Back to Journals » Clinical Ophthalmology » Volume 12
Treatment outcomes in the DRy Eye Amniotic Membrane (DREAM) study
Authors McDonald MB, Sheha H , Tighe S, Janik SB, Bowden FW, Chokshi AR, Singer MA, Nanda S, Qazi MA , Dierker D , Shupe AT, McMurren BJ
Received 11 January 2018
Accepted for publication 1 March 2018
Published 9 April 2018 Volume 2018:12 Pages 677—681
DOI https://doi.org/10.2147/OPTH.S162203
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Scott Fraser
Marguerite B McDonald,1 Hosam Sheha,2–5 Sean Tighe,2,3 Susan B Janik,6 Frank W Bowden,7 Amit R Chokshi,8 Michael A Singer,9 Seema Nanda,10 Mujtaba A Qazi,11 Damon Dierker,12 Adam T Shupe,13 Brittany J McMurren14
1Ophthalmic Consultants of Long Island, Lynbrook, NY, USA; 2Ocular Surface Center and TissueTech, Inc., Miami, FL, USA; 3Florida International University Herbert Wertheim College of Medicine, Miami, FL, USA; 4Hofstra University School of Medicine, Hempstead, NY, USA; 5Research Institute of Ophthalmology, Cairo, Egypt; 6Solinsky Eye Care, Kensington, CT, USA; 7Bowden Eye & Associates, Jacksonville, FL, USA; 8Florida Eye Specialists, Jacksonville, FL, USA; 9Medical Center Ophthalmology Associates, San Antonio, TX, USA; 10TX Eye Institute, Houston, TX, USA; 11Pepose Vision Institute, Chesterfield, MO, USA; 12Eye Surgeons of Indiana, Indianapolis, IN, USA; 13Royo Eye Care, Marysville, CA, USA; 14Gordon and Weiss Vision Institute, San Diego, CA, USA
Purpose: To evaluate the efficacy of cryopreserved amniotic membrane (CAM) in reducing signs and symptoms of dry eye disease (DED) in a large patient population.
Methods: A retrospective chart review at 10 clinical sites was done of patients with refractory DED who received CAM and completed at least 3 months of follow-up. Data collected were demographics; medical history including previous and current ocular treatment, diagnosis, clinical presentations, comorbidity, duration and frequency of treatment with CAM; and concomitant medications. The primary outcome was the change in dry eye workshop (DEWS) score after treatment.
Results: A total of 97 eyes of 84 patients exhibited severe dry eye despite maximal medical treatments including topical artificial tears, cyclosporine-A, serum, antibiotics, and steroids. Patients manifested with superficial punctate keratitis (86%), filamentary keratitis (13%), exposure keratitis (19%), neurotrophic keratitis (2%), and corneal epithelial defect (7%). After CAM treatment for 5.4±2.8 days, 74 (88%) patients demonstrated an improved ocular surface along with a notable reduction of the severity as the overall DEWS score was significantly reduced from 3.25±0.5 at baseline to 1.44±0.6 at 1 week, 1.45±0.6 at 1 month, and 1.47±0.6 at 3 months (p<0.001). Ten eyes (10%) required repeated treatment to complete healing. Apart from discomfort during CAM placement, there were no adverse events.
Conclusion: Placement of CAM is promising to enhance the recovery of ocular surface health and reduce signs and symptoms in patients with moderate-to-severe DED.
Keywords: amniotic membrane, dry eye, ocular surface, severity, DEWS
Introduction
Dry eye disease (DED) is one of the most common ocular surface disorders in the USA and worldwide. It affects nearly 30% of the population, and its symptoms, such as ocular discomfort and visual fluctuations, represent the most frequent complaints in ophthalmic practice.1–4 DED is comprised of tear film insufficiency and ocular surface involvement. Despite different underlying pathogenic processes, inflammation is a common denominator in DED, which in turn induces further damage to the corneal epithelium and its underlying structures.5 Various treatment modalities, such as steroids and cyclosporine, have been used to suppress inflammation. However, results are variable and refractory in some cases. In these cases, DED not only negatively impacts the quality of life,2 but also increases the burden on health economics.
