Back to Journals » ClinicoEconomics and Outcomes Research » Volume 8
A budget impact analysis of Spiromax® compared with Turbuhaler® for the treatment of moderate to severe asthma: a potential improvement in the inhalation technique to strengthen medication adherence could represent savings for the Spanish Healthcare System and five Spanish regions
Authors Darbà J , Ramírez G, García-Rivero JL, Mayoralas S, Pascual JF, Roger A, Vargas D, Bijedic A
Received 26 April 2016
Accepted for publication 22 June 2016
Published 6 September 2016 Volume 2016:8 Pages 435—444
DOI https://doi.org/10.2147/CEOR.S111453
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Giorgio Colombo
Josep Darbà,1 Gabriela Ramírez,2 Juan L García-Rivero,3 Sagrario Mayoralas-Alises,4 José Francisco Pascual,5 Albert Roger,6 Diego Vargas,7 Adi Bijedic8
1Department of Economics, Universitat de Barcelona, 2BCN Health Economics & Outcomes Research SL, 3Hospital Laredo, Cantabria, 4Hospital Ramón y Cajal, Madrid, 5Hospital General Universitario de Alicante, Alicante, 6Hospital Universitario Germans Trias i Pujol, Barcelona, 7Hospital de Alta Resolución el Toyo, Andalusia, 8Market Access and HEOR Department, TEVA Pharmaceutical, Madrid, Spain
Objective: To assess the economic impact of the introduction of DuoResp® Spiromax® by focusing on a potential improvement in the inhalation technique to strengthen medication adherence for the treatment of moderate to severe asthmatics in Spain and five Spanish regions including Andalusia, Catalonia, Galicia, Madrid, and Valencia.
Methods: A 4-year budget impact model was developed for the period 2015–2018 from the Spanish Healthcare System perspective. Budesonide–formoterol fixed-dose combination delivered by Turbuhaler® was considered to be the most appropriate comparator for assessing the budget impact with the introduction of DuoResp® Spiromax®. National and regional data on asthma prevalence were obtained from the literature. Input parameters on health care resources were obtained by consulting experts from different Spanish hospitals. Resources used included medical visits, emergency room visits, and hospitalizations. The average numbers of primary care and specialist visits per year were also gathered. Based on health care resource use per patient, the total treatment cost per patient was estimated.
Results: The population with moderate to severe asthma treated with budesonide–formoterol fixed-dose combinations delivered by Turbuhaler® in 2015 was estimated to be 166,985 in Spain. Region-specific prevalence data resulted in 25,081, 12,392, 16,097, 17,829, and 15,148 patients in Andalusia, Catalonia, Galicia, Madrid, and Valencia, respectively. Based on the forecast uptake of DuoResp® Spiromax®, the total budget savings in Spain were expected to be €1.509 million over the next 4 years. Region-specific rates imply that the total savings were expected to be €229,706 in Andalusia, €90,145 in Catalonia, €188,327 in Galicia, €122,669 in Madrid, and €165,796 in Valencia over 2015–2018.
Conclusion: The introduction of DuoResp® Spiromax®, which represents a potential improvement in the inhalation technique to strengthen medication adherence for the treatment of moderate to severe asthma, could represent savings for the Spanish National Health Society and five Spanish regions.
Keywords: dry powder inhaler, economic evaluation, region-specific estimates, payers perspective
Introduction
Asthma is a leading cause of morbidity, mortality, and economic burden and a significant public health problem worldwide.1,2 This chronic condition is characterized by inflammation of respiratory airways, hypersensitivity of airway path, and variable airflow limitation for short periods of time.
Daily medication is used to prevent or improve asthma symptoms. Inhaler therapy for asthma delivers therapeutic drug doses into the airways, leading to local efficacy within the lungs.3 Effective use of inhaler requires proper inhalation technique, which implies a good medication adherence, because it is a critical factor of self-management of the disease. Dry powder inhalers (DPIs) overcome the difficulties between inhaler actuation and inspiration, one of the most common errors with pressurized metered dry power; however, recent reviews have shown that misuse of DPIs is also common in real life.4,5
There are factors such as low inhaler complexity and patient preferences that can help to optimize the inhaler device by improving adherence to medication.6 Strengthening these factors would contribute to good disease management and better use of health care resources, given the excessive costs required to treat chronic obstructive lung diseases.
