Back to Journals » Vascular Health and Risk Management » Volume 15
The association between sedentary behavior, physical activity and hyperuricemia
Authors Park DY , Kim YS, Ryu SH , Jin YS
Received 3 January 2019
Accepted for publication 14 June 2019
Published 13 August 2019 Volume 2019:15 Pages 291—299
DOI https://doi.org/10.2147/VHRM.S200278
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Harry Struijker-Boudier
Doo Yong Park,1 Yeon Soo Kim,1,2 Seung Ho Ryu,3 Yu Sun Jin1
1Department of Physical Education, College of Education, Seoul National University, Seoul, Republic of Korea; 2Department of Physical Education, College of Education, Republic of Korea & Institute of Sport Science, Seoul National University, Seoul, Republic of Korea; 3Department of Occupational and Environmental Medicine, Kangbuk Samsung Hospital, Sungkyunkwan University School of Medicine, Seoul, Republic of Korea
Background: A significant discovery was recently made in which participation in physical activity and sedentary behavior, two contrasting lifestyles, was found to be related to the frequency of hyperuricemia diagnosis. The purpose of this study was to identify the association between sedentary behavior and physical activity levels in South Korean men and women diagnosed with hyperuricemia.
Methods: This study included 161,064 healthy men and women participants who had obtained a complete health examination. Physical activity levels and sitting time were assessed by the validated International Physical Activity Questionnaire Short Form Korean version. The presence of hyperuricemia in the subjects was determined by measuring serum uric acid (SUA) concentration (SUA ≥6 mg/dL [male], SUA ≥7 mg/dL [female]). Logistic regression analysis, adjusting other confounding factors, was conducted to identify the association of sedentary behavior and physical activity levels with hyperuricemia (p<0.05).
Results: Subjects who spent ≥10 hr/day in sedentary behavior were more likely to have hyperuricemia than those who spent <5 hour/day in sedentary behavior (OR=1.08, 95% CI=1.03–1.12). The subject group that more frequently participated in health enhanced physical activity (HEPA) had a lower hyperuricemia odds ratio than the subject group with lower physical activity participation rate (OR=0.90, 95% Ci=0.86–0.93). From the analysis of sex (male, female), age (young, middle, older), methods of measuring obesity (body mass index, waist circumference, body fat percentage), the association of sedentary behavior and physical activity levels with hyperuricemia was shown differently in different multivariable models.
Conclusion: Participation in regular physical activity and reduced sedentary time is highly recommended in order to reduce the prevalence of hyperuricemia.
Keywords: epidemiology, healthy lifestyle, uric acid, cardiovascular diseases
Introduction
Hyperuricemia is known as a condition that leads to oxidative stress which can lead to other illnesses such as gout and cardiovascular disease.1 According to the National Health and Nutrition survey released in 2007–2008, 21.4% of adults suffer from hyperuricemia.2 Owing to studies that reveal an increase in the number hyperuricemia patients, there has been a great interest in hyperuricemia.3,4 Present-day changes in living patterns, such as increased alcohol intake, high-calorie diets, decreased physical activity, as well as increased sedentary behavior, are causes of increased prevalence of hyperuricemia.5,6 Of these, physical activity and sedentary behavior guidelines for the elderly recommend shortening the daily average sitting time as well as living a more active life in order to prevent illness and rapid aging.7 In particular, cardiovascular disease is of national interest due to the fact that it is related to metabolic syndromes.8 In previous studies, it has been shown that the general population spends most of their awake time in sedentary behavior.9 As a result, 9.4% of the world population death rate is due to increased sedentary lifestyles. Thus, not only is a lack of physical activity considered to be an issue but also awareness and solution of time spent in sedentary behavior is deemed necessary.10 In the past, sedentary behavior was included under the term “lack of physical activity.” However, these are now considered to be two independent issues, and therefore, further research on sedentary lifestyles is recommended.11
Although lack of physical activity and sedentary behavior are being emphasized as the causes of multiple metabolic diseases in modern society, overall there are very few studies on hyperuricemia and physical activity and hardly any regarding sedentary lifestyles. Although in previous studies, the difference in prevalence rates of hyperuricemia due to the different lifestyles of people according to gender12 and age13 was reported, there is a lack of studies on hyperuricemia prevalence according to sedentary behavior and physical activity by gender and age. Further, different methods of measuring obesity, which is a major cause of hyperuricemia, such as body mass index, (BMI, kg/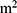 ), a waist measurement of central obesity, and bioelectrical impedance, each have distinct qualities. Therefore, independent studies should be performed on the relationship between obesity and hyperuricemia for each obesity indicator.14 In this study, we observed the effect of physical activity and sedentary behavior on hyperuricemia for different gender, age groups, and obesity indicators for measuring obesity.