Recent progress has been made in understanding the pathogenesis of DED, and different treatment modalities have been introduced. Cryopreserved amniotic membrane (CAM) was used to treat DED with ocular surface involvement, and its short-term efficacy was attributed to its known potent anti-inflammatory effect.6 More recently, John et al7 further evaluated the potential effect of CAM in restoring corneal nerves in DED using in vivo confocal microscopy due to the known correlation between corneal nerve density and the severity of DED.8–10 They showed CAM treatment significantly increased corneal nerve density which was correlated with improved corneal sensitivity and reduced dry eye symptoms.7 This treatment effect was seen for 3 months and may be attributed to CAM’s rich composition of neurotrophic factors, particularly nerve growth factor (NGF).11–13 However, these studies were conducted on a relatively small patient population, and further studies are needed to substantiate the findings. Hence, in this study, we retrospectively reviewed the effect of CAM in a larger patient population with moderate-to-severe dry eye.
Methods
Study design and participants
This is a retrospective study to evaluate the efficacy of self-retained CAM (PROKERA® Slim, Bio-Tissue, Miami, FL, USA) in reducing signs and symptoms of DED associated with ocular surface involvement. The study was exempted under 45 CFR §46.101(b)(4) by the Western Institutional Review Board (Puyallup, WA, USA), and patient consent was not required. The study was conducted at ten clinical sites across the USA in accordance with the Health Insurance Portability and Accountability Act and Declaration of Helsinki. The medical records of patients with DED associated with ocular surface disorders who were treated with CAM as a temporary bandage (PROKERA® Slim) before June 1, 2016, and completed 1 week, 1 month and 3 months of follow-up were reviewed. Inclusion criteria also included subjects aged 21 years and older who had moderate-to-severe DED, grades 2–4, as defined by the Report of the International Dry Eye WorkShop (DEWS).1 Exclusion criteria included symblepharon, recent ocular surgery or injury within 3 months, contact lens wearers, and those who had undergone previous brain surgery or trigeminal nerve damage.
Data collected were demographics, medical history including previous and current ocular treatment, diagnosis, clinical presentations, comorbidity, duration and frequency of treatment with CAM, and concomitant medications. The DEWS score and the severity of DED including discomfort, visual symptoms, corneal staining, and corneal signs were graded from 1 (mild) to 4 (severe) as previously described.1,14 Posttreatment results were evaluated at 1 week, 1 month, and 3 months of follow-up and compared to the baseline. All data were recorded in such a manner that subjects could not be identified, directly or through identifiers linked to their records.
Statistical analysis
Descriptive statistics for continuous variables are reported as the mean ± SD and were analyzed using SPSS software, version 24.0 (SPSS Inc., Chicago, IL, USA). Differences between parameters before and after treatment were analyzed with the analysis of variance test and Student t-test. A p-value <0.05 was considered statistically significant.
Results
A total of 97 eyes of 84 patients [12 (14%) male, 69 (82%) female, and 3 (4%) unknown] were included in this study. They exhibited severe dry eye (DEWS 3.25±0.5) despite maximal medical treatments such as artificial tears (82%), steroids (44%), cyclosporine-A (40%), antibiotics (30%), serum drops (8%), and nonsteroidal anti-inflammatory drugs (5%). Punctal plugs were also noted in 29 cases (35%). The majority of patients presented with ocular discomfort (83%) and blurry vision (60%). Other symptoms included ocular pain (35%), redness (29%), and light sensitivity (14%). Most of the cases manifested with superficial punctate keratitis (86%) followed by exposure keratitis (19%), filamentary keratitis (13%), epithelial defect (7%), and neurotrophic keratitis (2%). Comorbidities included blepharitis (39%), cataract (36%), glaucoma (20%), lagophthalmos (7%), and conjunctivitis (5%).