Asthma is a significant public health problem among inner city populations. In Spain, asthma prevalence varies widely, and studies have shown that divergence is explained by genetic factors, proportion of immigrants, and environmental, organizational, and health care factors of regional health care services.7 It has been reported that half of the treated patients do not comply correctly with their treatment.8 Medication adherence is a key factor for controlling progression of chronic disease.
The lack of adherence is associated with increased health care costs due to emergency room visits and hospitalizations as well as additional diagnostic tests and stepping up therapy compared to the original less costly therapy, which also indicates higher consumption of primary care (PC) and specialist consultations.5,9 Given the developments in inhalation devices such as pressurized metered dry powder and DPI, one would expect that these imply better clinical outcomes; however, most of the current inhalers are often poorly used and are not intuitive, which implies that an extensive training is needed in the long term.10,11 Therefore, optimization of the inhaler device needs to be oriented to meet the current unmet needs associated with medication adherence in the management of obstructive lung disease.
In the current study, we evaluated the expected economic impact from the increase in the market share of Spiromax®, a brand-new inhalation device, for the maintenance therapy with budesonide–formoterol fixed-dose combination (FDC), by using a budget impact model. This new inhalation device has been shown to reduce common utilization errors such as dose preparation errors, adequate flow rates, and even environmental conditions that might limit the delivery of the drug directly to the lungs.12 We included data from the perspective of the Spanish Healthcare System (NHS) as well as regional data from five Spanish Autonomous Communities (hereafter known as regions): Andalusia, Catalonia, Galicia, Madrid, and Valencia.
Methods
Model development and structure
According to legislation of scientific studies (SAS/3470/2009 Order of December 16, to make public guidelines on post-authorization studies that are observational for drugs for human use) this project did not require approval from an ethical review board as we did not have access to patient level data, we did not need to interview patients to obtain estimates, and this was not a clinical study. The budget impact model was developed in Microsoft Excel from the perspective of the Spanish NHS, and for this purpose a time horizon of 4 years (2015–2018) was used. Spain and five Spanish regions were included in our analysis. Budesonide–formoterol FDCs delivered by Turbuhaler® whose complete brand names can be Rilast® Turbuhaler® and Symbicort® Turbuhaler® were considered to be the most appropriate comparator for assessing the budget impact with the introduction of DuoResp® Spiromax®, a new inhaler for delivering budesonide–formoterol FDC. We decided to compare these two different inhalation devices that delivered budesonide–formoterol FDC because changes in prescription regimens were not hypothesized in this study.
Input parameters were obtained by expert panel consultation from different Spanish hospitals. Therefore, the model analyzed health care resource utilization per patient based on their daily maintenance treatment for asthma and the number of days with events such as hospitalizations and visits to the emergency room, PC visits, and specialist visits. All cost estimates were reported in Euros (EUR 2015), and a discount rate of 3% was applied.
It is worth mentioning that during the time this study was being completed, there was a significant change in prices of the drugs included in this analysis. From October 2015 onward, prices of both drugs were set at the same level by the Spanish Ministry of Health; therefore, price effect was no longer useful to calculate the economic impact of the introduction of DuoResp® Spiromax®.13
This model included diagnosed patients who control asthma with a maintenance therapy. Proportion of FDC of budesonide–formoterol utilization that was delivered by a DPI was based on the national and regional sales data for Spain reported by IMS. Therefore, data on Symbicort® Turbuhaler® and Rilast® Turbuhaler® utilization were used to estimate the target population. Forecast of DuoResp® Spiromax® uptake rates was reported by Teva Pharma.
The model generated estimates for the costs per patient and the total direct costs of treatment including drug and medical cost based on market shares and other input parameters. The potential effect of a strengthening of adherence to the total costs of asthma was generated from savings in medical resources utilization associated with an improvement in the inhalation technique. All patients were assumed to receive treatment during the whole year.
