Back to Journals » Clinical Ophthalmology » Volume 13
Systemic exposure to intracameral vs topical mydriatic agents: in cataract surgery
Authors Guell J, Behndig A, Pleyer U , Jaulerry S, Rozot P, Pisella PJ, Robert PY, Lanzl I, Pourjavan S , Aguiar C, Fernandez J, Grabner G, Mencucci R, Chiambaretta F , Labetoulle M
Received 4 October 2018
Accepted for publication 21 January 2019
Published 3 May 2019 Volume 2019:13 Pages 811—819
DOI https://doi.org/10.2147/OPTH.S189671
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Jose Guell,1 Anders Behndig,2 Uwe Pleyer,3 Stéphane Jaulerry,4 Pascal Rozot,5 Pierre-Jean Pisella,6 Pierre-Yves Robert,7 Ines Lanzl,8 Sayeh Pourjavan,9 Carlos Aguiar,10 Joaquin Fernandez,11 Gunther Grabner,12 Rita Mencucci,13 Frédéric Chiambaretta,14 Marc Labetoulle15
On behalf of the Intracameral Mydrane (ICMA) and Ethics Group
1Cornea and Refractive Surgery Unit, Instituto de Microcirugía Ocular, Barcelona, Spain; 2Department of Clinical Science/Ophthalmology, Umeå University Hospital, Umeå, Sweden; 3Universitäts-Augenklinik, Charité Campus Virchow-Klinik, Berlin, Germany; 4Centre Hospitalier de Bigorre, Tarbes, France; 5Clinique Monticelli, Marseille, France; 6Hôpital Bretonneau, CHRU de Tours, Tours, France; 7Hôpital Dupuytren, CHU de Limoges, Limoges, France; 8Chiemsee Augen Tagesklinik, Prien, Germany; 9Cliniques Universitaires Saint-Luc, Bruxelles, Belgium; 10Hospital de Santo Antonio, Porto, Portugal; 11Hospital Torrecárdenas, Almeria, Spain; 12Universitätsklinik Für Augenheilkunde Und Optometrie, Der Paracelsus, Austria; 13AOU Careggi, Clinica Oculistica, Università Degli Studi di Firenze, Firenze, Italy; 14Hôpital Gabriel Montpied, CHU de Clermont-Ferrand, Clermont-Ferrand, France; 15Hôpital Bicêtre, Université Paris Sud, Paris, France
Objective: The objective of this study was to compare systemic exposure to tropicamide/phenylephrine following intracameral or topical administration before cataract surgery.
Patients and methods: Mydriatics exposure was calculated in patients randomized to intracameral fixed combination of mydriatics and anesthetic ([ICMA]: tropicamide 0.02%, phenylephrine 0.31%, and lidocaine 1%, N=271) or mydriatic eye drops ([EDs]: tropicamide 0.5% and phenylephrine 10%, N=283). Additional doses were permitted if required. Mydriatic plasma levels were determined by mass spectrometric HPLC in 15 patients per group before and after administration.
Results: Most ICMA patients (73.6%) received a single dose (200 µL) representing an exposure to tropicamide of 0.04 mg and phenylephrine of 0.62 mg. None of these patients received additional mydriatics. In the control group (three administrations), the exposure was 0.45 (11.3-fold higher than ICMA) and 10.2 (16.5-fold higher) mg. When additional ED was used in this group (9.2% of patients), it was 37.5-fold higher for tropicamide (10 drops, 1.5 mg) and 54.8-fold higher for phenylephrine (10 drops, 34 mg) than the recommended ICMA dose. Tropicamide plasma levels were not detectable at any time point in ICMA patients while it was detectable in all ED patients at 12 and 30 minutes. Phenylephrine was detectable in 14.3% of ICMA patients compared to all ED patients at least at one time point. More ED patients experienced a meaningful increase in blood pressure and/or heart rate (11.2% vs 6.0% of ICMA patients; P=0.03).
Conclusion: Systemic exposure to tropicamide/phenylephrine was lower and cardiovascular (CV) effects were less frequent with ICMA. This could be of particular significance in patients at CV risk.
