factors, including symptom severity. By understanding which treatment characteristics are most important to patients, physicians can learn why some patients choose not to select treatment and can develop tailored treatment plans, which may result in more effective OAB management.5
Patient preferences and trade-offs for OAB therapies have been quantitatively assessed among individuals with OAB;6–8 however, little is known on how treatment preferences change based on severity of OAB or other clinical and patient characteristics. Although past discrete choice experiments (DCEs) have included a mix of treated and untreated patients,6,8 specific preferences among treatment-naïve patients have not been examined.5 Furthermore, previous DCEs on patient preferences for OAB therapies have been conducted in Europe and the United Kingdom,6–8 and applicability of these results to other countries is unknown. The primary objective of this study was to characterize the pharmacotherapy treatment preferences of treatment-naïve individuals with symptoms of OAB in the US. Additional analyses were performed to further examine how treatment preferences differ by both patient demographics and disease comorbidities.
Materials and methods
Study design and population
The DCE was conducted between October and November 2017. Respondents were sampled from a large independent respondent panel (Toluna, Inc.) that was designed to be demographically representative of the US general population. As traditional statistical power calculations cannot offer accurate estimates of sample size in DCE studies,1,10–12 the present study relied on the work conducted by Yang et al9 to identify the minimum sample size necessary. Based on the 10-attribute grid with three levels per attribute and no opt-out option, a target sample size of 500 respondents was set, with minimum recruitment quotas to ensure there was an analyzable sample in each group: sex, age, disease severity and physician diagnosis, urge urinary incontinence status, and nocturia status (Table 1). Respondents were eligible if they were ≥18 years of age, able to read and understand English, able to use a computer with Internet access, and have self-reported physician OAB diagnosis or have self-reported symptoms of OAB. Symptoms of OAB were defined using the criteria developed by Hall et al,10 which included self-reported assessment of urinary daytime frequency, urgency, and incontinence. Individuals who reported any of the following were excluded: current or previous use of oral OAB treatment (antimuscarinics or β3-adrenoreceptor agonists) or topical gels (oxybutynin), current or previous invasive treatment of OAB (onabotulinumtoxinA injections, peripheral tibial nerve stimulation, sacral neuromodulation, surgery), current diagnosis of urinary tract infection or interstitial cystitis, current use of indwelling catheter or practicing intermittent self-catheterization, current or previous malignant disease of the pelvic organs, previous pelvic radiation therapy, and women who are currently pregnant or have given birth in the past 3 months. Potential respondents were emailed an invitation to participate. The email invitation did not specify the specific topic of interest in order to minimize self-selection into the survey. Respondents received reward points in exchange for their time, which could be redeemed for a variety of items including magazines or coupons. Prior to beginning the survey, all respondents were informed that the survey was completely voluntary and that they could withdraw at any time. Completion of the survey was deemed to be informed consent.
  | Table 1 Minimum sample size quotas |
Demographic and OAB-specific information was collected pertaining to predefined comparator groups to facilitate subgroup analysis: sex, age, frailty, disease state, incontinence status, nocturia status, and physician diagnosed OAB.
Ethical approval was obtained for this study from the University of Mississippi Institutional Review Board.
Survey development
A DCE was designed to quantify the strength of preference for, as well as trade-offs between, pharmacotherapy treatment attributes among treatment-naïve OAB respondents and was developed in line with the International Society for Pharmacoeconomics and Outcomes Research best practice guidelines.11 A DCE can capture preferences of patients regarding different treatment characteristics. In a DCE, respondents are presented with a series of hypothetical treatment profile sets that describe attributes such as treatment efficacy, treatment side effects, dosage, costs, or health states and are then asked to select the profile they prefer. The resulting respondent choices reveal the relative importance (RI) of these attributes and an underlying utility function.