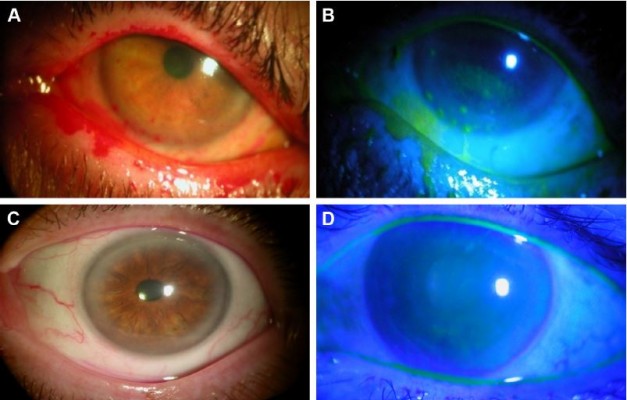
Placement and removal of CAM were uneventful in all cases. Tape-tarsorrhaphy was used in 26 cases (31%) to alleviate discomfort at the time of insertion. The average duration of CAM placement was 5.4±2.8 days, range 2–11 days. CAM was removed from 4 eyes (4%) after 2 days due to CAM intolerance, and another CAM fell out of one eye after 2 days. Upon removal, the AM was intact (28%), partially dissolved (20%), totally dissolved (42%), or not stated (10%). After removal, 74 patients (88%) demonstrated an improved ocular surface (Figure 1) along with notable reduction of the severity of dry eye symptoms. The overall DEWS score was significantly reduced from 3.25±0.5 at the baseline to 1.44±0.6 at 1 week (p<0.001), 1.45±0.6 at 1 month (p<0.001), and 1.47±0.6 at 3 months (p<0.001) (Figure 2). Specifically, ocular discomfort scores improved from 3.0±0.8 at baseline to 1.3±0.7 at 3 months (p<0.001); visual symptoms scores improved from 2.6±0.9 to 1.0±1.0 (p<0.001); corneal staining scores improved from 2.6±0.7 to 1.0±1.0 (p<0.001); and the overall corneal signs scores improved from 3.5±0.7 at baseline to 2.0±1.0 at 3 months (p<0.001). Although there was significant improvement in visual symptom, the change in distant visual acuity was not statistically significant.
Ten patients (10%), who had exposure keratitis or epithelial defects, did not heal at the time of CAM removal and required repeat treatment to complete the healing. For these patients, the overall DEWS score was significantly reduced from 3.60±0.7 at the baseline to 2.5±0.7 at 1 week, 1.90±0.9 at 1 month, and 1.90±0.9 at 3 months. The corneal staining scores improved from 3.3±1.0 at baseline to 1.9±1.2 at 3 months.
The ocular surface remained stable during the follow-up period while the patients continued to use conventional treatment including artificial tears (96%), cyclosporine-A (57%), steroids (32%), antibiotics (23%), serum drops (11%), and nonsteroidal anti-inflammatory drugs (8%). There was no significant change in the number of topical medications after CAM placement. Apart from CAM intolerance, there were no adverse events.
Discussion
This retrospective study demonstrates that self-retained CAM can accelerate the recovery of corneal surface health in patients with moderate and severe DED. Single placement of CAM for 5.4±2.8 days resulted in a significant improvement of DED signs and symptoms with an overall significant reduction in DEWS scoring from 3.25±0.5 (baseline) to 1.44±0.6 at 1 week, 1.45±0.6 at 1 month, and 1.47±0.6 at 3 months. This improvement was associated with restoration of corneal surface health as evidenced by resolution of corneal punctuate staining and improvement of visual symptoms. These findings are consistent with previous studies.6,7
The therapeutic effect of CAM in the treatment of DED can be attributed to multiple mechanisms of action. First, CAM acts as a therapeutic bandage that keeps the eye moist by retaining tears and protects the ocular surface from the surrounding environment. The second mechanism is by controlling ocular surface inflammation6 since it is well established that inflammation triggered by both innate and adaptive immune responses is critical to the pathogenesis and chronicity of DED.15,16 Taking the anti-inflammatory action as an example, CAM has been demonstrated to induce apoptosis of neutrophils,17,18 monocytes, and macrophages;19 reduce infiltration of neutrophils,17,18 macrophages,20,21 and lymphocytes;22 and promote polarization of M2 macrophages.23 Such anti-inflammatory action exerted by CAM is retained in the water-soluble extract24,25 and replicated by HC-HA/PTX3 purified from AM.11,12 A third mechanism is CAM’s ability to regenerate corneal nerves as previously reported,7,26 and this may explain the lasting effect. This notion is supported by the fact that NGF is abundantly present in CAM and is known to play an important role in nerve regeneration and epithelial healing.8,13,27 Other conventional topical anti-inflammatory therapies such as cyclosporine,28 corticosteroids,29 or nonsteroidal anti-inflammatory drugs30 have been found to compromise corneal nerves and may explain why some cases do not respond. Collectively, these actions mentioned above of CAM seem to be beneficial in treating DED.
In this study, the ocular surface did not heal in 10% of cases following single placement of CAM and required repeat treatment. These cases had exposure keratopathy, neurotrophic conditions, or persistent epithelial defect. These results are comparable with what has been reported previously by Suri et al,31 who have published a recurrence rate of 14.3%. This recurrence may be explained by the nature of the underlying disease or associated comorbidities. In fact, 39% of the cases had associated blepharitis in this study, and the improvement of lid hygiene helped improve the dry eye symptoms. Therefore, it is essential to look for other comorbidities and treat them accordingly.