Keywords: cataract surgery, intracameral mydriasis, topical mydriasis, systemic influence, cardiovascular safety
Introduction
Cataract surgery is the second most commonly undertaken surgical procedure after intravitreal injections.1 Cataract is associated with old age and, as the population ages, the demand for cataract surgery is increasing and will likely continue to increase. Additionally, elderlies are at an increased risk of developing multiple pathologies and are consequently polymedicated. Moreover, diagnosis and screening for cataract are improving, putting further pressure on health care resources that may not be keeping pace with the demand.1,2
A stable mydriasis is required for successful cataract surgery and lens implantation. The combination of tropicamide and phenylephrine eye drops (EDs) is the current gold standard for most surgeons worldwide, but the usual regimen requires multiple instillations that could possibly expose to overdose associated with increased risk of cardiovascular (CV) events. Moreover, a particular side effect of local overdose is the ocular surface toxicity leading to toxic keratopathy that may interfere with the quality of visualization during surgery.3 An alternative could be the use of a mydriatic insert that ensures a better control of the delivered dose of mydriatics,4,5 but the potential disadvantage is the longer time required for obtaining the optimal mydriasis, and the risk of local toxicity remains. The drawback of all these approaches is the need to start the administration at least 30 minutes before the surgery, and the prolonged supervision of patients by nurses to first instill the ED and monitor the mydriasis. Today, mydriatic EDs are most frequently initiated by a nurse in the ambulatory surgical center (82% of cases). For the 14% of patients who administer their mydriatic EDs at home,6 the medical supervision of mydriatic ED application is limited and it also increases the risk of CV side effects (for the remaining 4%, dilation is performed in the operating room). Changes in surgical scheduling are also common and may call for multiple applications of mydriatic preparations to ensure the pupil is appropriately dilated at the time of the surgery. Thus, for a variety of reasons cataract patients may be exposed to higher level than anticipated levels of mydriatics with possible topical and systemic implications.
In order to improve the process of mydriasis in routine cataract surgery, the intracameral application of mydriatic drugs has been explored and found to be safe and effective.7–9 Based on these pioneering studies, an intracameral formulation to be injected at the start of the surgery has been developed, comprising a mix of two synergistic mydriatics (tropicamide, a lipophilic anticholinergic agent and phenylephrine, a sympathomimetic agent) in addition to an anesthetic (lidocaine) to improve preoperative intraocular anesthesia.
A particular benefit of intracameral mydriatics/anesthetic is the excellent bioavailability directly in the target tissues and the putative lower systemic absorption compared to ED formulations. The synergistic effect of the two mydriatic components also permits lower doses of each to be used, further limiting the local toxicity of these drugs.10
A pivotal Phase III study has been completed comparing a standardized (quality-controlled) intracameral fixed combination of mydriatics and anesthetic (ICMA) with standard topical mydriatics regimen that confirmed excellent efficacy in terms of quickly initiating and maintaining mydriasis and a good safety profile for ICMA.11 Patients who received ICMA were significantly more comfortable during surgery and spent less time overall in the preoperative and surgical room than those who received standard mydriatic EDs. Moreover, surgeons found the insertion of the intraocular lens technically less demanding with ICMA compared to EDs.11
In the present report, we provide additional information on the systemic exposure to mydriatic components with the ICMA formulation in comparison to the standard topical administration. This study is a supplementary analysis of data collected during the Phase III clinical study on received doses and plasma pharmacokinetics that were not previously reported.11
Patients and methods
Study design
Data from the Phase III European Union approval study of ICMA (Mydrane®; Laboratoires Théa, Clermont-Ferrand, France) were used to compare patient systemic exposure to mydriatic agents. Immediately after the arrival in the surgery unit, patients were randomized to receive either ICMA at the beginning of cataract surgery or standard topical mydriatics.11 Mydrane® is a proprietary injectable salt- and pH-balanced solution of two mydriatics (tropicamide 0.02% and phenylephrine 0.31%) and one anesthetic agent (lidocaine 1%) for intracameral delivery at the beginning of cataract surgery. The standard topical mydriatic regimen consisted of tropicamide 0.5% (Mydriaticum® 0.5%; Laboratoires Théa) and phenylephrine 10% (Neosynephrine® 10% Faure; Les Laboratoires Europhta, Gabian, Monaco). The parameters reported in this study formed part of the secondary objectives of that study.
Ethics
The study was performed in compliance with the ethical principles having their origins in the Declaration of Helsinki (2004) regarding biomedical research on human patients. The study was conducted in compliance with applicable regulatory requirements and in accordance with Good Clinical Practice using the guidance documents and practices of the International Conference on Harmonization and the European directive 2001/20/CE.
Independent ethics committee approval was obtained for each participating center prior to patient recruitment in each center (Supplementary material). Written and oral information was provided to each patient by the local investigator, and signed written consent was obtained before the patient was enrolled.