Attributes and levels were selected based on reviews of the literature, as well as in consultation with a clinical expert and patients.7 Attributes and levels were included specific to incontinence, nocturia, urgency of urination, daytime micturition frequency, renal side effects, cardiovascular side effects, drug delivery method, and out-of-pocket costs (Figure 1). Risk levels of kidney or bladder-related side effects and heart rate or blood pressure-related side effects (low, moderate, and high risk) were also included. All treatment profiles were based on current pharmacotherapy treatment attributes; however, no reference to specific treatments was included.
  | Figure 1 Attribute grid. |
In addition to the DCE, a descriptive survey and validated questionnaires were also administered. Validated instruments included Overactive Bladder Questionnaire Short Form (OAB-q sf)12 assessing bother associated with OAB and impact on health-related quality of life (HRQoL); Overactive Bladder Satisfaction Questionnaire (OAB-S Pre-medication Module)13 assessing OAB control expectations and impact on life; Bladder Health Section of the Boston Area Community Health study questionnaire14 assessing symptomology; Fatigue, Resistance, Ambulation, Illness, and Loss of Weight (FRAIL) Questionnaire15 assessing frailty status; and the Work Productivity and Activity Impairment – Specific Health Problem Questionnaire (WPAI-SHP)16 assessing work productivity which was administered only to respondents who indicated working full or part time. Additional survey topics included diagnosis and initial perceptions; current symptoms and perceptions; impact of OAB on personal, social, and professional life; current health and lifestyle; and demographics. Scale scores for the OAB-q sf, OAB-S, and the WPAI-SHP range from 0 to 100. For symptom bother a higher score on the OAB-q SF is indicative of greater symptom bother while in the measurement of HRQoL, a higher score represents greater HRQoL. For the OAB-S, a higher score represents higher expectations from treatment and greater satisfaction with ability to perform daily activities. Across the different aspects of the WPAI-SHP, a higher score is indicative of greater impairment and more missed work. The FRAIL scale scores range from 0 to 5: a score of 0=robust, 1–2=pre-frail, and 3–5=frail health status. Prior to the deployment of the DCE survey, cognitive interviews were conducted among four OAB patients (two mild and two moderate/severe as per the criteria developed by Hall et al10), who were not included in the full DCE analysis, to identify potential sources of response error in the DCE and the descriptive survey. These respondents received a $50 honorarium. The cognitive interviews were conducted via WebEx. No sources of response error were identified.
Experiment and survey design
SAS 9.4 (SAS Institute Inc., Cary, NC, USA) choice model design macros were used to optimize the DCE design efficiency, correlations between attribute levels (Figure 1), and number of choice tasks.17–19 A balanced and orthogonal fractional factorial design was selected. SAS 9.4 choice model design macros – %mktruns, %mktex, and %mkteval – were used to select the number of choice sets, the number of treatments presented in each choice set, and which attribute levels should be shown within each of those choice sets. The %mktruns macro suggests possible design sizes, the %mktex macro creates a design with the maximum D efficiency (to maximize the precision of the estimated parameters), and the %mkteval macro is used to evaluate the design.19 Based on the 10-attribute grid with three levels per attribute, a 27-hypothetical profile design was finalized. Respondents were provided with nine choice tasks, each containing three hypothetical treatments to select between based on the values from the attribute grid. No “opt-out” option was provided.
Statistical analysis
The burden of illness associated with OAB in the overall sample as well as within various subgroups (eg, male vs female, age <65 years vs ≥65 years, <75 years vs ≥75 years, mild vs moderate-to-severe disease), with respect to various clinical and nonclinical outcomes such as levels of worry/anxiety, depression, fatigue/insomnia, mental health, impact on social life, and HRQoL was described. Scale scores were calculated for the OAB-q SF (OAB severity score, HRQoL score), OAB-s Pre-Medication Module (control expectation score, impact on daily living with OAB score), WPAI questionnaire specific to OAB (absenteeism score, presenteeism score, work productivity loss score, activity impairment score), and FRAIL (frailty score), by using specific algorithms for each scale. Differences in these scale scores between the comparative groups were tested using multivariable linear regression models.
A hierarchical Bayesian random-effects-only model was used to estimate relative preferences of OAB treatment attributes.20 In this model, choices made on each choice set functioned as the dependent variable, and the attribute levels were the independent variables. This analysis generated respondent-level relative preference weights, which can be interpreted as utilities, and converted into respondent-level RI scores. These respondent-level relative preference weights were used to calculate sample mean relative preference weights. A mean RI >10 indicated higher importance compared to baseline, where all attributes would have the same importance. The posterior distributions from the hierarchical Bayesian models were displayed as mean ± SD.
The relationship between the validated scales and mean RI scores was assessed using multivariable linear regression models using the scale scores as independent variables and RI scores associated with each attribute as the dependent variable. The analysis utilized an effect coding framework for categorical variables, whereby a coefficient value for each level of each attribute was calculated relative to a weighted average of all levels. The results of the multivariable linear regression analysis are presented as parameter estimates (PEs) with 95% CIs.