), a waist measurement of central obesity, and bioelectrical impedance, each have distinct qualities. Therefore, independent studies should be performed on the relationship between obesity and hyperuricemia for each obesity indicator.14 In this study, we observed the effect of physical activity and sedentary behavior on hyperuricemia for different gender, age groups, and obesity indicators for measuring obesity.
Method
This study included a population of 300,984 men and women who were asked to complete a physical activity questionnaire and underwent a comprehensive health examination between June 2012 and August 2014. We excluded 142,271 participants from the total for the following reasons: missing data, a past history of malignant diabetes, cardiovascular disease, and cancer. A total of 158,713 participants were eligible for this study. The Institutional Review Board of Kangbuk Samsung Hospital approved this study, and since de-identified retrospective data (which was routinely collected during the health screening process) was used, the requirement for informed consent was waived.
Data on demographic characteristics, smoking status (never, past, current, or unknown), alcohol consumption (0, <10, 10 g/day, or unknown), education levels (high school graduate, none), medical history, and medication intake were collected by a standardized self-questionnaire.15
Self-administered questionnaires, as previously described, assessed physical activity levels and sitting time by the validated Korean version of the International Physical Activity Questionnaire Short Form (IPAQ-SF).16 Categories of physical activity level are derived from the following criteria: inactive (meeting neither minimally active nor health enhanced physical activity [HEPA]), minimally active (meeting any of the following three criteria: (i) three or more days of vigorous intensity activity for 20 min/day; (ii) five days or more of activity of moderate intensity or walking 30 min/day; or (iii) five days or more of any combination of walking, moderate, or vigorous intensity activities achieving 600MET min/week), and HEPA (which meets either of two criteria: (i) vigorous intensity activity on three or more days per week to accumulate to 1500 MET min/week; or (ii) seven days of any combination of walking, moderate, or vigorous intensity activities reaching at least 3000 MET min/week).16
The total sitting time accumulated during the weekdays was measured by time spent sitting in various domains (ie, leisure, work, and recreation). This study categorized sitting time by the following groups using cut-offs as described in previous studies: <5 h per day, 5–9 h per day, and >10 h/day.16
Usual dietary intake was assessed using a designed and validated Korean version of a 103-item, a self-administered food frequency questionnaire (FFQ). Total calorie intake was assessed by the Korean Nutrition Society Can pro 4.0 standardized food composition database.
Sleep quality was assessed by the Pittsburgh Sleep Quality Index (PSQI)17 and sleep duration was used to assess Component 3, the number of hours of actual night-time sleep during the past month. Trained nurses measured the height, weight, and body composition of participants who wore only a lightweight hospital gown during examinations. Body mass index (BMI) was calculated as weight (kg) divided by height (m) squared (kg/m2). Body fat percentage was estimated using a multi-frequency bioimpedance analyzer with eight-point tactile electrodes (InBody 720, Biospace Co., Seoul Korea).
Serum biochemical parameters measurements, including uric acid and insulin, are described in detail elsewhere.18 Insulin resistance was measured by the homeostatic model assessment, insulin resistance (HOMA-IR) equation,19 which is fasting blood insulin (lU/mL) * fasting blood glucose (mmol/L)/405.