Study population
Patients
The study population comprised male or female cataract patients aged 40–88 years undergoing scheduled unilateral cataract surgery under topical anesthesia with clear corneal self-sealing incisions by phacoemulsification. Patients were only included if they demonstrated good mydriasis (≥7 mm) with topical mydriatics at the preoperative (selection) visit. Main noninclusion criteria included: a history of intraocular surgery or combined procedures, iatrogenic, traumatic, or congenital cataract, corneal, epithelial, stromal, or endothelial residual, progressive corneal disease, history of ocular trauma, infection or inflammation within the previous 3 months, and pseudoexfoliation and exfoliation syndromes.
Setting
Subjects were recruited from outpatient cataract surgery clinics in nine countries. Of 609 patients screened, 591 were randomized, 555 underwent the surgery, and 554 provided data for safety. Thirty patients from two centers ([Centre Hospitalier Régional Universitaire; CHRU Bretonneau of Tours and Centre Hospitalier Universitaire Dupuytren of Limoges, France) were selected for analysis of plasma tropicamide and phenylephrine levels.
Study interventions
To perform the mandatory preoperative step of antiseptics (5% povidone-iodine, Betadine®, in the ophthalmic area), all patients received topical anesthesia with one or two drops of tetracaine 1% instilled into the eye 5 minutes before surgery and a second identical instillation 1 minute before surgery.
Patients randomized to ICMA received the currently approved dose of ICMA, ie, a single injection of 200 μL administrated intracamerally through the side port or principal port at the beginning of the surgery. At this stage of the drug development, a supplementary 100-μL injection could be performed at the operating surgeon’s discretion.
Reference therapy comprised one drop of tropicamide 0.5% and one drop of phenylephrine 10%, instilled into the eye 30, 20, and 10 minutes prior. Further mydriatic EDs could be given at the operating surgeon’s discretion, in case of insufficient mydriasis.
Study outcomes
Determination of total exposure
Total exposure to their respective treatment was recorded in terms of intracameral dose of ICMA or number of drops of reference therapy in the overall population. For ICMA, the received dose was calculated based on the recorded volume injected in the anterior chamber and the concentration of each mydriatic in the ICMA solution. The patient’s exposure to the reference therapy was deduced from the amount of phenylephrine and tropicamide delivered with each instilled drop (0.15 and 3.4 mg, respectively, according to their respective summaries of product characteristics).3 To remain in a conservative hypothesis (sensibility analysis), it was assumed that only one drop was instilled per administration.
Sample collection and analysis (subgroup of patients)
Plasma levels of tropicamide and phenylephrine were measured in 30 patients (15 patients in each group) from two centers having appropriate technical and logistic ability. Blood samples (6 mL) were collected in heparinized tubes before and 2, 12, and 30 minutes after injection of ICMA or instillation of reference treatment. Blood samples were immediately centrifuged at 1,500× g for 10 minutes at 5°C. Plasma was harvested and transferred into polypropylene tubes for storage at −20°C. The frozen samples were shipped to SGS Cephac Europe SAS (Saint-Benoît, France) for bioanalysis. Plasma concentrations of tropicamide and phenylephrine were determined by HPLC with mass spectrometric detection (threshold of detection was 0.1 ng/mL). For both mydriatics, means of plasma levels were analyzed as well as the rate of patients presenting with plasma level above the limit of detection.
Vital signs
SBP (in mmHg), DBP (in mmHg), and heart rate (HR, radial pulses in beats/minute, bpm) were measured at the selection visit (60–70 days prior to surgery), then on the day of surgery (Day 0; before, during [5 minutes after the first incision], and at the end of surgery) and after 1, 8, and 28 days in the overall population. Two measurements were performed (with a resting time of 5 minutes between the two measurements), except during and at the end of the surgical procedure (for practical reasons). Means of the CV parameters were analyzed, and a post hoc analysis was performed to compare the rate of patients presenting with either at least one DBP value >100 mmHg, one SBP value >200 mmHg, or one HR value >120 bpm between groups.
Results
A total of 271 patients were exposed to ICMA and 283 patients to the reference treatment (hereinafter referred to as the safety set). Baseline characteristics of the overall study population were previously reported.11 For the subgroup of 30 patients who underwent blood sampling, mean age was 71.3±8.8 years (range: 51.3–85.5 years), with a large majority >65 years (80.0%) and more females than males (66.7% vs 33.3%, respectively).
