Back to Journals » Journal of Pain Research » Volume 10
A double-blind, randomized, comparative study of the use of a combination of uridine triphosphate trisodium, cytidine monophosphate disodium, and hydroxocobalamin, versus isolated treatment with hydroxocobalamin, in patients presenting with compressive neuralgias
Authors Goldberg H ![]() , Mibielli MA
, Mibielli MA ![]() , Nunes CP
, Nunes CP ![]() , Goldberg SW, Buchman L, Mezitis SG, Rzetelna H, Oliveira L
, Goldberg SW, Buchman L, Mezitis SG, Rzetelna H, Oliveira L ![]() , Geller M
, Geller M ![]() , Wajnsztajn F
, Wajnsztajn F
Received 23 September 2016
Accepted for publication 30 December 2016
Published 15 February 2017 Volume 2017:10 Pages 397—404
DOI https://doi.org/10.2147/JPR.S123045
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Micheal Schatman
Henrique Goldberg,1 Marco Antonio Mibielli,2 Carlos Pereira Nunes,2 Stephanie Wrobel Goldberg,3 Luiz Buchman,4 Spyros GE Mezitis,5 Helio Rzetelna,6 Lisa Oliveira,2 Mauro Geller,2 Fernanda Wajnsztajn7
1UERJ Medical School, Rio de Janeiro, Brazil; 2UNIFESO Medical School, Teresópolis, Brazil; 3Washington University School of Medicine, St Louis, MO, USA; 4Instituto de Pós-Graduação Médica Carlos Chagas (ICC), Rio de Janeiro, Brazil; 5New York-Presbyterian Hospital/Weill-Cornell Medical Center, New York, NY, USA; 6Santa Casa da Misericórdia do Rio de Janeiro, Rio de Janeiro, Brazil; 7New York-Presbyterian Hospital/Columbia University Medical Center, Neurology, New York, NY, USA
Context: This paper reports on the results of treatment of compressive neuralgia using a combination of nucleotides (uridine triphosphate trisodium [UTP] and cytidine monophosphate disodium [CMP]) and vitamin B12.
Objectives: To assess the safety and efficacy of the combination of nucleotides (UTP and CMP) and vitamin B12 in patients presenting with neuralgia arising from neural compression associated with degenerative orthopedic alterations and trauma, and to compare these effects with isolated administration of vitamin B12.
Methods: A randomized, double-blind, controlled trial, consisting of a 30-day oral treatment period: Group A (n=200) receiving nucleotides + vitamin B12, and Group B (n=200) receiving vitamin B12 alone. The primary study endpoint was the percentage of subjects presenting pain visual analog scale (VAS) scores ≤20 at end of study treatment period. Secondary study endpoints included the percentage of subjects presenting improvement ≥5 points on the patient functionality questionnaire (PFQ); percentage of subjects presenting pain reduction (reduction in VAS scores at study end in relation to pretreatment); and number of subjects presenting adverse events.
Results: The results of this study showed a more expressive improvement in efficacy evaluations among subjects treated with the combination of nucleotides + vitamin B12, with a statistically significant superiority of the combination in pain reduction (evidenced by VAS scores). There were adverse events in both treatment groups, but these were transitory and no severe adverse event was recorded during the study period. Safety parameters were maintained throughout the study in both treatment groups.
Conclusion: The combination of uridine, cytidine, and vitamin B12 was safe and effective in the treatment of neuralgias arising from neural compression associated with degenerative orthopedic alterations and trauma.