Hyperuricemia was defined as having a serum uric acid concentration of ≥6.0 mg/dL (men) and ≥7.0 mg/dL (women).20 Blood pressure was assessed by an automated oscillometric device (53000, Welch Allyn, New York, USA) in which subjects placed their arm at heart level while in a seated position.
In 2017, we analyzed data using STATA 13.1(STATA corp., College Station, TX, USA). We used logistic regression with robust variance to evaluate the association of hyperuricemia with sitting time and physical activity as well as to estimate odds ratios (OR) with 95% confidence intervals (CIs) for hyperuricemia. To progressively reduce confounding associations, we used a multivariable model which included both sitting time and physical activity as well as age, sleep duration, smoking, alcohol intake, history of hypertension, BMI, total protein, total energy intake, education level, and HOMA-IR.
Additionally, we tested the association between sitting time and physical activity with the risk of hyperuricemia by BMI, waist circumference, and percent fat mass (%). We included these variables in the multivariable models. Several categories were used as continuous variables and were tested on each multivariable model. All p-values were two-tailed, and values of p<0.05 were considered statistically significant.
Result
Characteristics of the participants are presented in Table 1. Those with a sitting time of ≥10 h were more likely to be male young adults and have a high BMI, low current smoking rate, high rate of high education level, high HOMA-IR, high waist circumference, high alcohol consumption, low percent body fat, low sleep duration, low rate of hypertension, high calorie intake, and high protein intake compared to those with a sitting time of <5 h/day.
 |
Table 1 Baseline characteristics of study participants |
Participants who participated in HEPA were more likely to be older male adults (≥40 years) and have high BMI, low current smoking rate, high waist circumference, high alcohol consumption, low percent body fat, low sleep duration, low HOMA-IR, low rate of high education level, high rate of hypertension, high calorie intake, and high protein intake in comparison to those who were physically inactive.
Table 2 shows the impact of sedentary behavior and physical activity participation on hyperuricemia according to gender. In multivariable logistic regression adjusting for potential confounders, including physical activity level, subjects who spent ≥10 h/day in sedentary behavior were more likely to have hyperuricemia than those who spent <5 h/day (OR=1.08, 95% CI=1.03–1.12). Participation in HEPA declined prevalence of hyperuricemia compared to the minimally active group (OR=0.90, 95% CI=0.86–0.93). Sitting time of ≥10 h (OR=1.07, 95% CI=1.02–1.11) and HEPA (OR=0.90, 95% CI=0.86–0.93) were associated with hyperuricemia in men, but not in women (≥10 h/day sitting time OR=1.31, 95% CI=0.98–1.72; HEPA OR=1.04, 95% CI=0.77–1.41).
 |
Table 2 Association of sedentary behavior and physical activity level with hyperuricemia in healthy adult by gender |
Table 3 shows the impact of sedentary behavior and physical activity participation on hyperuricemia according to age. The young adult group (age 18–39 years), who spent 5–9 h/day and ≥10 h/day in sedentary behavior, showed an increase in hyperuricemia OR, compared to those who spent <5 h/day in sedentary behavior (5–9 h/day OR=1.09, 95% CI=1.03–1.15; ≥10 h/day OR=1.11, 95% CI=1.04–1.17), and participation in HEPA declined hyperuricemia compared to the minimally active group (OR=0.92, 95% CI=0.87–0.97). The middle age group participants (age 40–64 years) who participated in HEPA (OR=0.88, 95% CI=0.83–0.94), showed association with the hyperuricemia OR. However, the hyperuricemia OR in the elderly group (age ≥65 years) was only associated with HEPA participation (OR=0.69, 95% CI=0.50–0.96).