Identification of potential confounders (ie, willingness to try pharmacological treatment, flavored drinks consumption [20 oz bottles], WPAI work productivity loss score) involved investigating bivariate relationships between the covariates and the independent and dependent variables, using independent sample t-tests with Bonferroni correction for multiple comparisons.
Initial multivariable models included covariates found to be at least moderately statistically significant (P<0.2) in the bivariable analyses. Backward variable selection was used to arrive at the most parsimonious multivariable model for each relationship.
Results
Patient characteristics
A total of 18,445 individuals were invited to participate in the online survey, among whom 776 (4.2%) met the eligibility criteria and 514 (66.2%) completed the survey (Table 2). Overall, respondents were predominantly female (68%) and <65 years of age (66%). Most respondents reported moderate/severe OAB (64.2%) and experienced incontinence (79.4%) and/or nocturia (59.1%). Only 184 (35.8%) reported being diagnosed by a physician with OAB, among whom 157 (85.3%) reported moderate/severe OAB and 27 (14.7%) reported mild OAB. Among respondents <65 years of age, 49% reported full-time employment, compared to 5.1% among respondents ≥65 years of age. Ten percent of respondents <65 years of age were retired, compared to 83.4% of respondents ≥65 years of age. The majority of respondents had some college or AA degree (39.3%), followed by college graduate level or above (36.4%) and high school graduate/General Education Diploma or equivalent (22.2%).
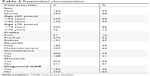  | Table 2 Respondent characteristics |
Burden of OAB among respondents
The burden of OAB among the study respondents are summarized in Table S1. Statistically significant associations were noted in all of the following: Female respondents were more likely than male respondents to report greater symptom bother (mean ± SD OAB-q scores: 48.3±8.0 vs 37.1±7.6) that OAB symptoms bother them most of the time (OAB-S scores: 61.8±25.7 vs 72.0±23.2) and dissatisfaction with their QoL (OAB-q scores: 63.2±26.5 vs 72.0±24.5) measures. Symptom bother increased with the level of frailty, with reported OAB-q scores for frail, pre-frail, and robust respondents of 54.4±6.6, 48.0±8.1, and 37.4±7.8, respectively. Furthermore, respondents identified as frail were more likely to report OAB symptoms impact them some or all of the time (OAB-S scores: 49.9±22.3 vs 74.1±21.5) and greater impacts on satisfaction with QoL compared to robust respondents (OAB-q scores: 54.0±24.3 vs 75.2±22.7) and WPAI activity impairment (56.3±26.8 vs 35.8±26.6). Moderate/severe respondents reported that bladder symptoms impact them most of the time (OAB-S scores: 56.3±24.4 vs 80.6±18.6) and lower QoL (OAB-q scores: 56.1±25.4 vs 83.6±16.7), as well as a greater impact on work productivity compared to respondents with mild OAB symptoms. Respondents reporting incontinence (50.2±7.7 vs 23.8±6.2) and nocturia (54.6±7.4 vs 30.5±7.0) were more likely to report greater symptom bother on the OAB-q, compared to respondents without incontinence or nocturia. Overall, respondents who had been diagnosed with OAB were more likely than respondents with undiagnosed OAB to report that bladder symptoms impact them most of the time (OAB-S scores: 53.5±24.1 vs 71.4±23.7) and lower QoL (OAB-q scores: 53.3±25.5 vs 73.0±23.8) as well as greater symptom bother (OAB-q scores: 59.7±6.9 vs 36.4±7.5). Symptom bother appeared to decrease with increasing age, with respondents <65 years of age reporting greater symptom bother compared to respondents ≥65 years of age. A similar trend was observed for QoL and work productivity, where respondents <65 years of age reported worse QoL and work productivity impairment compared to respondents ≥65 years of age.
After controlling for potential confounders, results from the multivariable linear regression analysis highlighted that among female respondents, neither age nor frailty was observed to be strongly associated with symptom bother (data not shown). However, a strong association was observed among female respondents reporting severe/moderate OAB severity, incontinence, nocturia, diagnosed OAB, and greater symptom bother. Furthermore, reporting moderate/severe OAB symptoms was associated with a 13.8 decrease in QoL scores.
