Back to Journals » International Journal of Nanomedicine » Volume 14
Small interfering RNAs (siRNAs) in cancer therapy: a nano-based approach
Authors Mahmoodi Chalbatani G, Dana H, Gharagouzloo E, Grijalvo S, Eritja R , Logsdon CD , Memari F, Miri SR, Rad MR, Marmari V
Received 4 January 2019
Accepted for publication 23 March 2019
Published 2 May 2019 Volume 2019:14 Pages 3111—3128
DOI https://doi.org/10.2147/IJN.S200253
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Thomas Webster
Ghanbar Mahmoodi Chalbatani,1,* Hassan Dana,1,2,* Elahe Gharagouzloo,1 Santiago Grijalvo,3,4 Ramon Eritja,3,4 Craig D Logsdon,5,6 Fereidoon Memari,1 Seyed Rouhollah Miri,1 Mahdi Rezvani Rad,7 Vahid Marmari2
1Cancer Research Center, Cancer Institute of Iran, Tehran University of Medical Science, Tehran, Iran; 2Department of Biology, Damghan Branch, Islamic Azad University, Damghan, Iran; 3Institute of Advanced Chemistry of Catalonia (IQAC-CSIC), Barcelona 08034, Spain; 4Biomedical Research Networking Center in Bioengineering, Biomaterials and Nanomedicine (CIBER BBN), Madrid, Spain; 5Department of Cancer Biology, University of Texas, M.D. Anderson Cancer Center, Houston, TX, USA; 6Department of GI Medical Oncology, University of Texas, M.D. Anderson Cancer Center, Houston, TX, USA; 7Department of Biology, Zabol University, Zabol, Iran
*These authors contributed equally to this work
Abstract: Cancer is one of the most complex diseases that has resulted in multiple genetic disorders and cellular abnormalities. Globally, cancer is the most common health concern disease that is affecting human beings. Great efforts have been made over the past decades in biology with the aim of searching novel and more efficient tools in therapy. Thus, small interfering RNAs (siRNAs) have been considered one of the most noteworthy developments which are able to regulate gene expression following a process known as RNA interference (RNAi). RNAi is a post-transcriptional mechanism that involves the inhibition of gene expression through promoting cleavage on a specific area of a target messenger RNA (mRNA). This technology has shown promising therapeutic results for a good number of diseases, especially in cancer. However, siRNA therapeutics have to face important drawbacks in therapy including stability and successful siRNA delivery in vivo. In this regard, the development of effective siRNA delivery systems has helped addressing these issues by opening novel therapeutic windows which have allowed to build up important advances in Nanomedicine. In this review, we discuss the progress of siRNA therapy as well as its medical application via nanoparticle-mediated delivery for cancer treatment.
Keywords: delivery strategies, lipoplexes, nanovectors, polymeric nanoparticles, siRNA
Introduction
Cancer has become one of the most complex diseases due to numerous genetic disorders and cellular abnormalities. Cancer disease continues to be a major health concern worldwide being the second leading cause of death in the world. According to the World Health Organization (WHO), nearly 10 million people are estimated to die of cancer by the year 2020. Cancer has been known as the number one cause of deaths in developed countries in the current century.1–3 While remarkable efforts have been developed during the past few decades in the detection, prevention, and treatment,4,5 signaling pathway complexities that regulate cancer progression together with its tumor microenvironment heterogeneity and metastasis remain as serious obstacles to find efficient cancer treatments.6,7 Nowadays, treatments based on single chemotherapeutic drugs generally face lack effectiveness in cancer therapy, whereas two or more combined therapeutic methods involving various mechanisms of action are needed to achieve certain effectiveness in cancer therapy.
The currently used therapies are non-selective leading to side effects responsible for prolonged and expensive recovery, often followed by relapse at later time points. In this context, the selective targeting may provide a platform for the development of novel, more effective diagnostic tools and/or less harmful treatments.8 Targeted therapies using monoclonal antibodies (mAb) against overexpressed receptors (eg, herceptin) have improved clinical outcomes.9 Moreover, nanomaterials have also become an interesting alternative approach for the administration of drugs reducing the side effects (eg, biodegradable nanoparticles loaded with docetaxel).10 The impact of these nanomedicines in human health may be enormous whether the right combination of targeting molecules and drugs become available with defined chemical structures. This has been demonstrated with the explosion of the antibodies-drug conjugates field.11 However, the control of the number and position of the drugs on the antibodies, as well as the design of autoimmolative linkers to help trigger drug release after internalization is still far to be solved although this is an active field of development.12
Recently, much attention has been paid to the potential application of RNA interference (RNAi) for cancer treatment.8,13 RNAi is the term given to the ability of a double-stranded RNA (dsRNA) containing a homologous sequence to a specific gene leading to sequence-specific gene silencing. RNAi is an endogenous post-transcriptional regulation process that consists of small regulatory RNAs including microRNAs (miRNAs) or small interfering RNAs (siRNAs) which are able to silence target messenger RNAs (mRNAs) in a sequence-specific procedure.14 After the discovery of RNAi in Caenorhabditis elegans15,16 and subsequent demonstration of siRNA activity in mammalian cells, RNAi has received considerable attention as an effective therapy for multiple diseases like cancer and viral infections, particularly for those diseases with “undruggable” molecular targets.17,18
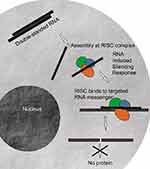
Today, RNAi has become a powerful technology for gene function studies and has been recently used in therapeutic applications.18–20 The main role of the RNAi in cells is the down-regulation of specific proteins (Figure 1). RNAi mechanism is triggered initially by the enzyme Dicer which cleaves double-stranded RNAs (dsRNAs) into short double-stranded siRNAs of 21–25 nt. The siRNA passenger strand is then unwound, and the siRNA guide strand is loaded into the RNA-induced silencing (RISC) complex leading to cleavage of target mRNAs by Argonaute 2 (Ago2) when the guide strand sequence is paired with an mRNA complementary sequence.21–23 This important mechanism has allowed to open novel therapeutic approaches by designing oligonucleotide molecules through using mRNA transcripts sequences found in the existing human genomic data. Therefore, a careful sequence selection and synthesis of tailored siRNAs may have enormous repercussion in therapy as almost all genes might be down-regulated as well as splice variants, separate transcripts, or mutations might also be specifically targeted. As a consequence, this powerful approach might help circumvent the limitations exhibited by small molecule drugs in conventional cancer therapy treatments leading to drug development processes based on gene functionality.21 Therefore, the development of this therapeutic strategy may have a high impact on modern medicine.20,24,25
However, siRNA technology faces multiple obstacles regarding efficient delivery and effectiveness. To overcome this issue, siRNA intracellular delivery strategies should be nontoxic and stable into the site of action for succeeding therapeutic applications of RNAi.26–28 Currently, different methods including mechanical (ultrasound), physical methods,29 electroporation,30 hydrodynamic tail vein injections in mice,31 and the use of a gene gun have been carried out for delivering siRNA in vivo. In addition, local administrations (eg, intraperitoneal, intravenous, subcutaneous injections) and chemical methods based on synthesizing non-viral vehicles (eg, polymers,32–34 cationic lipids,35,36 and peptides37,38) have also been successfully used among others. To demonstrate the potential of RNAi-based therapeutics, several proof-of-concept studies including bio-distribution, efficiency of delivery, and toxicity caused by cationic delivery vehicles have been carefully analyzed.
siRNA technology in cancer therapy
siRNA has been investigated as an effective treatment for the viral disease as well as cancer extensively with the aim of blocking multiple disease-causing genes.39 RNAi was discovered for the first time in the nematode Caenorhabditis elegans; however, this process has been also found in many different eukaryotes species including, vertebrates, plants, and insects. In mammalian cells, researchers reported that synthetic siRNAs were also able to promote RNAi and therefore silence the expression of altered proteins.15–17,40
Numerous chemical modifications have been proposed in order to increase efficacy and potency in RNAi for in vivo use.41 In this regard, rational chemical designs have allowed siRNA passenger strands to be more likely to modification than siRNA guide strands. These synthetic strategies have enabled to replace either non-bridging oxygen on the phosphate linkage with a sulfur atom, the 2′-hydroxyl group modification of the sugar ring with a methyl group (2′-OCH3) and ethyl group (2ʹ-OCH2CH3), among others.42–45 In addition, other strategies have been developed to deliver siRNAs safely in the cytoplasm. While most naked siRNAs have been effective for a good number of tumor cells in vitro, these siRNAs have unfortunately failed when have injected in vivo by systemic administrations.46
Immunotherapy has gained increasing attention as a promising strategy for treating cancer by promoting the host immune system activation. This activation may occur by simply introducing cytokines, cancer vaccines, monoclonal antibodies, or using antigen-presenting cells (APC) through Toll-like receptors.47 Dendritic cells (DCs) are the strongest antigen exhibiting cells that have the capacity to stimulate naïve T cells and stimulate differentiation and growth of B cells. Furthermore, they include a system of leukocytes broadly distributed in all tissues. DC therapy may offer a novel and promising immunotherapeutic method for prevention and treatment of advanced cancer as well as autoimmune disorders.48,49
DC immunization has been broadly studied in clinical trials involving several types of malignancies like melanoma, prostate cancer, and renal cell carcinoma (RCC), among others.50–54 The use of DC therapy has proved to be effective and a safe strategy in inducing antitumor immunity to patients containing advanced-stage cancer diseases.51,55 Several models have been proposed with the aim of isolating DCs in vitro. For example, Zhang et al developed a method to favor DC differentiation through contact circulating monocytes with natural killer (NK) cell subsets in order to find optimal protocols for DC vaccination.56 According to in vivo studies, DCs can promote T cell-mediated tumor eradication and controlled tumor-bearing hosts when ex vivo isolation have been implemented with tumor antigens. These perceptions have prompted clinical trials to explore both immunologic and clinical impacts of antigen-loaded DCs regulated as therapeutic vaccines to patients containing specific tumors. Some promising outcomes reported from clinical trials in patients with melanoma and malignant prostate tumors showed that immunotherapeutic strategies involving the development of antigen presenting cells in DCs might demonstrate the effectiveness of this process to certain human tumors.50 However, DC clinical effectiveness has not been as good as researchers expected in some specific cancers probably due to the heterogeneity and different methods to monocytes production as well as DC differentiation.