Model input variables
Target population
To estimate the target population diagnosed with a maintenance therapy with budesonide–formoterol FDC, the following algorithm was applied, as shown in Figure 1. A literature review was performed to identify the prevalence of asthma among the adult population of Spain and regions included in this analysis.14–17 Estimates for asthma were extrapolated to the adult populations obtained from the population projections conducted by the National Institute of Statistics (INE).18 We had to take into account that >50% of asthmatic patients are not diagnosed and 26% of those do not receive treatment.19 A percentage was applied to distinguish patients using an FDC and among these patients, those who take budesonide–formoterol FDC delivered by DPI were determined.20 The proxy for capturing these patients was the percentage of patients using Symbicort®/Rilast® Turbuhaler®.
Inhalation technique, medication adherence, medical resource utilization, and costs
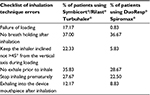
Before identifying directly variations in health care resources associated with the use of each inhaler, experts were asked to specify percentage of utilization errors with Spiromax® and Turbuhaler®, and the differences between use of inhalation devices were mainly those regarding the incorrect dose loading and keeping the inhaler inclined not >45° from the vertical axis (Table 1). These results were not used for calculations, but they were relevant to confirm that the misuse of inhaler is seen in clinical practice.
Obtaining input data on health care resource utilization associated with potential strengthening of adherence was the basis to estimate the economic consequences of the introduction of Spiromax® in Spain. The use of health care resources was estimated based on data from clinical practice by consulting a panel of five clinical experts in pneumology, allergy, and a general practitioner from different Spanish hospitals. Accordingly with our expert panel, the proportions of patients who visit the emergency room and those who are hospitalized due to suboptimal inhalation are estimated to be 6.96% and 2.64%, respectively. Among these patients we also observed whether there were differences in the number of these events between patients using Turbuhaler® versus patients using Spiromax® (Table 2).
Regarding number of PC visits and specialist visits, it was not possible to obtain a direct percentage of visits due to problems with the inhalers. One visit to a PC provider can be used to solve various patients’ concerns, including problems with the device. Thus, it was necessary to ask the overall number of PC and specialist visits that a regular asthmatic patient has. On average patients who used Turbuhaler® should not be different from those using Spiromax® because they share indication with the same FDC of budesonide–formoterol.21 Therefore, we assume that this variation between health care resource utilization associated with Spiromax® versus Turbuhaler® might be related to the inhalation device (Table 2). Given that there is no scientific literature published on this topic, experts’ consultation was needed to obtain input data on health care resource utilization. Monitoring test and its cost were also included in the model. This information was used to calculate the costs for medical resources associated with each drug.
Costs of health care resources included in this analysis were obtained from regional tariff lists.22–33 Cost of health care resources at the national level are the mean of 12 regions, including those in this analysis.
Budgetary impact analysis
Based on the annual drug cost and health care resource use per patient, the total treatment cost per patient was estimated in EUR 2015. With the annual average cost per patient for each treatment option, the target population, and the market shares for budesonide–formoterol FDC delivered by Turbuhaler®, the overall economic impact of the maintenance treatment of asthma for 2015–2018 was obtained. In the base case scenario or current scenario, a market without DuoResp® Spiromax® was estimated. The current scenario was compared with an alternative in which the economic impact was calculated by taking into account DuoResp® Spiromax® and its potential effects toward improved medication adherence. The budget impact model was based on differences between a scenario without DuoResp® Spiromax®, current scenario, and an alternative scenario (Table 3). The uptake of DuoResp® Spiromax® increases during the period of study.
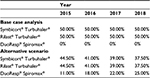  | Table 3 Distribution of treatments (%): base case analysis and alternative scenario Note: Data provided by Teva Pharma and it was prepared by the authors (unpublished data, 2015). |
Results
In our model, region-specific prevalence data were used, which vary across all five regions. Prevalence of asthma in Spain (9.90%) differs substantially from Catalonia (5.60%), Madrid (7%), or Galicia (12.9%), while it is similar to Andalusia (9.90%) and Valencia (9.80%).14–17 Based on demographics, the only region where slight increases in the asthmatic population were observed was Andalusia (Table 4).