DCE results
Relative mean preference weights
The relative mean preference weights for the attribute levels are summarized in Table 3. Patient preferences were largely driven by a low preference for drug delivery method via bladder injection (−89.4±63.1), as well as treatments with high daytime micturition frequency (16 times/24 hours: −59.5±37.4). Respondents stated preferences for the lowest incidence of nocturia (30.1±47.1), kidney/bladder-related side effects (27.4±40.7), dry mouth (28.4±29.3), and heart rate/blood pressure-related side effects (21.7±39.7), and for those that offered the most benefit with respect to urgency of urination (18.9±35.0). Respondents also stated a preference for treatments with a moderate level of incontinence (ie, 1–2 times/24 hours: 17.2±25.8) and a moderate risk (5%) of constipation (27.6±33.7). Respondents were also more likely to prefer treatments with low out-of-pocket costs (34.0±53.1).
  | Table 3 Relative preference weights, overall |
Relative mean importance scores
The relative mean importance scores are presented in Figure 2. Three attributes achieved RI scores >10, indicating higher importance. Overall, drug delivery method emerged as the most important attribute, with respondents stating a strong preference for oral and patches over injectable therapies (RI: 18.8±8.5). Respondents also placed great importance on reduced daytime micturition frequency (RI: 11.9±4.5) and lower out-of-pocket costs (RI: 11.3±7.6).
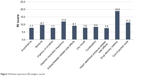  | Figure 2 Relative importance (RI) weights, overall. |
Bivariate and multivariable regression results
Preferences were fairly consistent across subgroups (data not shown). Female respondents were more risk averse in their treatment preferences and were less likely than their male counterparts to prefer injectable treatments (RI: 19.4 vs 17.5). Statistically significant associations were observed between respondents <65 years of age and treatments that reduced nocturia (RI: 10.0) and incontinence (RI: 8.1). Respondents ≥65 years of age were statistically significantly associated with a preference for treatments that offered reduced daytime micturition frequency (RI: 12.4). Respondents with moderate/severe OAB considered out-of-pocket costs the most important treatment attribute, with a statistically significant association compared to respondents with mild OAB (RI: 12.0 vs 10.0). Respondents with incontinence were more likely to state a preference for a mild level of urgency (RI: 7.7 vs 7.6) than those without incontinence. Similarly, there was a statistically significant association among respondents with nocturia and their preference for treatments that reduced nocturia (RI: 10.2).
Results from the multivariable regression analyses are summarized in Table 4. After controlling for potential confounders, female respondents continued to consider drug delivery method to be more important compared to males (PE: 2.57; 95% CI: 0.29–4.86). Respondents with greater symptom bother had low preference for drug delivery method (PE: −0.07; 95% CI: −0.11 to −0.03). Respondents who reported greater work productivity loss were less concerned with daytime micturition frequency (PE: −0.04; 95% CI: −0.07 to −0.01) and, likewise, were willing to compromise in favor of better reduction of incontinence (PE: 0.03; 95% CI: 0.00–0.06) and urgency (data not shown). Respondents with moderate/severe OAB continued to consider out-of-pocket costs as the most important treatment attribute (PE: 2.38; 95% CI: 0.90–3.85) and considered reduced urgency of urination as less important (PE: −1.05; 95% CI: −1.91 to −0.20) compared to mild patients. Respondents with nocturia continued to consider reduced nocturia as the most important treatment attribute (PE: 1.60; 95% CI: 0.49–2.72) and when compared to respondents with no nocturia were less concerned with a better kidney/bladder side effect profile (PE: −0.99; 95% CI: −1.90 to −0.07). Compared with robust respondents, pre-frail respondents considered reduced incontinence as an important treatment attribute (PE: 1.49; 95% CI: 0.05–2.94). Also, respondents ≥65 years of age were less concerned with cardiovascular side effects, as compared to respondents <65 years of age (PE: −1.12; 95% CI: −2.22 to −0.02).
Discussion
The results from this study highlight OAB treatment attributes that are important to treatment-naïve individuals with symptoms of OAB residing in the US, with drug delivery method being the most important feature, followed by reduced daytime micturition frequency and out-of-pocket costs. Preferences for OAB treatment were also found to vary depending on patient demographics and disease comorbidities, which have previously not been explored.