Interestingly, novel approaches have been implemented involving dendritic cell-based therapies and other therapies like RNAi in order to overcome these drawbacks and find a reliable vaccine for cancer therapy.57 For example, Qian et al reported that siRNA may be combined to DC-based therapy in order to manipulate CD40 expression levels and therefore reduce the functional capability of DC to stimulate allogeneic T cells. In this regard, silencing of CD40 expression on DC might be used for generating tolerance-promoting DC with the aim of applying in autoimmunity and transplantation processes.58
Recently, Liu et al prepared a specific cationic nanoparticle in order to deliver indoleamine 2, 3-dioxygenase (IDO) siRNA and tyrosine-related protein 2 (Trp2) to DCs.59 The authors were able to inhibit IDO expression, trigger T cell immune reaction and consequently favor the secretion of cytokines like IL-6, IFN-ϒ and TNF-α as well as promote DC differentiation when compared to traditional control DC vaccines. Thus, in vitro and in vivo studies in B16-F10 mouse model proved this approach to be effective in reducing melanoma tumor growth and therefore enhancing immune response in mice.
siRNA-based gene silencing
Great efforts have been made to understand how dsRNAs, siRNAs, and microRNAs (miRNAs) are able to affect control expression in eukaryotic cells. As described previously, siRNA technology mediates specifically gene silencing at different levels like mRNA degradation, chromatin modification, and translational repression.60–64 Remarkable findings were also found and confirmed transcriptional gene silencing was conserved in mammalian cells when were mediated by siRNAs.65 Despite advantages and interest showed by siRNA-based technology as an effective therapeutic approach, many obstacles like efficient cellular uptake, long-term stabilities, and off-target effects have been reported. These important issues have reduced siRNA effectiveness when used in vitro and/or injected in animal models as well.
While the enhancement of siRNA stability has been successfully solved by introducing chemical modifications at the level of sugar and phosphate groups, advances in efficient siRNA delivery systems to transport siRNAs safely into the cytoplasm of targeted cells is a vital element in cancer therapy.66,67 For this purpose, viral vectors (eg, lentivirus, retrovirus and adenovirus) have become potential vectors for siRNA delivery due to their ability to encapsulate and deliver genetic materials into cells.68,69 Despite such viral vectors having been shown highly transfection efficacies, their clinical usage is still limited due to possible risks of immune responses, mutation, and inflammation. To minimize such undesirable effects and exploit the potential of RNAi-based technology, a good number of synthetic non-viral vectors have been proposed to impart siRNA delivery safely and effectively.22,35
Off-target effects and stimulation of immune response
The specificity of action carried out by siRNA molecules depends upon the uniqueness of the selected sequences; however, many other factors may also have an impact on a lack of specificity.70 Several types involving siRNA off-target effects have been described and thoroughly reviewed.71 It is well known that mammalian immune cells tend to express a family of receptors so-called Toll-like receptors (TLRs) which have also the ability to recognize pathogens and molecules derived from microbes. In addition, some synthetic siRNA sequences as well as their non-viral vehicles have proved to promote the activation of immune system cells producing inflammatory cytokines and Type I interferon in vitro and in vivo by activating TLR7 and TLR8 in a sequence-dependent manner.72
Other significant off-target effect associated to siRNA delivery falls into the miRNA-like off-target silencing. miRNAs are small non-coding endogenous RNA molecules which have the ability to regulate gene expression of several genes in the same way as siRNAs do. This undesirable sequence-specific activity is mainly produced via siRNA base-pairing with sequences located at the 3ʹ-UTR regions of mRNAs. As a consequence, many transcripts might be affected dealing with multiple-site cleavage translational blocks.73,74
Finally, the RNAi machinery saturation mediated by synthetic siRNAs is another source of off-target. Some data have shown that synthetic siRNAs when entered the RNAi pathway tend to compete with endogenous miRNAs for RISC. This process has been observed in certain model studies in which both transcript upregulation and target script downregulation have been observed. Efforts to mitigate or reduce such off-target effects have been effectively studied by designing effective modified siRNAs.75–79
Delivery systems
Lipid-based nanovectors for siRNA delivery
Various kinds of systems have been employed for delivering siRNA such as antibody conjugates, micelles, natural polysaccharides, peptides, synthetic cationic polymers, and microparticles among others. Nevertheless, some lipid-based formulations and other lipid-like materials such as liposomes, niosomes, and stable nucleic acid lipid particles (SNALPs) have proved to be effective drug delivery systems as promising strategies for in vivo siRNA delivery80–85 (Figure 2).
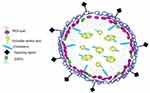 | Figure 2 A schematic image of LNPs siRNA showing a nanostructured core. |
Liposomes and lipoplexes
As non-viral vectors, liposomes have become a powerful platform as a pharmaceutical carrier to facilitate the delivery of small molecule drugs and macromolecules.86 Liposomes are made up of phospholipids which tend to form closed lipid bilayers in aqueous solvents dealing with particles formation of nanometric size. In addition to loading a good number of substances like active drugs, proteins, peptides, antibodies, and nucleic acids, liposomes have been also used in the transport of photosensitizers needed for promoting photo-dynamic therapy.87 Second and third generation of liposomes have also been proposed by including certain ligands or loading polymers which have the ability to recognize specific receptors or enhance liposome stabilities, respectively.88 These advances have allowed liposomes to be used in a good number of clinical applications and be also injected according to different administration routes.
Nucleic acids in particular antisense oligonucleotides (ASOs) and siRNAs have been widely incorporated into liposomal nanocarriers (Table 1).35,62 This electrostatic combination has resulted in obtaining the corresponding lipoplexes which have helped nucleic acids increase both their long-term stabilities and cellular internalization. Positive and negatively charged together with neutral liposomes have been used as vehicles for transporting efficiently siRNA molecules89 (Figure 3). In this regard, cationic lipids have attracted significant attention as appropriate non-viral vehicles. This interest has allowed to designing convenient synthetic strategies and characterizing a plethora of cationic lipids. Structurally, cationic lipids consist of three general components: i) the lipid tail(s); ii) the cationic head group and iii) the linkers and/or backbones that connect to both parts. Some studies have suggested a relationship between the efficiency and lipid structure; however, this involvement still remains a central goal in siRNA delivery.82
 | Table 1 Summary of siRNA-loaded encapsulated liposomes utilized for siRNA delivery |
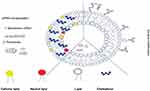 | Figure 3 A schematic representation of several strategies for encapsulating siRNA in liposomes. |
While anionic liposomes are usually cleared from circulation, cationic liposomes (eg, DC-Cholesterol, DOTAP, and AtuFECT01 among others) in combination with helper lipids (eg, Cholesterol, DOPE, and DPhyPE) have proved efficient in siRNA delivery at optimized N/P ratios. However, reducing toxicity levels still remains crucial when forming these types of lipoplexes.90–92 To solve these issues, neutral liposomes [eg, 1,2-dioleoyl-sn-glycero-3-phosphatidyl-choline (DOPC)] have been prepared in order to reduce such toxicity and therefore increase in biocompatibility although their entrapment efficiency might be compromised.93
The use of neutral lipid-based formulations has enabled the successful siRNA delivery in vivo in mouse models showing tumor growth inhibition and consequently the down-regulation of targeted genes.67 Several siRNA-based treatments against ovarian cancer were suggested by Sood et al.94 In this study, DOPC-encapsulated siRNA targeting the oncoprotein Ephrin Type-A receptor 2 was highly effective (65%) in reducing its expression after 48 hrs of being administered in a single dose in an orthotopic model. Interestingly, the authors found this liposomal formulation when injected both intraperitoneal and intravenous reduced the ovarian tumor size in mice xenograft models with the same efficiency. In addition, treatments with DOPC-based liposomes were also studied in order to target interleukin-8, β-2 adrenergic receptor, and focal adhesion-kinase by mediating intraperitoneal delivery of siRNAs in an ovarian cancer mouse model.94
In spite of obtaining powerful and effective nanovehicles based on formulating lipid molecules and siRNAs, liposomes and niosomes85 have been properly tuned in order to promote targeted delivery in specific organs and tissues. This strategy has enabled the preparation of unique lipid-based platforms bearing the arginine-glycine-aspartic acid (RGD) peptide95 or decorated with an octa-arginine (R8) cell-penetrating peptide (CPPs).96 These two strategies have confirmed increased particle stability, biocompatibility, and good siRNA therapeutic response without affecting remarkably cellular viability.
SNALPs
Stable nucleic acid lipid particles (SNALPs) has become a promising delivery platform for siRNA molecules in different animal models. This formulation is made up of a lipid bilayer based on a mixture of fusogenic and cationic lipids which enables endosomal release and therefore facilitates the siRNA cellular uptake. In addition, the SNALP surface is also coated during the formulation process with a PEG-lipid conjugate which provides hydrophilicity and a neutral layer with the aim of stabilizing these particles in the bloodstream when injected in vivo.35 The first use of SNALPs was reported by Zimmermann et al to silence the apolipoprotein B (Apo B) gene expression in non-human primates with a single dose (2.5 mg/kg) of siRNA.97 Optimized formulations based on modifying SNALPs chemically increased their siRNA efficacy in vivo by reducing doses until 0.01 mg·Kg−1 when targeted a hepatic endogenous gene.82
Other important applications of SNALPs have been reported and reviewed in literature.98 For example, McLachlan et al formulated a siRNA targeting the polymerase gene in vivo in order to fully protect guinea pigs against Ebola virus.99 More recently, SNALP technology has been successfully employed to favor the silencing of mTTR expression.100 Clinical results confirmed the suitability and therapeutic effect of this siRNA-SNALP combination in humans giving rise to the first RNAi therapeutic in the market.68,101
Polymeric nanoparticles
The last decades have been witnessed the large development on polymeric nanosized materials as drug carriers becoming some of the top selling drugs.100,102 Synthetic or naturally-occurring nanopolymers are colloidal solid materials specially designed to be degraded in vivo without producing toxic components.103 Several polymeric nanoparticles have been approved as drug carriers for human use.104 Due to their excellent properties, a variety of polymers including cationic polymers such as polyethylenimine (PEI), polysaccharides such as cyclodextrin (CD) and chitosan have been studied for siRNA delivery.105,106 As a general rule, polymer nanoparticles exhibit positively charged units to facilitate electrostatic binding of siRNA; however, the use of covalent strategies involving siRNA and polymers have been frequently prepared by using degradable linkers such as disulfide or thiol-maleimide bonds.107
Cyclodextrins (CDs) nanoparticles
Cyclodextrins are naturally occurring oligosaccharides that are produced during the bacterial digestion of cellulose. They have been well studied and characterized as pharmaceutical excipients with a favorable toxicological profile108–110 (Figure 4). Cyclodextrin-containing polycation nanoparticles are exciting polymer-based siRNA delivery systems, as they are able to self-assemble with siRNA dealing to shape colloidal particles of about 50 nm in diameter. In addition, their terminal imidazole groups help in the release and intracellular trafficking of nucleic acids. A complex system consisting of a siRNA, the human transferrin protein to engage transferrin receptors on the surface of the cancer cells, a cyclodextrin-based polymer, and polyethylene glycol (PEG) has been described to increase the stability of nanoparticles in biological fluids.111,112
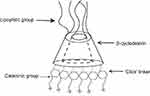 | Figure 4 Schematic illustration of a Cyclodextrin structure. |
An example of this system is CALAA-01, which is a targeted nanocomplex which includes an anti-R2 siRNA.113 This targeted therapeutic system has been designed to inhibit tumor growth. The selected siRNA prevents the tumor from growing via RNAi to decrease expression of the M2 subunit of R2 (ribonucleotide reductase). This system was used in clinical studies in patients bearing solid tumors.114
Chitosan and inulin nanoparticles
Chitosan and inulin are naturally occurring polysaccharides used for the preparation of siRNA formulations. Chitosan, that contains hydroxyl and amino groups, is widely used as a drug carrier due to its low cost, easy degradability and biocompatibility. Chitosan has positive charges under slightly acidic conditions allowing the formation of nanoparticles or complexes by interaction with siRNA molecules.115,116 Interestingly, optimized chitosan formulations containing folic acid as a targeting ligand resulted in increasing siRNA cellular uptake to tumor cells producing an enhancement in gene silencing properties in HeLa and OV-3 cell lines.116 Furthermore, grafting chitosan with polyethylene glycol117 and polyethylenenimine118 enable them for in vivo use through intravenous or intraperitoneal administration.117,118 Chitosan is frequently used in combination with other carriers such as poly(L-lactide) (PLLA) porous microparticle119,120 or the triblock polymer poly(L-lactide)-poly(ethyleneglycol)-poly(L-lactide) (PLLA-PEG-PLLA) to increase storage stability and decrease immunogenicity.121 These formulations, prepared by supercritical fluid technology,122 allowed the addition of multiple polymers as well as other chemotherapeutic drugs such as paclitaxel121 and doxorubicin.120
Another polysaccharide used recently in siRNA delivery is inulin. This polysaccharide made up of fructose and glucose units is usually processed with oligoamines such as ethylenediamine123 or diethylenetriamine and imidazole124,125 in order to generate cationic groups for siRNA binding and therefore favor endosomal escape.