Keywords: uridine triphosphate trisodium, cytidine monophosphate disodium, hydroxocobalamin, compressive neuralgia
Introduction
Uridine and cytidine are substances belonging to the pyrimidine nucleotide family, which in turn comprises one of four main classes of biological molecules (along with carbohydrates, lipids, and proteins) that play several fundamental roles in cellular metabolism. Aside from being components of DNA and participating in genome transcription and translation, functions of these nucleotides include energy transfer for chemical bonding, intracellular signaling, neurotransmission, and transfer of biochemical radicals from intermediate metabolism.1
Vitamin B12 is essential in the human body for growth and maintenance of a variety of processes, including cell reproduction and synthesis of myelin and nucleoproteins, and must be supplied from an exogenous source.2 Adequate vitamin B12 levels in the body depend on adequate ingestion and absorption (requiring an acid-pepsin complex in the stomach that releases cobalamin from its binding proteins, pancreatic proteins that release cobalamin from the R factor bond, secretion of intrinsic factor by gastric parietal cells in order to bind cobalamin, and the presence of cobalamin and intrinsic factor receptors in the ileum.3,4 Vitamin B12 is important to nerve metabolism via the remethylation of homocysteine to methionine for de novo synthesis of s-Adenosyl methionine.5 Vitamin B12 deficiency can be caused by numerous factors, usually some combination of inadequate ingestion and absorption.6 Deficiency can lead to megaloblastic anemia and neurological alterations, the latter likely linked to the absence of s-Adenosyl methionine formation.7,8
This trial assessed treatment outcomes following administration of a combination of uridine triphosphate trisodium (UTP), cytidine monophosphate disodium (CMP), and vitamin B12 in the treatment of patients with compressive neuralgias. There is a body of clinical and preclinical evidence in the literature, supporting use of CMP and UTP in neuropathic pain, with potential symptomatic benefits.9-22
Several preclinical studies of peripheral neuropathies of varying etiologies have indicated beneficial effects of UTP and CMP administration in situations such as experimentally induced crush injury. Reported preclinical observations include accelerated nerve and muscle fiber regeneration, increased nerve conduction velocity, increased levels of neuritic protein neurofilaments, increased axon myelin surface area and thickness, and increased levels of neuronal cell membrane phospholipids phosphatidylcholine and phosphatidylethanolamine following nucleotide administration.19-22 In the clinical setting, the combination of UTP and CMP with vitamin B12 has been investigated in several painful conditions with favorable results, among which are diabetic neuropathy, acute non-traumatic pain, lumbar pain, cervical pain, and traumatic-compressive lesions.10-13,18
Objectives
The objectives of this study were to assess the safety and efficacy of the combination of nucleotides (UTP and CMP) and vitamin B12 in patients presenting with neuralgia arising from neural compression associated with degenerative orthopedic alterations and trauma, and to compare these effects with isolated administration of vitamin B12.
Materials and methods
This was a randomized, double-blind, comparative, controlled trial performed at UNIFESO Medical School facilities and hospital. The study was registered with the Brazilian National Database GGMED/GESF-ANVISA under the following numbers: NOTIFICAÇÃO EM PESQUISA CLÍNICA Nº 61/2014 Expediente do CE: 0331070141. After study approval by the UNIFESO ethical committee (approval no 509.682) and written informed consent of all participants, subjects meeting inclusion criteria were randomized to a 30-day oral treatment period. Inclusion criteria included patients of both genders, ≥18 years of age, with a clinical presentation of degenerative orthopedic alteration with neural compression. Female patients performed a urine pregnancy test prior to inclusion and were required to maintain adequate birth control throughout the study period. Exclusion criteria included need for surgical treatment and need for other analgesics.
Enrolled subjects were randomized by order of arrival to the study center into two treatment groups: Group A received the combination of nucleotides and vitamin B12, while subjects in Group B received vitamin B12 monotherapy. The study drug and treatment regimen was two capsules, three times per day for Group A. Each capsule contained: CMP 2.5 mg; UTP 1.5 mg; and hydroxocobalamin 1.0 mg. Study treatment regimen for subjects in Group B was two capsules, three times per day, each capsule contained hydroxocobalamin 1.0 mg. There was no visible difference between the study drugs, in accordance with blinding procedures for double-blind trials. Subjects were instructed to swallow the capsules whole at breakfast, lunch, and dinner.
The study hypothesis was that the combination of nucleotides and vitamin B12 would show superior efficacy in relation to B12 monotherapy. The study protocol outlined three patient visits to the study center: pretreatment for screening, randomization, and study drug distribution; visit two took place after 15 days of treatment; and visit three occurred at the end of the 30-day treatment period. At each of these study visits, subjects underwent a series of efficacy and tolerability assessments.