This study builds the findings of Heisen et al7 in the evaluation of patient and physician preferences for oral pharmacotherapy for OAB across five European countries, which suggest that while physicians place greater importance on increasing the benefits associated with oral pharmacotherapy, patient preferences emphasize limiting risks of side effects. An earlier DCE conducted by Swinburn et al6 found that respondents place particular emphasis on improving clinical symptoms (eg, incontinence, urgency micturition, constipation, and dry mouth). Lastly, Decalf et al8 explored the importance of treatment-related side effects and cognitive effects of oral antimuscarinics among older (≥65 years) individuals with OAB. In that study, respondents placed the greatest importance on the avoidance of severe cognitive effects, followed by constipation, blurred vision, and dry mouth. These DCEs have only considered oral pharmacotherapies, thereby excluding treatment attributes specific to onabotulinumtoxinA.
The present study was limited to treatment-naïve individuals with OAB and therefore the relationship between treatment history and preference for drug delivery methods could not be examined. However, the strong preference for oral and patches over injectable therapies reported in this study highlights how important drug delivery method was among treatment-naïve individuals with OAB. Similar to the present study, in a time trade-off study that examined patient preferences among female patients with different levels of OAB severity, a greater preference was stated for less invasive treatments (ie, anticholinergic and β3 adrenergic receptor medications) over more invasive treatments, such as botulinum toxin injections and sacral neuromodulation.21 With a greater understanding of relationships between treatment preferences and patient characteristics, physicians can develop tailored treatment plans accordingly, which may result in more effective OAB management.5
This study does have several limitations. Current guidelines on the design of DCE studies highlight that most DCEs include between three and seven attributes.11 Although the present study included ten attributes, these were carefully reviewed against clinical criteria and determined to best represent the hypothetical treatment profiles necessary for the DCE. As the study was limited to individuals with Internet access, the generalizability of the results is limited to this population. Internet access may have also been a contributing factor in narrowly missing the quota for respondents 75–80 years of age (target 80, actual 72). Generalizability is also impacted in several important ways: 1) the use of convenience sampling to recruit respondents, 2) the reliance on self-reported physician OAB diagnosis or self-reported symptoms of OAB reported, and 3) the inherent selection biases in the populations that participate in surveys. To overcome these limitations, the included sample was selected to reflect US sociodemographic characteristics, and several questions were included to identify OAB, including preestablished criteria to define OAB symptoms developed by Hall et al.10 Furthermore, all responses relating to the respondents’ diagnosis experience and initial perceptions of OAB and OAB treatment were also subject to recall bias, although questions were carefully worded to minimize recall issues. Lastly, as the present study limited its examination to pharmacotherapy treatments for OAB, patient preferences for non-pharmacologic treatments, such as behavior modification, were not examined. Despite these limitations, the results from this study provide important information on patient preferences for OAB pharmacotherapy treatment in a novel patient population.
Conclusion
The results from this study highlight treatment attributes that are of the greatest importance to treatment-naïve OAB patients in general, but also how these preferences vary depending on clinical and patient-related factors. Overall, drug delivery method emerged as the most important treatment attribute, a preference that remained constant in the subgroup analysis for respondents who were female, older, and having mild OAB. Additionally, reduced daytime micturition frequency and out-of-pocket costs arose as important OAB treatment attributes. The multivariable regression analyses revealed that females were less likely to select injectable therapies, that symptom control of incontinence was most important to respondents who reported greater work productivity loss, and that out-of-pocket cost was most important to respondents with moderate/severe OAB. A better understanding of which treatment characteristics are most important to patients can assist physicians in the treatment decision-making process for OAB and provide important insight for future pharmacotherapy treatment preferences for OAB.
Acknowledgments
We would like to thank Meagan Harwood, MPH, and Dr Karissa Johnston, PhD, for drafting, reviewing, and editing this manuscript. Meagan Harwood and Dr Johnston are employees of Broadstreet Health Economics & Outcomes Research, which received funding from Astellas in the conduct of this study. The abstract of this paper was presented at the ICS as an eposter presentation. Both the abstract and eposter are available on the congress website at https://www.ics.org/2018/abstract/154.
Disclosure
Walker and Gooch are/were employees of Astellas Pharma Global Development, Inc., at the time of study completion. Athavale, Suh, Scaife, Haber, and Hadker are employees of Trinity Partners LLC, which received funding from Astellas for this study. Dmochowski provided consultation services in the conduct of this study, which was funded by Astella. The authors report no other conflicts of interest in this work.
