Back to Journals » International Medical Case Reports Journal » Volume 7
A case of thyroid storm with cardiac arrest
Authors Nakashima Y, Kenzaka T , Okayama M, Kajii E
Received 4 March 2014
Accepted for publication 31 March 2014
Published 8 May 2014 Volume 2014:7 Pages 89—92
DOI https://doi.org/10.2147/IMCRJ.S63475
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Yutaka Nakashima,1 Tsuneaki Kenzaka,2 Masanobu Okayama,3 Eiji Kajii3
1Department for Support of Rural Medicine, Yamaguchi Grand Medical Center, 2Division of General Medicine, Center for Community Medicine, Jichi Medical University School of Medicine, Shimotsuke, Japan; 3Division of Community and Family Medicine, Center for Community Medicine, Jichi Medical University School of Medicine, Shimotsuke, Japan
Abstract: A 23-year-old man became unconscious while jogging. He immediately received basic life support from a bystander and was transported to our hospital. On arrival, his spontaneous circulation had returned from a state of ventricular fibrillation and pulseless electrical activity. Following admission, hyperthyroidism led to a suspicion of thyroid storm, which was then diagnosed as a possible cause of the cardiac arrest. Although hyperthyroidism-induced cardiac arrest including ventricular fibrillation is rare, it should be considered when diagnosing the cause of treatable cardiac arrest.
Keywords: hyperthyroidism, ventricular fibrillation, treatable cardiac arrest, cardiac arrest, cardiopulmonary arrest
Introduction
There are few reports on cases of thyroid storm-induced cardiac arrest including ventricular fibrillation.1–5 We report a case of undiagnosed hyperthyroidism that was diagnosed as a result of thyroid storm-induced cardiac arrest.
Case presentation
The subject was a 23-year-old Japanese man with no abnormalities identified on examination and no particular medical history. Two weeks prior to onset of cardiac arrest, he noticed slight fever, coughing, loss of appetite, and weight loss of 3 kg. He did not have symptoms such as nausea, vomiting, or diarrhea. While jogging, the patient became aware of back pain and lost consciousness. A bystander immediately administered basic life support. Then, emergency medical technician-paramedics continuously administered cardiac pulmonary resuscitation using an automated external defibrillator to the subject, who was thereafter transported by ambulance to our hospital, Yamaguchi Grand Medical Center.
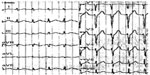
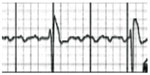
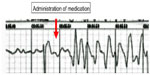
At the time of transfer to the hospital, the blood pressure of the subject could not be measured. An electrocardiogram (ECG) monitor indicated that his heart rate was 30 bpm (pulseless electrical activity) (Figure 1). In addition, his body temperature was 37.3°C and he had a Glasgow coma scale score of E1M1V1 (no eye opening, no verbal response, no motor response). Physical examination revealed swelling of the thyroid gland on the anterior side of the neck; however, there were no signs of jaundice or peripheral edema. In addition, there were no signs of exophthalmos, finger tremor, loss of hair, or nail lesions. Because of pulseless electrical activity, cardiac arrest was diagnosed using an ECG, and administration of medication (epinephrine and atropine) led to ventricular fibrillation (Figures 2 and 3). As a result, defibrillation was administered using a direct current (DC) type 360-J monophasic defibrillator (Nihon Kohden Corporation, Tokyo, Japan) and the sinus rhythm was restored. Nonetheless, heart rate and breathing sounds were normal.
  | Figure 1 Electrocardiogram at the time of transfer to the hospital. There was pulseless electrical activity. |
  | Figure 2 An electrocardiogram registered by the automated external defibrillator. Pulseless electrical activity turned into ventricular fibrillation. |
  | Figure 3 An electrocardiogram after administration of medication. Ventricular fibrillation was observed. |
The laboratory findings were as follows: a white blood cell count of 13,800/μL; hemoglobin, 12.0 g/dL; platelets, 434,000/μL; C-reactive protein, 0.2 mg/dL; aspartate aminotransferase, 51 IU/L; alanine aminotransferase, 74 IU/L; lactic dehydrogenase, 217 IU/L; creatine kinase 61 IU/L; creatine kinase MB 46 IU/L; troponin T, <0.05 ng/mL; blood urea nitrogen, 16.4 mg/dL; creatinine, 0.81 mg/dL; blood glucose, 77 mg/dL; sodium, 143 mmol/L; potassium, 5.8 mmol/L; chloride, 97 mmol/L; calcium, 9.2 mg/dL; arterial blood gases (FiO2:100%, timing of return of spontaneous circulation and post-tracheal intubation); pH 7.53; PaCO2 30.8 mmHg; PaO2 184 mmHg; and HCO3 11.4 mEq/L.