Efficacy assessments included a 100 visual analog pain scale (VAS), and the Patient Functionality Questionnaire (PFQ), a 12-item yes or no questionnaire about management of daily needs. The assessment included sleep quality, need to rest, ability to get up, standing and climbing stairs, washing, drying, and dressing, picking up things from the floor, changing posture, carrying weight, and need for assistance from other people. At each assessment, the patient and the investigating physician also evaluated the patient’s overall condition on a scale of 1-10 points. Safety assessments took into account vital signs at each assessment and occurrence, severity, and duration of any adverse effects, including changes in laboratory tests.
The primary study endpoint was the percentage of subjects presenting VAS ≤20 mm at end of study treatment period. Secondary study endpoints included the percentage of subjects presenting improvement ≥5 points on the PFQ; percentage of subjects presenting pain reduction (reduction in VAS scores at study end in relation to pretreatment); and number of subjects presenting adverse events.
Study sample size took into account the results of a previous study,10 evaluating 80 subjects (40 per treatment group), in which mean VAS scores at end of treatment were 21.11 in the combination group versus 41.75 in the B12 monotherapy group. Thus, a study sample size of 400 was determined, taking into account the mean between group difference of 20.6mm and standard deviation of 10.3mm, from the previous study, with a standard error of 1.03. The 95% confidence interval was 18.58–22.62, with a power of 1.0 and a two-tailed alpha of 0.05. Data were statistically analyzed using the software GraphPad Prism 5.0. Overall clinical efficacy and tolerability were analyzed via comparison of the results of each assessment in relation to pretreatment values, within and between treatment groups. For categorical variables, we used the χ2 or Fisher’s test, while continuous variables were analyzed using the repeated measures ANOVA or Student’s t-test.
Results
A total of 400 subjects were randomized to treatment, with 200 subjects per treatment group. Figure 1 shows the flow of subjects through the trial. No significant between-group differences were noted at pretreatment in the demographic data and pretreatment characteristics of the study population, as summarized in Table 1.
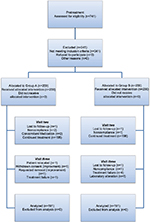  | Figure 1 Flowchart of subjects through the study |
The safety measures monitored during the study are summarized in Table 2. Pretreatment safety measures did not vary significantly between Groups A and B: weight (p=0.148), body mass index (BMI) (p=0.532), blood pressure (p=1.0 for systolic blood pressure and p=0.849 for diastolic blood pressure), and heart rate (p=0.413). Throughout the treatment period within the treatment groups, with the exception of systolic blood pressure in both groups and diastolic blood pressure in Group B, there was no statistically significant change in weight (Group A, p=0.855; Group B, p =0.0603), BMI (Group A, p =0.6; Group B, p =0.257), blood pressure (Group A, p = 0.0013 for systolic blood pressure and p=0.133 for diastolic blood pressure; Group B, p =0.0046 for systolic blood pressure and p=0.0032 for diastolic blood pressure), and pulse (Group A, p=0.541; Group B, p=0.337).
Pretreatment PFQ scores were significantly lower among patients in Group B (median for Group A, 9.0; median for Group B, 8.0; p=0.0036). Median PFQ scores were 5.0 at visit two in both treatment groups and 0.0 at visit three in both treatment groups. Both groups showed statistically significant improvement at the end of the study in relation to pretreatment PFQ scores (p<0.0001 for both treatment groups). In relation to the secondary study endpoint, there was no between-group difference in the percentage of subjects presenting improvement ≥5 points on the PFQ (p=0.135).
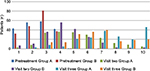
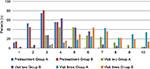
Pretreatment patient overall assessment scores were homogenous between treatment groups (p=0.29). At visits two and three, the scores of this assessment improved within each treatment group, but Group A patients showed a greater improvement in scores at both visit two (p=0.014) and visit three (p=0.0024) in comparison to Group B scores (Figure 2). Physician overall assessment scores were also homogenous between treatment groups at pretreatment (p=0.717), but at visits two and three scores were significantly lower in Group A as compared to Group B (p=0.0029 for visit two and p=0.006 for visit three) (Figure 3).