References
Coyne KS, Sexton CC, Vats V, Thompson C, Kopp ZS, Milsom I. National community prevalence of overactive bladder in the United States stratified by sex and age. Urology. 2011;77(5):1081–1087. | ||
Abrams P, Cardozo L, Fall M, et al. The standardisation of terminology in lower urinary tract function: report from the standardisation sub-committee of the International Continence Society. Urology. 2003;61(1):37–49. | ||
Abrams P, Kelleher CJ, Kerr LA, Rogers RG. Overactive bladder significantly affects quality of life. Am J Manag Care. 2000;6(11 Suppl):S580–S590. | ||
Helfand BT, Evans RM, Mcvary KT. A comparison of the frequencies of medical therapies for overactive bladder in men and women: analysis of more than 7.2 million aging patients. Eur Urol. 2010;57(4):586–591. | ||
Drutz HP. Overactive bladder: the importance of tailoring treatment to the individual patient. J Multidiscip Healthc. 2011;4:233–237. | ||
Swinburn P, Lloyd A, Ali S, Hashmi N, Newal D, Najib H. Preferences for antimuscarinic therapy for overactive bladder. BJU Int. 2011;108(6):868–873. | ||
Heisen M, Baeten SA, Verheggen BG, et al. Patient and physician preferences for oral pharmacotherapy for overactive bladder: two discrete choice experiments. Curr Med Res Opin. 2016;32(4):787–796. | ||
Decalf VH, Huion AMJ, Benoit DF, Denys MA, Petrovic M, Everaert K. Older People’s Preferences for Side Effects Associated with Antimuscarinic Treatments of Overactive Bladder: A Discrete-Choice Experiment. Drugs Aging. 2017;34(8):615–623. | ||
Yang J-C, Johnson FR, Kilambi V, Mohamed AF. Sample size and utility-difference precision in discrete-choice experiments: A meta-simulation approach. Journal of Choice Modelling. 2015;16:50–57. | ||
Hall SA, Link CL, Hu JC, Eggers PW, Mckinlay JB. Drug treatment of urological symptoms: estimating the magnitude of unmet need in a community-based sample. BJU Int. 2009;104(11):1680–1688. | ||
Reed Johnson F, Lancsar E, Marshall D, et al. Constructing experimental designs for discrete-choice experiments: report of the ISPOR Conjoint Analysis Experimental Design Good Research Practices Task Force. Value Health. 2013;16(1):3–13. | ||
Coyne KS, Thompson CL, Lai JS, Sexton CC. An overactive bladder symptom and health-related quality of life short-form: validation of the OAB-q SF. Neurourol Urodyn. 2015;34(3):255–263. | ||
Piault E, Evans CJ, Espindle D, Kopp Z, Brubaker L, Abrams P. Development and validation of the Overactive Bladder Satisfaction (OAB-S) Questionnaire. Neurourol Urodyn. 2008;27(3):179–190. | ||
Mckinlay JB, Link CL. Measuring the urologic iceberg: design and implementation of the Boston Area Community Health (BACH) Survey. Eur Urol. 2007;52(2):389–396. | ||
Morley JE, Malmstrom TK, Miller DK. A simple frailty questionnaire (FRAIL) predicts outcomes in middle aged African Americans. J Nutr Health Aging. 2012;16(7):601–608. | ||
Reilly MC, Zbrozek AS, Dukes EM. The validity and reproducibility of a work productivity and activity impairment instrument. Pharmacoeconomics. 1993;4(5):353–365. | ||
Huber J, Zwerina K. The Importance of Utility Balance in Efficient Choice Designs. Journal of Marketing Research. 1996;33(3):307–317. | ||
Kuhfeld WF, Tobias RD, Garratt M. Efficient Experimental Design with Marketing Research Applications. Journal of Marketing Research. 1994;31(4):545–264. | ||
Kuhfeld WF. Marketing Research Methods in SAS: Experimental Design, Choice, Conjoint, and Graphical Techniques. 2010. Available from: http://support.sas.com/techsup/technote/mr2010.pdf. Accessed December 1, 2017. | ||
Hauber AB, González JM, Groothuis-Oudshoorn CG, et al. Statistical Methods for the Analysis of Discrete Choice Experiments: A Report of the ISPOR Conjoint Analysis Good Research Practices Task Force. Value Health. 2016;19(4):300–315. | ||
Wu JM, Fulton RG, Amundsen CL, Knight SK, Kuppermann M. Patient preferences for different severities of and treatments for overactive bladder. Female Pelvic Med Reconstr Surg. 2011;17(4):184–189. |
Supplementary material
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