 |
Table 3 Association of sedentary behavior and physical activity level with hyperuricemia in healthy adult by age |
Table 4 shows the impact of sedentary behavior and physical activity participation on hyperuricemia according to the obesity index. Model 1, in which the body mass index was taken into account, shows that when sitting time is 5–9 h/day and ≥10 h/day, the hyperuricemia OR relationship is increased by 4% (95% CI=1.00–1.08) and 8% (95% CI=1.03–1.12), respectively, compared to that when sitting time is <5 h/day. Model 2, in which waist circumference measurements were taken into account, shows that when sitting time is ≥10 h/day, the hyperuricemia OR increased by 6% (95% CI=1.02–1.11) compared to that when sitting time is <5 h/day. However, similar results did not appear in the model in which body fat percentage was taken into account. The hyperuricemia OR in model 1 that was calibrated according to the body mass index showed association only with the participants’ HEPA participation (OR=0.90, 95% CI=0.86–0.93).
 |
Table 4 Association of sedentary behavior and physical activity level with hyperuricemia in healthy adult by obesity index |
Discussion
In this study, the following results were obtained regarding the relationship between physical activity and sedentary behavior individually with hyperuricemia. Increased sitting time can increase the OR relationship of hyperuricemia. For males, in particular, it has been shown that an increase in physical activity participation is an individual factor that can decrease the OR for hyperuricemia. According to different age ranges, the hyperuricemia OR increased for those between 18–39 years of age with >5 h of sitting time. For all age group ranges, the hyperuricemia OR decreased when higher levels of physical activity were met.
Further, this study showed that regular participation in physical activity and a decrease in sedentary time is related to decreasing the odds rate for hyperuricemia by obesity mediation.
Recent studies have reported that the increase in serum uric acid levels can be affected by not only higher alcohol intake rates, high-calorie diets, decreased physical activity, and increased sedentary behavior, but also biological processes of humans through genetic diversity as explained by the Mendelian randomization studies.21
According to one study, the genes associated with uric acid reabsorption in the kidneys are SLC2A9 and ABCG2, showing a 0.373 mg/dL and 0.217 mg/dL increase in uric acid, respectively.22 The increase in serum uric acid levels is determined by the interaction with environmental factors such as genetic factors and lifestyle habits.21 Management of lifestyle habits is important for the management of hyperuricemia.
Physical inactivity is thought to be a strong individual factor of all types of death in people with cardiovascular disease.23 Several previous studies have shown that according to the amount of physical activity, there is a positive impact on energy expenditure increase as well as weight loss, regardless of other factors that cause obesity.7 Additionally, according to research that shows getting plenty of physical activity can prevent weight gain, physical activity is shown to play a key factor in obesity prevention.24 A study report in which, regardless of physical activity, adults with >10 h of sitting time versus adults with <1 h of sitting time have a 34% higher chance of mortality. Therefore, it strengthens the notion that sitting time is an independent factor impacting the health index, apart from physical activity.25 Moreover, according to recent studies, when the daily average sitting time is exceeded, positive health outcomes provided by regular exercise disappear and can cause the chances of chronic illness to increase.26 Thus, whether physical activity and sitting time are independent causes of illness is still under debate.
A meta-analysis of the association between physical activity and hyperuricemia has shown that the risk of elevated uric acid levels is reduced by 12% in low and moderate intensity physical activity and reduced by 29% in high-intensity physical activity participation.27
Although it has been confirmed in this study that sedentary behavior and physical activity are independent causes of hyperuricemia, the exact mechanism of this is unclear. However, according to previous studies, physical activity, sedentary behavior, and hyperuricemia are thought to have a relationship with insulin resistance and obesity.
Previous studies have shown that an increase in blood insulin levels promotes sodium reabsorption in the renal tubule, which decreases uric acid clearance in the tubule and increases uric acid levels in the blood.28 Because the pentose phosphate pathway (which is associated with fat synthesis) is associated with the activation of xanthine oxidase, which produces uric acid,29 insulin resistance, and obesity are known to increase serum uric acid production.24,25
In relation to this phenomenon, physical activity can cause a decrease in obesity and insulin resistance by the contraction of active muscles due to increase in energy expenditure and glucose usage.7 Physical activity itself seems to decrease uric acid release as well as the decomposition of purine.24,30 Unlike physical activity, a sedentary lifestyle will lead to the deactivation of maintenance muscles due to leaned posture which means that there is lesser energy expenditure than when in standing posture.31,32 This suggests that the deactivation of lipoprotein in the muscles as well as the decrease in glucose usage,31,32 will cause an increase in insulin resistance.33,34 Accordingly, involvement in physical activity and decrease in everyday sitting time leads to an improvement in obesity35 and insulin resistance36 due to increase in energy consumption which can decrease the risk of hyperuricemia.