Polyethylenimine (PEI)
Polyethylenimine (PEI) is one of the most studied cationic polymers for transfection of oligonucleotides, siRNA, and plasmid DNA.126 It is accessible either in branched or in a linear shape and many molecular weights.127 The high charge density of PEI is considered a good property for complexation of siRNA and facilitates the endosomal escape by the proton sponge effect.126 However, high molecular weight PEI has exhibited significant toxicity in many cell lines.127 The use of PEI as a non-viral vehicle to deliver siRNAs was demonstrated to have an efficient antiviral effect in a guinea pig model of Ebola virus infection128 and a murine model of influenza infection.129 The first successful applications of PEI delivery of siRNA in cancer was the inhibition of human EGF receptor 2 (HER2) in mouse models of ovarian cancer.130 If provided the appropriate molecular weight polymer and structure to avoid toxicities, for systemic use, PEI appears to be a promising siRNA delivery system.131
At present, research on PEI is directed towards the development of mixed polymers especially with PEG,132 and polysaccharides such as chitosan118 to reduce potential toxic effects and decrease the removal of nanoparticles by the reticuloendothelial system (RES). To direct PEI-siRNA complexes to specific target cells, PEI has been modified with receptor-mediated ligands such as folic acid, mannose, N-acetylgalactosamine, and others.132
Anionic polymers
Poly-L-lactic-co-glycolic acid (PLGA) is a biocompatible, well-studied, and biodegradable polymer used for decades in pharmaceutical applications.133,134 PGLA polymer has the advantage of exhibiting lower toxicity when compared with cationic polymers and cationic lipids. On the other site, PLGA cannot form electrostatic complexes with siRNA as both are negatively charged. One way to overcome this issue is to use PLGA nanocapsules in order to encapsulate oligonucleotides bearing 5ʹ-lipophilic molecules. These resulting polymer microspheres have provided sustained release of the modified siRNAs and antisense oligodeoxynucleotides within 24 hrs when administered subcutaneously.135 Another interesting approach is to react polyamines or positively charged dendrimers with some of the carboxyl groups of PLGA in order to generate additional positive charges that can be used for electrostatic binding.134 In addition, siRNAs may also be covalently linked to PLGA via intracellular cleavable disulfide linkers.136
Hyaluronic acid (HA) is also an anionic polymer that plays a pivotal role as a ligand against CD44 receptor which is highly overexpressed in tumor cells. The combination of PLGA and hyaluronic acid has also been used to co-deliver paclitaxel together with a siRNA against focal adhesion kinase (FAK) that is overexpressed in breast, colon and ovarian cancers.137 The resulting PLGA nanoparticles were shown to possess a highly selective delivery of the paclitaxel and siRNA to CD44+ cells.
Cationic dendrimers
Cationic dendrimers with extremely branched peripheral chain ends are well-defined artificial macromolecules. These can be synthesized by adding several layer branches. Each branched layer represents a superior generation molecule.133 The precise core-shell nanostructures of dendrimers enables drug loading by surface adsorption, interior encapsulation, or chemical conjugation (Figure 5). The dendrimer surface can be functionalized to build a variety of functions for a good number of applications. Dendrimers have a well-defined chemical structure and size in comparison with alternative linear polymers with a spherical form. To deliver negatively charged plasmid DNA, siRNAs, and antisense oligonucleotides, dendrimers containing high density of positive charges on the surface have been used.138,139 Polycationic dendrimers including poly-propylenimine (PPI) and poly-(amidoamine) (PAMAM) dendrimers have been investigated for siRNA delivery in past years. PAMAM-mediated siRNA delivery is usually found at nucleolus and even perinuclear locations. In a study designed for increasing the siRNA loading capacity, dendrimers were additionally improved with magnetofluorescent nanoworms to create “dendriworms.”140 After adding the dendriworms carrying siRNAs to human glioblastoma cells, the siRNA-dendrimers quickly internalized into the cells and escaped into the cytosol. The delivered siRNAs were demonstrated to silence expression of the targeted gene in vivo.141
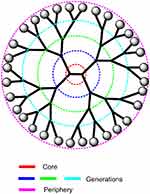 | Figure 5 A schematic representation of a dendrimer showing the central core, the peripherical sites, and the consecutive generations. |
Carbon nanotubes (CNTs)
Carbon nanotubes (CNTs) have been considered as potential nucleic acid and drug delivery vehicles within the nanomedicine field.142,143 They can be either single-walled CNTs (a single layer of graphene sheets) or multiwalled CNTs which can be prepared using multiple layers. In addition, CNTs can be modified with additional functional groups and also offer a structural advantage due to their very large surface. This can be used for loading therapeutic drugs like proteins and nucleic acids, and they. In order to form complexes with siRNA, positively charged functionalized single-walled CNTs (SWCNTs+) have been prepared. In a model study, siRNA designed to inhibit telomerase reverse transcriptase (TERT) was complexed with SWCNTs+ and incubated with tumor cells. The complexes TERT siRNA:SWCNTs+ were effective in delivering siRNA and knockdown the expression of TERT in a variety of tumor cells.144
Inorganic nanoparticles (INPs)
In the last decades, inorganic nanoparticles (INPs) have emerged as alternative nanomaterials to traditional lipid formulations for siRNA delivery.145 Specifically, magnetic nanoparticles and gold nanoparticles have been extensively studied for siRNA delivery and in vivo imaging because of their biocompatibility and reduced toxicity.145,146 For example, mesoporous silica nanoparticles (MSNPs) have been used as interesting vehicles for delivering siRNAs in cancer cells. Recently, Ngamcherdtrakul et al successfully prepared 50 nm INPs decorated with PEI, PEG, and an antibody.147 These authors were able to establish a lyophilization protocol for these modified INPs and therefore promote the formation of complexes with siRNAs. Interestingly, these siRNA nanoconstructs were stable, showed luciferase silencing inhibition, and were able to display anti-proliferative effect in vitro. Other INPs like carbonate apatite have also studied by Tiash et al. In this work, the authors used these NPs as vehicles for delivering siRNA targeting GFR genes simultaneously (egfr1 and erbb2) in mouse models showing the ability of these siRNA complexes to reduce tumor growth and reduce cell viability.148
Magnetic nanoparticles (MNPs)
Magnetic nanoparticles (MNPs) have been extensively employed as magnetic resonance imaging (MRI) contrast agents for tumor imaging and drug delivery as well. Iron oxide nanoparticles can be synthesized to obtain particles of small nanometric size with narrow and high magnetization values. In addition, MNPs have shown to induce cancer cell apoptosis by magnetic heating because of their excellent contrast signal in MRI.149 In addition, MNPs can be coated with siRNAs as well as other chemotherapeutic agents in order to promote delivery to the tumor site by applying external magnetic fields. Some representative examples inducing cellular death by magnetic heating have been reported.150,151
Gold nanoparticles (AuNPs)
Gold nanoparticles (AuNPs) have been used as effective nanomaterials for siRNA delivery applications for several reasons.146,152,153 First, functional diversity can be easily obtained with the creation of multifunctional monolayers and second, AuNPs can be prepared in a scalable fashion with low size disparity. AuNPs have been widely utilized for gene therapy targets in preclinical animal model and in vitro studies because of their low toxicity, rapid endosomal escape, high payload, efficient uptake, increased half-life; specific, efficient, extensive transcriptional activation of the innate immune response, and selective gene silencing and transfection.154 AuNPs were effectively employed as a platform for the delivery of Bcl-2 (B-cell lymphoma 2) or VEGF (vascular endothelial growth factor) siRNAs to a human cervical carcinoma cell line or into a human glioma cell line, for silencing of EGFP (enhanced green fluorescent protein) and LUC (luciferase) reporter genes.155,156
Limitations to the siRNA therapeutic approach
Table 2 summarizes some of the limitations described for siRNA delivery.157 Degradation in serum of siRNA therapeutic molecules is one of the major limitations for the therapeutic use of these molecules. Double-stranded siRNA molecules are more stable than single-stranded antisense oligonucleotides. For this reason, unmodified siRNA in saline buffer has been described to be successful for human treatments. However, these cases have been mostly focused for the treatment of ocular diseases by local administration.158
 | Table 2 Intracellular and extracellular limitations of RNAi |
Most of the siRNA molecules used for therapeutic uses are partially modified since a large number of modifications may block RISC binding to siRNA and therefore prevent RNA interference mechanism.159 Frequently these modifications are located near the 3ʹ and 5ʹ-ends of the passenger and guide strands where it has been demonstrated that have a strong impact in the following properties: i) avoid exonuclease degradation action;74,159 ii) increase affinity to RISC;76 iii) prevent the passenger strand loading;76 iv) lower innate immunostimulation response;79 and v) avoid miRNA-like effects.75 Another interesting siRNA chemical modification is the preparation of siRNA conjugates covalently,160 especially lipid conjugates such as cholesterol. Such lipid siRNA conjugates have exhibited exonuclease resistance have promoted cellular uptake and also increased serum circulation time by binding to serum proteins.161,162
Carbohydrate-siRNA conjugates and especially N-acetyl-galactosamine (GalNAc)-siRNA conjugates have also being used for in vivo targeting to hepatocytes. A large impact on cellular uptake was observed with siRNAs that were conjugated with triantennary GalNAc residues. Transfection experiments corroborated high specificity and gene silencing levels in liver.163 Peptide-siRNA conjugates especially cell-penetrating peptides (CPPs) and fusogenic peptides160 have been also described to potentiate cellular uptake and facilitate endosomal escape being a good alternative for anti-cancer siRNA delivery.