DED presents as a public health problem32,33 with substantial economic implications including increased prescription medication use and expenditures for DED. It has been shown that CAM can alleviate DED accompanied with a significant reduction in usage of concomitant topical medications.6 However, the patients in the current study continued to use conventional treatment as per their usual routine even though their symptoms were refractory before CAM placement. Hence, it may be advisable to reduce the number and frequency of concomitant medications to minimize potential toxicity, economic burden, and impact on quality of life.
Conclusion
In conclusion, CAM is a promising treatment to enhance the recovery of ocular surface health and reduce signs and symptoms in patients with moderate-to-severe DED. Further studies are needed to determine longer-term effects (>3 months) and whether repetitive use of CAM generates a more lasting effect.
Acknowledgments
The abstract of this paper was partially presented at the American Society of Cataract and Refractive Surgery (ASCRS) annual meeting, Los Angeles, CA, May 2017, as a conference talk with interim findings. The abstract of this presentation is available at http://www.ascrs.org/node/29198. The ASCRS abstract was also discussed at the 34th Annual Cornea, Contact Lens, Contemporary Vision Care Symposium, Houston, TX, Dec 2017 available at https://ce.opt.uh.edu/live/2017/cornea-contact-lens-contemporary-vision-care-symposium/course-notes/. This work was also presented at the American Academy of Ophthalmology (AAO) annual meeting, New Orleans, LA, November 2017 as a poster presentation. The poster’s abstract is available at https://aao.scientificposters.com/epsAbstractAAO.cfm?id=1. This study was supported in part by a research grant from TissueTech, Inc. Miami, FL.
Disclosure
Dr McDonald and Dr Nanda are consultants and members of the speaker bureau of Bio-Tissue Inc., that distributes PROKERA®. Dr Sheha and Mr Tighe are employees of TissueTech Inc. The authors report no other conflicts of interest in this work.
References
The definition and classification of dry eye disease: report of the Definition and Classification Subcommittee of the International Dry Eye WorkShop. Ocul Surf. 2007;5:75–92. | ||
Galor A, Feuer W, Lee DJ, et al. Depression, post-traumatic stress disorder, and dry eye syndrome: a study utilizing the national United States Veterans Affairs administrative database. Am J Ophthalmol. 2012;154:340.e2–3466.e2. | ||
Schaumberg DA, Dana R, Buring JE, Sullivan DA. Prevalence of dry eye disease among US men: estimates from the Physicians’ Health Studies. Arch Ophthalmol. 2009;127:763–768. | ||
Schaumberg DA, Sullivan DA, Buring JE, Dana MR. Prevalence of dry eye syndrome among US women. Am J Ophthalmol. 2003;136:318–326. | ||
Sheha H, Tseng SCG. The role of amniotic membrane for managing dry eye disease. In: Ocular Surface Disorder. 39th ed. London: JP Medical; 2013:325–329. | ||
Cheng AM, Zhao D, Chen R, et al. Accelerated restoration of ocular surface health in dry eye disease by self-retained cryopreserved amniotic membrane. Ocul Surf. 2016;14:56–63. | ||
John T, Tighe S, Sheha H, et al. Corneal nerve regeneration after self-retained amniotic membrane in dry eye disease. J Ophthalmol. 2017:6404918. | ||
Lambiase A, Sacchetti M, Bonini S. Nerve growth factor therapy for corneal disease. Curr Opin Ophthalmol. 2012;23:296–302. | ||
Jain P, Li R, Lama T, Saragovi HU, Cumberlidge G, Meerovitch K. An NGF mimetic, MIM-D3, stimulates conjunctival cell glycoconjugate secretion and demonstrates therapeutic efficacy in a rat model of dry eye. Exp Eye Res. 2011;93:503–512. | ||
Labbe A, Liang Q, Wang Z, et al. Corneal nerve structure and function in patients with non-Sjogren dry eye: clinical correlations. Invest Ophthalmol Vis Sci. 2013;54:5144–5150. | ||