Table 2 shows that the physical activity, sitting time, and hyperuricemia OR relationship in men are shown to be related. This may be due to the biological difference between the lifestyle patterns of men and women. Due to renal clearance of uric acid and low uric acid reabsorption in the renal tubule that results from high estrogen levels in women, men who have comparatively low estrogen levels are more prone to develop hyperuricemia.37 Further, Table 1 of this research shows that compared to women, men have a 10 hr-longer sedentary lifestyle but are more involved in physical activity. Therefore, according to previous research, it can be said that physical activity and sedentary lifestyle are independent causes that lead to disease. Gender-based differences in physical activity participation and sitting time can cause the difference in disease diagnosis rates.38 In conclusion, high obesity index as well as low estrogen levels in men, biological proneness of uric acid buildup in the blood, longer sitting time, and more active lifestyles have a greater and clearer relationship between physical activity and sedentary lifestyles with hyperuricemia in men than in women.39
Table 3 shows that unlike adults under the age of 65, elders above the age of 65 showed a decrease in the hyperuricemia OR for participation in physical activity. This means that an increase in protein synthesis rate due to participation in high-quality physical activity is more likely to decrease the hyperuricemia OR than protein decomposition rate due to aging. According to previous studies, regular participation in physical activity can help increase muscle mass and contraction protein synthesis ratio,40 as well as decrease all-cause mortality and disease rate due to aging.41 Therefore, an increase in physical activity participation, apart from sitting time in adults over the age of 65, will slow down the rapid aging of cells. The level of nucleic acid, which is a decomposition product of cells, decreases causing uric acid composition in the blood to decrease, thereby, lowering the risk of hyperuricemia.42,43
In Table 4, the relationship between all kinds of activity (sitting time and physical activity participation) and the hyperuricemia OR relationship could not be found in the body fat percentage model, unlike other models. In previous studies, increased leptin levels in the blood due to continuous body fat buildup caused uric acid concentration to increase.44 Additionally, other research has shown that increased sedentary time and decreased physical activity causes a restriction in muscle usage and thus decreased energy expenditure increases body fat. This shows that obesity is related to physical activity, sedentary lifestyles, and hyperuricemia.32 The most common forms of obesity measurement are BMI using height and weight, measuring waist circumference for abdominal obesity, and the impedance technique. These obesity measurement techniques show different characteristics for different diseases of which,45–47 according to previous research on the relationship between obesity and uric acid concentrations, BMI and waist circumference measurements did not show any relationship to uric acid concentrations although it showed relationship to body fat percentage.44 In conclusion, increased sedentary behavior and decreased physical activity can cause a decrease in energy consumption due to restricted muscle movement which can cause an increase in body fat percentage.32 Therefore, increased body fat percentage due to increased sedentary behavior and decreased participation in physical activity can cause a higher risk of hyperuricemia.
This study contains several limitations. First, because we assessed hyperuricemia with only one measurement of serum uric acid, it was not possible to confirm that the state of oxidation-induced hyperuricemia persisted for a long time. However, this study is a cross-sectional study using large-scale cohort data, and it confirms the relationship between hyperuricemia, physical activity, and sedentary time. Further studies should be done to investigate the relationship between hyperuricemia and physical activity by periodically measuring serum uric acid levels every 3 months. Second, measurement of participation in physical activity was done by a self-assessment questionnaire which can be affected by recall bias. In future studies, a more objective method of measurement such as an accelerometer should be used. Third, because the questionnaire results for physical activity did not indicate the type of exercise the participants performed (aerobic or resistance exercise), it was not possible to elucidate which type of physical activity had an effect on hyperuricemia. In further studies, the type of physical activity should be specified in the questionnaire. Fourth, the food frequency questionnaire used in this study has problems with recall bias or lack of data. In future studies, it is necessary to study energy intake by doubly-labeled water (a gold standard for energy intake) and use it as a confounding variable that can affect hyperuricemia. Fifth, because this research was a cross-sectional study, it was not possible to find the causal relationship between the independent variable and the intermediate variable. Further, a longitudinal study should be done on this same topic.