  | Table 4 Target population (n) for asthma treatment Abbreviations: FDC, fixed-dose combination; DPI, dry powder inhaler. |
Asthma treatment resulted to be very costly for the Spanish NHS. For instance, it has been estimated that treating patients using Symbicort® Turbuhaler® cost €64.21 million in 2015. Specifically, in the base case analysis of Spain before the introduction of DuoResp® Spiromax®, the total economic impact was estimated to be €524.87 million over 4 years (Table 5). Taking into account the increasing market share of DuoResp® Spiromax® in the alternative scenario, the total economic impact for Spain was calculated to be €523.36 million. Overall, at the national level the total budget savings with the market share of DuoResp® Spiromax® was expected to be €1.509 million over the next 4 years, given the difference in number of days of hospital stay and PC visits (Table 6).
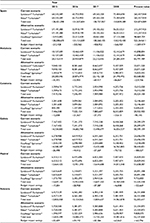  | Table 5 Results of the base case budget impact analysis in Euro 2015 (€) |
  | Table 6 Specific results: savings due to reduction in health care resource utilization Euro 2015 (€) Note: negative quantities mean savings. Abbreviation: PC, primary care. |
This model also provides region-specific estimates. In the case of Andalusia, treating patients who take budesonide–formoterol FDC delivered by Turbuhaler® would lead to a health care expenditure of €83.79 million between 2015 and 2020 (Table 5). In the alternative scenario, costs were estimated to be €83.56 million in the same time period. Differences between both scenarios would allow Andalusia to save €229,706, which might be due to assumption of fewer PC visits and shorter hospital stays, associated with an assumption of better medication adherence (Table 6). Savings of Andalusia are higher compared with the results of the rest of the regions included in this study.
Regarding Catalonia, the total economic impact of the current scenario was estimated to be €30.02 million, while in the alternative scenario, we estimated €29.93 million over 2015–2018. Health care expenditure for asthma in Catalonia is the smallest one compared with the other regions included in this analysis. Given the results of both scenarios, the overall savings in Catalonia were estimated to be €90,145, which are obtained through reduction of health care resource, specifically fewer days of hospital stay and PC visits (Table 6).
The total economic impact without the introduction of DuoResp® Spiromax® in Galicia was estimated to be €58.59 million between 2015 and 2018, while in the alternative scenario was calculated to be €58.41 million (Table 5). Therefore, Galicia might save €188,327, principally due to fewer PC visits and a small proportion of fewer days of hospital stay (Table 6).
Referring to Madrid, the total economic impact for the current scenario throughout 2015–2018 was €51.66 million; meanwhile, the economic impact with the introduction of DuoResp® Spiromax® summed up to €51.53 million (Table 5). The total savings were expected to be €122,669 over the next 4 years, given the potentially positive effect of DuoResp® Spiromax® by reducing the days of hospitalization and PC visits (Table 6).
Finally, with regard to Valencia, economic impact without DuoResp® Spiromax® onto the market leads to €52.30 million throughout 2015–2018, while economic impact with DuoResp® Spiromax® is estimated to be €52.14 million (Table 5). Differences between both scenarios gave total savings of €165,796 (Table 5). These savings might be due to a reduction of hospitalization resource (length of stay) and PC visits (Table 6).
Discussion
Treating asthma is very costly for the regional health care budget, given the results illustrated in Table 5. Health care resources used in clinical practice included drug cost, medical visits, hospital resource utilization, and other interventions. However, a potential improvement in medication adherence due to a more intuitive device could impact economically by reducing medical visits and hospital resource use. Therefore, any intervention that could enhance medication adherence in asthmatic patients would be important to lower the economic impact of the disease. If the inhalation technique related to the inhaler does not help the drug-taking pattern because of lack of easy-to-use design, the drug-taking history does not coincide with the prescribed treatment. Indeed, we took into account the use of different inhalers in order to quantify the economic impact, keeping constant the drug effect (budesonide–formoterol). Finally, Turbuhaler presentations (Rilast® and Symbicort® Turbuhaler®) presented higher proportion of errors compared to DuoResp® Spiromax® according to our expert panel.