Most of the clinical trials involving systemic delivery of siRNA molecules have used lipid formulations including liposomes, SNALPs, and other types of lipid formulations that contain cationic lipids to form complexes with siRNA molecules. One of the first human clinical trials for cancer treatment used two siRNA molecules formulated in lipid nanoparticles (LNP) to treat liver metastasis of colon cancer.164 These formulations were actively taken up by the liver but treatment of cancer to other tissues was not demonstrated.
As shown in this review, the development of novel approaches based on nanomaterials is one of the most active areas of development for future medicines for cancer treatment. Polymeric nanoparticles are one of the most promising alternatives for the near future. The knowledge generated in the last decades for the delivery of chemotherapeutic agents using biocompatible nanopolymers will trigger the development of novel formulations that may include combinations of chemotherapeutic agents together with one or several siRNA molecules targeting overexpressed proteins for a more efficient and less toxic medicines. These nanomaterials as well as gold and SPION nanoparticles also allow the introduction of specific targeting molecules that will direct the accumulation of the therapeutic molecules near the tumors increasing the passive accumulation of nanoparticles by the enhanced permeability and retention (EPR) effect. The addition of acid responsive polymers such as PEI and peptides may also increase the efficacy by facilitating endosomal escape. A limitation on the use of nanomaterials is the potential in vivo aggregation with serum proteins and subsequent removal by the RES. This undesired effect may be prevented by the incorporation of PEG.136
Finally, a novel approach with high potential is the use of DNA scaffolds for the delivery of siRNA.165 For example, DNA dumbbell,166 DNA nanoribbons,167 and spherical nucleic acids (SNA)168 have been described as alternative systems which are biodegradable, non-toxic, and also non-immunogenic. These DNA scaffolds have proved to be effective when have incorporated targeting ligands, siRNA drugs as well as chemotherapeutic drugs with high precision.169,170
siRNA in clinical trials for cancer therapy
Recently, RNAi-based treatment has been rapidly developed into clinical trials. In addition, it has been studied for treating diverse diseases, such as cancer, respiratory infection, AMD (age-related macular degeneration), glaucoma, and hypercholesterolemia, among others.171 So far, at least 20 clinical trials have been initiated using siRNA- and miRNA-based therapeutics.172 Although there are many ways to engage RNAi pathways, currently the majority of the clinical trials involve siRNA technology.114,173–175
In a recent report from a phase I study on patients with refractory or relapsed solid cancer, Davis et al provided the first clinical evidence that RNAi could be achieved by administering siRNA against the M2 subunit of ribonucleotide reductase (RRM2).109
Recently, the therapeutic potential of RNAi as a revolutionary modern class of medicine was demonstrated in cancer therapy for the treatment of inaccessible solid tumors via systemic administration by delivering Atu027176 and CALAA-01114 developed by Silence Therapeutics and Calando Pharmaceuticals Inc, respectively (Table 3). In 2008, CALAA-01 was employed for treating the first patient in a phase I clinical trial. In these studies, a siRNA nanoparticle targeting protein kinase N3 (PKN3) or RRM2 was administered intravenously for the treatment of solid tumors.114 Recently Atu027, a siRNA targeting protein kinase N3 (PKN3), is being used in a clinical trial together with gemcitabine for the treatment of advanced or metastatic pancreatic cancer (clinicaltrials.gov Identifier: NCT01808638).
 | Table 3 Selection of clinical trials against cancer using siRNA drugs |
An interesting therapeutic approach has been shown by Tabernero et al.177 In this study, two different siRNAs targeting two genes (vascular endothelial growth factor, VEGF and kinesin spindle protein, KSP) involved in angiogenesis were formulated in the same lipid formulation. The authors showed that siRNAs were able to promote mRNA cleavage and antitumor activity in humans. This lipid formulation (ALN-RSV) (Alnylam Pharmaceuticals) demonstrated complete regression of liver metastases and endometrial cancer in some cases.
A similar lipid formulation is being used for silencing Myc oncoprotein which is deregulated in over half of human malignancies. In a recent dose-escalation work, the clinical activity of DCR-MYC was evaluated in patients with advanced solid tumors, multiple myeloma, or lymphoma showing good clinical and metabolic responses over various dose levels.178
Recently, the use of 1,2-dioleoyl-sn-glycero-3-phosphatidylcholine liposomes for systemic siRNA delivery was evaluated in phase 1 clinical trial to inhibit Ephrin type-A receptor2 (EphA2) gene.179 The liposomal formulation was well tolerated, and clinical trials are ongoing.
In another approach, Silenseed has used the insertion of a specialized bio-polymeric “scaffold” containing the siRNA drug into the solid tumor core. The LODER® polymer matrix has been used in a clinical trial for silencing a mutated K-RAS oncogene relevant for pancreatic ductal adenocarcinoma.180
Although there are not a large number of siRNAs in advanced clinical trials, RNAi is a noteworthy mechanism for advanced novel therapeutics because in cancer cells, the functionality and expression of overexpressed damaging genes are significantly reduced by siRNAs. Nonetheless, once applying RNAi techniques in vivo, the most obstacle to achieving gene silencing is the delivery of therapeutic siRNA molecules.178 Thus, the clinical application of siRNA therapy faces several challenges to achieve effective dosages in target cells at safe and maintaining oligonucleotide stability in circulation.
The strategies for the observation of the distribution and therapeutic effects as well as techniques for enhancing cellular uptake also are essential.18,155 First clinical trials have shown encouraging results. An interesting direction is the use of combinations of chemotherapeutic agents and siRNA as well as the combination of several siRNA targeting overexpressed proteins involved in different metabolic routes such as oncogenes, control of the nucleotide pool, angiogenesis, telomeres, antiapoptotic genes, and so on. Finally, the use of exosomes derived from mesenchymal stromal cells will be evaluated in a clinical trial for the delivery KrasG12D siRNA in patients of pancreatic cancer with KrasG12D mutation (clinicaltrials.gov Identifier: NCT03608631). Exosomes are natural lipid-particles secreted by cells that play a crucial role in protecting and transporting macromolecules such as endogenous microRNA and mRNA. These interesting properties have triggered the interest in the use of such exosomes as potential nanovehicles for internalization of therapeutic molecules.
Conclusions
Cancer is accounted for one of the most prevalent diseases with high mortality rates in the world. It is considered one of the major challenges of medical treatment and health.181 For instance, colorectal cancer with more than 1.2 million new cases causes 600 thousand deaths per year and is the fourth cause of deaths worldwide.182 Nearly 50,000 people will die in the United States per year and nearly 135,000 new cases will be diagnosed.183
In addition to surgery and classical chemotherapeutic and radiation methods, there is a need for novel and less aggressive treatments. RNA interference can be considered a promising alternative for cancer therapy as it is less toxic than classical chemotherapy. In addition, siRNAs have been studied for the treatment of various human diseases such as genetic disorders, ocular conditions, cardiovascular diseases, viral infections, and cancers. The ability to target virtually any gene(s) is one of the most attractive aspects of siRNA therapeutics when treatments involving protein-based drugs or small molecules cannot be properly used.
However, a major restriction within the therapeutic applications of siRNA is the low cellular uptake of unmodified siRNAs as they cannot penetrate the cells with high efficiency. For this reason, siRNA molecules need to be complexed or conjugated with an appropriate carrier system. In addition, their fast degradation in cellular cytoplasm and plasma lead to short half-lives. For this reason, numerous strategies involving the combination of biocompatible and versatile non-viral carriers with siRNAs containing modifications and optimized sequences have resulted in potential RNAi-based drugs as efficient medicines into the clinic.