Touhami A, Grueterich M, Tseng SC. The role of NGF signaling in human limbal epithelium expanded by amniotic membrane culture. Invest Ophthalmol Vis Sci. 2002;43:987–994. | ||
Banerjee A, Nurnberger S, Hennerbichler S, et al. In toto differentiation of human amniotic membrane towards the Schwann cell lineage. Cell Tissue Bank. 2014;15:227–239. | ||
Aloe L, Tirassa P, Lambiase A. The topical application of nerve growth factor as a pharmacological tool for human corneal and skin ulcers. Pharmacol Res. 2008;57:253–258. | ||
Maychuk DY. Prevalence and severity of dry eye in candidates for laser in situ keratomileusis for myopia in Russia. J Cataract Refract Surg. 2016;42:427–434. | ||
Wei Y, Asbell PA. The core mechanism of dry eye disease is inflammation. Eye Contact Lens. 2014;40:248–256. | ||
Yagci A, Gurdal C. The role and treatment of inflammation in dry eye disease. Int Ophthalmol. 2014;34:1291–1301. | ||
Park WC, Tseng SCG. Modulation of acute inflammation and keratocyte death by suturing, blood and amniotic membrane in PRK. Invest Ophthalmol Vis Sci. 2000;41:2906–2914. | ||
Wang MX, Gray TB, Parks WC, et al. Corneal haze and apoptosis is reduced by amniotic membrane matrix in excimer laser photoablation in rabbits. J Cat Refract Surg. 2001;27:310–319. | ||
Shimmura S, Shimazaki J, Ohashi Y, Tsubota K. Antiinflammatory effects of amniotic membrane transplantation in ocular surface disorders. Cornea. 2001;20:408–413. | ||
Bauer D, Wasmuth S, Hermans P, et al. On the influence of neutrophils in corneas with necrotizing HSV-1 keratitis following amniotic membrane transplantation. Exp Eye Res. 2007;85:335–345. | ||
Heiligenhaus A, Bauer D, Meller D, Steuhl KP, Tseng SC. Improvement of HSV-1 necrotizing keratitis with amniotic membrane transplantation. Invest Ophthalmol Vis Sci. 2001;42:1969–1974. | ||
Bauer D, Wasmuth S, Hennig M, Baehler H, Steuhl KP, Heiligenhaus A. Amniotic membrane transplantation induces apoptosis in T lymphocytes in murine corneas with experimental herpetic stromal keratitis. Invest Ophthalmol Vis Sci. 2009;50:3188–3198. | ||
Bauer D, Hennig M, Wasmuth S, et al. Amniotic membrane induces peroxisome proliferator-activated receptor-gamma positive alternatively activated macrophages. Invest Ophthalmol Vis Sci. 2012;53:799–810. | ||
Li W, He H, Kawakita T, Espana EM, Tseng SCG. Amniotic membrane induces apoptosis of interferon-gamma-activated macrophages in vitro. Exp Eye Res. 2006;82:282–292. | ||
He H, Li W, Chen SY, et al. Suppression of activation and induction of apoptosis in RAW264.7 cells by amniotic membrane extract. Invest Ophthalmol Vis Sci. 2008;49:4468–4475. | ||
Morkin MI, Hamrah P. Efficacy of self-retained cryopreserved amniotic membrane for treatment of neuropathic corneal pain. Ocul Surf. 2018;16(1):132–138. | ||
Lambiase A, Mantelli F, Sacchetti M, Rossi S, Aloe L, Bonini S. Clinical applications of NGF in ocular diseases. Arch Ital Biol. 2011;149:283–292. | ||
Namavari A, Chaudhary S, Chang JH, et al. Cyclosporine immunomodulation retards regeneration of surgically transected corneal nerves. Invest Ophthalmol Vis Sci. 2012;53:732–740. | ||
Lee HK, Ryu IH, Seo KY, Hong S, Kim HC, Kim EK. Topical 0.1% prednisolone lowers nerve growth factor expression in keratoconjunctivitis sicca patients. Ophthalmology. 2006;113:198–205. | ||
Gaynes BI, Onyekwuluje A. Topical ophthalmic NSAIDs: a discussion with focus on nepafenac ophthalmic suspension. Clin Ophthalmol. 2008;2:355–368. | ||
Suri K, Kosker M, Raber IM, et al. Sutureless amniotic membrane prokera for ocular surface disorders: short-term results. Eye Contact Lens. 2013;39:341–347. | ||
Brewitt H, Sistani F. Dry eye disease: the scale of the problem. Surv Ophthalmol. 2001;45(Suppl 2):S199–S202. | ||
The epidemiology of dry eye disease: report of the Epidemiology Subcommittee of the International Dry Eye WorkShop. Ocul Surf. 2007;5:93–107. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.