This study compares the cost of budesonide–formoterol FDC delivered by two different DPIs and estimates the budget impact for the asthma treatment in Spain. Results suggest that the increasing utilization of budesonide–formoterol FDC delivered by Spiromax®, that is DuoResp® Spiromax®, would result in a saving for the Spanish NHS of €1.509 million as well as savings for Andalusia, Catalonia, Galicia, Madrid, and Valencia of €229,706, €90,145, €188,327, €122,669, and €165,796, respectively.
Maintenance treatments are widely known and FDCs have become an advantage for controlling progression and symptomatology of the disease.34 As with any treatment for a chronic condition adherence is a requirement to achieve pharmacological efficacy, delay the progression of the disease, and avoid health care resource waste. Our results suggest that savings for Spain and five Spanish regions are possible given the estimate parameters based on clinical expert consultation. These outcomes are in line with the results of former studies that investigated the relationship between nonadherence and the use of health care resources in a population suffering from a pulmonary disease.5,8,35 A proportion of events such as hospitalization and excessive PC visits are avoidable and could provide cost savings if current adherence problems are addressed (Table 1).
One of the strengths for our analysis is that due to the lack of published clinical input data from Spanish populations, this study was based on the real-world utilization of these treatments and consequences of nonadherence, considering that input data were based on the expert opinion of specialists working in different Spanish hospitals. Furthermore, our analysis also includes results of five representative regions in Spain, which allowed us to have various regions involved by providing input to the estimated real-world utilization. Differences in asthma prevalence rates are significant and, for instance, health care resources for treating asthmatics in Catalonia are lower than those expended in Galicia although its population is smaller than the Catalonia population. Furthermore, Galicia might obtain greater savings if adherence improves in their population by using a more intuitive inhaler according to our estimations.
There are several limitations that should be noted. First, the model was developed to estimate the potential budget impact when increasing the market share of DuoResp® Spiromax® due to a reduced utilization of Symbicort® Turbuhaler® and Rilast® Turbuhaler®, whose FDC is budesonide–formoterol. This outcome may not reflect totally real-world change. Moreover, there are other FDCs such as salmeterol–fluticasone and formoterol/beclomethasone whose delivery system is also a DPI, for which Spiromax® could be an alternative although we did not hypothesize switching prescriptions. The quantification of gains in adherence leads to conservative quantities in our study, which in the end may be acceptable because we understand that the lack of adherence involves many factors,5,8,10,36 besides problems with the inhalation technique. Additionally, underdiagnosis of asthma is frequent, which is barrier for avoiding increasing prevalence rates of severe asthma. In Spain, underdiagnosis rates are high and half of the asthmatic population remains unaware of their condition.31 This would imply that asthma health care expenditure might be even higher as only a proportion of asthmatics are currently treated.
Conclusion
Enhancement of adherence is a key factor to guarantee the efficacy of treatment in multiple diseases, especially those with a chronic profile. In our study, we quantify the economic impact of a potential improvement in the inhalation technique in order to strengthen medication adherence. Results from our analysis suggest that an increase in utilization of DuoResp® Spiromax®, while the use of budesonide–formoterol FDC delivered by other devices Turbuhaler® (Symbicort® Turbuhaler® and Rilast® Turbuhaler®) decreases, could result in €1.509 million decrease in the overall budget over the period 2015–2018. These results could also be possible in the five Spanish regions included in this analysis, which are Andalusia, Catalonia, Galicia, Madrid, and Valencia.
Acknowledgments
BCN Health Economics and Outcomes Research SL provided statistical analysis and editorial support. The authors wish to thank Laura García-Bujalance, Rainel Sánchez-de la Rosa, and Saku Torvinen for their participation in this study. Preliminary results of this study were presented at the 19th Annual International Meeting of the International Society for Pharmacoeconomics and Outcomes Research (ISPOR) in Milan, Italy, November 9–12, 2015. This study was sponsored by Teva Pharma SLU.
Disclosure
JD is an employee of the University of Barcelona. GR is an employee of BCN Health Economics & Outcomes Research SL, Barcelona, Spain, an independent contract health economic organization. AB is an employee of Teva Pharma SLU. The authors report no other conflicts of interest in this work.