Although a large number of excellent works have been described, future studies are required to concentrate on the in vivo safety profiles of the nanoparticle-based delivery systems like polymers, cationic lipids, dendrimers, and inorganic nanoparticles, including undesirable cytotoxicity and immune stimulation. For the clinical usage of siRNA-based cancer therapeutics, the development of biocompatible, biodegradable, and safe biodegradable nanoparticle delivery systems is still necessary together with the development of simple and reproducible protocols for the production of batches for regulatory assessment and clinical trials.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Ebrahimi M, Moazzen F, Marmari V, et al. In silico analysis, cloning and expression of recombinant CD166 in E. coli BL21 (DE3) as a marker for detection and treatment of colorectal cancer. J Med Microbiol Diagnosis. 2017;06(01):1–6. doi:10.4172/2161-0703.1000249
2. Vries RGJ, Huch M, Clevers H. Stem cells and cancer of the stomach and intestine. Mol Oncol. 2010;4(5):373–384. doi:10.1016/j.molonc.2010.05.001
3. Dana H. An overview of cancer stem cell. J Stem Cell Res Ther. 2017;1(4):169–174. doi:10.15406/jsrt.2016.01.00029
4. Zhang X, Li X, You Q, Zhang X. Prodrug strategy for cancer cell-specific targeting: a recent overview. Eur J Med Chem. 2017;139:542–563. doi:10.1016/j.ejmech.2017.08.010
5. Hassanpour SH, Dehghani M. Review of cancer from perspective of molecular. J Cancer Res Pract. 2017;4(4):127–129. doi:10.1016/j.jcrpr.2017.07.001
6. Saraswathy M, Gong S. Recent developments in the co-delivery of siRNA and small molecule anticancer drugs for cancer treatment. Mater Today. 2014;17(6):298–306. doi:10.1016/j.mattod.2014.05.002
7. Endoh T, Ohtsuki T. Cellular siRNA delivery using cell-penetrating peptides modified for endosomal escape. Adv Drug Deliv Rev. 2009;61(9):704–709. doi:10.1016/j.addr.2009.04.005
8. Siegel R, Ma J, Zou Z, Jemal A. Cancer statistics, 2014. CA Cancer J Clin. 2014;64(1):9–29. doi:10.3322/caac.21208
9. Spano J-P, Azria D, Gonçalves A. Patients’ satisfaction in early breast cancer treatment: change in treatment over time and impact of HER2-targeted therapy. Crit Rev Oncol Hematol. 2015;94(3):270–278. doi:10.1016/j.critrevonc.2015.01.007
10. Wee Gan C, Chien S, Feng -S-S. Nanomedicine: enhancement of chemotherapeutical efficacy of docetaxel by using a biodegradable nanoparticle formulation. Curr Pharm Des. 2010;16(21):2308–2320. doi:10.2174/138161210791920487
11. Panowski S, Bhakta S, Raab H, Polakis P, Junutula JR. Site-specific antibody drug conjugates for cancer therapy. MAbs. 2013;6(1):34–45. doi:10.4161/mabs.27022
12. Leal M, Sapra P, Hurvitz SA, et al. Antibody–drug conjugates: an emerging modality for the treatment of cancer. Ann N Y Acad Sci. 2014;1321(1):41–54. doi:10.1111/nyas.12499
13. Jain S, Pathak K, Vaidya A. Molecular therapy using siRNA: recent trends and advances of multi target inhibition of cancer growth. Int J Biol Macromol. 2018;116:880–892. doi:10.1016/j.ijbiomac.2018.05.077
14. Mahmoodzad H, Ardaneh M, Zeinalinia E, et al. Microrna a new gate in cancer and human disease: a review. J Biol Sci. 2017;17(6):247–254. doi:10.3923/jbs.2017.247.254
15. Fire A, Xu S, Montgomery MK, Kostas SA, Driver SE, Mello CC. Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature. 1998;391:806. doi:10.1038/35888
16. Caplen NJ, Parrish S, Imani F, Fire A, Morgan RA. Specific inhibition of gene expression by small double-stranded RNAs in invertebrate and vertebrate systems. Proc Natl Acad Sci. 2001;98(17):9742–9747. doi:10.1073/pnas.171251798
17. Elbashir SM, Harborth J, Lendeckel W, Yalcin A, Weber K, Tuschl T. Duplexes of 21-nucleotide RNAs mediate RNA interference in cultured mammalian cells. Nature. 2001;411(6836):494–498. doi:10.1038/35078107
18. Kim DH, Rossi JJ. Strategies for silencing human disease using RNA interference. Nat Rev Genet. 2007;8(3):173–184. doi:10.1038/nrg2006
19. Dana H, Chalbatani GM, Mahmoodzadeh H, et al. Molecular mechanisms and biological functions of siRNA. Int J Biomed Sci. 2017;13(2):48–57.
20. Yang J. Patisiran for the treatment of hereditary transthyretin-mediated amyloidosis. Expert Rev Clin Pharmacol. 2019;12(2):95–99. doi:10.1080/17512433.2019.1567326
21. Lorenzer C, Dirin M, Winkler AM, Baumann V, Winkler J. Going beyond the liver: progress and challenges of targeted delivery of siRNA therapeutics. J Control Release. 2015;203:1–15. doi:10.1016/j.jconrel.2015.02.003
22. Hong CA, Nam YS. Functional nanostructures for effective delivery of small interfering RNA therapeutics. Theranostics. 2014;4(12):1211–1232. doi:10.7150/thno.8491
23. Lee S-Y, Lee SJ, Oh Y-K, et al. Stability and cellular uptake of polymerized siRNA (poly-siRNA)/polyethylenimine (PEI) complexes for efficient gene silencing. J Control Release. 2009;141(3):339–346. doi:10.1016/j.jconrel.2009.10.007
24. Sjouke B, Balak DMW, Beuers U, Ratziu V, Stroes ESG. Is mipomersen ready for clinical implementation? A transatlantic dilemma. Curr Opin Lipidol. 2013;24:4. doi:10.1097/MOL.0b013e328362dfd9
25. Burnett J, Rossi J. RNA-based therapeutics: current progress and future prospects. Chem Biol. 2012;19(1):60–71. doi:10.1016/j.chembiol.2011.12.008
26. Biswas S, Deshpande PP, Navarro G, Dodwadkar NS, Torchilin VP. Lipid modified triblock PAMAM-based nanocarriers for siRNA drug co-delivery. Biomaterials. 2013;34(4):1289–1301. doi:10.1016/j.biomaterials.2012.10.024
27. Whitehead KA, Langer R, Anderson DG. Knocking down barriers: advances in siRNA delivery. Nat Rev Drug Discov. 2009;8(2):129–138. doi:10.1038/nrd2742
28. Rettig GR, Behlke MA. Progress toward in vivo use of siRNAs-II. Mol Ther. 2012;20(3):483–512. doi:10.1038/mt.2011.263
29. Tsunoda S, Mazda O, Oda Y, et al. Sonoporation using microbubble BR14 promotes pDNA/siRNA transduction to murine heart. Biochem Biophys Res Commun. 2005;336(1):118–127. doi:10.1016/j.bbrc.2005.08.052
30. Kishida T, Asada H, Gojo S, et al. Sequence-specific gene silencing in murine muscle induced by electroporation-mediated transfer of short interfering RNA. J Gene Med. 2004;6(1):105–110. doi:10.1002/jgm.456
31. Morrissey DV, Blanchard K, Shaw L, et al. Activity of stabilized short interfering RNA in a mouse model of hepatitis B virus replication. Hepatology. 2005;41(6):1349–1356. doi:10.1002/hep.20702
32. Zhang S, Zhao B, Jiang H, Wang B, Ma B. Cationic lipids and polymers mediated vectors for delivery of siRNA. J Control Release. 2007;123(1):1–10. doi:10.1016/j.jconrel.2007.07.016
33. Kim WJ, Kim SW. Efficient siRNA delivery with non-viral polymeric vehicles. Pharm Res. 2009;26(3):657–666. doi:10.1007/s11095-008-9774-1
34. Gary DJ, Puri N, Won YY. Polymer-based siRNA delivery: perspectives on the fundamental and phenomenological distinctions from polymer-based DNA delivery. J Control Release. 2007;121(1–2):64–73. doi:10.1016/j.jconrel.2007.05.021
35. Tseng YC, Mozumdar S, Huang L. Lipid-based systemic delivery of siRNA. Adv Drug Deliv Rev. 2009;61(9):721–731. doi:10.1016/j.addr.2009.03.003
36. Hassani Z, Lemkine GF, Erbacher P, et al. Lipid-mediated siRNA delivery down-regulates exogenous gene expression in the mouse brain at picomolar levels. J Gene Med. 2005;7(2):198–207. doi:10.1002/jgm.659
37. Simeoni F, Morris MC, Heitz F, Divita G. Insight into the mechanism of the peptide-based gene delivery system MPG: implications for delivery of siRNA into mammalian cells. Nucleic Acids Res. 2003;31(11):2717–2724. doi:10.1093/nar/gkg385
38. Simeoni F, Morris MC, Heitz F, Divita G. Peptide-based strategy for siRNA delivery into mammalian cells BT - RNA silencing: methods and protocols. In: Carmichael GG, Totowa NJ, editors. Vol. 309. Humana Press; 2005:251–260. doi:10.1385/1-59259-935-4:251.
39. Farrow B, Evers BM, Iwamura T, Murillo C, O’Connor KL, Rychahou P. Inhibition of pancreatic cancer cell growth and induction of apoptosis with novel therapies directed against protein kinase A. Surgery. 2003;134(2):197–205. doi:10.1067/msy.2003.220
40. Schwarz DS, Hutvágner G, Haley B, Zamore PD. Evidence that siRNAs function as guides, not primers, in the Drosophila and human RNAi pathways. Mol Cell. 2002;10(3):537–548. doi:10.1016/S1097-2765(02)00651-2
41. Behlke MA. Chemical modification of siRNAs for in vivo use. Oligonucleotides. 2008;18(4):305–320. doi:10.1089/oli.2008.0164
42. Chiu Y, Rana TM. siRNA function in RNAi: a chemical modification analysis. Rna. 2003;9:1034–1048. doi:10.1261/rna.5103703.2000
43. Hall AHS, Wan J, Shaughnessy EE, Shaw BR, Alexander KA. RNA interference using boranophosphate siRNAs: structure-activity relationships. Nucleic Acids Res. 2004;32(20):5991–6000. doi:10.1093/nar/gkh936
44. Dowler T, Bergeron D, Tedeschi AL, Paquet L, Ferrari N, Damha MJ. Improvements in siRNA properties mediated by 2′-deoxy-2′-fluoro-β-D-arabinonucleic acid (FANA). Nucleic Acids Res. 2006;34(6):1669–1675. doi:10.1093/nar/gkl033
45. Watts JK, Deleavey GF, Damha MJ. Chemically modified siRNA: tools and applications. Drug Discov Today. 2008;13(19–20):842–855. doi:10.1016/j.drudis.2008.05.007
46. Xu C, Wang J. Delivery systems for siRNA drug development in cancer therapy. Asian J Pharm Sci. 2015;10(1):1–12. doi:10.1016/j.ajps.2014.08.011
47. Ghafouri-Fard S, Ghafouri-Fard S. siRNA and cancer immunotherapy. Immunotherapy. 2012;4(9):907–917. doi:10.2217/imt.12.87
48. Klippstein R, Pozo D. Nanotechnology-based manipulation of dendritic cells for enhanced immunotherapy strategies. Nanomedicine Nanotechnology Biol Med. 2010;6(4):523–529. doi:10.1016/j.nano.2010.01.001
49. Banchereau J, Steinman RM. Dendritic cells and the control of immunology. Nature. 1998;392:245–252. doi:10.1038/32588
50. Fong L, Engleman EG. Dendritic cells in cancer immunotherapy. Pathology. 2000;245–273. doi:10.1146/annurev.immunol.21.120601.141040
51. Constantino J, Gomes C, Falcão A, Neves BM, Cruz MT. Dendritic cell-based immunotherapy: a basic review and recent advances. Immunol Res. 2017;65(4):798–810. doi:10.1007/s12026-017-8931-1
52. Verra N, De Jong D, Bex A, et al. Infiltration of activated dendritic cells and T cells in renal cell carcinoma following combined cytokine immunotherapy. Eur Urol. 2005;48(3):527–533. doi:10.1016/j.eururo.2005.03.031
53. Van Poppel H, Joniau S, Van Gool SW. Vaccine therapy in patients with renal cell carcinoma. Eur Urol. 2009;55(6):1333–1344. doi:10.1016/j.eururo.2009.01.043
54. Asemissen AM, Brossart P. Vaccination strategies in patients with renal cell carcinoma. Cancer Immunol Immunother. 2009;58(7):1169–1174. doi:10.1007/s00262-009-0706-7
55. Anguille S, Smits EL, Lion E, Van Tendeloo VF, Berneman ZN. Clinical use of dendritic cells for cancer therapy. Lancet Oncol. 2014;15(7):257–267. doi:10.1016/S1470-2045(13)70585-0
56. Zhang AL, Purath U, Tarner IH, et al. Natural killer cells trigger differentiation of monocytes into dendritic cells. Blood. 2007;110(7):2484–2493. doi:10.1182/blood-2007-02-076364
57. Wang Z, Rao DD, Senzer N, Nemunaitis J. RNA interference and cancer therapy. Pharm Res. 2011:2983–2995. doi:10.1007/s11095-011-0604-5
58. Li M, Qian H, Ichim TE, et al. Induction of RNA interference in dendritic cells. Immunol Res. 2004;30(2):215–230. doi:10.1385/IR:30:2:215
59. Liu S, Liu D, Ji M, et al. An indoleamine 2, 3-dioxygenase siRNA nanoparticle-coated and Trp2-displayed recombinant yeast vaccine inhibits melanoma tumor growth in mice. J Control Release. 2018;273:1–12. doi:10.1016/j.jconrel.2018.01.013
60. Filipowicz W, Jaskiewicz L, Kolb FA, Pillai RS. Post-transcriptional gene silencing by siRNAs and miRNAs. Curr Opin Struct Biol. 2005;15:331–341. doi:10.1016/j.sbi.2005.05.006
61. He L, Hannon GJ. MicroRNAs: small RNAs with a big role in gene regulation. Nat Rev Genet. 2004;5(7):522–531. doi:10.1038/nrg1379
62. Castanotto D, Rossi JJ. The promises and pitfalls of RNA-interference-based therapeutics. Nature. 2009;457(7228):426–433. doi:10.1038/nature07758
63. Tatiparti K, Sau S, Kashaw S, Iyer A. siRNA delivery strategies: a comprehensive review of recent developments. Nanomaterials. 2017;7(4):77. doi:10.3390/nano7040077
64. Massadeh S, Al Aamery M. Nano-materials for gene therapy: an efficient way in overcoming challenges of gene delivery. J Biosens Bioelectron. 2016;07(01):1–12. doi:10.4172/2155-6210.1000195
65. Morris KV, Chan SW-L, Jacobsen SE, Looney DJ. Small interfering RNA-induced transcriptional gene silencing in human cells. Cell. 2004;305(1):1289–1293.