Conclusion
Lifestyle changes in modern-day society can cause various diseases which could lead to the risk of death. Although different according to gender and age, physical activity and sitting time are two easily changeable lifestyle patterns that with constant effort, anyone can fix in order to maintain one’s health. In conclusion, since increased physical activity as well as decreased sitting time in Korean adults has a relationship with hyperuricemia which causes various complications, to ensure healthy lifestyles, not emphasis must be placed on increasing physical activity and cutting down sitting time
Acknowledgment
The abstract of this paper was presented at the ACSM Annual Meeting 2017 as a poster presentation/conference talk with interim findings. The poster’s abstract was published in “Poster Abstracts” in Medicine & Science in Sports & Exercise: DOI (10.1249/01.mss.0000519087.35023.d1).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Bengtsson C, Lapidus L, Stendahl C, Waldenström J. Hyperuricaemia and risk of cardiovascular disease and overall death: a 12-year follow-up of participants in the Population Study of Women in Gothenburg, Sweden. Acta Med Scand. 1988;224(6):549–555.
2. Zhu Y, Pandya BJ, Choi HK. Prevalence of gout and hyperuricemia in the US general population: the National Health and Nutrition Examination Survey 2007–2008. Arthritis Rheumatism. 2011;63(10):3136–3141. doi:10.1002/art.30520
3. Trifirò G, Morabito P, Cavagna L, et al. Epidemiology of gout and hyperuricaemia in Italy during the years 2005–2009: a nationwide population-based study. Ann Rheum Dis. 2013;72(5):694–700 .
4. Chen L-Y, Zhu W-H, Chen Z-W, et al. Relationship between hyperuricemia and metabolic syndrome. J Zhejiang Univ Sci B. 2007;8(8):593–598. doi:10.1631/jzus.2007.B0831
5. Choi HK, Atkinson K, Karlson EW, Willett W, Curhan G. Alcohol intake and risk of incident gout in men: a prospective study. Lancet. 2004;363(9417):1277–1281. doi:10.1016/S0140-6736(04)16000-5
6. Choi HK, Atkinson K, Karlson EW, Willett W, Curhan G. Purine-rich foods, dairy and protein intake, and the risk of gout in men. N Engl J Med. 2004;350(11):1093–1103. doi:10.1056/NEJMoa035700
7. Bouchard C, Blair SN, Katzmarzyk PT, et al. Less sitting, more physical activity, or higher fitness? Mayo Clinic Proc. 2015. Elsevier. doi:10.1016/j.mayocp. 2015;90(11):1533–1540
8. Owen N, Healy GN, Matthews CE, Dunstan DW. Too much sitting: the population-health science of sedentary behavior. Exerc Sport Sci Rev. 2010;38(3):105–113. doi:10.1097/JES.0b013e3181e373a2
9. Tremblay MS, Colley RC, Saunders TJ, Healy GN, Owen N. Physiological and health implications of a sedentary lifestyle. Appl Physiol Nutr Metab. 2010;35(6):725–740. doi:10.1139/H10-079
10. Lee I-M, Shiroma EJ, Lobelo F, Puska P, Blair SN, Katzmarzyk PT. Effect of physical inactivity on major non-communicable diseases worldwide: an analysis of burden of disease and life expectancy. Lancet. 2012;380(9838):219–229. doi:10.1016/S0140-6736(12)61031-9
11. Barnes J, Behrens TK, Benden ME, et al. Letter to the editor: standardized use of the terms” sedentary” and” sedentary behaviours”. Appl Physiol Nutr Metab. 2012;37(3):540–542. doi:10.1139/h2012-024
12. Wang S-F, Shu L, Wang S, et al. Gender difference in the association of hyperuricemia with hypertension in a middle-aged Chinese population. Blood Press. 2014;23(6):339–344. doi:10.3109/08037051.2014.906131
13. Ryu S, Chang Y, Zhang Y, et al. A cohort study of hyperuricemia in middle-aged South Korean men. Am J Epidemiol. 2011;175(2):133–143. doi:10.1093/aje/kwr291
14. Ehrampoush E, Arasteh P, Homayounfar R, et al. New anthropometric indices or old ones: which is the better predictor of body fat? Diabetes Metab Syndrome. 2017;11(4):257–263. doi:10.1016/j.dsx.2016.08.027
15. Aadahl M, Andreasen AH, Hammer-Helmich L, et al. Recent temporal trends in sleep duration, domain-specific sedentary behaviour and physical activity. A survey among 25–79-year-old Danish adults. Scand J Public Health. 2013;41(7):706–711. doi:10.1177/1403494812466989
16. IPAQ Research Committee. Guidelines for data processing and analysis of the International Physical Activity Questionnaire (IPAQ)-short and long forms. 2005.