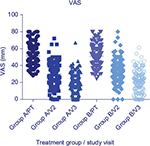
At pretreatment, mean VAS scores among the patients in Group A was 51.26mm (±12.38) while in Group B mean pretreatment VAS score was 51.06 (±11.72) (p=0.87). Visit two VAS scores for Group A and Group B were 29.86 mm (±12.94) and 33.88 mm (±11.86) (p=0.0014); At visit three, mean Group A VAS scores were 16.06 mm (±12.03) and 20.40 mm (±11.42) for Group B (p=0.0003). The percentage of subjects presenting VAS scores ≤20 mm (primary study endpoint) at visit three was significantly higher in Group A as compared to Group B (p=0.0425). Within the treatment groups, VAS score reduction was statistically significant for both Group A and Group B (p<0.0001 for both groups) (Figure 4).
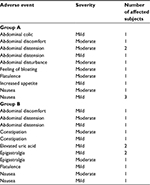
Adverse events were recorded in both treatment groups, though for the most part these were not serious and were transient. Table 3 summarizes the adverse events determined by the investigating physician to be related to the study drug. There was no between-group difference in the percentage of subjects presenting adverse events during the treatment period (p=0.6009).
Discussion
We report the results of a clinical trial using the combination of uridine, cytidine, and vitamin B12 in the treatment of neuralgias caused by degenerative orthopedic alterations with neural compression. This combination is commercially available in Brazil under the brand name Etna®. The combination of the nucleotides with vitamin B12 yielded a more significant pain reduction as measured by the VAS. Assessments performed by both the patients and the investigating physician had higher scores after 15 and 30 days of treatment among subjects treated with the combination. Treatment with the combination and with the control of vitamin B12 monotherapy resulted in significant improvement in patient functionality throughout the treatment period, as measured by the patient functionality questionnaire.
The findings of this study are in agreement with those previously described in the literature. A previous study performed by Goldberg et al (2009) assessed the use of this combination in the treatment of back pain and hip pain, reporting significant reduction in pain assessments as well as functionality.10 As with the study of Goldberg and also another recent study in alcoholic polyneuropathy in which the authors compared the use of the combination of UTP + CMP + vitamin B12 with the use of vitamin B12 monotherapy,17 there was a significant improvement over the treatment period in pain manifestations as compared to the monotherapy group, although the patients within the monotherapy group also displayed significant improvement throughout the treatment as compared to pretreatment status. This finding and that of our study highlights the importance the vitamin B12 component in the treatment of these painful conditions.
A limitation of this study was the inclusion of three distinct pain locations: lower back, hip, and carpal tunnel syndrome which ultimately reduced the number of subjects in each pain subcategory. Future studies should focus on a single pain location in order achieve more specific results. Nevertheless, we believe the results of this study are in concordance with findings in the literature and serve to contribute to the growing body of evidence indicating the therapeutic value of the combination of UTP,CMP and vitamin B12 for the treatment of peripheral neuropathies. It is also important to point out that unlike vitamin B12 (which has been confirmed to exhibit antinociceptive effects in both animal and human studies) the nucleotides do not have any direct role in anti-inflammation or analgesia and thus the symptomatic improvements observed in our study and in previous studies are suggestive of structural neuroregeneration by the nucleotides as a mechanism of action.10,11,14,15 Indeed, previous studies on animal models give credence to this notion, with previous observations of the nucleotides playing a positive role in axon and myelin sheaths of nerve and muscle fibers during regeneration following experimentally-induced damage, in addition to improved nerve fiber conduction velocity.19-21
A combination of uridine, folic acid, and vitamin B12 was recently reported to be effective in the treatment of carpal tunnel syndrome. Although in a small scale exploratory trial of 48 subjects, authors reported reduced pain scores and improvement in symptom intensity following 60 days of treatment with the combination therapy.23 An important limiting factor in our study with regards to carpal tunnel syndrome is the relatively small sample size in relation to the total patient population, as mentioned in the preceding paragraph. However, these findings together with the results of our study warrant further investigation into the use of the nucleotides and vitamin B12 in the treatment of this very common ailment.