66. Li J, Xue S, Mao ZW. Nanoparticle delivery systems for siRNA-based therapeutics. J Mater Chem B. 2016;4(41):6620–6639. doi:10.1039/c6tb01462c
67. Oh YK, Park TG. siRNA delivery systems for cancer treatment. Adv Drug Deliv Rev. 2009;61(10):850–862. doi:10.1016/j.addr.2009.04.018
68. Chen M, Du Q, Zhang H-Y, Wahlestedt C, Liang Z. Vector-based siRNA delivery strategies for high-throughput screening of novel target genes. J RNAi Gene Silencing. 2005;1(1):5–11.
69. Lee NS, Dohjima T, Bauer G, et al. Expression of small interfering RNAs targeted against HIV-1 rev transcripts in human cells. Nat Biotechnol. 2002;20(5):500–505. doi:10.1038/nbt0502-500
70. Moffatt S. siRNA-based nanoparticles for cancer therapy: hurdles and hopes. MOJ Proteomics Bioinforma. 2017;4(6):4–6. doi:10.15406/mojpb.2016.04.00142
71. Jackson AL, Linsley PS. Recognizing and avoiding siRNA off-target effects for target identification and therapeutic application. Nat Rev Drug Discov. 2010;9(1):57–67. doi:10.1038/nrd3010
72. Marques JT, Williams BRG. Activation of the mammalian immune system by siRNAs. Nat Biotechnol. 2005;23:1399. doi:10.1038/nbt1161
73. Alagia A, Eritja R. siRNA and RNAi optimization. Wiley Interdiscip Rev RNA. 2016;7(3):316–329. doi:10.1002/wrna.1337
74. Bramsen JB, Pakula MM, Hansen TB, et al. A screen of chemical modifications identifies position-specific modification by UNA to most potently reduce siRNA off-target effects. Nucleic Acids Res. 2010;38(17):5761–5773. doi:10.1093/nar/gkq341
75. Suter SR, Ball-Jones A, Mumbleau MM, et al. Controlling miRNA-like off-target effects of an siRNA with nucleobase modifications. Org Biomol Chem. 2017;15(47):10029–10036. doi:10.1039/c7ob02654d
76. Alagia A, Jorge AF, Aviñó A, et al. Exploring PAZ/3′-overhang interaction to improve siRNA specificity. A combined experimental and modeling study. Chem Sci. 2018;9(8):2074–2086. doi:10.1039/c8sc00010g
77. Kang HS, Lee D, Li CJ, et al. Asymmetric shorter-duplex siRNA structures trigger efficient gene silencing with reduced nonspecific effects. Mol Ther. 2009;17(4):725–732. doi:10.1038/mt.2008.298
78. Sioud M. Advances in RNA sensing by the immune system: separation of siRNA unwanted effects from RNA interference. Methods Mol Biol. 2010;629:33–52. doi:10.1007/978-1-60761-657-3_3
79. Eberle F, Peter M, Richert C, et al. Modifications in small interfering RNA that separate immunostimulation from RNA interference. J Immunol. 2014;180(5):3229–3237. doi:10.4049/jimmunol.180.5.3229
80. Slobodkin G, Wilkinson L, Pence C, et al. Versatile cationic lipids for siRNA delivery. J Control Release. 2011;158(2):269–276. doi:10.1016/j.jconrel.2011.11.006
81. Tam YYC, Chen S, Cullis PR. Advances in lipid nanoparticles for siRNA delivery. Pharmaceutics. 2013;5(3):498–507. doi:10.3390/pharmaceutics5030498
82. Semple SC, Akinc A, Chen J, et al. Rational design of cationic lipids for siRNA delivery. Nat Biotechnol. 2010;28:172. doi:10.1038/nbt.1602
83. Love KT, Mahon KP, Christopher G, et al. Correction for Love et al., Lipid-like materials for low-dose, in vivo gene silencing. Proc Natl Acad Sci. 2010;107(21):9915. doi:10.1073/pnas.1005136107
84. Abrams MT, Koser ML, Seitzer J, et al. Evaluation of efficacy, biodistribution, and inflammation for a potent siRNA nanoparticle: effect of dexamethasone co-treatment. Mol Ther. 2010;18(1):171–180. doi:10.1038/mt.2009.208
85. Grijalvo S, Puras G, Zárate J, et al. Cationic niosomes as non-viral vehicles for nucleic acids: challenges and opportunities in gene delivery. Pharmaceutics. 2019;11:2. doi:10.3390/pharmaceutics11020050
86. Pattni BS, Chupin VV, Torchilin VP. New developments in liposomal drug delivery. Chem Rev. 2015;115(19):10938–10966. doi:10.1021/acs.chemrev.5b00046
87. Derycke A. Liposomes for photodynamic therapy. Adv Drug Deliv Rev. 2003;56(1):17–30. doi:10.1016/j.addr.2003.07.014
88. Minko T, Pakunlu RI, Wang Y, Khandare JJ, Saad M. New generation of liposomal drugs for cancer. Anticancer Agents Med Chem. 2014;6(6):537–552. doi:10.2174/187152006778699095
89. Ozpolat B, Sood AK, Lopez-Berestein G. Liposomal siRNA nanocarriers for cancer therapy. Adv Drug Deliv Rev. 2014;66:110–116. doi:10.1016/j.addr.2013.12.008
90. Duchemin A, Evrard B, Piel G, Sanna V, Lechanteur A, Mottet D. Cationic liposomes carrying siRNA: impact of lipid composition on physicochemical properties, cytotoxicity and endosomal escape. Nanomaterials. 2018;8(5):270. doi:10.3390/nano8050270
91. Santel A, Aleku M, Keil O, et al. A novel siRNA-lipoplex technology for RNA interference in the mouse vascular endothelium. Gene Ther. 2006;13(16):1222–1234. doi:10.1038/sj.gt.3302777
92. Aleku M, Schulz P, Keil O, et al. Atu027, a liposomal small interfering RNA formulation targeting protein kinase N3, inhibits cancer progression. Cancer Res. 2008;68(23):9788–9798. doi:10.1158/0008-5472.CAN-08-2428
93. Wu SY, McMillan NAJ. Lipidic systems for in vivo siRNA delivery. Aaps J. 2009;11(4):639–652. doi:10.1208/s12248-009-9140-1
94. Landen CN, Merritt WM, Mangala LS, et al. Intraperitoneal delivery of liposomal siRNA for therapy of advanced ovarian cancer. Cancer Biol Ther. 2006;5(12):1708–1713. doi:10.4161/cbt.5.12.3468
95. Cheng Y, Ji Y. RGD-modified polymer and liposome nanovehicles: recent research progress for drug delivery in cancer therapeutics. Eur J Pharm Sci. 2019;128:8–17. doi:10.1016/j.ejps.2018.11.023
96. Mattern-Schain SI, Fisher RK, West PC, et al. Cell mimetic liposomal nanocarriers for tailored delivery of vascular therapeutics. Chem Phys Lipids. 2019;218:149–157. doi:10.1016/j.chemphyslip.2018.12.009
97. Zimmermann TS, Lee ACH, Akinc A, et al. RNAi-mediated gene silencing in non-human primates. Nature. 2006;441(1):111–114. doi:10.1038/nature04688
98. Ho W, Zhang XQ, Xu X. Biomaterials in siRNA delivery: a comprehensive review. Adv Healthc Mater. 2016;5(21):2715–2731. doi:10.1002/adhm.201600418
99. Sood V, Honko AN, MacLachlan I, et al. Postexposure protection of non-human primates against a lethal Ebola virus challenge with RNA interference: a proof-of-concept study. Lancet. 2010;375(9729):1896–1905. doi:10.1016/s0140-6736(10)60357-1
100. Nikam RR, Gore KR. Journey of siRNA: clinical developments and targeted delivery. Nucleic Acid Ther. 2018;28(4):209–224. doi:10.1089/nat.2017.0715
101. Lin PJC, Tam YK. Chapter 9 - controlling protein expression by delivery of RNA therapeutics using lipid nanoparticles. In: Filice M, Ruiz-Cabello JBT-NA-N editors. Micro and Nano Technologies. Amsterdam, the Netherlands: Elsevier; 2019:277–310. doi:10.1016/B978-0-12-814470-1.00009-5.