17. Buysse DJ, Reynolds III CF, Monk TH, Berman SR, Kupfer DJ. The Pittsburgh sleep quality index: a new instrument for psychiatric practice and research. Psychiatry Res. 1989;28(2):193–213.
18. Sung KC, Wild SH, Kwang HJ, et al. Fatty liver, insulin resistance, and features of metabolic syndrome: relationships with coronary artery calcium in 10,153 people. Diabetes Care. 2012;35(11);2359–2364.
19. Takir M, Kostek O, Ozkok A, et al. Lowering uric acid with allopurinol improves insulin resistance and systemic inflammation in asymptomatic hyperuricemia. J Invest Med. 2015;63(8):924–929. doi:10.1097/JIM.0000000000000242
20. Yu S, Yang H, Guo X, et al. Prevalence of hyperuricemia and its correlates in rural Northeast Chinese population: from lifestyle risk factors to metabolic comorbidities. Clin Rheumatol. 2016;35(5):1207–1215. doi:10.1007/s10067-015-3051-6
21. Merriman TR. An update on the genetic architecture of hyperuricemia and gout. Arthritis Res Ther. 2015;17(1):98. doi:10.1186/s13075-015-0609-2
22. Köttgen A, Albrecht E, Teumer A, et al. Genome-wide association analyses identify 18 new loci associated with serum urate concentrations. Nat Genet. 2013;45(2):145. doi:10.1038/ng.2500
23. US Department of Health and Human Services. Physical Activity and Health: A Report of the Surgeon General. DIANE Publishing; 1996.
24. Katzmarzyk P, Janssen I, Ardern C. Physical inactivity, excess adiposity and premature mortality. Obesity Rev. 2003;4(4):257–290.
25. Chau JY, Grunseit AC, Chey T, et al. Daily sitting time and all-cause mortality: a meta-analysis. PLoS One. 2013;8(11):e80000. doi:10.1371/journal.pone.0080000
26. Thyfault JP, Du M, Kraus WE, et al. Physiology of sedentary behavior and its relationship to health outcomes. Med Sci Sports Exerc. 2015;47(6):1301. doi:10.1249/MSS.0000000000000651
27. Yuan H, Yu C, Li X, et al. Serum uric acid levels and risk of metabolic syndrome: a dose-response meta-analysis of prospective studies. J Clin Endocrinol Metab. 2015;100(11):4198–4207. doi:10.1210/jc.2015-2527
28. Facchini F, Chen Y-DI, Hollenbeck CB, Reaven GM. Relationship between resistance to insulin-mediated glucose uptake, urinary uric acid clearance, and plasma uric acid concentration. Jama. 1991;266(21):3008–3011.
29. Tsushima Y, Nishizawa H, Tochino Y, et al. Uric acid secretion from adipose tissue and its increase in obesity. J Biol Chem. 2013;288(38):27138–27149.