Total exposure
In the ICMA group, all patients received 200 μL of ICMA in the operated eye, and the patient’s total exposure (without additional injection) was calculated to be 0.04 mg tropicamide and 0.62 mg phenylephrine. In addition, 72 (26.6%) patients received an additional injection of 100 μL of ICMA, and a single patient received a third 100-μL injection. Patients receiving one additional 100 μL of ICMA received maximum doses of 0.06 mg tropicamide and 0.93 mg phenylephrine.
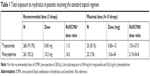
In the reference group, the regimen was one drop of 0.5% tropicamide plus one drop of 10% phenylephrine thrice in the eye to be operated. These EDs were administered alternately 10 minutes apart. The patients’ exposure to tropicamide and phenylephrine was, respectively, 0.45 and 10.2 mg. This represents 11.3 and 16.5 times more drug exposure compared to ICMA at a dose of 200 μL (Table 1). In the reference treatment group, 23 (8.1%) patients received additional drops of tropicamide and 22 (7.8%) patients received extra drops of phenylephrine. In these patients, the total dose varied between 4 and 10 drops (0.6–1.5 mg tropicamide and 13.7–34.0 mg phenylephrine). In case of 10 drops, this represents a dose ratio of 37.5 and 54.8 as compared to ICMA for tropicamide and phenylephrine, respectively.
Plasma levels
All 30 patients with blood sampling received the standard regimen of mydriatics (200 μL of ICMA or three drops each of tropicamide and phenylephrine), with the exception of one patient in the ICMA group who received an additional 100-μL injection.
Tropicamide plasma levels were below the limit of detection at all time points (2, 12, and 30 minutes) following ICMA injection in all patients (Figure 1). In patients receiving the reference regimen tropicamide was detectable in the plasma (73.3%) at 2 minutes, and in all patients at 12 and 30 minutes. The maximum plasma tropicamide concentration was 3.16 ng/mL at 30 minutes in the reference group.
Phenylephrine was only detectable in the plasma of two patients in the ICMA group (Figure 2); the maximum plasma phenylephrine concentration observed in these two patients were 0.140 and 0.587 ng/mL. In contrast, phenylephrine was detectable in plasma in all patients in the reference treatment group at least at one time point (depending on the patient, the maximum concentration ranged between 0.109 and 1.42 ng/mL). Maximal plasma phenylephrine concentration exceeded 0.59 ng/mL in 38.5% of patients.
Vital signs
When mean blood pressure or HR was considered, no changes of note were detected among the 271 patients of the ICMA group and the 283 patients treated with reference treatment (Table 2). However, when CV events were analyzed based on the rate of patients presenting with either at least one DBP value >100 mmHg, one SBP value >200 mmHg, or one HR value >120 bpm, instances of hypertension or tachycardia events were statistically more common in patients who received reference treatment (11.2%) than in the ICMA group (6.0%; P-value =0.033).
Discussion
While cataract surgery cannot proceed without adequate mydriasis, mydriatic drugs have the potential to cause CV adverse events. This is a particular concern in the generally older patient population undergoing cataract surgery. In a busy presurgical room with a number of patients being treated with EDs, the possibility for overdose errors is ever present. Surgery may be delayed and extra drops may be required to provide adequate mydriasis at the time of the rescheduled surgery. Moreover, some patients who commence mydriasis at home before coming to the ophthalmic surgical unit may either inadvertently or forgetfully administer more than one drop, or may be inadequately dilated at the commencement of surgery. Lastly, imperfect instillation technique may result in patients receiving more than one drop per administration.
The present shows that the total exposure to mydriatic agents is different between the reference topical regimen and the intracameral administration, resulting in differences in plasma levels. Even in the controlled environment of this clinical trial, ~9% of ED patients received additional drops of the reference ED formulation, with some receiving as many as 10 drops. None of these patients were included in the subset analyzed for plasma phenylephrine levels, but it is reasonable to assume that their exposure to phenylephrine would be higher than those who received the recommended dose. In daily practice, the recommendation for one drop per instillation is not universally respected during the presurgical phase of the patient’s care, especially when the patient self-instill the EDs.