102. Zhao J, Weng G, Li J, Zhu J, Zhao J. Polyester-based nanoparticles for nucleic acid delivery. Mater Sci Eng C. 2018;92:983–994. doi:10.1016/j.msec.2018.07.027
103. Rozema DB, Lewis DL, Wakefield DH, et al. Dynamic PolyConjugates for targeted in vivo delivery of siRNA to hepatocytes. Proc Natl Acad Sci. 2007;104(32):12982LP–12987. doi:10.1073/pnas.0703778104
104. Wagner E. polymers for sirna delivery: inspired by viruses to be targeted, dynamic, and precise. Acc Chem Res. 2012;45(7):1005–1013. doi:10.1021/ar2002232
105. Cheng R, Feng F, Meng F, Deng C, Feijen J, Zhong Z. Glutathione-responsive nano-vehicles as a promising platform for targeted intracellular drug and gene delivery. J Control Release. 2011;152(1):2–12. doi:10.1016/j.jconrel.2011.01.030
106. Farra R, Musiani F, Perrone F, et al. Polymer-mediated delivery of siRNAs to hepatocellular carcinoma: variables affecting specificity and effectiveness. Molecules. 2018;23(4). doi:10.3390/molecules23040777
107. Parmar RG, Poslusney M, Busuek M, et al. Novel endosomolytic poly(amido amine) polymer conjugates for systemic delivery of siRNA to hepatocytes in rodents and nonhuman primates. Bioconjug Chem. 2014;25(5):896–906. doi:10.1021/bc400527e
108. Kanasty R, Dorkin JR, Vegas A, Anderson D. Delivery materials for siRNA therapeutics. Nat Mater. 2013;12(11):967–977. doi:10.1038/nmat3765
109. Davis ME. The first targeted delivery of siRNA in humans via a self-assembling, cyclodextrin polymer-based nanoparticle: from concept to clinic. Mol Pharm. 2009;6(3):659–668. doi:10.1021/mp900015y
110. O’Mahony AM, Godinho BMDC, Ogier J, et al. Click-modified cyclodextrins as nonviral vectors for neuronal siRNA delivery. ACS Chem Neurosci. 2012;3(10):744–752. doi:10.1021/cn3000372
111. Díaz-Moscoso A, Le Gourriérec L, Gómez-García M, et al. Polycationic amphiphilic cyclodextrins for gene delivery: synthesis and effect of structural modifications on plasmid DNA complex stability, cytotoxicity, and gene expression. Chem A Eur J. 2009;15(46):12871–12888. doi:10.1002/chem.200901149
112. Miele E, Spinelli GP, Miele E, et al. Nanoparticle-based delivery of small interfering RNA: challenges for cancer therapy. Int J Nanomedicine. 2012;7:3637–3657. doi:10.2147/IJN.S23696
113. Sica G, Chen Z, Wang Z, et al. RRM2 regulates Bcl-2 in head and neck and lung cancers: a potential target for cancer therapy. Clin Cancer Res. 2013;19(13):3416–3428. doi:10.1158/1078-0432.ccr-13-0073
114. Davis ME, Seligson D, Tolcher A, et al. Evidence of RNAi in humans from systemically administered siRNA via targeted nanoparticles. Nature. 2010;464(7291):1067–1070. doi:10.1038/nature08956
115. Hovgaard MB, Rahbek UL, Glud SZ, et al. RNA interference in vitro and in vivo using a novel chitosan/siRNA nanoparticle system. Mol Ther. 2006;14(4):476–484. doi:10.1016/j.ymthe.2006.04.010
116. Dai K, Winnik F, Qiu X, et al. Low molecular weight chitosan conjugated with folate for siRNA delivery in vitro: optimization studies. Int J Nanomedicine. 2012;7:5833. doi:10.2147/ijn.s35567
117. De Smedt SC, Nysten B, Le Duff CS, et al. Chitosan nanoparticles for siRNA delivery: optimizing formulation to increase stability and efficiency. J Control Release. 2014;176:54–63. doi:10.1016/j.jconrel.2013.12.026
118. Kim YK, Minai-Tehrani A, Lee JH, Cho CS, Cho MH, Jiang HL. Therapeutic efficiency of folated poly(ethylene glycol)-chitosan-graft-polyethylenimine-Pdcd4 complexes in H-ras12V mice with liver cancer. Int J Nanomedicine. 2013;8:1489–1498. doi:10.2147/IJN.S42949
119. Kankala RK, Lin XF, Song HF, et al. Supercritical fluid-assisted decoration of nanoparticles on porous microcontainers for codelivery of therapeutics and inhalation therapy of diabetes. ACS Biomater Sci Eng. 2018;4(12):4225–4235. doi:10.1021/acsbiomaterials.8b00992
120. Xu P-Y, Kankala RK, Pan Y-J, Yuan H, Wang S-B, Chen A-Z. Overcoming multidrug resistance through inhalable siRNA nanoparticles-decorated porous microparticles based on supercritical fluid technology. Int J Nanomedicine. 2018;13:4685–4698. doi:10.2147/IJN.S169399
121. Wang S-B, Chen A-Z, Tang N, Su X-Q, Kang Y-Q. Preparation and antitumor effect evaluation of composite microparticles co-loaded with siRNA and paclitaxel by a supercritical process. J Mater Chem B. 2015;3(31):6439–6447. doi:10.1039/c5tb00715a
122. Kankala RK, Chen B-Q, Liu C-G, Tang H-X, Wang S-B, Chen A-Z. Solution-enhanced dispersion by supercritical fluids: an ecofriendly nanonization approach for processing biomaterials and pharmaceutical compounds. Int J Nanomedicine. 2018;13:4227–4245. doi:10.2147/IJN.S166124
123. Licciardi M, Li Volsi A, Sardo C, Mauro N, Cavallaro G, Giammona G. Inulin-ethylenediamine coated SPIONs magnetoplexes: a promising tool for improving siRNA delivery. Pharm Res. 2015;32(11):3674–3687. doi:10.1007/s11095-015-1726-y
124. Sardo C, Farra R, Licciardi M, et al. Development of a simple, biocompatible and cost-effective Inulin-Diethylenetriamine based siRNA delivery system. Eur J Pharm Sci. 2015;75:60–71. doi:10.1016/j.ejps.2015.03.021
125. Sardo C, Craparo EF, Porsio B, Giammona G, Cavallaro G. Improvements in rational design strategies of inulin derivative polycation for siRNA delivery. Biomacromolecules. 2016;17(7):2352–2366. doi:10.1021/acs.biomac.6b00281
126. Aigner A. Delivery systems for the direct application of siRNAs to induce RNA interference (RNAi) in vivo. J Biomed Biotechnol. 2006;2006:1–15. doi:10.1155/JBB/2006/71659
127. Akhtar S, Benter IF. Review series nonviral delivery of synthetic siRNAs in vivo. J Clin Invest. 2007;117(12):3623–3632. doi:10.1172/JCI33494.following
128. Judge A, Fritz EA, Phelps JR, et al. Postexposure protection of guinea pigs against a lethal ebola virus challenge is conferred by RNA interference. J Infect Dis. 2006;193(12):1650–1657. doi:10.1086/504267
129. Bai A, Chen J, Filip L, Nguyen T, Ge Q, Eisen HN. Inhibition of influenza virus production in virus-infected mice by RNA interference. Proc Natl Acad Sci. 2004;101(23):8676–8681. doi:10.1073/pnas.0402486101
130. Urban-Klein B, Werth S, Abuharbeid S, Czubayko F, Aigner A. RNAi-mediated gene-targeting through systemic application of polyethylenimine (PEI)-complexed siRNA in vivo. Gene Ther. 2004;12:461. doi:10.1038/sj.gt.3302425
131. Akhtar S, Benter I. Toxicogenomics of non-viral drug delivery systems for RNAi: potential impact on siRNA-mediated gene silencing activity and specificity. Adv Drug Deliv Rev. 2007;59(2–3):164–182. doi:10.1016/j.addr.2007.03.010
132. Pandey AP, Sawant KK. Polyethylenimine: A versatile, multifunctional non-viral vector for nucleic acid delivery. Mater Sci Eng C. 2016;68:904–918. doi:10.1016/j.msec.2016.07.066
133. Wang J, Lu Z, Wientjes MG, Au JL-S. Delivery of siRNA therapeutics: barriers and carriers. Aaps J. 2010;12(4):492–503. doi:10.1208/s12248-010-9210-4
134. Fornaguera C, Grijalvo S, Galán M, et al. Novel non-viral gene delivery systems composed of carbosilane dendron functionalized nanoparticles prepared from nano-emulsions as non-viral carriers for antisense oligonucleotides. Int J Pharm. 2015;478(1):113–123. doi:10.1016/j.ijpharm.2014.11.031
135. Khan A, Benboubetra M, Sayyed PZ, et al. Sustained polymeric delivery of gene silencing antisense ODNs, siRNA, DNAzymes and ribozymes: in vitro and in vivo studies. J Drug Target. 2004;12(6):393–404. doi:10.1080/10611860400003858
136. Svenson S, Case RI, Cole RO, et al. Tumor selective silencing using an RNAi-conjugated polymeric nanopharmaceutical. Mol Pharm. 2016;13(3):737–747. doi:10.1021/acs.molpharmaceut.5b00608
137. Byeon Y, Lee J-W, Choi WS, et al. CD44-targeting PLGA nanoparticles incorporating paclitaxel and FAK siRNA overcome chemoresistance in epithelial ovarian cancer. Cancer Res. 2018;78(21):6247LP–6256. doi:10.1158/0008-5472.CAN-17-3871
138. Winkler J. Nanomedicines based on recombinant fusion proteins for targeting therapeutic siRNA oligonucleotides. Ther Deliv. 2011;2(7):891–905. doi:10.4155/tde.11.56
139. Kaneshiro TL, Lu ZR. Targeted intracellular codelivery of chemotherapeutics and nucleic acid with a well-defined dendrimer-based nanoglobular carrier. Biomaterials. 2009;30(29):5660–5666. doi:10.1016/j.biomaterials.2009.06.026
140. Bhatia S, Zhu H, Birjiniuk A, et al. Functional delivery of siRNA in mice using dendriworms. ACS Nano. 2009;3(9):2495–2504. doi:10.1021/nn900201e
141. Zhou J, Shum KT, Burnett JC, Rossi JJ. Nanoparticle-based delivery of RNAi therapeutics: progress and challenges. Pharmaceuticals. 2013;6(1):85–107. doi:10.3390/ph6010085
142. Cheung W, Pontoriero F, Taratula O, Chen AM, He H. DNA and carbon nanotubes as medicine. Adv Drug Deliv Rev. 2010;62(6):633–649. doi:10.1016/j.addr.2010.03.007