Chest radiography revealed no congestion or pneumothorax in the lung field. After the sinus rhythm improved, an ECG revealed no significant changes in ST-T or QT interval prolongation (Figure 4). In the echocardiography results, there was no cardiac wall motion dyssynchrony suggestive of ischemic heart disease, and the ejection fraction was 70%. There were no right side of the heart findings indicative of cardiac tamponade and thromboembolism. Thyroid ultrasound imaging showed that the parenchyma was marginally irregular and heterogeneous with rich blood flow.
On examination of thyroid function, the following findings were obtained: thyroid-stimulating hormone (TSH) level: <0.005 (reference range: 0.24–3.72), free thyroxine (T4) level: 5.03 (reference range: 0.94–1.52), free triiodothyronine (T3) level: >30.00 (reference range: 2.01–3.72), TSH receptor antibody: 30.2% (reference values: <15), and TSH-stimulating receptor antibody: 146% (reference values: <180).
After spontaneous circulation was returned, the reduced level of consciousness persisted and artificial respiration and cerebral hypothermia were administered. Hyperthyroidism-induced thyroid storm was strongly suspected and the patient was treated with thiamazole 60 mg/day, inorganic iodine (iodine solution 150 mg/day), hydrocortisone 300 mg/day, and metoprolol tartrate (beta-blocker) 120 mg/day for 2 weeks. After the initial 2 weeks, thiamazole 60 mg/day and metoprolol tartrate (beta-blocker) 120 mg/day were taken.
On day 22 of hospitalization, the subject was weaned from the artificial respirator; however, hypoxic encephalopathy- induced higher cerebral dysfunction persisted.
On day 30 of hospitalization, follow-up testing revealed that free T4 and free T3 levels had returned to normal (free T4 level: 1.46 and free T3 level: 3.43), but TSH suppression persisted (TSH <0.005). Because of higher cerebral dysfunction, coronary angiogram and magnetic resonance imaging (MRI) of the heart were not performed in accordance with the wishes of his family. On day 55, the subject was transferred to another hospital for long-term medical care. TSH level was 0.14 at the time.
At the present 1-year follow-up, no signs of recurrent ventricular fibrillation or other cardiac arrest rhythms have been observed in the other hospital in which the patient is receiving long-term medical care.
Discussion
Our case subject was diagnosed with TSH receptor antibody-positive hyperthyroidism, otherwise known as Basedow’s disease.6,7 Arrhythmia due to hyperthyroidism often manifests itself as sinus tachycardia, atrial fibrillation, or ventricular tachycardia,8 but rarely as a cardiac arrest including ventricular fibrillation.1–5 A cardiac arrest is rarely led by hypokalemia.3 In contrast, Ueno et al,1 Ando et al,4 and Jao et al5 described cases of a cardiac arrest with a thyroid storm where the patients did not have hypokalemia or other signs of a complicating disorder.
In this case, congenital coronary artery anomalies, myocarditis, drug intoxication, and anaphylaxis from an insect bite were considered as the cause of cardiac arrest. Because serum creatine kinase, 12-lead ECG, and echocardiographic evaluation did not change over time, it is unlikely that our patient had myocarditis. Because of the absence of a history of drug abuse and the lack of injection scars, bite marks, or a skin rash, it is unlikely that drug intoxication or anaphylaxis from an insect bite were involved. Because a coronary angiogram and MRI of the heart were not performed, the patient might have congenital coronary artery anomalies. Nonetheless, an additional factor was necessary for the sudden cardiac arrest to occur, even if the patient has congenital heart disease.