The plasma levels were not excessive in the patients included in the relatively small subset of 30 patients analyzed for plasma mydriatic levels, and CV events were absent in the controlled context of this clinical study. In the less rigorously controlled environment of everyday clinical practice, however, extra drops are frequently used (indeed were used in this study). In this Phase III clinical trial, levels measured in the plasma of patients who received ICMA were much lower than those in patients who received the reference treatment. As reported in previous clinical studies on intracameral mydriatics, there were no systemic adverse effects reported in this study.12,13 Bearing in mind that patients undergoing cataract surgery are generally elderly and frequently have preexisting CV pathologies, any additional sympathetic activation consequent on the systemic passage of phenylephrine from mydriatic medication is to be avoided. The patient population in the present study may have been at lower risk of CV adverse effects than cataract surgery patients in general since patients with uncontrolled diabetes, excessive anxiety, uncontrolled hypertension, and other CV diseases were not included in this study.
A number of other studies have reported CV adverse effects with both 2.5% and 10% phenylephrine EDs.14 Our study results on HR and blood pressure measurements suggested that more patients might present with one meaningful CV event defined as one DBP value >100 mmHg, one SBP value >200 mmHg, or one HR value >120 bpm in the group receiving mydriatic EDs than in the ICMA group. Similar to our clinical trial, a comparative study of different mydriatic approaches in cataract surgery showed that the intracameral route is likely the safest from a CV perspective, presumably a reflection of the low doses of direct and indirect sympathomimetics used.15 However, in our study, no major differences were detected when the mean CV parameters were considered. This might be due to the fact that cataract surgery is a stressful experience for patients, especially in the context of a clinical trial. Therefore, difference in the mean CV parameters might have been diluted by patients experiencing an increase in their vital signs due to this stressful environment. This phenomenon was notable in the ICMA group where many patients already experienced an increase in blood pressure before receiving the study product. This stressful environment is well recognized in the literature since around half of patients report anxiety, even panic attacks, associated with the sensations of surgery and such reactions are likely to induce a sympathetic response with elevation of HR and blood pressure.16–18
Intracameral mydriasis provides auxiliary benefits; patients spend less time in the waiting room7 (potentially reducing stress and anxiety) and the risk of inadequate mydriasis jeopardizing operating room schedules is diminished. On the other hand, topical mydriatic EDs need to be instilled 30 minutes to 2 hours prior to cataract surgery (either in the waiting room or at home) in order to ensure adequate mydriasis at the time of capsulorhexis and further steps of phacoemulsification and lens implantation. Obviously, ED administration at home is entirely unsupervised; moreover, in a busy surgical department the degree of supervision of each patient is necessarily limited and the potential for medication errors and overdose exists.19,20 In addition, ocular surface toxicity may occur after multiple dosages of EDs including tropicamide, phenylephrine, and topical anesthetics (eg, oxybuprocaine or tetracaine).3,21 The toxically induced superficial punctate keratopathy may lead to suboptimal visualization during surgery (this is being prospectively evaluated in another ongoing study). By contrast, injection in the anterior chamber, directly to the target tissue, permits the induction of mydriasis in the operating room by the surgeon so the patient is under supervision throughout the entire procedure, and no side effects on the corneal epithelium can occur.
Two recent studies have attested to the benefits of intracameral mydriasis in terms of pre- and intraoperative operations and patient experience.22,23
Conclusion
In conclusion, ICMA result has lower systemic ocular surface exposure to phenylephrine and tropicamide than the reference topical treatment. It permits the correct dose of the appropriate agents to be administered consistently at a convenient time (at the start of surgery), in a safe and monitored environment (the operating room) at an appropriate site (anterior chamber) with less CV side effects compared to the usual dilation procedure.
Data sharing
Since the data were collected from a clinical trial, these are not intended to be shared by the authors, and no other study-related documents will be made available.
Acknowledgment
This study was conceived and funded by Laboratoires Théa, Clermont Ferrand, France.
Disclosure
JG, UP, PR, PJP, PYR, FC, and ML have been engaged as consultants for Laboratoires Théa. FC and ML have received honoraria from Laboratoires Théa. The authors report no other conflicts of interest in this work.