References
Eisner M, Katz P, Yelin E, Shiboski S, Blanc P. Risk factors for hospitalization among adults with asthma: influence of sociodemographic factors and asthma severity. Respir Res. 2001;2(1):53–60. | ||
Wisnivesky J, Leventhal H, Halm E. Predictors of asthma-related healthcare utilization and quality of life among inner-city patients with asthma. J Allergy Clin Immunol. 2005;116(3):636–642. | ||
Laube B, Janssens H, de Jongh F, et al. What the pulmonary specialists should know about the new inhalation therapies. Eur Respir J. 2011;37(3):1308–1331. | ||
Lavorini F, Magnan A, Dubus J, et al. Effect of incorrect use of dry powder inhalers on management of patients with asthma and COPD. Respir Med. 2008;102(4):593–604. | ||
Darba J, Ramirez G, Sicras A, Francoli P, Torvinen S, Sanchez-de la Rosa R. The importance of inhaler devices: the choice of inhaler device may lead to suboptimal adherence in COPD patients. Int J Chron Obstruct Pulmon Dis. 2015;10:2335–45. | ||
Global Initiative for Asthma (GINA). Global Strategy for Asthma Management and Prevention [updated 2015]. Available from: http://ginasthma.org/. Accessed August 1, 2015. | ||
SEMG [webpage on the Internet]. Guía española para el Manejo del Asma [Spanish Guidelines for the management of asthma] – GEMA 4.0. Available from: http://www.semg.es/documentos-semg/guias/1164-gema-4-0-2015.html. Accessed August 1, 2015. | ||
Gonzalez F, de la Fuente-Cid R, Álvarez-Gil R, Tafalla M, Nuevo J, Caamaño-Isorna F. Factors associated with asthma control in primary care patients: the CHAS Study. Arch Bronconeumol. 2010;46(7):358–363. | ||
Dalcin Pde T, Grutcki DM, Laporte PP, et al. Impact of short-term educational intervention on adherence to asthma treatment and on asthma control. J Bras Pneumol. 2011;37(1):19–27. | ||
Lokke A, Ahlbeck L, Bjermer L, et al. Expert Nordic perspectives on the potential of novel inhalers to overcome unmet needs in the management of obstructive lung disease. Eur Clin Respir J. 2015;2:29445. | ||
Virchow JC, Akdis CA, Darba J, et al. A review of the value of innovations in inhalers for COPD and asthma. J Mark Access Health Policy. 2015;3:28760. | ||
Canonica GW, Arp J, Keegstra JR, Chrystyn H. Spiromax, a new dry powder inhaler: dose consistency under simulated real-world conditions. J Aerosol Med Pulm Drug Deliv. 2015;28(5):309–319. | ||
BOE [webpage on the Internet]. Ministerio De Sanidad, Servicios Sociales E Igualdad. Orden SSI/2160/2015, October 17th, Where Public Prices Were Updated. Available from: https://www.boe.es/boe/dias/2015/10/17/pdfs/BOE-A-2015-11177.pdf. Accessed November 18, 2015. | ||
Carvajal-Ureña I, García-Marcos L, Busquets-Monge R, et al. Geographic variation in the prevalence of asthma symptoms in Spanish Children and Adolescents. International Study of asthma and allergies in childhood (ISAAC) Phase 3, Spain. Arch Bronconeumol. 2005;41(12):659–666. | ||
Urrutia I, Aguirre U, Sunyer J, et al. Changes in the prevalence of asthma in Spanish cohort of the European Community Respiratory Health Survery (ECRHS-II). Arch Bronconeumol. 2007;43(8):425–430. | ||
Colmenarejo J, Calle B, López A, Cisneros C [webpage on the Internet]. Programa Integrado de Continuidad Asistencial en Enfermedades Respiratorias. Integra ASMA Neumomadrid. Available from: www.neumomadrid.org/descargas/379-v2-integra-asma.pdf. Accessed January 2015. Accessed July 1, 2015. | ||
Lopez A, Pertega S, Rueda S, et al. Prevalencia de síntomas de asma en los niños y adolescentes de la Comunidad Autónoma de Galicia (España) y sus variaciones geográficas [Prevalence of asthma symptoms in children and teenagers in the Autonomous Community of Galicia (Spain) and its geographical variations]. Arch Bronconeumol. 2011;47(6):274–282. | ||