In the present case, there was no underlying heart disease according to echocardiography. Moreover, the treatment of hyperthyroidism prevented recurrence of ventricular fibrillation and other cardiac arrest rhythms. This observation led to the suspicion of a thyroid storm, which was then diagnosed as a possible cause of the cardiac arrest.
It has been reported that a thyroid storm may affect the heart owing to early repolarization and coronary artery spasms; in addition, a smoking habit can also contribute to early repolarization and coronary artery spasms.4 The effects of thyroid hormones on the heart are as follows: accelerated protein and messenger RNA synthesis in myocardial cells; Na–K adenylpyrophosphatase (ATPase) hyperactivity; increased Ca2+ transport; an increased number of β-receptors; a reduced refractory phase; and a low stimulus threshold.8
Our subject had a score of 70 points (temperature, 5; central nervous system effects, 30; cardiovascular dysfunction, 25; and precipitant history, 10) according to the diagnostic criteria (Table 1); these data were indicative of a thyroid storm.7
  | Table 1 Diagnostic criteria of a thyroid storm |
A thyroid storm is a rare condition affecting 1%–2% of patients with hyperthyroidism. It is a life-threatening condition and is reported to have a fatality rate of 50%–90% if left untreated or 20%–30% if treated.9
Treatable causes of a cardiac arrest include the H’s and T’s, namely hypoxia, hypovolemia, hydrogen ion (acidosis), hypo- or hyperkalemia, hypothermia, toxins, tamponade (cardiac), tension pneumothorax, pulmonary thrombosis, and coronary thrombosis.10 Although a hyperthyroidism-induced cardiac arrest is rare, it is a treatable arrhythmia.
Despite persistent higher cerebral dysfunction, our subject survived and exhibited no recurrence of ventricular fibrillation and other cardiac arrest rhythms because of the treatment of thyroid function. In the event of a cardiac arrest in the absence of the H’s and T’s, hyperthyroidism should be considered as another possible treatable cause.
Conclusion
We experienced a case of cardiac arrest associated with hyperthyroidism. In the event of cardiac arrest in the absence of a treatable cause (one of the H’s and T’s), hyperthyroidism should be considered as another potential treatable cause.
Author contributions
Yutaka Nakashima: manuscript editing.
Tsuneaki Kenzaka: management of the case, manuscript editing and correction.
Masanobu Okayama: clinical management of the case and revision of the manuscript.
Eiji Kajii: manuscript correction, editing of the captions for the illustrations.
All authors contributed to conception and design, acquisition of data, or analysis and interpretation of data, and read and approved the final version of the manuscript.
Disclosure
The authors declare that they have no conflicts of interest related to this work.
References
Ueno A, Yamamoto T, Sato N, Tanaka K. Ventricular fibrillation associated with early repolarization in a patient with thyroid storm. J Interv Card Electrophysiol. 2010;29(2):93–96. | |
Wei JY, Genecin A, Greene HL, Achuff SC. Coronary spasm with ventricular fibrillation during thyrotoxicosis: response to attaining euthyroid state. Am J Cardiol. 1979;43(2):335–339. | |
Boccalandro C, López-Penabad L, Boccalandro F, Lavis V. Ventricular fibrillation in a young Asian man. Lancet. 2003;361(9367):1432. | |
Ando T, Henmi T, Haruta D, et al. Graves’ disease complicated by ventricular fibrillation in three men who were smokers. Thyroid. 2011;21(9):1021–1025. | |
Jao YT, Chen Y, Lee WH, Tai FT. Thyroid storm and ventricular tachycardia. South Med J. 2004;97(6):604–607. | |
Matthews DC, Syed AA. The role of TSH receptor antibodies in the management of Graves’ disease. Eur J Intern Med. 2011;22(3):213–216. | |
Burch HB, Wartofsky L. Life-threatening thyrotoxicosis. Thyroid storm. Endocrinol Metab Clin North Am. 1993;22(2):263–277. | |
Klein I, Ojamaa K. Thyroid hormone and the cardiovascular system. N Engl J Med. 2001;344(7):501–509. | |
Fisher JN. Management of thyrotoxicosis. South Med J. 2002;95(5):493–505. | |
Neumar RW, Otto CW, Link MS, et al. Part 8: adult advanced cardiovascular life support: 2010 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation. 2010;122(18 Suppl 3):S729–S767. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.