30. Wannamethee SG, Shaper AG, Alberti KGM. Physical activity, metabolic factors, and the incidence of coronary heart disease and type 2 diabetes. Arch Intern Med. 2000;160(14):2108–2116.
31. Bey L, Hamilton MT. Suppression of skeletal muscle lipoprotein lipase activity during physical inactivity: a molecular reason to maintain daily low‐intensity activity. J Physiol. 2003;551(2):673–682. doi:10.1113/jphysiol.2003.045591
32. Hamilton TM, Hamilton GD, Zderic WT. Exercise physiology versus inactivity physiology: an essential concept for understanding lipoprotein lipase regulation. Exerc Sport Sci Rev. 2004;32(4):161–166.
33. Hu FB, Leitzmann MF, Stampfer MJ, et al. Physical activity and television watching in relation to risk for type 2 diabetes mellitus in men. (Original Investigation). Arch Internal Med. 2001;161(12):1542. doi:10.1001/archinte.161.12.1542
34. Hu F, Li T, Colditz G, Willett W, Manson JE. Television watching and other sedentary behaviors in relation to risk of obesity and type 2 diabetes mellitus in women. JAMA. 2003;289(14):1785–1791. doi:10.1001/jama.289.14.1785
35. Ching PLYH, Willett WC, Rimm EB, et al. Activity level and risk of overweight in male health professionals. Act Leve Risk Overweight Male Health Professionals. 1996;86(1):25–30.
36. Assah F, Brage S, Ekelund U, et al. The association of intensity and overall level of physical activity energy expenditure with a marker of insulin resistance. Clin Exp Diabetes Metab. 2008;51(8):1399–1407.
37. Mateos Antón F, García Puig J, Ramos T, González P, Ordás J. Sex differences in uric acid metabolism in adults: evidence for a lack of influence of estradiol-17β (E 2) on the renal handling of urate. Metabolism. 1986;35(4):343–348.
38. Balkau B, Mhamdi L, Oppert J-M, et al. Physical activity and insulin sensitivity the RISC study. Diabetes. 2008;57(10):2613–2618. doi:10.2337/db07-1605
39. Lamonte M, Blair S, Church T. Physical activity and diabetes prevention. J Appl Physiol. 2005;99(3):1205–1213. doi:10.1152/japplphysiol.00193.2005
40. Desouza AC, Shapiro FL, Clevenger MC, et al. Regular aerobic exercise prevents and restores age-related declines in endothelium-dependent vasodilation in healthy men. Circulation. 2000;102(12):1351–1357. doi:10.1161/01.CIR.102.12.1351
41. Morey MC, Pieper CF, Crowley GM, et al. Exercise adherence and 10-year mortality in chronically ill older adults. J Am Geriatr Soc. 2002;50(12):1929–1933.
42. Roddy E, Doherty M. Epidemiology of gout. Arthritis Res Ther. 2010;12(6):223. doi:10.1186/ar3199
43. Álvarez-Lario B, Macarrón-Vicente J. Uric acid and evolution. Rheumatology. 2010;49(11):2010–2015. doi:10.1093/rheumatology/keq204
44. Fruehwald-Schultes B, Peters A, Kern W, et al. Serum leptin is associated with serum uric acid concentrations in humans. Metabolism. 1999;48(6):677–680.
45. World Health Organization. Netlibrary I, Netlibrary I. Obesity [electronic Resource]: Preventing and Managing the Global Epidemic: Report of a WHO Consultation. Geneva: World Health Organization; 2000.
46. He S, Chen X, Miele C. Could the new body shape index predict the new onset of diabetes mellitus in the Chinese Population? (Research article). PLoS One. 2013;8(1):e50573. doi:10.1371/journal.pone.0050573
47. Sun G, French CR, Martin GR, et al. Comparison of multifrequency bioelectrical impedance analysis with dual-energy X-ray absorptiometry for assessment of percentage body fat in a large, healthy population. (Author abstract). Am J Clin Nutr. 2005;81(1):74–78. doi:10.1093/ajcn/81.1.74
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