Instituto Nacional de Estadístico (INE) [webpage on the Internet]. Proyecciones de población a corto plazo; 2011–2021 [Short-term projects of population 2011–2012]. Available from: http://www.ine.es/jaxi/menu.do?type=pcaxis&path=%2Ft20%2Fp269%2F2011-2021&file=pcaxis&L=. Accessed March 5, 2015. | ||
Plaza V, Fernández M, Gómez F, et al. Guía Española para el Manejo de Asma (GEMA). Arch Bronconeumol. 2009;45(Suppl 7):2–35. | ||
IMS. National Sales Data 2013–2014 for Asthma Maintenance Treatment with Fixed-Dose-Combinations for Spain. Not publicly available. | ||
Azouz W, Chetcuti P, Hosker H, Saralaya D, Chrystyn H. Inhalation characteristics of asthma patients, COPD patients and healthy volunteers with the Spiromax and Turbuhaler devices: a randomised, cross-over study. BMC Pulm Med. 2015;15:47. | ||
Junta de Andalucía [webpage on the Internet]. Orden de 14 de octubre de 2005, por la que se fijan los precios públicos de los servicios sanitarios prestados por Centros dependientes del Sistema Sanitario Público de Andalucía [Order of 14 October 2005 establishing public prices of services provided by health centers under the Public Health System of Andalusia]. Available from: http://www.juntadeandalucia.es/boja/2005/210/28. Accessed March 5, 2015. | ||
Resolució SLT 353/2013. DOGC Núm 6326. 13/02/2013. Available from: http://www.comb.cat/home.aspx. Accessed April 2015. | ||
Madrid Autonomous Community [webpage on the Internet]. Orden 731/2013, de 6 de septiembre, del Consejero de Sanidad, por la que se fijan los precios públicos por la prestación de los servicios y actividades de naturaleza sanitaria de la Red de Centros de la Comunidad de Madrid [Order 731/2013, of September 6, of the Adviser of Health, for which public prices are set for the provision of health services and activities related to healthcare at the Network of Centres of the Community of Madrid]. Available from: http://www.madrid.org/wleg/servlet/Servidor?opcion=VerHtml&nmnorma=8275&cdestado=P. Accessed April 1, 2015. | ||
DIARI OFICIAL [webpage on the Internet]. Llei 14/2007, de 26 de desembre, de la Generalitat, de Mesures Fiscals, de Gestió Administrativa i Financera, i d’Organització de la Generalitat de la Comunitat Valenciana [Law 14/2007, the 26th of December, of the Government, about measures on taxes, and public management and financial from the Government of the Autonomous Community of Valencia]. Available from: www.docv.gva.es/datos/2007/12/28/pdf/2007_15710.pdf. Accessed April 1, 2015. | ||
DOG [Webpage on the Internet]. Decreto 56/2014. DOG Núm - 96 21/05/2014 Diario Oficial de la Xunta de Galicia [Order 56/2014 DOG number 9621/05/2014 Official jorunal of the Government of Galicia]. Available from: http://www.xunta.es/dog/Publicados/2014/20140521/AnuncioC3K1-140514-0001_es.html. Accessed April 1, 2015. | ||
SALUD [Webpage on the Internet]. Decreto 25/2010, de 17 de junio, por el que se actualizan los precios públicos por actos asistenciales y servicios sanitarios prestados por la Gerencia Regional de Salud de Castilla y León [Order 25/2010, 17th of June, to update public prices for care events and healthcare services provided by Regional Health Management of Castilla and Leon]. Available from: http://www.saludcastillayleon.es/institucion/es/resumen-bocyl-legislacion-sanitaria/decreto-25-2010-17-junio-actualizan-precios-publicos-actos-. Accessed April 1, 2015. | ||
Euskadi.eus [webpage on the Internet]. Tarifas para facturación de servicios sanitarios y docentes de Osakidetza para el año 2014 [Tariffs for the invoicing of healthcare and educational services from the Basque Country Government for 2014]. Available from: www.osakidetza.euskadi.eus/contenidos/./libro_tarifas/es./tarifas2014.pdf. Accessed April 1, 2015. | ||