Clinical trials of painful conditions attributable to neuropathic pain (pain following neurological surgical procedures, neural compression-induced neuralgias, chronic neuropathic lumbar pain, peripheral neuropathies, and pain and paresthesia related to B12 deficiency anemia) have consistently yielded encouraging results with use of the combination of uridine, cytidine, and vitamin B12, and further studies may reveal additional indications where this combination or others like it may be of use, while also expanding our understanding of the interaction between the nucleotides and vitamin B12 in painful conditions.9-21 For instance, the combination of uridine, folic acid, and vitamin B12 was also reported to be effective in neuropathic pain of peripheral neuropathies, with a reduction in pain intensity and also allowing for reduction in concomitant medication use.23,24 These types of study are important to explore new alternatives for addressing neuropathic pain – with the aim of providing treatment options that reduce pain, decreased need for other drugs with more unwanted side effects, and improve patient care and quality of life.
Conclusion
The results of this study showed a more expressive improvement in efficacy evaluations among subjects treated with the combination of nucleotides + vitamin B12, with a statistically significant superiority of the combination in pain reduction (evidenced by VAS scores). There were adverse events in both treatment groups, but these were transitory and no severe adverse event was recorded during the study period. Safety parameters were maintained throughout the study in both treatment groups. The combination of uridine, cytidine, and vitamin B12 was safe and effective in the treatment of neuralgias arising from neural compression associated with degenerative orthopedic alterations and trauma. Considering these results, a prospective, multi-center randomized controlled clinical trial, comparing the activity of vitamin B12 alone to the activity of vitamin B12 in combination with nucleotides in pain reduction, is warranted.
Acknowledgements
The authors thank Renato Kaufman and Adenilson de Souza da Fonseca for participation in the study with subject recruitment, treatment, data review and contributions to the drafts of this paper. Thanks also to Alessandra Santos for clinical research form and data verification, and Daiane Bergamim, Silvia Maciel, Breno Lorch and Aline Pacheco for study monitoring.
Disclosure
The authors report no conflicts of interest in this work.
References
Voet D, Voet JG, Pratt CW. Fundamentals of biochemistry: Life at the molecular level. Hoboken, NJ: Wiley, 2008. | ||
Oh R, Brown DL. Vitamin B12 deficiency. Am Fam Phys. 2003;67:979–986. | ||
Schjonsby H. Vitamin B12 absorption and malabsorption. Gut. 1989;30:1986–1991. | ||
Wilson JA. Vitamin deficiency and excess. In: Harrison’s Principles of Internal Medicine. 14th Ed. Fauci AS, Braunwald E, Isselbacher K (eds) et al. McGraw-Hill, New York. 1998:481. | ||
Gubler CJ. In: Handbook of vitamins: Nutritional, biochemical, and clinical aspects. Machlin, L.J. (ed), Marcel Dekker, New York. 1984:245. | ||
Allen RH, Stabler SP, Savage DG, Lindenbaum J. Metabolic abnormalities in cobalamin (vitamin B12) and folate deficiency. FASEB J. 1993;7:1344–1348. | ||
Carmel R. Current concepts in cobalamin deficiency. Ann Rev Med. 2000;51:357–75. | ||
Sechi G, Sechi ES, Fois C, Kumar N. Advances in clinical determinants and neurological manifestations of B vitamin deficiency in adults. Nutr Rev. 2016;74(5):281–300. | ||
Gallai V, Mazzotta G, Montesi S, Sarchielli P, Del Gatto F. Effects of uridine in the treatment of diabetic neuropathy: an electrophysiologic study. Acta Neurol Scandinav. 1992;86:3–7. | ||