References
OECD (2014), health at a glance: Europe 2014, OECD publishing. Available from: http://dx.doi.org/10.1787/health_glance_eur-2014-en | ||
Lundström M, Barry P, Henry Y, Rosen P, Stenevi U. Evidence-based guidelines for cataract surgery: guidelines based on data in the European Registry of quality outcomes for cataract and refractive surgery database. J Cataract Refract Surg. 2012;38(6):1086–1093. | ||
Base de Données Publique des medicaments. Available from: http://base-donnees-publique.medicaments.gouv.fr/affichageDoc.php?specid=67313123&typedoc=R. Accessed March 22, 2017. | ||
Saenz-de-Viteri M, Fernández-Robredo P, de Nova E, et al. Comparative study measuring the dilatory effect of a mydriatic device (Mydriasert(®)) versus topical drops. Int J Ophthalmol. 2013;6(6):801–804. | ||
Levet L, Touzeau O, Scheer S, Borderie V, Laroche L. [A study of pupil dilation using the Mydriasert ophthalmic insert]. J Fr Ophtalmol. 2004;27(10):1099–1108. French. | ||
Behndig A, Cochener-Lamard B, Güell J, et al. Surgical, antiseptic, and antibiotic practice in cataract surgery: results from the European Observatory in 2013. J Cataract Refract Surg. 2015;41(12):2635–2643. | ||
Lundberg B, Behndig A. Intracameral mydriatics in phacoemulsification cataract surgery – a 6-year follow-up. Acta Ophthalmol. 2013;91(3):243–246. | ||
Lundqvist O, Behndig A. Posterior chamber injection of intracameral mydriatics increases the durability of the mydriatic response. Acta Ophthalmol. 2013;91(7):657–659. | ||
Mori Y, Miyai T, Kagaya F, et al. Intraoperative mydriasis by intracameral injection of mydriatic eye drops: in vivo efficacy and in vitro safety studies. Clin Exp Ophthalmol. 2011;39(5):456–461. | ||
Behndig A, Lundberg B, Bäckström G. Intracameral mydriatics in cataract surgery, cataract surgery, Farhan Zaidi (ED.). InTech. 2013;12:149–172. Available from: http://www.intechopen.com/books/cataract-surgery/intracameral-mydriatics-in-cataract-surgery | ||
Labetoulle M, Findl O, Malecaze F, et al; Intracameral Mydrane Study 2 Group. Evaluation of the efficacy and safety of a standardised intracameral combination of mydriatics and anaesthetics for cataract surgery. Br J Ophthalmol. 2016;100(7):976–985. | ||
Lundberg B, Behndig A. Intracameral mydriatics in phacoemulsification cataract surgery. J Cataract Refract Surg. 2003;29(12):2366–2371. | ||
Behndig A, Eriksson A. Evaluation of surgical performance with intracameral mydriatics in phacoemulsification surgery. Acta Ophthalmol Scand. 2004;82(2):144–147. | ||
Stavert B, McGuinness MB, Harper CA, Guymer RH, Finger RP. Cardiovascular adverse effects of phenylephrine eyedrops: a systematic review and meta-analysis. JAMA Ophthalmol. 2015;133(6):647–652. | ||
Morgado G, Barros P, Martins J, Lima A, Martins N. Comparative study of mydriasis in cataract surgery: topical versus Mydriasert versus intracameral mydriasis in cataract surgery. Eur J Ophthalmol. 2010;20(6):989–993. | ||
Tranos PG, Wickremasinghe SS, Sinclair N, et al. Visual perception during phacoemulsification cataract surgery under topical and regional anaesthesia. Acta Ophthalmol Scand. 2003;81(2):118–122. | ||
Tan CS, Au Eong KG, Kumar CM, Rengaraj V, Radhakrishnan M. Fear caused by intraoperative visual sensations during cataract surgery. Acta Ophthalmol Scand. 2005;83(5):631–632. | ||
Khan MA, Tang WK, Tahhan M. Patient perceptions during phacoemulsification cataract surgery: a proposal for routine preoperative patient counselling. Br J Ophthalmol. 2014;98(8):1132–1133. | ||
Fraunfelder FT, Scafidi AF. Possible adverse effects from topical ocular 10% phenylephrine. Am J Ophthalmol. 1978;85(4):447–453. | ||
Wellwood M, Goresky GV. Systemic hypertension associated with topical administration of 2.5% phenylephrine HCl. Am J Ophthalmol. 1982;93(3):369–370. | ||
Base de Données Publique des medicaments. 2017. Available from: http://base-donnees-publique.medicaments.gouv.fr/affichageDoc.php?specid=61200510&typedoc=R | ||
Chiambaretta F, Pleyer U, Behndig A, et al. Intracameral Mydrane (ICMA) and ethics group. Pupil dilation dynamics with an intracameral fixed combination of mydriatics and anesthetic during cataract surgery. J Cataract Refract Surg. 2018;44:341–347. | ||
Schulz CB, Goverdhan SV, Humphry RC. An evaluation of intracameral mydriasis for routine cataract surgery. Br J Ophthalmol. 2018;102(6):784–789. |
Supplementary material
Ethics committees
  |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.