BOC [webpage on the Internet]. Servicio Canario de la Salud.- Resolución de 1 de febrero de 2013, de la Directora, por la que se modifica la cuantía de los precios públicos de servicios sanitarios previstos en el Decreto 81/2009, de 16 de junio, por el que se establecen los precios públicos de los servicios sanitarios prestados por el servicio Canario de la Salud y se fijan sus cuantías [Healthcare service from Canary Islands. Order 1st of February 2013, from the Head of the department, which modifies public prices and set a fixed amount from healthcare services provided by the Healthcare service from Canary Islands]. Available from: http://www.gobiernodecanarias.org/boc/2013/051/001.html. Accessed April 1, 2015. | ||
Derecho [webpage on the Internet]. Resolución de 03/09/2012, de la Dirección Gerencia, sobre precios a aplicar por sus centros sanitarios a terceros obligados al pago o a los usuarios sin derecho a asistencia sanitaria [03/09/2015 resolution of the Management Board, on prices to apply by their health centers to users without the right to health care.]. [2012/12714]. Available from: http://legislacion.derecho.com/resolucion-03-09-2012-13-setiembre-2012-servicio-de-salud-de-castilla-la-mancha-sescam-4473664. Accessed April 1, 2015. | ||
ASESORIA & EMPRESA [webpage on the Internet]. Orden de 5 de febrero de 2013 de la Consejería de Economía y Hacienda, por la que se publican las tarifas de las tasas y precios publicos aplicables en el 2013 [Order of February 5, 2013 of the Adviser of Economy and Finance, for which the rates of fees and public prices are set to be applicable in 2013.]. Available from: http://portaljuridico.lexnova.es/legislacion/JURIDICO/188905/orden-de-5-de-febrero-de-2013-de-la-consejeria-de-economia-y-hacienda-por-la-que-se-publican-las-t. Accessed April 1, 2015. | ||
Saludinforma.es [webpage on the Internet]. Resolución de 30 de julio de 2012, de la Dirección Gerencia del Servicio Aragonés de Salud, sobre revisión de las tarifas a aplicar por la prestación de servicios sanitarios a terceros obligados al pago o a usuarios sin derecho a asistencia sanitaria en la Comunidad Autónoma de Aragón [Resolution of 30 July 2012, management of Aragon Health Service on revision of tariffs to be applied for the provision of health services to users without the right to health care in the Autonomous Community of Aragon]. Available from: https://www.saludinforma.es/portalsi/web/salud/servicios-prestaciones/modificaciones-aseguramiento/legislacion;jsessionid=UO8tW+KzATD4PEs6Bza5XtWd.mov-saludinforma-01. Accessed April 1, 2015. | ||
Caib.es [webpage on the Internet]. Consejería de Salud, Familia y Bienestar Social. Núm. 4814. Resolución del director general del Servicio de Salud de modificación de los anexos 1 y 2 de la Orden de la consejera de Salud y Consumo de 22 de diciembre de 2006 [Ministry of Health, Family and Social Welfare. No. 4814. Resolution of the General Director of Health Service amending Annexes 1 and 2 of the Order of the Minister of Health and Consumption of December 22, 2006.]. Available from: http://www.caib.es/eboibfront/es/2012/7826. Accessed April 1, 2015. | ||
Frois C, Wu E, Ray S, Colice G. Inhaled corticosteroids or long-acting β-agonists alone or fixed-dose combinations in asthma treatment: a systematic review of flucticasone/budesonide formoterol/salmeterol. Clin Ther. 2009;31(12):2779–2803. | ||
Mäkela M, Backer V, Hedegaard M, Larsson K. Adherence to inhaler therapies, health outcomes and costs in patients with asthma and COPD. Respir Med. 2013;107:1481–1490. | ||
Bender BG, Bender SE. Patient-identified barriers to asthma treatment adherence: responses to interviews, focus groups, and questionnaires. Immunol Allergy Clin North Am. 2005;25:107–130. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.