Goldberg H, Scussel Jr AR, Cohen JC, et al. Neural compression-induced neuralgias: clinical evaluation of the effect of nucleotides associated with vitamin B12. Rev Bras Med. 2009;66(11):380–385. | ||
Lauretti GR, Omais M, Pereira AC, Mattos AL. Avaliação clínica do efeito analgésico do complexo citidina-uridina-hidroxocobalamina como coadjuvante no tratamento da dor lombar crônica neuropática [Clinical evaluation of the cytidine-uridine-hydroxicobalamine complex as adjuvant in the management of the chronic neuropathic low back pain]. Coluna. 2004;3(2):73–76. Portuguese. | ||
Mibielli MA, Nunes CP, Cohen JC, et al. Treatment of acute, non-traumatic pain using a combination of diclofenac-cholestyramine, uridine triphosphate, cytidine monophosphate, and hydroxocobalamin. Proc West Pharmacol Soc. 2010;53:5–12. | ||
Mibielli MA, Nunes CP, Scussel Jr AB, Suchmacher Neto M, Oliveira L, Geller M. Symptomatic improvement in an acute, non-traumatic spine pain model with a combination of uridine triphosphate, cytidine monophosphate, and hydroxocobalamin. PST. 2014;2(1):6–10. | ||
Müller D. Weniger Schmerzen bei diabetischer polyneuropathie. [Pilot study with pyrimidine nucleotides. Less pain in diabetic polyneuropathy]. MMW Fortschr der Med. 2002;144(46):61. German. | ||
Müller D. Therapie de neuropathischen schemerzsyndroms: Ergebnisse einer offenen studie zur wirksamkeit eines pyrimidinnukleotid-Praparats. [Treatment of neuropathic pain syndrome. Results of an open study on the efficacy of a pyrimide nucleotide preparation]. Fortschr der Med Orig. 2002;120(4):131–133. German. | ||
Nunes CP, Higashi R, Ribeiro MG, et al. Efficacy and tolerability of a combination of uridine, cytidine, and vitamin B12 in anemia. A double-blind, comparative study versus nucleotide monotherapy. Rev Bras Med. 2008;65:15–21. | ||
Nunes CP, Scussel Jr AB, Goldberg H, et al. Alcoholic polyneuropathy: clinical assessment of treatment outcomes following therapy with nucleotides and vitamin B12. Res Neurol Inter J. 2013;2013. | ||
Serra JP, Veciana MG, Bordas LB. Efectos terapéuticos de una asociación de C.M.P., U.T.P., y vitamina B12 en cincuenta casos de neuropatias perifericas. [Therapeutic results of an association of CMP, UTP and vitamin B12 in 50 cases of peripheral neuropathy]. N Engl J Med (Spanish Edition). 1972;VI(64):1–7. Spanish. | ||
Wattig B, Heydenreich F, Schalow G, Radzewitz B, Warzok R, Cervós-Navarro J. Nucleotide beschleunigen die nervenregeneration [Nucloetides accelerate nerve regeneration]. Z Klin Med. 1991;46:1371–1373. German. | ||
Wattig B, Schalow G, Heydenreich F, Warzok R, Cervós-Navarro J. Enhancement of nerve fibre regeneration by nucleotides after peripheral nerve crush damage. Electrophysiologic and morphometric investigations. Arzneimittel-Forschung/Drug Research. 1992;42(II)9:1075–1078. | ||
Wattig B, Schalow G, Madauss M, Heydenreich F, Warzok R, Cervos-Navarro J. Acceleration of nerve and muscle regeneration by administration of nucleotides – electroneurophysiological and morphometrical investigations. Acta Histochemica. 1992;42:333–339. | ||
Wurtman RJ, Ulus IH, Cansev M, Watkins CJ, Wang L, Marzloff G. Synaptic proteins and phospholipids are increased in the gerbil brain by administering uridine plus docosahexanoic acid orally. Brain Res. 2006;1088:83–92. | ||
Negrão L, Almeida P, Alcino S, Duro H, Libório T, et al. Effect of the combination of uridine nucleotides, folic acid and vitamin B12 on the clinical expression of peripheral neuropathies. Pain Manag. 2014;4(3):191–196. | ||
Negrão L, Nunes P. Uridine monophosphate, folic acid and vitamin B12 in patients with symptomatic peripheral entrapment neuropathies. Pain Manag. 2016;6(1):25–29. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.