143. Lynn Kirkpatrick D, Weiss M, Naumov A, Bartholomeusz G, Bruce Weisman R, Gliko O. Carbon nanotubes: solution for the therapeutic delivery of siRNA? Materials (Basel). 2012;5(2):278–301. doi:10.3390/ma5020278
144. Zhang Z, Yang X, Zhang Y, et al. Delivery of telomerase reverse transcriptase small interfering RNA in complex with positively charged single-walled carbon nanotubes suppresses tumor growth. Clin Cancer Res. 2006;12(16):4933–4939. doi:10.1158/1078-0432.CCR-05-2831
145. Jiang Y, Huo S, Hardie J, Liang X-J, Rotello VM. Progress and perspective of inorganic nanoparticle-based siRNA delivery systems. Expert Opin Drug Deliv. 2016;13(4):547–559. doi:10.1517/17425247.2016.1134486
146. Gindy ME, Prud’homme RK. Multifunctional nanoparticles for imaging, delivery and targeting in cancer therapy. Expert Opin Drug Deliv. 2009;6(8):865–878. doi:10.1517/17425240902932908
147. Sangvanich T, Bejan D, Gu S, Yantasee W, Reda M, Ngamcherdtrakul W. Lyophilization and stability of antibody-conjugated mesoporous silica nanoparticle with cationic polymer and PEG for siRNA delivery. Int J Nanomedicine. 2018;13:4015–4027. doi:10.2147/ijn.s164393
148. Tiash S, Kamaruzman NIB, Chowdhury EH. Carbonate apatite nanoparticles carry siRNA(s) targeting growth factor receptor genes egfr1 and erbb2 to regress mouse breast tumor. Drug Deliv. 2017;24(1):1721–1730. doi:10.1080/10717544.2017.1396385
149. Courty J, Latorre A, Villanueva A, et al. Efficient treatment of breast cancer xenografts with multifunctionalized iron oxide nanoparticles combining magnetic hyperthermia and anti-cancer drug delivery. Breast Cancer Res. 2015;17(1):1–17. doi:10.1186/s13058-015-0576-1
150. Cai D, Long R, Liu Y, et al. Bacterial magnetosomes-based nanocarriers for co-delivery of cancer therapeutics in vitro. Int J Nanomedicine. 2018;13:8269–8279. doi:10.2147/ijn.s180503
151. Wu Z, Shen J, Zhang L-M, et al. Magnetic cationic amylose nanoparticles used to deliver survivin-small interfering RNA for gene therapy of hepatocellular carcinoma in vitro. Nanomaterials. 2017;7(5):110. doi:10.3390/nano7050110
152. Roca M, Aj H. Probing cells with noble metal nanoparticle aggregates. Nanomedicine. 2008;3(4):555–565. doi:10.2217/17435889.3.4.555
153. Xu ZP, Zeng QH, Lu GQ, Yu AB. Inorganic nanoparticles as carriers for efficient cellular delivery. Chem Eng Sci. 2006;61(3):1027–1040. doi:10.1016/j.ces.2005.06.019
154. Wong JKL, Habib N, Seifalian AM, Mohseni R, Hamidieh AA, MacLaren RE. Will nanotechnology bring new hope for gene delivery? Trends Biotechnol. 2017;35(5):434–451. doi:10.1016/j.tibtech.2016.12.009
155. Mendes R, Fernandes AR, Baptista PV. Gold nanoparticle approach to the selective delivery of gene silencing in cancer-The case for combined delivery? Genes (Basel). 2017;8(3):94. doi:10.3390/genes8030094
156. Hou W, Wei P, Kong L, Guo R, Wang S, Shi X. Partially PEGylated dendrimer-entrapped gold nanoparticles: A promising nanoplatform for highly efficient DNA and siRNA delivery. J Mater Chem B. 2016;4(17):2933–2943. doi:10.1039/c6tb00710d
157. Tokatlian T, Segura T. siRNA applications in nanomedicine. Wiley Interdiscip Rev Nanomedicine Nanobiotechnology. 2010;2(3):305–315. doi:10.1002/wnan.81
158. Benitez-Del-Castillo JM, Moreno-Montañés J, Jiménez-Alfaro I, et al. Safety and efficacy clinical trials for SYL1001, a novel short interfering RNA for the treatment of dry eye disease. Investig Ophthalmol Vis Sci. 2016;57(14):6447–6454. doi:10.1167/iovs.16-20303
159. Perales JC, Marquez VE, Eritja R, Ocampo SM, Terrazas M. Effect of north bicyclo[3.1.0]hexane 2′-Deoxy-pseudosugars on RNA Interference: a novel class of siRNA modification. ChemBioChem. 2011;12(7):1056–1065. doi:10.1002/cbic.201000791
160. Grijalvo S, Alagia A, Jorge AF, Eritja R. Covalent strategies for targeting messenger and non-coding RNAs: an updated review on siRNA, miRNA and antimiR conjugates. Genes (Basel). 2018;9:2. doi:10.3390/genes9020074
161. Soutschek J, Akinc A, Bramlage B, et al. Therapeutic silencing of an endogenous gene by systemic administration of modified siRNAs. Nature. 2004;432(7014):173–178. doi:10.1038/nature03121
162. Manoharan M, Andree C, Amaya P, et al. Identification of siRNA delivery enhancers by a chemical library screen. Nucleic Acids Res. 2015;43(16):7984–8001. doi:10.1093/nar/gkv762
163. Nair JK, Willoughby JLS, Chan A, et al. Multivalent N-Acetylgalactosamine-conjugated siRNA localizes in hepatocytes and elicits robust RNAi-mediated gene silencing. J Am Chem Soc. 2014;136(49):16958–16961. doi:10.1021/ja505986a
164. Meyers RE, Weiss GJ, Clausen VA, et al. First-in-humans trial of an RNA interference therapeutic targeting VEGF and KSP in cancer patients with liver involvement. Cancer Discov. 2013;3(4):406–417. doi:10.1158/2159-8290.cd-12-0429
165. Donahoe JS, Nahrendorf M, Langer R, et al. Molecularly self-assembled nucleic acid nanoparticles for targeted in vivo siRNA delivery. Nat Nanotechnol. 2012;7(6):389–393. doi:10.1038/nnano.2012.73
166. Salvans C, Brun-Heath I, Ivani I, et al. Rational design of novel N-alkyl-N capped biostable RNA nanostructures for efficient long-term inhibition of gene expression. Nucleic Acids Res. 2016;44(9):4354–4367. doi:10.1093/nar/gkw169
167. He C, Weizmann Y, Lin W, Liu D, Gannett TR, Chen G. Enzymatic synthesis of periodic DNA nanoribbons for intracellular pH sensing and gene silencing. J Am Chem Soc. 2015;137(11):3844–3851. doi:10.1021/ja512665z
168. Ruan W, Zheng M, An Y, et al. DNA nanoclew templated spherical nucleic acids for siRNA delivery. Chem Commun. 2018;54(29):3609–3612. doi:10.1039/c7cc09257a
169. Jorge AF, Eritja R. Overview of DNA self-assembling: progresses in biomedical applications. Pharmaceutics. 2018;10:4. doi:10.3390/pharmaceutics10040268
170. Li H, Wang L, Fan C, Gu H, Hu Q. DNA nanotechnology-enabled drug delivery systems. Chem Rev. 2018. doi:10.1021/acs.chemrev.7b00663
171. Ni Z, Hui P. Emerging pharmacologic therapies for wet age-related macular degeneration. Ophthalmologica. 2009;223(6):401–410. doi:10.1159/000228926
172. Chakraborty C, Sharma AR, Sharma G, Doss CGP, Lee S-S. Therapeutic miRNA and siRNA: moving from bench to clinic as next generation medicine. Mol Ther Nucleic Acids. 2017;8:132–143. doi:10.1016/j.omtn.2017.06.005
173. Xin Y, Huang M, Guo WW, Huang Q, Zhang L, Jiang G. Nano-based delivery of RNAi in cancer therapy. Mol Cancer. 2017;16(1):1–9. doi:10.1186/s12943-017-0683-y
174. Shim MS, Wong S, Kwon YJ. SiRNA as a conventional drug in the clinic? Challenges and current technologies. Drug Discov Today Technol. 2012;9(2):e167–e173. doi:10.1016/j.ddtec.2012.01.003
175. Ferrari M. Vectoring siRNA therapeutics into the clinic. Nat Rev Clin Oncol. 2010;7:485. doi:10.1038/nrclinonc.2010.131
176. Santel A, Kaufmann J, Strumberg D, et al. First-in-human phase I Study of the liposomal RNA interference therapeutic Atu027 in patients with advanced solid tumors. J Clin Oncol. 2014;32(36):4141–4148. doi:10.1200/jco.2013.55.0376
177. Tabernero J, Shapiro GI, LoRusso PM, et al. First-in-humans trial of an RNA interference therapeutic targeting VEGF and KSP in cancer patients with liver involvement. Cancer Discov. 2013;3(4):406LP–417. doi:10.1158/2159-8290.CD-12-0429
178. Tolcher AW, Papadopoulos KP, Patnaik A, et al. Safety and activity of DCR-MYC, a first-in-class Dicer-substrate small interfering RNA (DsiRNA) targeting MYC, in a phase I study in patients with advanced solid tumors. J Clin Oncol. 2015;33(15_suppl):11006. doi:10.1200/jco.2015.33.15_suppl.11006
179. Wagner MJ, Sood AK, Lopez-Berestein G, et al. Preclinical mammalian safety studies of EPHARNA (DOPC Nanoliposomal EphA2-Targeted siRNA). Mol Cancer Ther. 2017;16(6):1114–1123. doi:10.1158/1535-7163.mct-16-0541
180. Golan T, Khvalevsky EZ, Hubert A, et al. RNAi therapy targeting KRAS in combination with chemotherapy for locally advanced pancreatic cancer patients. Oncotarget. 2015;6(27):24560–24570. doi:10.18632/oncotarget.4183
181. Mahmoodzadeh H, Moazzen F, Mehmandoost N, et al. Cloning and expression of C2 and V Domains of ALCAM Protein in E. coli BL21 (DE3). Clin Microbiol Open Access. 2017;06(01):1–6. doi:10.4172/2327-5073.1000271
182. Dana H, Marmari V, Mazraeh A, Ghamari A, Forghanifard MM. Cloning and expression of the V-domain of the CD166 in prokaryotic host cell. Int J Cancer Ther Oncol. 2017;5(1).
183. Dana H. CD166 as a stem cell marker? A potential target for therapy colorectal cancer? J Stem Cell Res Ther. 2017;1(6):6–9. doi:10.15406/jsrt.2016.01.00041
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.